Abstract
Objectives:
Obstructive sleep apnea syndrome is associated with cardiovascular diseases. Mean platelet volume has emerged as a marker of prothrombotic conditions and cardiovascular risk. The aim of this study was to investigate the association between the mean platelet volume and cardiovascular diseases in patients with obstructive sleep apnea syndrome.
Methods:
The medical records of 207 patients were analyzed. Obstructive sleep apnea syndrome was diagnosed by polygraphy, and patients were classified according to apnea–hypopnea index: control group: individuals with simple snoring (apnea–hypopnea index < 5), mild obstructive sleep apnea syndrome group (5 ⩽ apnea–hypopnea index < 15), moderate obstructive sleep apnea syndrome group (15 ⩽ apnea–hypopnea index < 30), and severe obstructive sleep apnea syndrome group (apnea–hypopnea index ⩾ 30). Mean platelet volume was obtained from medical records. Cardiovascular diseases were defined if patients had hypertension, heart failure, coronary artery disease, or arrythmia. The independent predictors related to cardiovascular diseases in obstructive sleep apnea syndrome were determined by using multiple logistic regression analysis.
Results:
Of the patients, 175 were included in the analysis. Sixty-three (36%) were males and 112 (64%) were females. The mean age was 51.85 ± 11 years. There were, 26 (14.9%), 53 (30.3%), 38 (21.7%), and 58 (33.1%) participants in the simple snoring, mild, moderate, and severe obstructive sleep apnea syndrome groups, respectively. Cardiovascular diseases were significantly different between the four groups (p = 0.014). Mean platelet volume in severe obstructive sleep apnea syndrome group was significantly higher than in mild or moderate obstructive sleep apnea syndrome group and simple snoring group (p < 0.05). Moreover, there was a positive correlation between mean platelet volume levels and apnea–hypopnea index (r = 0.424; p < 0.001). The independent predictors of cardiovascular diseases in obstructive sleep apnea syndrome were age (p < 0.001; odds ratio = 1.134; confidence interval: 1.072–1.2), body mass index (p = 0.012; odds ratio: 1.105; confidence interval: 1.022–1.194), and mean platelet volume (p < 0.001; odds ratio: 2.092; confidence interval: 1.386–3.158).
Conclusion:
The present study demonstrated that there is an association between mean platelet volume levels and cardiovascular diseases in patients with obstructive sleep apnea syndrome.
Introduction
Obstructive sleep apnea syndrome (OSAS) is a public health issue that affects 3%–7% of the general middle-aged population. 1 This syndrome is characterized by repeated upper airway collapse during sleep, which has the consequences of marked reduction or cessation of airflow, oxygen desaturation, and recurrent arousals. The absence of deep sleep leads to daytime sleepiness, cognitive impairment, and lower quality of life.2,3 Regardless of the presence of obesity or other confounding risk factors, OSAS is associated with an increased risk of myocardial infarction, heart failure, hypertension, and global cardiovascular morbidity. 4 It is not uncommon that undiagnosed cardiovascular diseases (CVD) coexist in patients with OSAS. This may have harmful consequences both on the cardiovascular system and the severity of OSAS. The pathophysiology of cardiovascular complications is complex, without being fully elucidated. It involves a sympathetic hyperactivation, chronic inflammation, excessive production of free radicals causing an oxidative stress and tissue inflammation, an endothelial dysfunction, and abnormalities in platelets aggregation and activation. 5 Mean platelet volume (MPV) is a measurement of an average platelet size, usually included in complete blood count testing. It has emerged as a relatively reliable marker of prothrombotic conditions. 6 In fact, increased average platelet size is a sign of increased platelet turnover and activity and is associated with inflammation. 7 It has also been linked with elevated risk of arterial thrombotic events. 8 Therefore, MPV is higher when there is destruction of platelets, as observed in patients with CVD.
The aim of this study was to investigate the association between the MPV and CVD in patients with OSAS.
Methods
In this retrospective study, we analyzed the medical records of 207 patients aged 18 years and older, who were referred to the Department of Otorhinolaryngology—Head and Neck Surgery with complaints of witnessing apnea, excessive daytime sleepiness, and snoring, between January 2019 and December 2021. Patients who had central sleep apnea syndrome, narcolepsy, and upper airway resistant syndrome were excluded. Patients using any drug (including aspirin, dipyridamole, clopidogrel, heparin, aminophylline, verapamil, nonsteroid anti-inflammatory drugs, steroid, furosemide, antibiotics, alcohol intake) that may affect platelet function were also excluded. Patients with cerebrovascular disease or a lung disease with hypoxemia such as chronic obstructive pulmonary diseases, interstitial lung disease, asthma, and patients with infection were also excluded. Demographic characters and medical history including metabolic and CVD were obtained with the use of standardized questionnaire. CVD were defined if patients had hypertension, heart failure, coronary artery disease, or arrythmia. Obesity was defined if body mass index (BMI) was greater than 30 kg/m2. A complete physical examination was performed for each participant. Respiratory polygraphy over a night period of at least 6 h was performed on all participants. It included measurement of blood oxygen saturation by oximetry, nasobuccal airflow, quantification of snoring with recording of tracheal noises, and position analysis. Polygraphy recordings were scored according to the criteria of the American Academy of Sleep Medicine.9–11 Apnea was defined as complete cessation of airflow for at least 10 s. Hypopnea was defined as reduction of more than 30% the airflow signal with an associated fall of at least 3% in oxygen saturation. Apnea–hypopnea index (AHI) was defined as the number of apneas and hypopneas per hour of sleep. Patients with AHI at least five times per hour were diagnosed as having OSAS. As a result of polygraphy, patients were classified into four groups according to AHI: control group: individuals with simple snoring (AHI < 5), mild OSAS group (5 ⩽ AHI < 15), moderate OSAS group (15 ⩽ AHI < 30), and severe OSAS group (AHI ⩾ 30). 12 A total of 32 patients were excluded from the study: 30 with missing data and 2 with lung disease with hypoxemia (chronic obstructive pulmonary diseases)
Blood samples
Data on blood cell counts including MPV were obtained in a retrospective fashion from the medical records. Blood cell counts were analyzed using an automated blood cell counter (Hematology Analyzer Coulter LH 750; Beckman Coulter).
Statistical analysis
Statistical analyses were performed using the Statistical Package for the Social Sciences for Mac (SPSS) version 20.0. Continuous variables were presented as mean (standard deviation) or median and categorical variables were reported as frequencies and group percentages. For comparisons among groups, the chi-square test was used for categorical variables and the unpaired student’s t test or the Mann–Whitney rank sum test for continuous variables after testing for normality which was performed using histograms and the Shapiro–Wilk test. If tests of normality were met, one-way ANOVA was used to compare more than two groups. Bivariate relationships between variables were determined by Pearson’s or Spearman’s rank correlation coefficients (ρ). The independent predictors related to CVD in OSAS were determined by using multiple logistic regression analysis. Univariate variables with p value less than 0.1 were included in the multiple logistic regression analysis for the calculation of odds ratios and 95% confidence intervals. Two-sided p values <0.05 were considered statistically significant.
Results
A total of 175 patients were included in the analysis. The sex ratio was 0.56 (63 males/112 females). The mean age was 51.85 ± 11 years. There were, 26 (14.9%), 53 (30.3%), 38 (21.7%), and 58 (33.1%) participants in the simple snoring, mild, moderate, and severe OSAS groups, respectively. The demographic and clinical characteristics and laboratory findings of the study population stratified by OSA severity are shown in Table 1. There was no statistical difference between the groups in terms of age, sex, and BMI. Desaturation index and lowest oxygen saturation increased with the severity of OSAS and therefore were different in each group as expected (p < 0.0001). CVD were significantly different between the four groups (p = 0.014). However, there were no significant differences among the groups in terms of diabetes mellitus, dyslipidemia, smoking, and BMI (p > 0.05). The current findings indicated that the MPV in severe OSAS group was significantly higher than in mild or moderate OSAS group and simple snoring group (p < 0.05). But there was no significant difference between mild and moderate OSAS patients (p = 0.085).
Characteristics of the study population.

BMI: body mass index; AHI: apnea hypopnea index; CVD: cardiovascular disease; MPV: mean platelet volume; OSAS: obstructive sleep apnea syndrome; WBC: white blood cell.
CVD is significantly more prevalent in moderate OSAS group than in simple snoring group (p < 0.05).
CVD is significantly more prevalent in severe OSAS group than in mild OSAS and simple snoring groups (p < 0.05).
The variable in severe OSAS group is significantly different from the other groups (p < 0.05).
The variable in moderate OSAS group is significantly different from mild OSAS and simple snoring groups (p < 0.05).
By Spearman’s correlation, there was a positive correlation between MPV levels and AHI (r = 0.424; p < 0.001). MPV was significantly higher in patients with CVD than whom without CVD among all groups except for mild OSAS (p < 0.05) (Figure 1). Furthermore, all factors that can determinate CVD were evaluated by univariate analysis (Table 2). Parameters associated with CVD were therefore introduced in a multiple regression analysis that included MPV, age, hyperlipidemia, diabetes mellitus, severe OSA, and BMI. The independent predictors of CVD in OSAS were age (p < 0.001; OR = 1.134; CI: 1.072–1.2), BMI (p = 0.012; OR: 1.105; CI: 1.022–1.194), and MPV (p < 0.001; OR: 2.092; CI: 1.386–3.158).
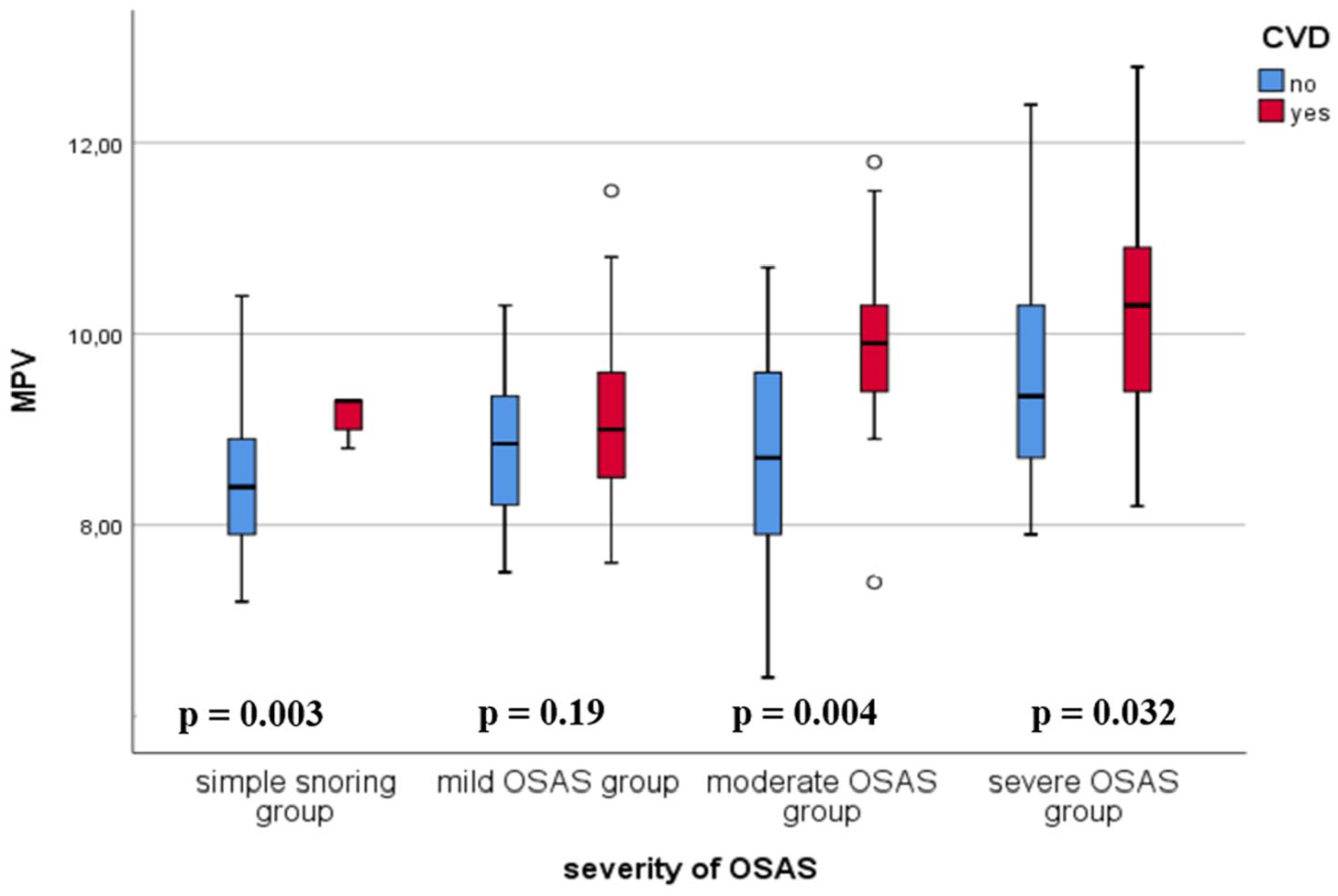
MPV in subjects with and without CVD in mild, moderate, and severe OSAS patients and simple snoring group.
Comparison between no-CVD group and CVD group.

BMI: body mass index; CVD: cardiovascular disease; MPV: mean platelet volume; OSAS: obstructive sleep apnea syndrome.
Discussion
There were two main findings of the present study. First, MPV, which is a marker of thrombopoiesis and platelet function, 7 was increased in severe OSAS patients when compared with mild, moderate, and simple snoring patients. Second, MPV was positively correlated with AHI, oxygen desaturation index, and minimum oxygen saturation values (p < 0.05). OSAS is not a simple respiratory failure that occurs during sleep. It generates a systemic inflammatory response that might lead to CVD.13,14 This not yet clear relation might be due to an increased platelets activation and aggregation in patients with both CVD and OSAS. The activation of platelets trigging the cardiovascular alterations and its relationship with AHI in OSAS may be explained by several mechanisms: Intermittent night hypoxia and sleep deprivation lead to an activation of sympathetic system characterized by epinephrine and norepinephrine increase.15,16 Epinephrine and norepinephrine cause dose-dependent activation of platelets. 17
Moreover, inflammation in OSAS releases interleukins IL-6 which stimulate megakaryocytes and therefore the production of platelets with larger granules resulting, in increased MPV. 18 Large platelets are generally indicative of increased platelet activation, they contain more granules and thromboxane A2 and express more glycoprotein Ib and IIb/IIIa receptors; therefore, larger platelets aggregate more rapidly and strongly to collagen, possibly increasing thromboembolic events.19,20 MPV is a simple parameter demonstrating the increased platelet activation. MPV increases in some diseases whose physiopathology includes inflammation, hypoxia, and vascular endothelial damage, such as CVD.17,21 Several studies have focused on highlighting the markers of cardiovascular risk associated with OSAS, in particular through the thickness of the intima media measured via an ultrasound of the vessels of the neck.22–24 MPV is measured by automated hematology analyzers. 8 Using it as a marker to assess the cardiovascular risk in OSAS is quite simple and a cheap method when compared to other methods. Previous limited studies focused on the interest of this marker in patients with both CVD and OSAS. An association of higher MPV with CVD in patients with OSAS and a significant positive correlation have been shown in a study involving 205 patients with exploration breathing disorders during sleep. It has also shown that the prevalence of high blood pressure or coronary artery disease was significantly higher than at control subjects or patients whose AHI was less than 15/h. 25 Also, inflammation stimulates oxidative stress, which may stimulate platelets, and increased cardiovascular mortality in patients with OSAS. 26
Another study including 699 subjects in whom a polysomnographic recording had been carried out as part of the suspicion of breathing disorders during sleep, confirmed the presence of a significant correlation between the value of MPV and AHI, highlighting the simultaneous correlation between MPV and night desaturation indices (SaO2 minimum and average, time spent below 90% of SaO2) which represent the main factors in the development of endothelial dysfunction. 27
In similar studies, Varol et al.28,29 investigated effects of hypertension, smoking, and severity of OSAS on the MPV level and showed that MPV is higher in hypertensive patients and increases as the severity of OSAS increases.
In our study, we showed, on one hand, that MPV is associated with CVD in patients with OSAS, and on the other hand, that CVD and MPV increase as the disease becomes more severe. These results are all the more important in the multivariate analysis because no association was not found between the MPV and hyperlipidemia, diabetes mellitus, sex, or smoking that might represent confounding factors. This study reinforces the importance of MPV as a quick, easy, and complementary tool assessing cardiovascular comorbidity associated with OSAS before applying other expensive and invasive procedures. Its mechanism of action as well as its influence in diagnosis and therapy are still not well known and will justify its use in larger and long-term studies. OSAS is related with many consequences such as daytime sleepiness, impairment of cognitive function, depression, altered quality of life, elevated risk for crashes while driving, and increased cardiovascular morbidity and mortality.30,31 For those reasons, predicting CVD and so that, high-risk patients may stimulate us to give priority to them in sleep laboratories, which could decrease morbidity and mortality by implementing the best treatment at the best moment.
This study had some limitations. First, the study population was relatively small, and the calculation of the sample size selected for this study was not done. Second, we did not evaluate the effect of continuous positive airway pressur (CPAP) treatment on MPV. Third, it was a retrospective study and therefore not the best way to investigate a causal relationship, but the design of our study was not to elucidate the mechanisms that lead to an association between MPV and CVD in patients with OSAS.
Conclusion
The present study demonstrated that there is an association between MPV levels and CVD in patients with OSAS. Consequently, MPV can be considered as a marker to predict CVD in OSAS.
Footnotes
Author contributions
Study conception and design: SK and KK. Data collection: SK and KK. Analysis and interpretation of results: SK, KK, and IC. Article preparation: SK, KK, IK, MAC, SA, and IC. Reviewing and editing the article: SK, KK, IK, MAC, SA, and IC.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
