Abstract
Thioamides have been used in the management of hyperthyroidism for over 50 years. Liver dysfunction is a rare but important side effect associated with their use. Recently, cases of liver failure associated with propylthiouracil have prompted the Federal Drug Administration to issue a Boxed Warning to the label of propylthiouracil regarding its risk of potentially fatal liver injury and acute liver failure in adults and children. Herein, we present a case to underline the importance of recognising the similar potential for severe hepatic dysfunction with the use of other thioamides.
Keywords
Introduction
Propylthiouracil (PTU) and methimazole (MMI) (or its prodrug, carbimazole) have played a role in the management of hyperthyroidism for over 50 years. Recently, there has been a recommendation by the US Endocrine Society to avoid the use of PTU in the paediatric population, and the Federal Drug Administration (FDA) has issued a Boxed Warning to the label of PTU regarding its risk of potentially fatal severe liver injury and acute liver failure in adults and children. 1 These recommendations follow a re-evaluation of the use of PTU, which identified 47 published reports of severe PTU-related liver failure in adults and children over a 17-year period, including 23 cases that resulted in liver transplantation. 2 During this period, there were no reports of liver transplants attributed to MMI. We report a case of severe cholestatic liver injury in a patient who was treated with carbimazole, to highlight the ongoing importance of recognising a similar potential for severe, potentially fatal hepatic injury in patients treated with other thioamides such as MMI and carbimazole. Furthermore, abnormalities of liver function can be seen in patients with untreated hyperthyroidism, as discussed below.
Clinical record
A 49-year-old man with no relevant past medical history presented with generalised pruritus for investigation. He was found to have mildly abnormal liver function tests (LFTs), with alanine transaminase (ALT) measuring 61 U/L (N: <41) and gamma-glutamyl transpeptidase (γGT) measuring 98 U/L (N: <51). Over the following month, he developed symptoms of thyrotoxicosis and was diagnosed with Graves’ disease. His thyroid function tests (TFTs) were as follows: thyroid stimulating hormone (TSH) <0.01 mU/L (N: 0.4–4.7), free thyroxine (fT4) 42.3 pmol/L (N: 9–25) and free triiodothyronine (fT3) 16.4 pmol/L (N: 2.5–5.5). A thyroid pertechnetate scan showed increased uniform uptake of 10% (N: 2%–7%). Hence, carbimazole 10 mg twice daily was commenced.
The patient’s pruritus worsened over the next 2 weeks, and he developed jaundice. He had received no other new medications or hepatotoxins during this time. LFTs now showed severe cholestasis with bilirubin measuring 581 µmol/L (N: <25), alkaline phosphatase (ALP) 147 U/L (N: <120), ALT 104 U/L (N: <41) and γGT 33 U/L (N: <51). Liver biopsy demonstrated prominent intrahepatic cholestasis with small bile duct canalicular bile plugs. These features were thought to represent a cholestatic drug reaction. Hepatitis serology and work-up for other causes of liver failure were negative. Carbimazole was ceased and the patient was treated with prednisolone 25 mg daily and propranolol 10 mg bd. His liver function quickly began to improve following cessation of carbimazole (Table 1) and normalised within 3 months (Figure 1). As expected, the patient’s thyrotoxicosis worsened following cessation of the carbimazole, and he was instead treated with radioactive iodine, which led to normalisation of his thyroid function within 2 months. Unfortunately, a set of TFTs was not obtained immediately before or after radioiodine administration, and therefore, predicted changes in fT4 are shown by the interrupted line in Figure 1.
Liver function tests during follow-up.
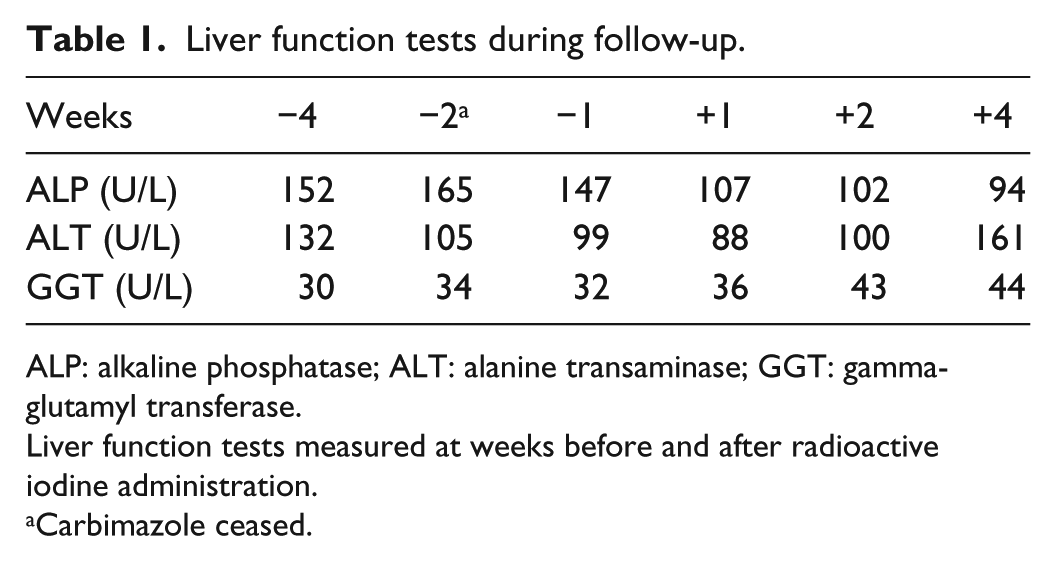
ALP: alkaline phosphatase; ALT: alanine transaminase; GGT: gamma-glutamyl transferase.
Liver function tests measured at weeks before and after radioactive iodine administration.
Carbimazole ceased.
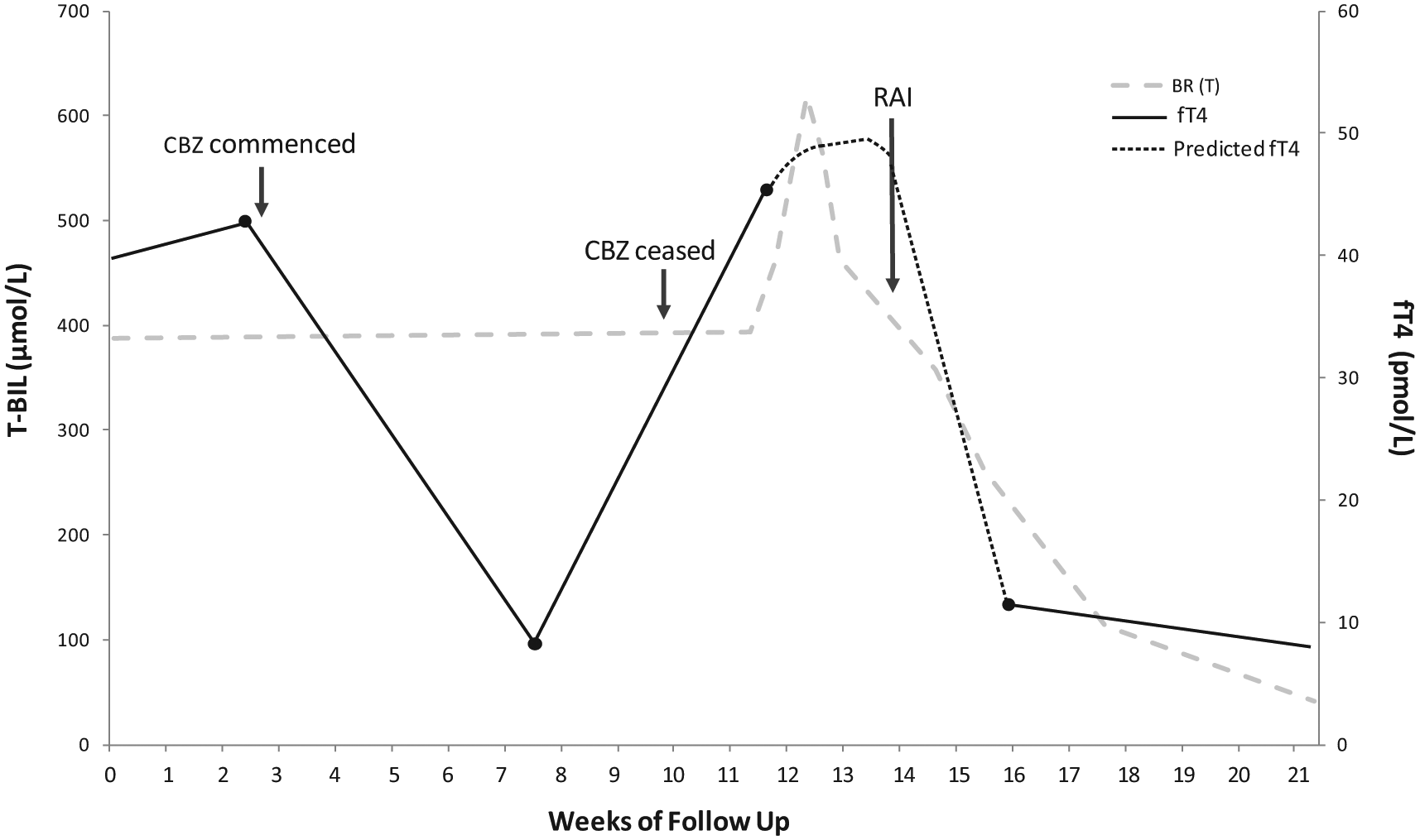
Changes in total bilirubin (T-BIL) and free thyroxine (fT4) in relation to carbimazole (CBZ) use and radioactive iodine (RAI) administration.
The St Vincent’s Hospital–Melbourne Human Research Ethics Committee does not require ethics approval for reporting this case and has waived the requirement for consent to be obtained from the patient in this particular setting.
Discussion
The liver plays an important role in thyroid hormone metabolism, and similarly, thyroid hormones regulate hepatic function and bilirubin metabolism. 3 Not surprisingly, disorders of either organ have the potential to affect function of the other. Therefore, it is important to consider liver function when interpreting TFTs and vice versa.
Hyperthyroidism and hepatic dysfunction
LFT abnormalities associated with hyperthyroidism were first reported in 1874. A hepatitic pattern of LFT derangement is more common than a cholestatic picture; however, abnormalities are usually mild, returning towards normal as the euthyroid state is restored.
Mild elevations in transaminases occur in up to 50% of patients with untreated hyperthyroidism. This is most likely a result of relative hypoxia and consequent hepatic injury due to the inability of the circulation to meet increased oxygen demands from the splanchnic circulation. 4 In mild cases, liver histological changes on biopsy range from mild lobular inflammation to centrizonal necrosis and perivenular fibrosis. 3 While commonly self-limiting, a small number of thyrotoxic patients have been reported to present with fulminant hepatic failure. 5 Elevation of serum ALP occurs in 60%–70% of patients with thyrotoxicosis, but this is predominantly accounted for by a rise in the bone isoform of the enzyme. 6 In contrast, other markers of liver dysfunction such as γGT and bilirubin have only been reported to be elevated in 17% and 5% of hyperthyroid patients, respectively. 7
Antithyroid therapy and hepatic dysfunction
It has long been documented that thioamides can induce liver dysfunction. Typically, MMI and carbimazole are associated with a cholestatic pattern of LFT derangement, and PTU is associated with a hepatitic pattern. 8 The incidence of severe liver failure with PTU is estimated to be approximately 1:10,000 in adults and 1:1000 in children. 2
Observational studies support MMI and its prodrug carbimazole as initial treatment for hyperthyroidism for its better efficacy, higher adherence rate, and more favourable toxicity profile. 8 In the first trimester of pregnancy, however, PTU is preferred, due to the risk of MMI-induced embryopathy. 9 It is now recommended that PTU is reserved only for cases where MMI is not tolerated, or for patients for whom radioactive iodine therapy or surgery is not appropriate treatment. 10
Recently, the use of PTU was re-evaluated by the National Institute of Child Health and Development, the American Thyroid Association and the FDA. These bodies identified 33 published reports of severe PTU-related liver failure in adults and 14 in children, including 16 cases in adults and 7 cases in children that resulted in liver transplantation. 2 In contrast, there were no reports of liver transplants attributed to MMI within the same 17-year period. As a result, the US Endocrine Society now recommends avoidance of PTU in children. 1 Additionally, in April 2010, the FDA issued a black box warning on PTU regarding its risk of potentially fatal severe liver injury and acute liver failure in adults and children. 10
Conclusion
Liver dysfunction associated with thyroid disease and its treatment is a rare but important clinical entity. In the case presented, we describe a man with Graves’ disease who presented with mildly abnormal liver dysfunction in the setting of hyperthyroidism, which within 2 weeks of treatment with carbimazole developed into severe cholestatic liver damage. The nature of the liver dysfunction, as well as the timing after commencement of carbimazole and the evidence of intrahepatic cholestasis, suggested an adverse drug reaction to carbimazole. Improvement of liver function following cessation of the medication also supported a thioamide-induced reaction. Clinicians should be aware of liver dysfunction as an important side effect of both carbimazole and PTU. Currently, routine monitoring of LFTs is not recommended, as liver dysfunction associated with anti-thyroid therapy still remains rare and unpredictable.
Footnotes
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
