Abstract
Diabetes mellitus is a growing concern in today’s population and a common cause of significant morbidity and mortality in those affected. Diabetic myonecrosis is an uncommon condition that can be seen in uncontrolled, long-standing diabetic patients who complain of localized, sudden-onset muscle pain. We present a 57-year-old woman with type 2 diabetes mellitus who presented to the emergency department with left-sided pelvic pain for the past 2 months. The pain had progressively gotten worse with a significant reduction in the range of motion of her left hip. Magnetic resonance imaging imaging revealed focal enhancement, edema, and enlargement of the left iliacus muscle. Assessment of serum glucose and hemoglobin A1c revealed a history of poor diabetic control, eventually giving rise to a diagnosis of diabetic myonecrosis. She was discharged with a new diabetic regimen, pain control, and outpatient physical therapy.
Introduction
Type 2 diabetes mellitus (T2DM) is an exceedingly common diagnosis that can chronically contribute to a variety of problems if left uncontrolled. 1 We present an unusual case of diabetic myonecrosis in a middle-aged woman complaining of widespread proximal muscle weakness with localized pelvic pain. Recognition and treatment of diabetic myonecrosis will be discussed, along with a review of inflammatory myopathies that may often present with similar complaints.
Case presentation
A 57-year-old Caucasian woman with a past medical history of a provoked pulmonary embolism (04/2017), T2DM, hypertension, hyperlipidemia, and chronic headaches presented to the emergency department complaining of left-sided pelvic pain for the past 2 months. At this time, the patient also complained of diarrhea and fever for the past 2 months. The patient states the pelvic pain had increased gradually and had progressively gotten more painful. The pain was sharp, 10 out of 10 and radiated to the back and to the left hip. The patient stated the pain was constant and aggravated by movement. She also complained of non-bloody, loose diarrhea for the past 2 months with no resolution. The patient admitted to an episode of fever at home registering at 102.4°F. Upon further questioning, the patient had experienced proximal muscle weakness, with difficulty combing her hair and reaching for items above her head. The proximal muscle weakness was present in the shoulders and the hips bilaterally. The patient had experienced gait abnormalities secondary to the muscle weakness, causing her to lose balance on multiple occasions, but denies any falls. She stated the proximal muscle weakness was present prior to the onset of her localized pelvic pain. The patient had completed therapy with an anticoagulant for her pulmonary embolism and denies recent antibiotic use. She further denied any trauma, vision changes, chest pain, dyspnea, or blood in the stool. Review of systems was otherwise unremarkable. Patient denies current or prior diagnosis of retinopathy, nephropathy, or peripheral neuropathy.
Family history was significant for a father with T2DM, a sister with Friedreich’s ataxia, a first cousin with an unknown autoimmune disorder, a maternal uncle with colon cancer, and a paternal aunt with breast cancer.
Vital signs on admission were as follows: blood pressure, 150/73 mmHg; heart rate, 101 beats/min; respiratory rate, 20 breathes/min, temperature, 101.9°F; and oxygen saturation, 97% on room air. General physical examination revealed a patient who is alert and oriented, cooperative but in mild distress. Head, eyes, ears, nose, and throat examination (HEENT) was negative for facial edema, rashes, or visual defects. Abdominal exam was significant for left lower quadrant pain on superficial and deep palpation. Musculoskelatal exam revealed tenderness to palpation in the left anterior and lateral hip, with reduced range of motion (ROM) in both hips; the left more affected than the right. Upper extremities were non-tender and had full ROM. No rashes, lesions, or signs of trauma noted on skin examination. Neurological exam revealed normal muscle strength and deep tendon reflexes (DTRs) in both upper extremities. Motor strength was 2/5 in the left lower thigh and 3/5 in the right lower thigh, and DTR was 2+ in both lower extremities, including achilles and patellar tendons. Bedside monofilament test revealed no sensory deficits in the lower extremities. Cardiovascular and pulmonary examinations were normal.
Initial lab work up upon admission revealed mild anemia (hemoglobin: 10.1 g/dL and hematocrit: 31.5%) but no leukocytosis (white blood cells 10.6 K/uL). A comprehensive metabolic panel (CMP) revealed a low albumin (2.8 g/dL), a total protein of 7.9 g/dL, calcium of 9.1 mg/dL (10 mg/dL when corrected), blood urea nitrogen (BUN) and creatinine of 21 and 0.9 mg/dL, respectively, and a high serum glucose (296 mg/dL). Hemoglobin A1c was 9.7%. Blood and urine cultures were obtained to rule out an infectious systemic process. Serum protein electrophoresis (SPEP) and urine protein electrophoresis (UPEP) were obtained for a mildly increased protein gap and anemia. Blood cultures, urine cultures, SPEP, and UPEP were all negative. At this point, a myositis workup had been initiated secondary to proximal muscle weakness and reduced ROM on physical exam. Serum levels of creatine kinase (CK) were obtained on two separate occasions. In addition, erythrocyte sedimentation rate (ESR), C-reactive protein (CRP), lactate dehydrogenase (LDH), and aldolase were also obtained. CK levels, LDH, and aldolase were within normal limits; however, ESR (112 mm/h) and CRP (125.5 mg/L) were markedly elevated. Furthermore, specific markers for inflammatory myositis and general autoimmune pathologies were negative, including anti-Mi-2, anti–signal recognition particle (SRP), anti-Jo-1, antinuclear antibody (ANA), and anti-Smith/ribonucleoprotien (Sm/RNP).
An abdominal and pelvic computed tomography (CT) scan was obtained on day of admission with benign, non-specific findings. A follow-up abdominal and pelvic magnetic resonance imaging (MRI) with and without contrast was subsequently obtained 48 h later and had revealed edema ventral to the psoas and iliacus muscles bilaterally, enhancement of the left iliacus muscles (Figure 1) and asymmetrically enlarged left iliacus in comparison to the right (Figure 2).

Coronal view of a pelvic MRI showing significant enlargement and enhancement of the left iliacus muscle in comparison to the right.
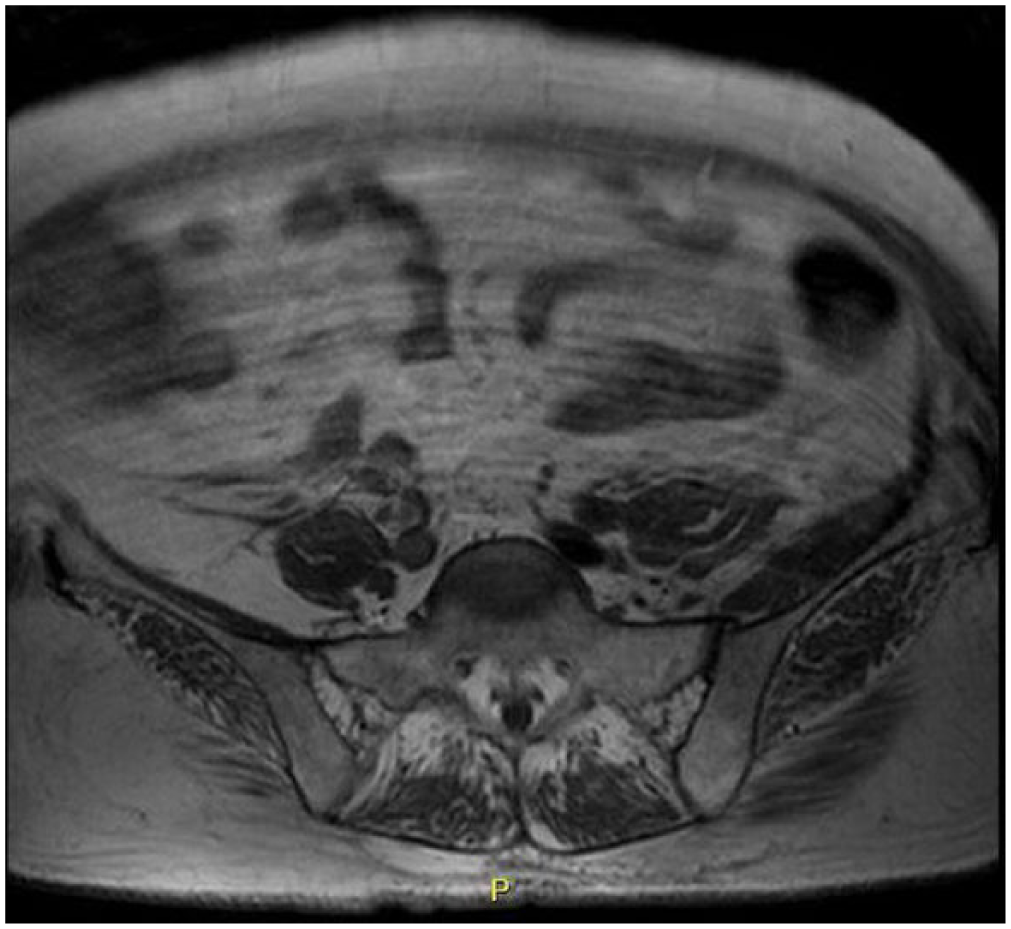
Transverse view of a pelvic MRI showing significant enlargement of the left iliacus muscle in comparison to the right.
Basic lab work and vitals were monitored daily since admission with no significant changes noted. An extensive workup resulting in lack of conclusions had warranted a rheumatology consult, leading to a diagnosis of diabetic myonecrosis of the left iliacus muscle. Further inspection of laboratory results revealed an average serum glucose of 250 mg/dL and an extensive history of a hemoglobin A1c averaging around 10%. The patient was made aware of the diagnosis and informed of the conservative management of her diabetic myonecrosis. The patient’s medication list was reassessed and her oral metformin was discontinued. In addition, the patient’s previous basal insulin dose was increased with the introduction of bolus/prandial injections for better glycemic control.
The patient was seen during her follow-up visit with her primary-care physician approximately 6 weeks after discharge. Patient continues to have mild discomfort in the left pelvic area but reports significant improvement in both pain and ROM.
Discussion
Diabetic myonecrosis is an uncommon complication of long-standing and uncontrolled diabetes mellitus. This case highlights a previously healthy middle-aged woman without any history of diabetes-related complications, diagnosed with iliacus muscle infarction. The odd presentation of muscle infarction can present in either type 1 (more common) or type 2 diabetics and is often seen in patients with established neuropathy, retinopathy, and/or nephropathy.2,3 Muscle infarction has been linked to poor chronic glycemic control with an average hemoglobin A1c level of 9.3% according to a 2015 systemic review. 4
Diabetic myonecrosis usually presents in a middle-aged patient complaining of an acute/sudden onset of pain in the hip, thigh, or calf. 4 Patients may experience localized edema, swelling, and tenderness secondary to internal muscle necrosis from spontaneous infarction. 5 Although acute in onset, patients may not present to a clinician until weeks later, with non-specific vitals and laboratory markers upon admission.2,4 Once clinically suspected, the diagnosis of diabetic myonecrosis is achieved by obtaining an MRI with intravenous (IV) contrast, depicting muscle enlargement, focal edema, and hyperintensity 6 in the setting of a fluid-sensitive sequence. Although not needed, a muscle biopsy can be obtained if results have been non-conclusive and clinical suspicion remains high. The pathophysiology behind spontaneous diabetic myonecrosis is hypothesized to be secondary to the effects of atherosclerosis and underlying diabetic microangiopathy. 7 Thankfully, diabetic muscle infarction has a good short-term prognosis and can be managed with rest, tightened glycemic control, and pain management.4,8 Patients should be monitored for recurrence, but resolution can be expected as early as a few weeks to a few months. 2
When dealing with patients with localized muscle pain, physicians should retain a high clinical suspicion for muscle trauma, localized myositis, thrombophlebitis, an infectious process, or abscess formation. 3 As in this patient, diabetic myonecrosis and non-specific muscle pain or weakness may have signs and symptoms similar to that of inflammatory myositis, including polymyositis (PM), dermatomyositis (DM), and inclusion body myositis (IBM).
Inflammatory myositis is a condition that commonly exhibits proximal muscle weakness, with muscles of the pelvis more severely affected than the shoulders. Age of presentation is variable, and patients complain of a chronic onset with little to no associated pain. Although different in pathophysiology, DM presents similarly to PM but with the addition of skin findings, including but not limited to Gottron’s papules, shawl sign, and a heliotrope rash. In addition, IBM commonly presents with local muscle involvement, often affecting the quadriceps, and is differentiated with findings of intracellular vacuoles and inclusions upon histopathological studies of a muscle biopsy. 9 Diagnosis of inflammatory myositis can be made with elevated laboratory markers such as CK, aldolase, LDH, AST/ALT, anti-Jo1, anti-SRP, and anti-Mi-2.10,11 Diagnosis is often confirmed with a muscle biopsy, and corticosteroids are the first-line therapy. 9
Conclusion
We present a middle-aged diabetic woman with muscle pain, who was diagnosed with a subacute diabetic muscle infarction of the left iliacus muscle. Clinicians should be aware of the causes of localized muscle pain and be able to differentiate an infectious process from an inflammatory process. A patient’s past medical history is of great importance, and we must ensure a history of diabetes is taken into account and integrated into the differential.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Our institution does not require ethical approval for reporting individual cases or case series.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
