Abstract
Objective:
Optimal oral health is an essential component of good quality of life. However, this may be hindered by dental anxiety (DA), thereby, affecting the utilization of dental services. DA could be alleviated by pre-treatment information; however, the method of delivering this information is yet to be explored. It is, therefore, necessary to assess the modes of presenting pre-treatment information to ascertain the one with significant effect on DA. This will improve treatment outcomes and quality of life for individuals. Hence, the primary objective is to assess the effect of audiovisual and written forms of pre-treatment information on DA, while the secondary objective will compare the subjective and objective methods of assessing DA with psychometric anxiety scale (Index of Dental Anxiety and Fear (IDAF)-4C+) and salivary alpha-amylase respectively.
Study design:
Single-centred, single-blind, parallel-group, four-arm randomized clinical trial.
Methods:
The study will compare the effects of audiovisual and written forms of pre-treatment information on DA among adults. Patients 18 years and above scheduled for dental treatment will be screened for eligibility. Written informed consent will be sought before participation. Participants will be allocated randomly using block randomization, to the groups; G1: audiovisual and G2: a written form of pre-treatment information. At the visit, participants will complete the DA questionnaires (IDAF-4C+, Modified Dental Anxiety Scale and Visual Analogue Scale). Physiological anxiety-related changes of salivary alpha-amylase will be measured using a point-of-care kit (iPro oral fluid collector) at baseline, and 10 min after intervention. Furthermore, blood pressure will be taken at baseline and 20 min into the treatment. The mean changes of physiologic anxiety levels and 95% confidence intervals will be compared between the methods of pre-treatment information.
Discussion:
This study hopes to establish pre-treatment information as a method for reducing DA among the populace. Also, to assess the relationship between questionnaire-based and physiologic methods of assessing DA.
Keywords
Introduction
Dental anxiety (DA) is multidimensional and is associated with many factors that are yet to be fully explored. Dental fear and DA are often used interchangeably in literature as they are closely related. However, dental fear is an actual response to a known danger or threatening object or situation, while DA is an emotional reaction and an unpleasant feeling to an unknown feared situation or object. Nevertheless, both involve physiological, behavioural, cognitive, and emotional components.1–4 Hence, in this study both terms will also be used interchangeably.
The fear displayed towards dental treatment is majorly of two factors5–7: the exogenous; this refers to external conditions such as sight, sound and information from relatives, which may predispose an individual to anxiety. The endogenous type, on the other hand, is related to internal/inherent effect of anxiety on a person as informed by past negative experiences from the provider of a treatment and the treatment complexity. 6
Fear for dental treatment can also be affected or compounded by some situations that can impact on the psychological well-being of the patient such as the COVID-19 pandemic. Studies8,9 have shown that the pandemic added to the anxiety and fear of visiting the dental clinic, due to the fear of meeting people and increasing their risk of contracting the disease. However, this study plans to focus on a normal situation where there is no external nor additional situation such as the pandemic.
In many parts of the world, including Nigeria, many people still report anxiety as a barrier to dental treatments.10–16 Moreover, inadequate knowledge of dental procedures such as endodontics/root canal treatment (RCT), invariably affects the perception of individuals and the outcome of the treatment.17,18
Endodontic treatment (RCT), conservative management of a complication arising from delay in accessing oral health care probably due to fear, is a fear factor by itself.12,19–22 Dental fear is known to take its root from DA. There is a dearth of literature, in Ibadan and Nigeria as a whole on the management of DA with endodontic treatment. Though RCT is a common dental treatment in Nigeria, the fear of the treatment is a special problem that has not been well explored. The anxiety towards the treatment is of public health concern and needs to be investigated, in order to increase the acceptance of the procedure and improve the treatment outcome.
Management of DA begins with an understanding and identification of dentally anxious individuals, finding a reliable and sensitive instrument and above all, fashioning out an appropriate treatment plan. The latter factors are mandatory for a positive treatment outcome, as they would ease the process of dental treatment for the dental team as a whole. Regrettably, DA is not routinely assessed in Nigeria, 23 thus creating a challenge of identification and planning for such individuals.
DA has been assessed using different instruments of which, the Corah Dental Anxiety Scale (DAS) and its modification (Modified Dental Anxiety Scale (MDAS)) are the most commonly employed.24–26 Though, the scales have some shortcomings. The DAS and MDAS scales do not measure the multicomponent of anxiety 27 while the questions in DAS measure different things, and the scale combines both uni- and bi-directional scales in the measurement of anxiety. Also, the questions are more of qualitative measures than quantitative but are scored quantitatively.1,2,28 Nevertheless, they can differentiate between mild, moderate levels of anxiety, and phobia (an extreme form of anxiety). However, it is necessary to have a sensitive scale or scales that can differentiate more between mild and moderate anxiety. Furthermore, use of instruments that can combine all the aspects (emotional, cognitive and behavioural) of DA in one scale like the Index of DA and Fear (IDAF-4C/IDAF-4C+)29–31 should be encouraged. Its applicability for regular /routine screening and monitoring of DA should be explored.
The aforementioned instruments are subjective assessments of DA and should be complemented with objective measures. The use of physiological responses to anxiety provides an objective measurement of anxiety which is independent of a person’s subjectivity.32,33 Physiological responses to anxiety include heart rate, skin reaction32,33 and use of salivary stress biomarkers; salivary alpha amylase and cortisol.34–38 Saliva as a means of diagnosing and monitoring systemic diseases, has the advantages of ease of collection, being non-invasive, elimination of fear of prick, low cost of collection, reduction of risk of spreading infection, requires little or no skill for the collection, and acceptable by both clinicians and patients.39,40 The recent use of diagnostic biomarkers has also increased the use of saliva for various analyses in assessment of diseases and monitoring of treatment outcomes. Hence, recent findings have shown salivary stress biomarkers as a good objective means of assessing DA.
Though, information about its use as a point of care mode of assessment41–43 for DA is sparse. The availability of a valid and objective point of care41–43 assessment of DA before treatment could facilitate appropriate treatment planning towards alleviating anxiety and preparing the patient psychologically for the treatment.
In terms of intervention, effective communication between the dentist and the patient about the procedure or the disease condition has been found to reduce anxiety among individuals.20,44 Thus, leading to an improvement of patient’s attitude and confidence in the dentist. 45 The mode of passing across the information may either aggravate or alleviate the anxiety in any individual. Audiovisual information was found to contribute to reduction of anxiety and increased uptake of endodontic procedure. 46 The audiovisual information used in that particular instance were videos chosen by the patients for relaxing the mind and not explanatory videos on the procedure. Altogether, there is little or no research on the use of information communication to reduce DA among adults in our environment and in Nigeria as a whole.
To the best of the authors’ knowledge, this study will be the first to consider pre-treatment information as a tool to assess and compare the effect of audiovisual and written information on patients’ anxiety levels. The outcome of this study should provide insight into the appropriate methods of identifying and controlling DA among patients.
In addition, the findings of this study could serve as a basis for the commencement and incorporation of the form of the information method that is found effective in reducing anxiety, in the regular protocol of treatment at the study centre. Furthermore, it is believed that the method of information communication will help patients to understand the details regarding the various treatments they are about to undergo. The study may also encourage the uptake of preventive measures for oral health and reduce symptom-driven clinic visits due to DA.
Aim: To compare the effects of audiovisual and written forms of pre-treatment information on DA among adult patients seeking dental care.
Objectives
Primary objective: To assess whether audiovisual form of pre-treatment information communication will be effective at reducing DA as against the written form of pre-treatment information.
Secondary objective: To assess whether the level of salivary alpha-amylase will increase due to DA in anticipation of dental treatment. The study will further assess the correlation between subjective psychometric scale (IDAF-4C+) and the objective saliva alpha-amylase in assessment of DA.
Research hypothesis
The primary hypothesis to be tested is:
Null: There is no difference between the audiovisual and written form of pre-treatment information on DA.
Alternate: There is a difference between the audiovisual and written form of pre-treatment information on DA.
Methods
This part includes World Health Organization trial registration data set as reported in Table 1.
World Health Organization trial registration data set for study protocol.

DA: dental anxiety; N/A: Not Applicable.
Trial registration no: PACTR202108664752830. https://pactr.samrc.ac.za
Also, the SPIRIT reporting guideline 47 was used in the development of the trial protocol.
Study design: This will be a randomized control trial. It is designed as a single-centred, single-blind, parallel-group, four arm trial.
Participants
The sample will consist of a minimum of 192 adult patients scheduled for dental treatment. These participants will be recruited from the Dental Centre, University College Hospital, Ibadan.
Study setting: Participants shall be eligible patients attending the Dental Clinic, an outpatient academic facility of the University College Hospital Ibadan, Nigeria. A tertiary, academic hospital responsible for both undergraduate and post graduate training in various specialities of Dentistry.
Selection criteria
Inclusion criteria
Individuals aged 18 years and above, individuals who have had record of dental pain and being planned for endodontic treatment
Patients for routine scaling and polishing
Patients planned for endodontic treatment
Patients with no history of psychological imbalance.
Patients with the American Society of Anesthesiologists (ASA) I and II where:
ASA I: A normal healthy patient, non-smoking with minimal or no alcohol use
ASA II: A patient with mild systemic disease, mild diseases only without substantive functional limitations
Exclusion criteria
Patients with ASA III and IV
ASA III: A patient with severe systemic disease
ASA IV: A patient with severe systemic disease that is a constant threat to life
Patients on medication that causes increase in alpha amylase including aspirin, diuretics, oral contraceptives, corticosteroids, indomethacin, ethyl alcohol.
Patients on adrenergic agonist and antagonists, for example, anti-hypertensive, anti-asthmatics.
Chronic alcoholics
Habitual smokers
Patients with psychiatric disease
Patients who have had the prescribed treatment done previously
Sample size calculation
To estimate the sample size of participants to be included in the randomized control trial, comparing the effects of audiovisual (multimedia) and written form of delivery of information on reduction of DA among adult patients planned for dental treatment. A sample size formula for comparative study with quantitative outcome was used. The means and standard deviations from a previous study 46 were used at power of 90, and two-sided significance level of 0.05. This yielded 192 participants to possibly give a difference between the two main study groups.
The calculation
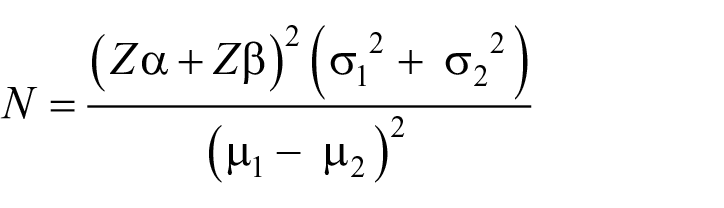
Where:
N = sample size
Zα = Z value for α level at 5% = 1.96
Zβ = Z value for β level at 90% = 1.28
Using the referenced article on effect of audiovisual information on anxiety 46
σ1 = standard deviation for control = 2.01
σ2 = standard deviation for test = 1.78
µ1–µ2 = Difference in mean for pre-treatment information
µ1 = mean for multimedia information (audiovisual) = 0.7
µ2 = mean for control = 1.64
By substitution:

N = 85.9
With adjusted sample size for 10% non-response (N = n/1−r)
N = 85.9/1−0.1 = 95.5 ≈ 96 participants in each group.
Study protocol
Study design: Figure 1 is the study flow diagram
Enrolment: Adult dental care seekers/patients that present to the dental centre and have been booked to have scaling and polishing shall constitute group A and those booked for RCT within the study period, shall be recruited into group B to participate. The target participants shall be those being exposed or booked for the aforementioned procedures for the first time and should not have been exposed to attending doctors before the treatment.
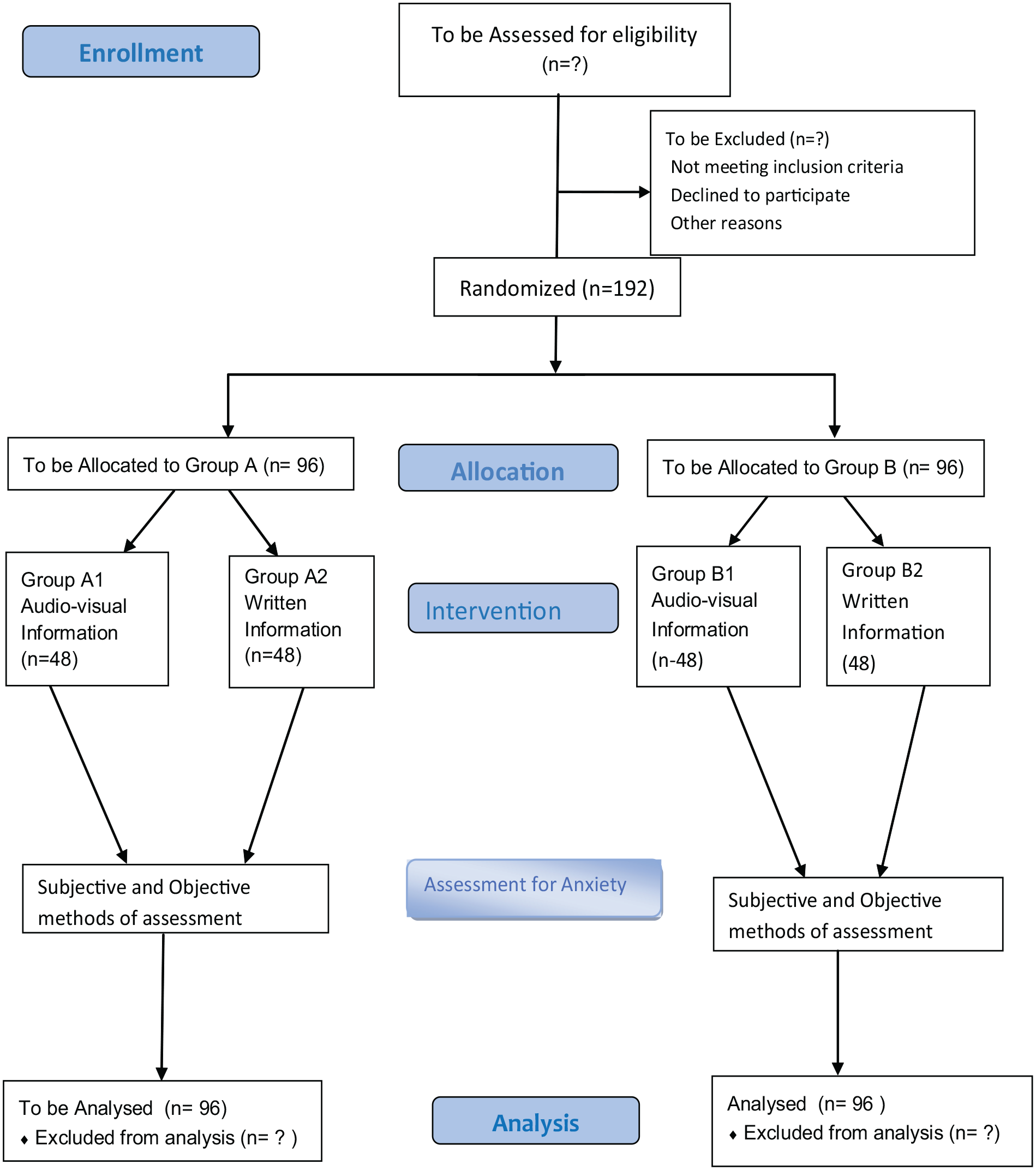
Study flow diagram.
The participants will be enrolled from the two clinics involved in the treatments mentioned above. The enrolment of participants shall be till the minimum calculated sample size is achieved. Study time will be for at least 5 months to accommodate and achieve this.
Consent: The primary investigator will obtain written informed consent from the participants after verifying their eligibility (consent form available as Supplemental Appendix I).
Pre-intervention
After written informed consent is taken by the primary investigator, baseline assessment will be done while the participants are seated on the dental chair. Participants included in the study shall have all the data on socio-economic variable such as age, gender, educational level, marital status and occupation collected with the data collection form/questionnaire. Thereafter, the patient will be required to rinse the mouth with water before taking the pre-intervention saliva sample. Saliva shall be collected for pre-treatment analysis of salivary alpha-amylase level at chair side, and the participants will also fill out the questionnaire (IDAF-4C+) (Supplemental Appendix II). Furthermore, the participants will be required to fill the MDAS (Supplemental Appendix III) and will be correlated with the IDAF-4C+. The blood pressure of the participant shall also be taken at baseline.
Randomization
Each group will be sub-divided into two sub-groups (A1, A2, B1, B2) based on the mode of pre-treatment information.
Intervention: The pre-treatment information (audiovisual) (A1, B1)
Control: Written information (A2, B2)
Group A will be those for non-anxiety provoking/less complex procedure such as routine scaling and polishing which do not require anaesthesia and manipulation or perforation of the gingiva, while the group B will be participants who are being planned for anxiety provoking procedures which require anaesthesia such as endodontics/RCT, ranked as intermediate/complex with treatment complexity score of 2 and 3 respectively, on the indicator scale for sedation need. 48
Block randomization using 1:1 allocation will be performed, to divide the main dental treatment group into sub-groups (A1, A2, B1, B2) based on the mode of pre-treatment information. The pre-treatment information test groups (A1, B1) will be given audiovisual information on the procedure they are about to undergo while the control group (A2, B2) will be given a written information note. The allocation will be concealed by using sealed, sequentially numbered, identical envelopes that will contain group assignments. After the envelope has been assigned to each participant (without replacement into the box), it will be opened by the dental assistant and the allocation will be unknown to the attending dentist.
Blinding: Postgraduate doctors who have been calibrated but blinded to the type of information received by participant will carry out the treatment procedure.
Calibration of dentists
They shall be postgraduate resident dentists in the clinic. Four dentists each in the two clinics shall be included as treating dentists. Training and briefing on the research protocol will be done for the attending dentists in the study clinics. The step-by-step requirements for the research and selection criteria of the participants shall be discussed before the start of the research.
Intervention
The participants in the intervention group will be shown a self-developed video of the procedure, while the control group will be given a written but similar information on the procedure. This will be carried out by the calibrated researcher assisted by a research assistant. The calibrated research assistants will ensure adherence of participants to the study protocol.
Post-intervention data collection
Ten minutes after the intervention, the participants shall have saliva sample taken again. 49 Also, the participants shall be asked to fill the modified psychometric questionnaire IDAF-4C+, while the Visual Analogue Scale (a 10cm line drawn) 50 will be used to assess overall anxiety as perceived by the participants (after intervention).
For each procedure, postgraduate doctors who have been calibrated but blinded to the type of information received by participant will carry out the procedure. The intra-operative assessment will be done 20 min into each procedure (taking the blood pressure). Overall anxiety as perceived by patient will be done after the treatment is completed, and this will complete the participation in the study.
Data collection form, set on software for data collection will be used to obtain participants’ data, readings from the tests and the questionnaire. ArcGIS Survey 123.version 3.16 data software (University of Ibadan) will be employed. All information collected will be transferred to a pass-worded computer for analysis.
Protocol for saliva collection
To prevent the diurnal effect on saliva, saliva shall be collected from the participants between 9 am and 11 am (data collection period minimum of 3 h after waking up and after eating). The participants will be asked to rinse their mouth with water and spit out. A swab will be used to collect 0.5ml of the saliva from the floor of the mouth or the tongue, till a colour change (according to the manufacturer) indicates sufficient amount of saliva is taken.
The swab will be placed in the Oral Fluid Collector (OFC) (Soma Bioscience, Oxfordshire, UK).50,51 Three drops of Saliva/buffer mix will be placed with the OFC dropper cap, on the Lateral Flow Device (LFD) (Soma Bioscience, Oxfordshire, UK) and the test is allowed to run (10 min).
The alpha amylase LFD is then scanned to give the quantitative result by reading with the LFD reader/Soma cube reader (iPro Cube, Soma Bioscience, Oxfordshire, UK). The reading shall be compared with the normal to check for increase or decrease.
The protocol for saliva described above will be done at chairside as a point of care collection for saliva analysis as prescribed by the manufacturer of the point of care saliva analysis kit.51,52
Protocol for development of pre-treatment information
Audiovisual information
The audiovisual information shall be self-developed by the researcher. It will comprise an audiovisual recording of the procedures with a clear explanation/illustration. The video will be pretested to check content validity, among other subjects that are not part of the study, but similar in biodata. This shall be loaded on an iPad for the participants to view while waiting for the treatment on the dental chair. The video will include all the stages involved in each procedure (anaesthesia, rubber dam placement during RCT etc.). The video will also be translated into the local language (Yoruba) and be made available for participants who do not understand the English language.
Written information document
Similar information on the procedures will be written by a specialist in the field and pretested for clarity and content validity among other subjects that are not part of the study, but similar in biodata. This will also be translated into the local (Yoruba) language. Validation of translation and back translation of the document will be done among patients who will not be participants in the main study. 53 This document shall be presented in hard copy form for the participants to read while waiting for the treatment on the dental chair.
Participation in this research will entirely be voluntary and of no harm to the participants. Any participants/patients who wish not to participate or wish to decline the prescribed treatment shall be free to decline to be part of the study.
Data management
Confidentiality of data
The data obtained from the questionnaire will be accessible to the investigators only. All information shall be given code numbers and no name will be recorded. All data will be transferred to a password protected personal computer. All published articles arising from this research will have no information that would reveal the identity of any participant.
Monitoring: The data collection, participant enrolment, eligibility, consent, allocation to study groups and adherence to trial intervention and completeness of data shall be monitored and reviewed on a day-to-day basis.
Statistical analysis
All randomized participants in the study will be included in the analysis. Each participant will be given a study code. Participants will be identified with the phone number and clinic file number. The code will be used to label the data recording sheets, and the Alpha Amylase data. The study is being taken at a single visit treatment with no follow up; it is expected that there will not be any missing data.
Data will be exported into a computer and analysed using SPSS IBM version 25 and the normalcy of the data will be tested using Kolmogorov-Smirnov test. 54
Mean anxiety scores and corresponding 95% confidence interval will be determined. This will be used to compare the treatments and the two interventions. If quantitative data is normally distributed, t-tests will be used for comparison, while Mann–Whitney U-test will be used if skewed. 47 Association between categorical data will be tested using chi-square test. Logistic regression will be used to test differences in categorical variables (binary outcome) while linear regression will be used to test the difference in continuous variables. The significance level will be set at 5% (p ⩽ 0.05). Pearson correlation will be used to assess relationship between pre-treatment information and anxiety. 55 Also, correlation between salivary alpha amylase and psychometric tools (IDAF-4C+, MDAS and Visual Analogue Scale) shall be done.
Results: The study result will be presented at both local and international conferences and will be submitted for publication in high impact journals. All the members that contribute to the conception, design and writing of the trial results shall be adequately mentioned in the authorship according to the contribution. There will be no engagement of professional writers in the writing of the final manuscript of the trial.
Discussion
The primary outcome expected in the study is a reduction in anxiety levels and better acceptance of procedure, while the primary outcome will be measured physiologically (objectively), using the levels of salivary alpha amylase. Anxiety scores will be taken at baseline (prior to intervention with information), after information and during treatment. Furthermore, as the secondary outcome, the study is set to assess the possible effect of complexity/invasiveness of dental treatment on DA and to correlate between the objective (salivary alpha amylase) method and subjective (psychometric) methods of DA assessment.
As an improvement over other studies on DA in Nigeria, this study hopes to establish ways to reduce the fear of dental treatment among the populace.
Strengths and limitations of the study
This study will be conducted as a randomized controlled trial involving the use of methods of pre-treatment information on dental procedures to reduce DA.
The study involves blinding the operators/clinicians to the methods of information used to communicate to the participant; this will reduce bias in the performance of the clinician.
The findings of the study will provide evidence for the adoption of a suitable form of pre-treatment information communication to reduce anxiety towards dental treatment.
The secondary outcome of this study will also give insight into a better form of assessment of DA between the objective method, using a salivary biomarker and the subjective method, using the questionnaire.
The study’s single centre is a possible limitation of the study design.
The study is not considering the learning style of the participants and this may be a limitation.
Though the type of food taken before participation is not considered, the time to the last food taken will be considered for participation.
Conclusion
The study is expected to add to the knowledge of what is presently known about DA. It will also give an insight into the non-pharmacological management of DA among adults. Furthermore, it is expected to be a step towards change in policy regarding DA assessment and management in Nigeria as a whole.
Supplemental Material
sj-docx-1-smo-10.1177_20503121231171995 – Supplemental material for A study protocol for a single-centred randomized trial to investigate the effect of pre-treatment communication methods on dental anxiety among adult dental patients
Supplemental material, sj-docx-1-smo-10.1177_20503121231171995 for A study protocol for a single-centred randomized trial to investigate the effect of pre-treatment communication methods on dental anxiety among adult dental patients by Shakeerah Olaide Gbadebo, Gbemisola Aderemi Oke and Oluwole Oyekunle Dosumu in SAGE Open Medicine
Supplemental Material
sj-docx-2-smo-10.1177_20503121231171995 – Supplemental material for A study protocol for a single-centred randomized trial to investigate the effect of pre-treatment communication methods on dental anxiety among adult dental patients
Supplemental material, sj-docx-2-smo-10.1177_20503121231171995 for A study protocol for a single-centred randomized trial to investigate the effect of pre-treatment communication methods on dental anxiety among adult dental patients by Shakeerah Olaide Gbadebo, Gbemisola Aderemi Oke and Oluwole Oyekunle Dosumu in SAGE Open Medicine
Footnotes
Author contributions
Concept, design and protocol writing by SOG, GAO and OOD, GAO and OOD were major contributors in writing of the manuscript. All authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study is being sponsored under a PhD fellowship grant to the first author (SOG), by the Consortium for Advanced Research Training in Africa (CARTA).
Ethics approval and consent to participate
Ethical Approval received from the University of Ibadan/University College Hospital Ibadan, Institution Review Board: NHREC/05/01/2008a. Written informed consent to participate and publish will be sought from the participants before inclusion in the study. Any changes to protocol modification shall be communicated to the ethics board and the trial registry.
Written informed consent for study and publication
Written informed consent taken for study shall include consent for presentation of result at national and international conferences and submission in high impact journals for publication.
Availability of data and material
The datasets to be generated during and/or analysed during the proposed study will not be publicly available due to need to take permission from the funders. However, the data will be made available from the corresponding author on reasonable request with the permission of funder.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
