Abstract
We review the chronic phase ventilation (CPV) strategy recommended in infants with the most severe bronchopulmonary dysplasia (msBPD, mechanically ventilated at 36–40 weeks post-menstrual age). The safety and efficacy of CPV was important to scrutinize because msBPD is increasingly common, and infants with msBPD are often transferred to the Pediatric Intensive Care Unit (PICU) where the CPV strategy started in neonatal intensive care is expected to continue as the standard of care. First, we describe the CPV strategy, and the supporting evidence given by expert proponents. Second, we subject the supporting evidence to critical scrutiny and explain flaws that weaken support. Third, we give evidence that the strategy is based upon unsound pathophysiology and hence may be harmful. Fourth, we put this all together by making unstated (and unsupported) premises in explaining the benefit of CPV explicit. We made four conclusions. First, the literature suggested that CPV is based upon circular referencing among chapters and narrative reviews written by the same respected experts, and therefore, upon literature inadequately subjected to critical scrutiny. Second, these reviews explained physiologic theory with little supportive evidence that had likely been misinterpreted, and referred to outcome studies that did not examine the effect of the CPV strategy. Third, when implicit assumptions are made explicit, there is evidence to show a lack of consensus about and potential harms of the CPV strategy, and inaccurate interpretations of msBPD physiology. Fourth, there was no rationale that withstands critical scrutiny to suppose ventilated children with msBPD might be an exception to using a standard of care ventilation strategy used in acute lung disease. If the CPV strategy is beneficial, we urgently need better data to that effect; otherwise, it is too early to widely adopt what may be a harmful strategy as standard of care.
Introduction
Bronchopulmonary dysplasia (BPD) is chronic lung disease after premature birth, defined by respiratory support duration, and graded as mild, moderate, or severe.1,2 Although there is debate about the best definition, this review refers to the most severe BPD (msBPD), those infants who are on invasive mechanical ventilation at 36–40 weeks postmenstrual age (PMA). The incidence of BPD is increasing as more extremely low gestational age newborns (<28 weeks gestation) and extremely-low birth weight newborns (<1000 g) survive the neonatal period.1,2 The strongest risk factors for BPD are prematurity and low birth weight, with approximately 88% of infants 22–24 weeks gestational age and >43% of infants born at <29 weeks of age developing BPD, and 95% of infants with BPD having birth weight <1500 g.2,3 In 2023 in Canada, 9% of those born at 23–24 weeks, and 4.5% of those born at <29 weeks, have msBPD, 3 and these infants required hospitalization for 150–300 days.4–8 This has led to “new BPD,” in infants born during the canalicular-saccular stage of lung development, having developmental arrest of alveolar and lung vascular development.1,2 In these vulnerable lungs, supplemental oxygen and ventilator-induced lung injury (VILI) contribute to inflammation and lung injury that culminate in BPD,2,9–11 and ongoing respiratory morbidity. 12
A chronic phase ventilation (CPV) strategy in established msBPD has been endorsed by many experts (Table 1).1,2,13–32 The safety and efficacy of CPV is important to examine because msBPD is becoming increasingly common, and the CPV strategy is affecting larger numbers of infants, and infants with msBPD are often transferred to the Pediatric Intensive Care Unit (PICU) where the CPV strategy started in neonatal intensive care by the referring neonatologists and parents is expected to continue as the standard of care. 54
Book chapters and reviews that claim the chronic ventilation strategy in established severe BPD is suggested.
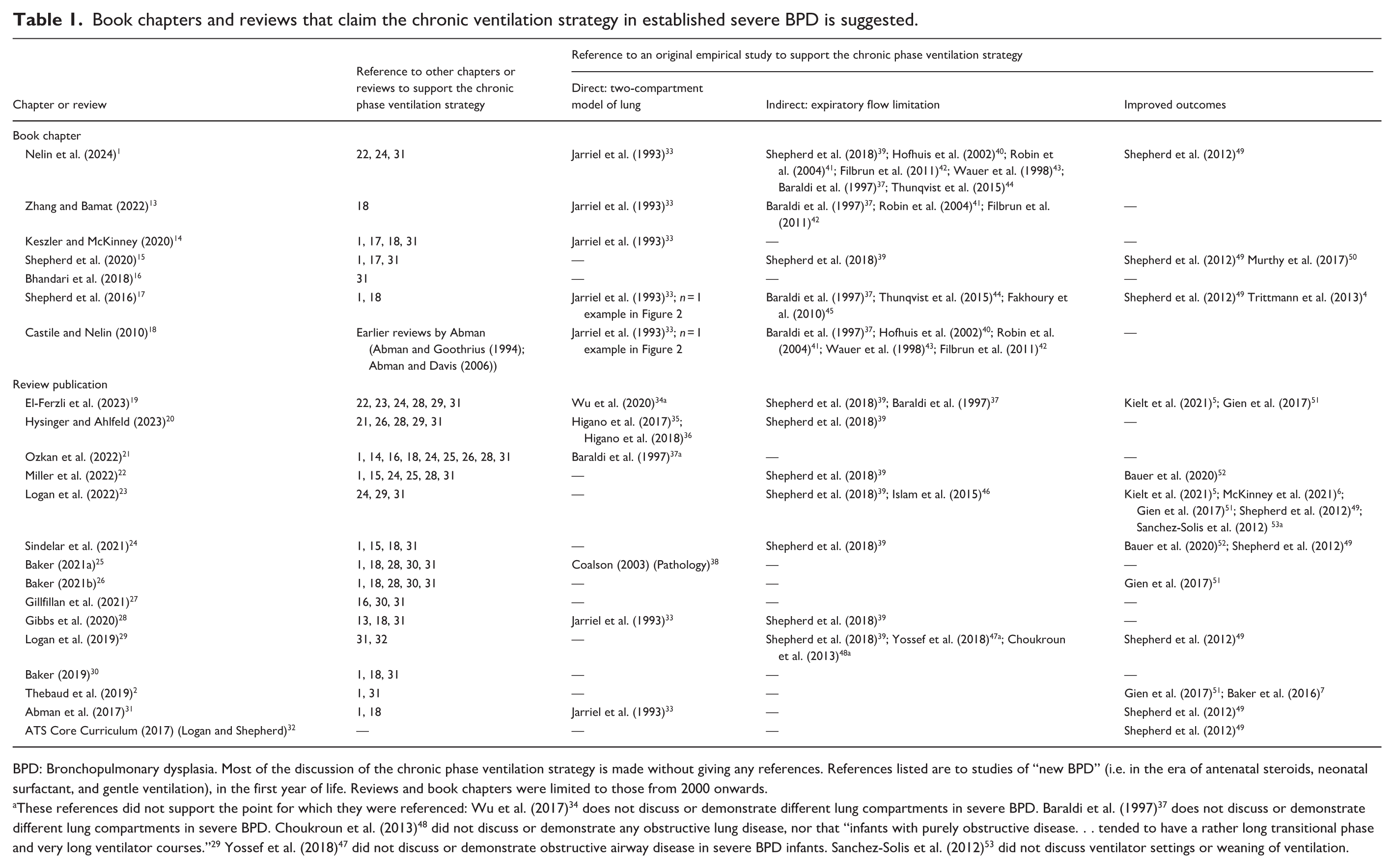
BPD: Bronchopulmonary dysplasia. Most of the discussion of the chronic phase ventilation strategy is made without giving any references. References listed are to studies of “new BPD” (i.e. in the era of antenatal steroids, neonatal surfactant, and gentle ventilation), in the first year of life. Reviews and book chapters were limited to those from 2000 onwards.
These references did not support the point for which they were referenced: Wu et al. (2017) 34 does not discuss or demonstrate different lung compartments in severe BPD. Baraldi et al. (1997) 37 does not discuss or demonstrate different lung compartments in severe BPD. Choukroun et al. (2013) 48 did not discuss or demonstrate any obstructive lung disease, nor that “infants with purely obstructive disease. . . tended to have a rather long transitional phase and very long ventilator courses.” 29 Yossef et al. (2018) 47 did not discuss or demonstrate obstructive airway disease in severe BPD infants. Sanchez-Solis et al. (2012) 53 did not discuss ventilator settings or weaning of ventilation.
This review is organized as follows: First, we describe the CPV strategy, and the supporting evidence given by expert proponents. Second, we subject this supporting evidence to critical scrutiny and explain flaws that weaken this support. Third, we give evidence that the CPV strategy is based upon unsound pathophysiology and may be harmful. Fourth, we put this all together by making unstated (and unsupported) premises in explaining the benefit of CPV explicit. Finally, we draw conclusions and give suggestions for management of ventilation in infants with msBPD.
The new CPV strategy in established msBPD
Literature search
A search of PubMed was done in April 2025 and updated in September 2025. Search terms were “bronchopulmonary dysplasia” AND “ventilation” in title/abstract, limited to review articles in the past 5 years in humans, and written in English. Titles and abstracts were screened to determine eligibility, defined as publications in which ventilation strategies in infants with established severe BPD were discussed. Review of the full publication was done for potentially eligible reviews to verify eligibility. All references in the included papers that were used for discussion of ventilation in infants with established severe BPD were examined, and any other reviews (including book chapters) referenced were screened for inclusion, regardless of year of publication. If a book chapter was updated over the years, only the most recent update of the chapter was used.
What is the CPV strategy?
The CPV strategy for “improving the distribution of ventilation, minimizing physiologic dead space and gas trapping, and improving gas exchange” is based upon the idea that there is “marked regional variability in time constants.” 1 The CPV is said to “reduce chronic retractions and respiratory distress and may decrease recurrent cyanotic spells in some patients,” which is important “in order to interact well with parents, caregivers, and environment or to be able to work on motor skills. . . These developmental treatments should focus on creating an age-appropriate sensory and social environment.” 1 The CPV strategy involves (1) larger tidal volumes (VT, 10–12 ml/kg) and longer inspiratory time (Ti, ⩾0.6 s) to improve regional heterogeneity of ventilation, and (2) slower respiratory rate (RR) with long expiratory time (Te) to allow adequate time for exhalation and avoid gas trapping. 1 This physiologically based CPV strategy was proposed by Castile and Nelin in a book chapter in 2010 based upon “heterogeneous airway obstruction with air trapping” compatible with a two-compartment model of the lung (i.e. a “fast compartment” with short time constants, and a “slow compartment” with long time constants due to high resistance) and expiratory flow limitation in severe BPD, and a “focus on exhalation” to prevent overinflation and resulting worsening ventilation to perfusion (V/Q) matching. 18 Importantly, Castile and Nelin suggested that “as the lung deflates areas of overinflation will decrease, which will bring the lung at end-expiration to a steeper portion of the compliance curve. Thereby, adequate tidal volumes can be generated at lower inspiratory pressure.” 18 There is a variable role for PEEP, to prevent end-expiratory alveolar and small and large airway collapse, and sometimes match auto-PEEP to improve ventilator synchrony and comfort.13–15,20,25,28,31 Spontaneous breaths are supported with moderate pressure support, to “ventilate the fast compartments with relatively faster respiratory rate and lower tidal volume” 14 (see also references 13, 21, 22, 28).
A number of book chapters and narrative reviews have championed this CPV strategy, which has evolved over time to include multiple lung compartments,14,19,21,24,30,31 and require very low RR of 10-20 breaths/minute,13–15,19–26,28,30,31 higher VT of 12-15 ml/kg and even higher,14,27,20–24 resulting in PIP 30-40 cm H2O and even higher.13,19,22,28 The CPV is suggested to foster growth and neurodevelopment due to improved comfort, less sedation, and better tolerance of stimulating interactions.13,14,17,19–26,28,31,32
What is the referenced evidence for the CPV Strategy?
Table 1 describes the evidence base provided in many book chapters and narrative reviews that recommend the CPV strategy.1,2,4–7,13–53 Several points can be made based on this evidence. First, there seems to be much circular referencing among many chapters and narrative reviews when evidence for the CPV strategy is provided (Figure 1). Most statements made in the chapters and narrative reviews do not have a reference provided other than to another chapter or narrative review. Second, the empirical evidence base for the CPV strategy is limited. The main pathophysiological basis of the CPV strategy, the two-compartment lung model, has only one empirical published study. 33 Indirect evidence for high resistance in the lung, describing expiratory forced flow limitation, included reference to several studies.37,39–46 Improved outcomes using the CPV strategy was suggested by reference to several studies.4–7,49–53

Relationship map of cross-referencing among the book chapters and narrative reviews recommending the chronic phase ventilation strategy in most severe bronchopulmonary dysplasia.
Critical scrutiny applied to the supporting evidence provided
We examine the details of the empirical evidence base referenced in the many book chapters and narrative reviews for the CPV strategy (Table 2).4–7,33–57
Original empirical studies that support the chronic phase ventilation strategy in established severe BPD.

BSID: Bayley Scales of Infant Development; CTA: CT angiography of the lungs; FEF: forced expiratory flow; FEV: forced expiratory volume; FIO2: fraction of inspired oxygen concentration; FRC: functional residual capacity; FVC: forced vital capacity; MEF: maximal expiratory flow; PEEP: positive end expiratory pressure; PHTN: pulmonary hypertension; PMA: postmenstrual age; RR: respiratory rate; RV: residual volume; RVRTC: raised-volume rapid thoracic compression; sBPD: severe BPD; SD: standard deviation; SGA: small for gestational age; Ti: inspiratory time; TLC: total lung capacity; TVRTC: tidal volume rapid thoracic compression; UET-MRI: ultra-short echo time magnetic resonance imaging of the lungs; VC: vital capacity; VLBW: very low birth weight; VmaxFRC: maximal expiratory flow at functional residual capacity; VT: tidal volume.
What is the evidence for the two-compartment model of the lung?
The evidence is based on modeling the flow-volume loop of the lung. The only published study to do this included six VLBW infants “that displayed clinical evidence of pulmonary gas trapping including, diminished breath sounds or wheezing, and chest radiographs that showed hyperexpanded lung fields” at 26 (Standard Deviation, SD 16) days of life, using passive flow-volume loops during exhalation to PEEP 2–5 cmH2O. 33 Despite the selection bias and small sample size, the long time-constant compartment was modeled as containing 60% of the exhaled VT. 33 Two other infants are described, one in each of two chapters, with no details of their clinical state; the long time-constant compartment was modeled as containing 62% and 67% of the exhaled VT.17,18 The other referenced studies did not provide data to suggest a two-compartment lung.32–36,38 One MRI lung imaging study found that lungs with higher scores (e.g. hyperexpansion, emphysema, and lung opacities) were associated with respiratory support at 40 weeks PMA; 36 yet, another MRI study found no significant difference in ventilation inhomogeneity (using differences in signal intensities) between mild, moderate, and severe BPD. 55
The relevant empirical data are limited by small sample size, selection bias, and modeling (i.e. it is unclear how to extrapolate from modeling the flow-volume loop to actual ventilation as if the lung has two independent compartments). This is particularly the case given that 60% of the lung is theorized to be in the long time-constant compartment, not a large majority of the lungs.15,17,22,23,31 Despite these limitations, the two-compartment model has been used to produce Figure 2, reproduced in many of the chapters and reviews as the main justification for the CPV strategy.1,2,14,21,31

Theoretical effects of mechanical ventilator strategies for severe bronchopulmonary dysplasia.
Does evidence for forced expiratory flow limitation indicate a high respiratory system resistance lung compartment?
There are many references for flow limitation in severe BPD (sBPD), detailed in Table 2.37,39–46,53 Forced expiratory flow was measured using the tidal volume rapid thoracic compression technique (TVRTC, measuring Vmax at functional residual capacity (FRC), that is, at low lung volume)37,40,44,45 or raised-volume rapid thoracic compression technique (RVRTC), measuring forced flows at various lung volumes.39,41,42,53 There are several limitations of these studies including selection, referral, and/or attrition bias,39,40–44 not being limited to msBPD (or even to sBPD),39–42,44,45 and small sample sizes.37,41,42 Many studies found the flow limitation was mild to moderate,37,41,42,44 most pronounced at low lung volumes,37,40,41,44,45,53 and unrelated to recurrent wheezing, 41 birthweight,37,41,42,45 GA,37,39,41,42,45 days of mechanical ventilation,37,41,42,45 days of oxygen therapy,37,41,42 somatic growth, 42 or resistance of the respiratory system, 37 that is, to the severity of BPD.44,53 There was little change of forced expiratory flow limitation over time,42,45,46 and often worsening over the first year of life,40,44 even when the clinical course was improving. 45 The flow limitation was less apparent when BPD infants were compared to preterm infants without BPD.43,46,53 A more detailed critique of the largest and frequently referenced study, particularly concerning selection bias, unexpected physiologic findings, and difficulties in performing RVRTC in invasively ventilated patients, are given in the Supplemental discussion.39,58–61
The relevance of forced expiratory flow to respiratory system resistance during tidal ventilation in infants with msBPD is unclear. This is particularly the case given the lack of association with many markers of the severity of BPD, the lack of improvement in forced expiratory flow as the respiratory clinical course improved, and minimal difference between BPD and non-BPD ex-preterm infants.37,39,42–46,53 Studies did not rule out intrathoracic tracheomalacia or bronchomalacia, which are common in msBPD, and will result in severe flow limitation on airway collapse during forced expiration. 62 Moreover, application of PEEP in invasively ventilated infants would be expected to prevent not just tracheal or bronchial collapse but also small airway collapse at low lung volumes responsible for the expiratory flow limitation.13–15,20,25,28,31 Finally, it is unclear how long Te is achieved with low set ventilator RR, when the spontaneous RR (usually well above 20 breaths/minute) will interrupt and shorten the predicted Te. Asserting that the pressure-supported spontaneous breaths only ventilate the fast time-constant compartment,13,14,21,22,28 not interrupting the long Te, does not make physiological sense. The relevance of these studies in relation to the theorized two-compartment lungs of msBPD infants is therefore, at best, unclear.
What is the evidence for improved outcomes using the CPV strategy?
There are many references suggesting improved outcomes (Table 2).4–7,49–52 These studies have limitations, including being retrospective,4–6,51,52 historical controls,6,7,49,51,52 single-center,4–7,49,51,52 with attrition6,52 and referral4,5 bias, and all with absent data on ventilator settings,4–7,49–52 and using multidisciplinary complex multipronged interventions.4–7,49,51,52 Some vaguely suggested they might be using the CPV strategy, highlighting the use of “minimal-impact respiratory support. . . necessitating a slow respiratory rate to allow full exhalation,” 49 admission to the referral NICU was “usually accompanied by a change in respiratory support. . . based on the physiology of two-compartment lung disease,” 5 “in general, patients were transitioned. . . to a more chronic ventilator approach,” 51 high PEEP, long Ti, high VT, and low RR “were recommended.” 6 A few of the referenced studies did not attempt to determine whether there were improvements in outcomes by comparing interventions or time-periods.4,5,50
Outcomes that improved with interdisciplinary teams included shorter NICU length of stay6,7 (one study focused on an intervention to improve the discharge process in infants with tracheostomy, and data were not reported separately for BPD patients 7 ); higher weight z-score at discharge, less failure to thrive, and lower likelihood to be discharged on oxygen (without difference in z-score for length at discharge, or in longer-term outcomes of readmissions in first 2 years, diagnosis with asthma in first 5 years, tracheostomy, or PHTN, and with an earlier first ED visit); 6 less neurodevelopmental impairment 52 and improved Bayley-III cognitive and language scores, 49 and improved survival. 51 These improvements must be considered in the context of study limitations, particularly that no study directly examined the effect of using the CPV strategy, and the use of historical controls. For example, the improved survival in one study 51 was similar to the contemporaneous survival reported across 348 NICUs among infants having tracheostomy for BPD, presumably with most not having an interdisciplinary ventilator care program. 56 A more recent example in two BPD commentaries is discussed in the Supplemental Material.54,63,64
Overall, studies did not examine or demonstrate an effect of the CPV strategy on any neurodevelopmental, lung, or survival outcome. The good outcomes among some of these centers are to be commended. Focusing on developmental care with an interdisciplinary BPD team,31,65 to “foster a physical and therapeutic environment that empowers the family to participate in the care,” with “developmental therapies three to five times per week” 49 is good practice. 66 However, this does not demonstrate that the CPV strategy was responsible for improved outcomes.
Is the CPV strategy based on sound pathophysiology: Might the strategy be harmful?
We present evidence to argue that the CPV strategy is based on unsound physiology and may potentially be harmful (Table 3).67–87
Publications suggesting the chronic phase ventilation strategy in established severe BPD is not supported by consensus, and can be harmful to lungs with heterogeneous time constants.
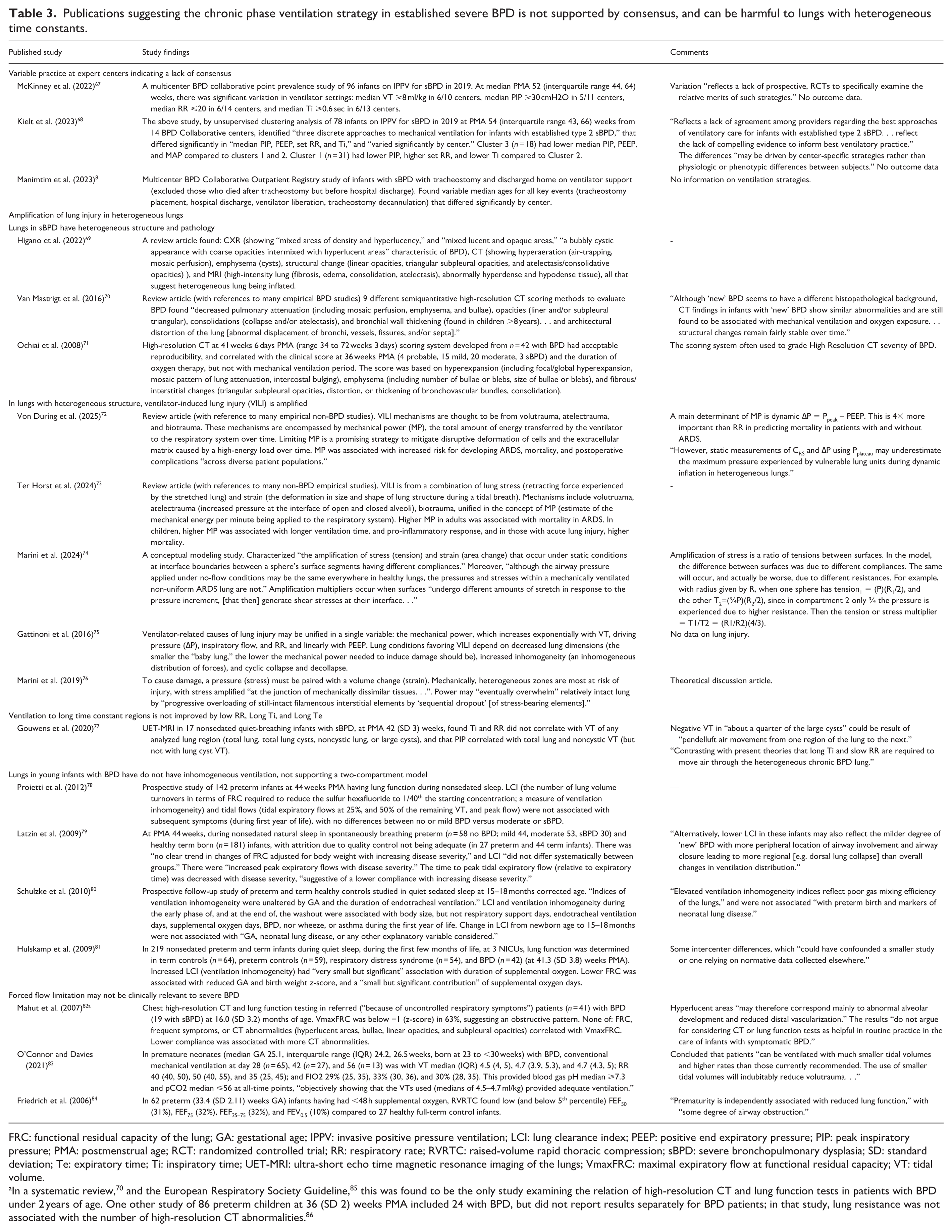
FRC: functional residual capacity of the lung; GA: gestational age; IPPV: invasive positive pressure ventilation; LCI: lung clearance index; PEEP: positive end expiratory pressure; PIP: peak inspiratory pressure; PMA: postmenstrual age; RCT: randomized controlled trial; RR: respiratory rate; RVRTC: raised-volume rapid thoracic compression; sBPD: severe bronchopulmonary dysplasia; SD: standard deviation; Te: expiratory time; Ti: inspiratory time; UET-MRI: ultra-short echo time magnetic resonance imaging of the lungs; VmaxFRC: maximal expiratory flow at functional residual capacity; VT: tidal volume.
In a systematic review, 70 and the European Respiratory Society Guideline, 85 this was found to be the only study examining the relation of high-resolution CT and lung function tests in patients with BPD under 2 years of age. One other study of 86 preterm children at 36 (SD 2) weeks PMA included 24 with BPD, but did not report results separately for BPD patients; in that study, lung resistance was not associated with the number of high-resolution CT abnormalities. 86
Is there consensus on the CPV strategy?
Most of the chapters and narrative reviews1,2,13–15,17–19,22,24–26,28–32 and outcome studies discussed above4–7,49,51,52 were written by members of the BPD Collaborative. However, there does not appear to be a clinical consensus within that Collaborative. There was significant variation in ventilator settings among centers, including in VT, PIP, RR, and Ti. 67 A clustering analysis suggested “three discrete approaches to mechanical ventilation for infants with established type 2 sBPD” differing in “median PIP, PEEP, set RR, and Ti” and that “varied significantly by center.” 68 There were also variable ages for tracheostomy and discharge among centers. 8 The variability “reflects a lack of prospective RCTs to specifically examine the relative merits of such strategies” 67 and “a lack of agreement among providers regarding the best approaches to ventilatory care for infants with established type 2 sBPD.” 68
Many of the chapters and reviews also stated, regarding the CPV strategy, that there are “no objective studies,”1,18 “absence of clinical trial evidence,” 14 “evidence from RCTs is lacking,” 13 “lack of high-level evidence,” 22 “no high-level evidence. . . no RCTs have been done,” 24 “limited existence of robust clinical trials,” 28 “no clinical trials to validate,” 31 “paucity of prospective human studies,” 30 “absence of evidence from prospective RCTs,” 19 and “not subject to any prospective trials.” 24 The implication seemed that the only option (other than CPV) is to await RCTs, which may not be feasible. However, when there are no RCTs, we should critically scrutinize other evidence available including observational studies, detailed pathophysiology, and indirect evidence, as we do below.
Might ventilator-induced lung injury be amplified in msBPD lungs with heterogeneous structure and pathology?
The lungs in msBPD have heterogeneous structure and pathology. The CXR, high-resolution CT scan (HRCT), and MRI of lungs in sBPD show “coarse opacities intermixed with hyperlucent areas”; hyperaeration, emphysema (cysts), and structural change (opacities); and abnormally hyperdense and hypodense tissue. 69 In particular, HRCT scoring methods have found decreased pulmonary attenuation (emphysema and bullae), opacities, consolidations, and architectural distortion, that remain fairly stable over time.60,61 This is compatible with sBPD pathology showing “unevenly expanded airspaces.” 38
Ventilator-induced lung injury (VILI), from volutrauma (the force (stress) causing excessive stretching and distension of airspaces (strain)) and atelectrauma (repeated opening and closing of airspaces causing shear injury), may be unified by the concept of mechanical power (MP), the total amount of energy transferred by the ventilator to the respiratory system over time, deforming cells and extracellular matrix to “eventually overwhelm” lung tissue.72,73,76 The MP has been associated with increased risk for developing ARDS, mortality, and postoperative complications in adults, 72 and with longer ventilation time, pro-inflammatory response, and, in those with acute lung injury, higher mortality in children. 73 A main determinant of MP is ∆Pdyn = Ppeak – PEEP, 72 and MP increases exponentially with VT, ∆P, inspiratory flow, and RR, and linearly with PEEP. 75 Moreover, stress (tension) and strain (area change) are highly amplified at “a sphere’s surface segments having different compliances [and, we calculated, these are even more amplified when due to different resistances].” 74 Surfaces that “undergo different amounts of stretch in response to the pressure increment, generate shear stresses at their interface.” 74
Importantly, static measurements of ∆P “using Pplat may underestimate maximum pressure experienced by vulnerable lung units during dynamic inflation in heterogeneous lungs.” 72 This is because “although the airway pressure applied under no-flow conditions may be the same everywhere in healthy lungs, the pressures and stresses within a mechanically ventilated non-uniform ARDS [and, we hypothesize, msBPD] lung are not,” 74 explaining the importance of using Ppeak in calculation of ∆Pdyn. Pendelluft describes the phenomenon where inhomogeneous inflation or deflation of the lungs can cause dynamic pressure differences between regions and lead to interregional airflows, concentrated in poorly ventilated regions of the lung (i.e. may be more prominent in diseases with significant heterogeneity in both resistance and compliance (such as emphysema)). 87 Pendelluft may lead to underestimation of the alveolar pressure using inspiratory hold to achieve zero flow 18 and may explain negative VT in some cysts in BPD lungs. 77
These considerations suggest that in the structurally heterogeneous lungs of msBPD, VILI will be amplified, be related to VT and ∆Pdyn, and this is the case in both the theorized two-compartment lung (due to high resistance in one compartment) and in lungs with emphysema, cysts, bullae, and opacities (as in msBPD), each with different compliances. Indeed, we speculate whether the two-compartment modeling might be modified to reflect not different resistances, but rather different compliances due to some areas of lung being cystic and emphysematous, with expanded airspaces that fill more rapidly. Many of the chapters and narrative reviews seemed to imply that since lung injury had already occurred in established msBPD, VILI was not (or much less) of concern.1,15,17,19,20,23–26,30–32 However, as stated in the European Respiratory Society guideline on long-term management of BPD, “studies have shown that children with BPD have an impaired lung structure, lower lung function, including declining lung function over time, and increased risk of respiratory symptoms in later life. . . Previous studies observed elevated neutrophils and oxidative stress in airways, measured by induced sputum and exhaled breath condensate respectively, in children aged 11 years or adolescents born preterm compared with children born at term. This suggests that BPD reflects an ongoing respiratory disease after birth with long-term consequences and not just stabilized structural lung damage after the neonatal period.” 85
Is ventilation in msBPD heterogeneously distributed as theorized by the CPV strategy and two-compartment model?
The lungs in msBPD have heterogeneous structure and pathology; however, this does not necessarily mean that ventilation is heterogeneously distributed. To our knowledge, the only study to empirically determine whether long Ti and slow RR improved the distribution of ventilation to the lungs of msBPD patients did not confirm this theory. 77 Using UET-MRI in 17 nonsedated quiet-breathing infants with sBPD at PMA 42 (SD 3) weeks, Ti and RR did not correlate with VT of any analyzed lung region (total lung, total lung cysts, noncystic lung, or large cysts), “contrasting with present theories that long Ti and slow RR are required to move air through the heterogeneous chronic BPD lung.” 77
Using lung clearance index (LCI) as a measure of ventilation inhomogeneity (the number of lung volume turnovers in terms of FRC required to reduce the inhaled sulfur hexafluoride to 1/40th the starting concentration), studies in BPD patients found no differences between mild, moderate, and severe BPD,78,79 no association with subsequent respiratory symptoms in the first year,78,79 no association with respiratory support days, endotracheal ventilation days, supplemental oxygen days, or BPD, 80 or only a ”very small but significant” association with duration of supplemental oxygen. 81
To our knowledge, the only study to examine the relationship between lung function testing and HRCT in BPD found VmaxFRC was below −1 z-score in 63%, suggesting an obstructive pattern; however, none of FRC, frequent symptoms, or HRCT abnormalities (hyperlucent areas, bullae, linear opacities, and subpleural opacities) correlated with VmaxFRC. 82 Studies also suggested that forced expiratory flow is similar in BPD when compared to non-BPD preterm infants,43,46,53 with forced flow limitation in many preterm infants having had <48 h of supplemental oxygen. 84 We conclude that forced flow limitation does not reflect inhomogeneous ventilation, and is compatible with structurally inhomogeneous lungs that are relatively homogeneously inflated.
Has critical care medicine found treatment based on physiology alone to be harmful?
The CPV strategy is based on physiology alone.1,13,14,17–22,24,25,28–32 This has often not served the field of critical care well, with practices based upon what, at one time, seemed to be sound physiology later proven to be ineffective, and even harmful (Supplemental Table S2).72,73,88–98 Sometimes this was based on inaccurate interpretation of physiology, and other times on attempting to achieve physiologic targets alone (Supplemental Table S2, and Supplemental discussion).72,73,88–98
In our opinion, based upon evidence reviewed above, we are concerned that both these errors are being made using the CPV strategy in msBPD. First, inaccurate physiology, based upon likely inaccurate interpretations of the two-compartment model and forced expiratory flow limitation, is being used to predict inhomogeneous distribution of lung inflation and better distribution with low RR and long Ti. How long Te is achieved with low set ventilator RR, when the spontaneous RR will interrupt and shorten the predicted Te, is unclear. Second, attempting ventilation to achieve proposed physiologic targets alone, is a strategy which may have the undesired effect of worsening VILI in the heterogeneous lungs of msBPD.
Putting it all together: making unstated premises explicit
Applying critical scrutiny to the suggested CPV strategy revealed several explicit premises and many implicit premises that were not supported (and sometimes refuted) by evidence (Supplemental Table S1). Based on this, we suggest several empirical questions that should be investigated in msBPD before the CPV strategy can be supported (Table 4).
Empirical questions warranting investigation before the chronic phase ventilation strategy can be supported.

What strategy do we suggest?
In infants and children with severe acute lung disease in the PICU ventilation strategies are aimed at physiology that has withstood critical scrutiny (minimizing volutrauma, atelectrauma, oxygen toxicity, and mechanical power), and have been associated with improved clinical outcomes. 73 This is also the experience in critically ill adults. 72 Our suggestions are based upon recent consensus publications considering systematic reviews of the literature (Supplemental Table S3).9,99–104 Surrogates used to minimize volutrauma include avoiding high VT (VT 4–8 ml/kg), PIP (Pplat ⩽30 cmH20), and driving pressure (∆P ⩽15 cmH20). Surrogates to minimize atelectrauma include PEEP adequate to maintain ventilated lung on the descending limb of the pressure-volume curve (i.e. maintain end-expiratory volume), associated with minimizing the required FIO2 to <60%. Unlike in msBPD, in lungs with more homogeneous obstructive disease, such as asthma, higher PIP is allowed because this pressure is dissipated (before reaching alveoli) in overcoming the high respiratory system resistance, and lower respiratory rate is used because this avoids breath-stacking (i.e. when the next breath occurs before end-expiratory flow is complete) causing auto-PEEP.
In msBPD, applying these practices to avoid VILI should be individualized using clinicians’ best judgment. During acute episodes of deterioration (e.g. ventilator-associated pneumonia or viral bronchiolitis), similar targets are aimed for, while temporarily increasing respiratory support as indicated, just as gradual decreasing of respiratory support is aimed for over time in the chronic setting (which often lasts for many months). The only study we are aware of to describe lung protective ventilation in BPD was O’Connor and Davies that described adequate chronic ventilation in evolving BPD (at days 28, 42, and 56 of life after birth at gestational age 25.1 (interquartile range 24.4, 26.7) weeks) using a strategy to minimize volutrauma (VT at 56 days PMA median 4.7 (IQR 4.3, 5) ml/kg) and not focused on slow RR (at 56 days PMA median 35 (IQR 25, 45) breaths/minute). 83 This does not prove lung-protective ventilation is necessary, but does show it was adequate for chronic ventilation at least in evolving BPD. We use this strategy in msBPD, and, as the infant’s lungs grow, the ventilator RR can be slowly weaned until settings appropriate for ward and then home ventilation are achieved, followed eventually by no need for ventilation.
Limitations
This review has limitations. First, this was not a systematic review of the literature, and important studies could have been missed. Examining all references in the eligible review articles may mitigate this concern. Second, we and authors that recommended CPV acknowledge that there is no evidence that conclusively describes the best practices for ventilation in msBPD. Our suggested strategy was not based on robust studies of ventilated infants with msBPD and was based on physiology and outcomes in children ventilated with acute lung disease. However, we found that the physiology in favor of CPV had, in our assessment, not been subjected to adequate critical scrutiny, and that was the main purpose of our review. Third, it may be that the CPV strategy was intended only for patients with msBPD who, still being ventilated 3–4 months after birth, have failed all other ventilation strategies. The reviews we included seemed to have meant to apply the CPV strategy in virtually all infants with established msBPD, and the few that gave descriptions of msBPD appropriate for the CPV strategy also seem to apply to the vast majority of infants ventilated for msBPD at term PMA (see Supplemental Discussion). Some intensivists may nevertheless consider the CPV strategy more selectively, only for msBPD patients that are not “thriving” or are clearly “failing” conventional lung-protective ventilation. This may be a better approach, but how to define the appropriate patients remains unclear, and evidence to support this more selective CPV strategy we still found lacking. We expand on some of these limitations below, by considering some objections.
Objection 1. There is no standard of care for managing ventilation in msBPD
There are no studies we are aware of that compare benefits and harms between the standard approach to ventilation and the CPV strategy in msBPD. We agree with a workshop that wrote “ventilation and respiratory management strategies for infants with established BPD have weak evidence to support current practices.” 99 In that sense, it could be stated that there is no standard of care for managing ventilation in msBPD, and we cannot make definitive recommendations. However, our concern is that this would imply that what is known about VILI in other ventilated pediatric and adult patients with acute lung disease does not apply to msBPD. The literature on ventilation to avoid VILI is extensive, and we did not independently review the strength of this evidence. However, the reviews referenced for our suggested strategy, which is the standard of care for ventilated children and adults with acute lung disease, were based upon systematic reviews of empirical studies in the literature and structured transparent consensus interpretation of this evidence.9,100–105 One can always choose a specific group of ventilated patients and claim that this evidence does not apply to that subgroup as there are few if any high-quality studies of ventilation in that subgroup. However, a rationale that withstands critical scrutiny for that subgroup to be an exception should be given (e.g. a reason why that group may not be susceptible to VILI). We remain unconvinced that msBPD, being a chronic lung disease, should be such an exception. First, why infants with a chronic lung disease would not be susceptible to VILI is unclear. Second, as discussed above, BPD likely has components of acute lung injury in that it “reflects an ongoing respiratory disease after birth with long-term consequences and not just stabilized structural lung damage after the neonatal period.” 85 Third, also discussed above, the physiology explaining VILI is plausible. This is supported by the finding that mechanical power has been associated with several outcomes in adults and children with acute disease,72,73 and that stress and strain are highly amplified at “a sphere’s surface segments having different compliances [and, we calculated, these are even more amplified when due to different resistances].” 74 Until convincing evidence is available, we suggest that the current standard of care for ventilation in children and adults with acute lung disease can be applied in msBPD to prevent ongoing VILI, and we respectfully disagree with calls for local expert endorsed standardization, consensus guidelines, and protocols for using the CPV strategy in the management of msBPD.106,107
Objection 2. Bias is evident in the critical evaluation of evidence without emphasizing the strengths of the CPV strategy
We relied on the published rationale and evidence provided by many experts for the CPV approach in order to discuss its strengths and weaknesses. We first transparently gave this rationale and evidence (i.e., what could be considered the strengths) for the CPV strategy, and then critically evaluated these. Bias perceived may be because we found this evidence unconvincing, based upon transparently stated evidence throughout the review. Whether our suggested strategy is best can and should be debated; however, the evidence to support CPV we found to be inadequate to recommend what may be harmful ventilator settings.
Objection 3. Ventilation in msBPD may be similar to that for asthma or chronic obstructive lung disease, closer to the CPV strategy
The ventilation strategy for asthma and chronic obstructive lung disease (COPD) is not similar to the CPV strategy. Evidence reviews of ventilation in obstructive lung disease in adults emphasized allowing sufficient time for exhalation to avoid dynamic hyperinflation, with short Ti allowing an inspiratory-to-expiratory ratio of 1:2 to 1:4..100,108,109 These reviews also emphasized that VT should be 6–8 ml/kg and Pplat <28–30 cmH2O to minimize VILI, with minute ventilation minimized further if Pplat is higher (i.e. to lower VT and/or lower respiratory rate).90,108,109 PEEP is set to counterbalance auto-PEEP and therefore reduce the effort needed to trigger the ventilator, and to prevent airway collapse at end-expiration.90,108,109 The same recommendations are made for asthma and bronchiolitis in children.110–113
In addition, msBPD has important differences from asthma and COPD. As discussed in detail above, differences include the degree of heterogeneity of lung structure, and the questionable relevance of evidence for forced expiratory flow limitation in suggesting a high respiratory system resistance lung compartment. We did not find description of Pplat in msBPD, likely because measurement with an inspiratory hold would be accurate only if there is no spontaneous respiratory effort (i.e. during pharmacologic paralysis). However, if there is zero flow on the ventilator flow-time trace at end-inspiration (i.e. just prior to expiration), this would mean that PIP=Pplat, which in our experience has been the case.
Objection 4. That lungs in msBPD have heterogeneous structure and pathology is evidence for a two- or multicompartment model of the lung
This is a misunderstanding of the two-compartment model of the lung. The two-compartment model suggested that there are two independently ventilated compartments of the lung, such that CPV will ventilate the slow compartment and not harm the fast compartment (Figure 2). However, despite the known structural heterogeneity, the lungs in msBPD appear to have relatively homogeneous distribution of ventilation.77–82 The high VT, high PIP (and Pplat), and long Ti (that, despite the low set RR, is associated with a faster spontaneous RR and hence short Te) affect all regions of the heterogeneous lungs, and, we have argued, will cause VILI.
Conclusions
We made four conclusions
We suggest (Table 1, Figure 1, and Figure 2) that CPV is based upon repeated circular referencing among chapters and narrative reviews often written by the same experts. Evidence reviewed above suggested that, in our opinion, the CPV strategy in msBPD is based upon literature that had not been adequately subjected to critical scrutiny, and therefore, CPV has prematurely become considered a suggested strategy.
We suggest that these reviews, in our assessment, explained physiologic theory that has little supportive evidence and that has likely been misinterpreted, and referred to outcome studies that did not examine the effect of the CPV strategy (Table 2).
When implicit assumptions are made explicit (Supplemental Table S1), there is evidence to show a lack of consensus about the CPV strategy, potential harms of the strategy, and what we argued were inaccurate interpretations of msBPD physiology (Table 3).
The standard approach to ventilation in children is based upon best evidence available for acute lung disease, and we have argued that no rationale that withstands our critical scrutiny has been given to suppose the subgroup of ventilated children with msBPD might be an exception to using this strategy (e.g. in our assessment, no rationale for why the msBPD group with chronic lung disease may not be susceptible to VILI).
We do not know the optimal strategy for ventilation in established msBPD. We acknowledge that these patients are difficult to manage using any strategy and that, in this context, teams that are working to make care better should be encouraged. We also acknowledge that CPV proponents may disagree with our critical interpretation of the available literature. We believe that this is how science moves forward, with reasoned debate that leads to work to obtain better evidence. In that spirit, we conclude that, if the CPV strategy is beneficial, we urgently need better evidence to that effect; otherwise, we have argued, it is too early to adopt this potentially harmful strategy as a standard of care. Moving forward, future well-designed observational studies of clinically relevant outcomes using different ventilation strategies in msBPD are a priority need. Then, if supported by this background work, a multicenter RCT enriched for certain subgroups of msBPD may be indicated to settle the issue.
Supplemental Material
sj-docx-1-smo-10.1177_20503121261431454 – Supplemental material for Chronic phase ventilation strategy in established severe bronchopulmonary dysplasia: A critical evaluation of the evidence
Supplemental material, sj-docx-1-smo-10.1177_20503121261431454 for Chronic phase ventilation strategy in established severe bronchopulmonary dysplasia: A critical evaluation of the evidence by Ari R. Joffe and Robert P. Lemke in SAGE Open Medicine
Footnotes
Acknowledgements
We have no acknowledgments to make.
Ethical considerations
Approval was not required for the narrative review as we used only already publicly available published data for review.
Author contributions
Both authors made substantial contributions to conception or design of the work, and interpretation of data for the work; reviewed the work critically for important intellectual content; made final approval of the version to be published; and agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. ARJ drafted the first version of the manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
All data used are given in the publication, and the original reference(s) for that data provided.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
