Abstract
In October 2015, a 74-year-old Caucasian male patient (past medical history of hyperlipidemia, paroxysmal atrial fibrillation, hypertension, and hypothyroidism) presented to the cardiologist for follow-up outpatient evaluation of exertional chest pain. The patient had recently been seen at the Emergency Department for the same complaint. At that time, the patient’s cardiac markers, EKG, and pharmacological nuclear stress testing were all reported as normal. At presentation to the cardiologist, the patient’s physical examination findings were unremarkable. Over the course of the following year, repeat electrocardiograms and myocardial perfusion imaging studies demonstrated no evidence of ischemia. Despite the persistence of symptoms, the patient was reluctant to undergo invasive testing. The cardiologist ordered a simple blood test: the Age, Sex, and Gene Expression Score, which provides the current likelihood of obstructive coronary artery disease in nondiabetic patients. Based on the high Age, Sex, and Gene Expression Score result, the patient underwent invasive coronary angiography and a 98% stenotic lesion in the proximal left anterior descending artery was discovered. A drug-eluting coronary stent was placed and resulted in the complete resolution of the patient’s symptoms.
Keywords
Introduction
Myocardial perfusion imaging (MPI) has been referred to as the “gatekeeper for invasive coronary angiography (ICA)” with more than 10 million stress tests (exercise and MPI) performed every year. 1 Of those tests, 20% are either false negatives or false positives. One study showed that more than 90% of MPIs are reported as normal. 2 A study of patients referred for angiography showed MPI positively identifies obstructive coronary artery disease (CAD) in fewer than 50% of men. 3 Another study found that 70,000 patients per year experience cardiac death or myocardial infarction after having a normal stress test. 1 It has been documented that patients with a negative MPI are rarely referred on for subsequent studies. 4 When adjusted for referral bias by providing the anatomical validation of disease (ICA or computed tomography angiography (CTA)) for all patients who received an MPI, the sensitivity of MPI drops from 85% to 38%. 5 The addition of a simple office-based lab test, such as the Age, Sex, and Gene Expression Score (ASGES), could minimize verification bias and uncover obstructive CAD in patients preventing costly and deadly cardiovascular events.
The ASGES (Corus CAD, CardioDX) provides a score on a scale of 1–40 that factors the patient’s age, sex, and gene expression levels of 23 genes associated with coronary atherosclerosis. The 23 genes measured in the ASGES are grouped into five terms representing different biological processes, including cellular and innate immunity, inflammation, cellular necrosis, and apoptosis. A detailed explanation of the algorithm calculation and score transformation is available in the PREDICT Validation Study. 6 The COMPASS Validation Study found that the sensitivity of ASGES at a score of ≤15 is 89% and has a 96% negative predictive value (NPV) to aid the clinician in ruling out the presence of obstructive CAD in symptomatic nondiabetic patients. 5 A recently published NIH funded study found that patients with a low ASGES have the same risk of Major Adverse Coronary Event (MACE) over 25 month median follow-up as a negative CTA or MPI. 7 Conversely, as the ASGES increases, so does the likelihood of obstructive CAD presence (defined as 50% or greater stenosis), the severity of the stenosis (70% vs 50% lesions), multiple vessel disease, and the risk of MACE.5–7 A score of 35 is associated with a 71% positive predictive value (PPV) to aid the clinician in ruling in the presence of obstructive CAD. 5 The PRESET clinical utility study had 566 patients who were followed for 1 year after initial assessment. It was found that as the ASGES increases, so does the likelihood of the patient being referred to cardiology for advanced testing. It also found that out of the patients with a low ASGES who were referred on to cardiology that none had significant findings on advanced cardiac testing. Finally, it found in all patients with a low ASGES that there were significantly fewer adverse cardiac events compared to patients with a high ASGES at 1 year follow-up. 8
Case
In October 2015, an established 74-year-old Caucasian male patient presented to the cardiologist for an outpatient evaluation of intermittent exertional chest pain. A recent ED visit included negative cardiac markers and serial electrocardiograms with no evidence of ischemia. Significant past medical history included hyperlipidemia, paroxysmal atrial fibrillation, hypertension, hypothyroidism, and a 35 pack-year history of cigarette smoking. Other past medical, social, and family histories were noncontributory. Relevant medication use included metoprolol for blood pressure control. The patient’s chest pain was characterized as a dull ache, pressure-like sensation. The patient’s physical exam was unremarkable and an EKG demonstrated sinus bradycardia with no signs of acute ischemia. A Regadenoson nuclear stress test demonstrated normal myocardial perfusion without evidence of ischemia. No change in the patient’s management was made at that time. At 2 month follow-up, the patient described that his previous symptoms, which he attributed to dyspepsia, had since resolved.
In May 2016, the patient presented to the Emergency Department complaining of radiating chest pain across his anterior chest wall. He was observed on telemetry overnight; serial EKGs and cardiac troponins were negative for ischemia. He was discharged the following day and subsequently underwent a repeat Regadenoson nuclear stress test (Figure 1) and an echocardiogram; both were reported as normal. At follow-up, in June 2016, the patient continued to complain of intermittent exertional chest pain coupled with a decrease in his endurance. The quality of his chest pain had both typical and atypical anginal features. Due to the recurring nature of the patient’s symptoms, discussion of further diagnostic testing included a recommendation for ICA. The patient was reluctant to proceed with any invasive procedure but agreed to have the ASGES blood test performed to help the cardiologist assess the patient’s current likelihood of obstructive CAD.
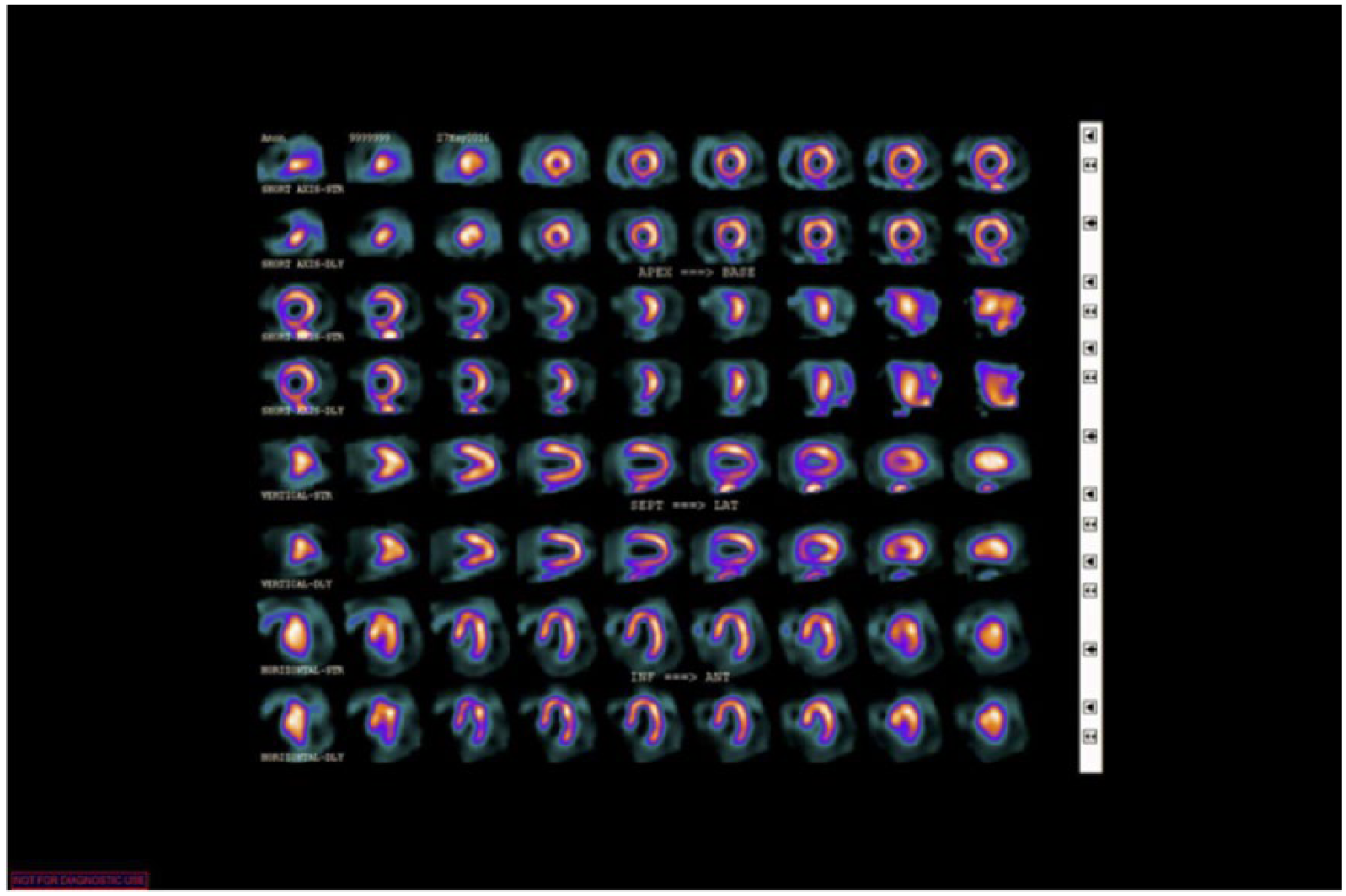
Regadenoson stress testing shows no signs of ischemia or obstructive coronary artery disease.
In July 2016, the patient received an ASGES of 35 which equates to a 53% likelihood of obstructive CAD. 5 Based upon the patient’s recent increase in symptoms and elevated ASGES, the patient agreed to proceed with ICA. The patient was already on chronic beta-blocker therapy (metoprolol), and amlodipine was added as a second anti-hypertensive agent, in order to optimize the empiric medical therapy for angina. A few days later, during catheterization, it was discovered that the patient had a 98% stenosis in the proximal left anterior descending (LAD) artery (Figure 2). The patient underwent placement of a drug-eluting coronary stent in the affected vessel with complete resolution of symptoms.

Invasive coronary angiography (ICA) reveals 98% stenosis of the left anterior descending (LAD) coronary artery.
Discussion/conclusion
In this case, we demonstrated the limitations of MPI for the discovery of obstructive CAD. In this patient, a comprehensive cardiac evaluation, including two MPIs over the preceding year, had been reported as normal. Despite the persistent increase of symptoms, the patient refused advanced testing.
The ASGES was performed on this patient to help the cardiologist assess the presence of underlying obstructive CAD, given his recurrence of chest pain in the setting of advanced age, mixed hyperlipidemia, and Afib. The COMPASS Validation study found, in patients with a negative core-lab read MPI and an elevated ASGES (≥28), that 38% of patients had obstructive CAD present. 5 This patient had a score of 35, which prompted the clinician and patient to seek an invasive angiography procedure that subsequently discovered and treated the severe 98% stenosis in the proximal LAD (Figure 2).
In conclusion, a simple blood-based ASGES can be a useful diagnostic tool for the clinician in the evaluation of a patient with a negative advanced imaging study yet ongoing concern around the presence of obstructive CAD.
Footnotes
Acknowledgements
R.J.P. contributed to (1) conception or design of the work, (2) data collection, (3) data analysis and interpretation, (4) drafting the article, (5) critical revision of the article, and (6) final approval of the version to be published.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: R.J.P. is a member of the Speaker Bureau program at CardioDx, Inc. CardioDX, Inc. is a molecular diagnostics company specializing in cardiovascular genomics and currently provides the Corus CAD (ASGES) blood test.
Ethical approval
Our institution does not require ethical approval for reporting individual cases or case series.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
