Abstract
Objective:
The objective of the present study was to evaluate the safety and tolerability of autoinjector devices (AIDs) in rabbits by intramuscular (i.m.) administration, using haematological and biochemical markers.
Introduction:
Emergency and mass casualty situations require immediate drug delivery for which AIDs are preferred. The tolerability of amikacin as antibacterial and buprenorphine as analgesic AID has been studied in rats by intraperitoneal administration. In the present study, it was evaluated in rabbits by i.m. administration.
Methods:
Water-filled glass cartridges (2.3–2.4 mL) were converted to amikacin (106 mg/mL) and buprenorphine (0.128 mg/mL) cartridges. Dual dose AID was used for i.m. administration (1.2 mL). The study was done as a crossover design on 12 rabbits. Initially, three rabbits each were given manually or AID, 57 mg/kg amikacin, and three rabbits each by manual or by AID, 0.07 mg/kg buprenorphine for 7 days. After 1 month, the injections were changed in the rabbits. In the place of manual injection, AID and in the place of amikacin, buprenorphine injection was given. This ensured that all rabbits received 14 injections, 7 manual and 7 AID consisting of 7 amikacin and 7 buprenorphine. 24 h before and 24 h after last drug administrations, blood was withdrawn from ear vein for haematological and biochemical estimations.
Results:
The rabbits were healthy, active and no sign of any injection-related changes were observed after administration of amikacin and buprenorphine by manually or by AID. The haematological and biochemical parameters showed similar changes in manual and AID administration of amikacin and buprenorphine.
Conclusion:
The present study of amikacin and buprenorphine by AID shows the safety of the device and is recommended for further experimentation. These AIDs are intended for self-administration during emergency and mass causality situation and are suitable for adults and children, as well as farm and pet animals.
Introduction
Many emergency and life-threatening situation arises in which immediate administration of the drugs are required. Although intravenous (i.v.) administration of the drugs is recommended, it may not be feasible in a non-clinical situation. Intramuscular (i.m.) injection is an alternative, but the absorption depends upon the drug, and may not be suitable in all the conditions. For rapid administration and faster absorption of the drug, autoinjector device (AID) is preferable.1 –3 The AID is a convenient method for quick administration of drugs. The drugs are delivered by spray effect by the AID, providing a larger area for absorption. Hence, the absorption of the drugs is faster, thus making the AID injection equivalent to that of i.v. injection.4,5
AIDs are in use for subcutaneous (s.c.) and i.m. administrations.2,3 Many AIDs are available with specific monoclonal antibodies for the treatment of myocardial infarction, hypercholesterolaemia, joint and bowel disease, ulcerative colitis, psoriasis, rheumatoid arthritis, multiple sclerosis and systemic lupus erythematosus. These AIDs are mostly for s.c. administration and are in various stages of clinical trials.2,3,6 Initially, the AID was developed for nerve agent exposure and that required immediate administration. Nerve agent exposure would inhibit acetylcholinesterase resulting in excess acetylcholine, leading to respiratory muscle paralysis and death. Since there will be bradycardia and hypotension, for faster absorption of the antidotes, i.m. injection of atropine and oxime is required. AID was developed for i.m. administration of atropine sulphate and oximes. 7 Similar emergency situations viz., anaphylaxis due to allergies by food, venoms and sports activities required immediate administration of epinephrine i.m., as AID. 8 The other i.m. AIDs available are diazepam and midazolam for seizures, naloxone for opioid overdose, and thrombolytic agents αIIbβ3 and αVβ3 for myocardial infarction. 2
Injuries due to calamities of natural origin such as earth quake, landslide, avalanche and flood, and man-made like accidents and terrorist attacks require an antibacterial drug and an analgesic drug. Since medical support may be difficult to control pain and infection, the drugs can be delivered by AID, on-site. Amikacin sulphate as AID was developed for i.m. administration with 500 mg amikacin in 2 mL as a broad-spectrum antibacterial agent. Buprenorphine hydrochloride as AID was also developed for i.m. administration with 0.6 mg buprenorphine in 2 mL. Extensive studies have been carried out in rats using the AID developed for human, by administering intraperitoneally after diluting the drug cartridge for animal use. The studies have shown that both the AIDs are convenient to use and the injections were tolerable.9 –11
Although human reports are available for s.c. and i.m, injections of AID for the acceptability and usability, studies are not well reported by i.m. in larger animals except for few studies of atropine and oxime. 4 For chronic diseases in humans, the AID is used for s.c. administration for the monoclonal antibodies and for acute symptoms, i.m. administration is recommended for the nerve gas poisoning, anaphylaxis and epilepsy. Since antibacterial and analgesic drugs are required for immediate effect, AID by i.m. administration is preferred. Hence, the aim of the present study was to evaluate the tolerability and injection capability of amikacin and buprenorphine AID, developed for humans, by i.m. injection in rabbits.
Materials and methods
Animals: The present study was carried out as a crossover design. In all, 12 New Zealand white rabbits (2.0–2.5 kg) were used. They were procured from Biogen (Bengaluru, India), housed individually in stainless steel cages and acclimatized for 1 month prior to the experiments. Pellet feed (Biogen) and filtered water were given ad libitum. They were maintained at 25 ± 2°C with natural light/dark cycle. The care and maintenance of the animals were as per the guidelines of Committee for the Purpose of Control and Supervision of Experiments on Animals (CPCSEA, India). This study has the approval of Institutional Animal Ethical Committee of Saveetha Medical College (SU/BRULAC/RD/001/2013).
AID and drugs: Water-filled cartridges for the AID were purchased from M/s Neon Laboratories (Mumbai, India). The cartridges were made of glass and filled with 2.3–2.4 mL of sterile water with a convoluted needle inside. Upon actuation by the AID, 2.1–2.2 mL would be delivered. Amikacin sulphate (500 mg of amikacin in 2 mL) and buprenorphine hydrochloride (0.6 mg of buprenorphine in 2 mL were purchased from M/s Neon Laboratories. AID produced by M/s Sigma Engineering (Hyderabad, India) with an option to select full-dose delivery or partial-dose delivery (dual dose AID) was used. 2 The water from the cartridge, 1.0 mL was withdrawn and 1.0 mL of amikacin was injected to make the concentration to 106 mg/mL. Similarly, 1.0 mL of water from the cartridge was withdrawn and 1.0 mL of buprenorphine was injected to make the concentration to 0.128 mg/mL. All the dilutions were made aseptically in a laminar flow (Figure 1).

Amikacin and buprenorphine AID with water-filled cartridges.
Experimental design: The 12 rabbits were divided into four groups of three animals each by block randomization using SigmaPlot 14.5 version (Systat software Inc., San Jose, CA, USA) – manual amikacin, AID amikacin, manual buprenorphine and AID buprenorphine. Twenty-four hours prior to the injections, the blood was collected in K3EDTA tubes. The ear vein was made prominent by gently scrubbing ethyl alcohol and using a butterfly needle, 2.0 mL of blood was withdrawn. They were given injections either manually or AID, daily once for 7 days. The animals were placed on a flat table. They were held gently on the side in a curved posture with all the four legs together exposing the thigh musculature. Amikacin or buprenorphine was administered manually or by AID. The volume administered by manual injection was 1.2 mL per rabbit by the conventional method and administered vertically by i.m. The drug cartridge was placed in the AID with the restrictor. This allows partial injection of the drug (1.2 mL) and restricting the ejected needle length (1.5 cm). The AID was unlocked, positioned gently on the thigh muscle vertically and the drug was injected. The AID was held in position for 10 sec and gently removed. The needle length that was protruding from the AID was measured. The drug cartridge was weighed before and after injection to estimate the quantity of the drug injected. The injections were delivered daily alternating the right and left side thigh. On the 8th day, blood was withdrawn (about 2 mL) from the ear vein by the same procedure. The animals were rehabilitated for 1 month and the drugs were administered in a crossover design. In the place of amikacin, buprenorphine was administered and in the place of manual, AID administration was given. This ensured that all rabbits received 14 injections, 7 manual and 7 AID consisting of 7 amikacin and 7 buprenorphine. The animals were monitored closely after each injection for signs of pain, and general observation on the food and water consumption. They were weighed daily. The rabbits were rehabilitated again after the completion of the experiment.
Haematological analyses: Various haematological parameters viz., haemoglobin (Hb), packed cell volume (PCV), red blood cell (RBC) count; white blood cell (WBC) count and platelet count were estimated using the whole blood by complete blood cell count by Beckman Coulter Analyser (Beckman Coulter India Pvt Ltd, Chennai, Tamil Nadu, India) as per the instrument guidelines.
Biochemical analyses: The plasma was separated, centrifuging at 2000 rpm for 15 min at 4°C. The following biochemical parameters were estimated viz., glucose, cholesterol, bilirubin, protein, albumin, aspartate amino transferase (AST), alanine amino transferase (ALT), alkaline phosphatase (ALP), gamma glutamyl transferase (GGT), urea, creatinine and uric acid using Roche Modular autoanalyzer, Mumbai, Maharashtra, India (Germany) as per the instrument manufacturer’s procedure. The principles followed for the biochemical estimations were as follows: Glucose was estimated by the ortho-toluidine method. The formation of green-coloured product was measured at 620 nm. Trinder’s method was used for the estimation of cholesterol. The oxidation of cholesterol by cholesterol-oxidase yields a pink-coloured product which was measured at 510 nm. Bilirubin estimation was done as per diazo method. Direct and indirect bilirubin produces azobilirubin in the presence of dimethyl sulfoxide. The colour produced by the reaction mixture was measured at 540 nm. Total protein was estimated by the biuret method. The cupric ion reacts with peptide bond resulting in blue to purple-coloured complex which was measured at 550 nm. Estimation of albumin was based on the bromocresol method. The colour changes from yellow to green which was measured at 610 nm. Urea estimation was based on the urease method. The hydrolysis of urea takes place in the presence of water and urease, forming ammonia and carbon dioxide. The ammonia combines with an α-oxoglutarate and Nicotinamide adenine dinucleotide phosphate (NADH) in the presence of glutamate-dehydrogenase that forms glutamate and NAD. This was measured at 340 nm. Jaffe’s method was employed for the estimation of creatinine. Creatinine in the presence of picric acid in an alkaline medium forms a yellow-coloured complex which was measured at 500 nm. Uric acid estimation was performed as per uricase method of Tinder. The enzymatic reaction between uricase and uric acid forms allantoin and hydrogen peroxide. This was catalysed by peroxidase to oxidize the 3,5-dicholoro-2-hydroxy benzene sulphonate and 4-aminophenazone to a red-violet quinonimine product, which was measured at 520 nm.
Enzyme assays: Measurement of AST, ALT, ALP and GGT was carried out as the per International Federation of Clinical Chemistry kinetic method.
Statistical analysis: The data were analysed using one-way analysis of variance with Student–Newman Keul’s multiple comparison test. A probability of 0.05 and less was taken as statistically significant. SigmaPlot 14.5 version (Systat software Inc., USA) was used for statistical analyses.
Results
The rabbits were healthy prior to the experiment, including the crossover experiment. The baseline haematological and biochemical values were within the normal limits. After the manual and AID administration, the rabbits were active with normal movements and no sign of injection-related changes were observed after the administration of amikacin and buprenorphine, throughout the 7-day experimental period. The food and water consumption were normal after the manual and AID injections. There was no change in the body weight of the rabbits. The injected dose of amikacin per day was approximately 57 mg/kg (1.2 mL injection of 106 mg/mL for 2.25 kg rabbit). The injected dose of buprenorphine per day was approximately 0.07 mg/kg (1.2 mL injection of 0.128 mg/mL for 2.25 kg rabbit).
Table 1 shows the mean and standard error of mean (SEM) of haematological parameters by manual and AID administration of amikacin and buprenorphine, before and after injection. A significant decrease in Hb was observed after the administration of amikacin and buprenorphine by manually and by AID (p < 0.001 and p = 0.023, respectively). In amikacin, there was 12.7% and 26.4% decrease by manual and AID administration, respectively. In buprenorphine, there was 6.8% and 16.1% decrease by manual and AID administration, respectively. The AID group showed a significant decrease in Hb compared to the manual group. A significant decrease in RBC was observed after the administration of amikacin and buprenorphine by manually and by AID (p = 0.059 and p = 0.034, respectively). However, the decrease was similar for the administration of amikacin and buprenorphine by manually and by AID. A significant decrease in PCV was observed after the administration of amikacin and buprenorphine by manually and by AID (p = 0.020 and p = 0.032, respectively). However, the decrease was similar for the administration of amikacin and buprenorphine by manually and by AID. No significant difference was observed in WBC by manually and by AID in amikacin and buprenorphine (p = 0.501 and p = 0.885, respectively), and platelet by manually and by AID in amikacin and buprenorphine (p = 0.312 and p = 0.079, respectively).
Haematological parameters after the administration of amikacin and buprenorphine by manually and by AID.

Mean ± SE (n = 6 each).
Significantly different from the respective before administration.
AID, autoinjector device; SE, standard error.
Table 2 shows the mean and SEM of serum parameters of glucose, cholesterol, bilirubin, protein and albumin by the administration of amikacin and buprenorphine by manually and by AID, before and after injection. No significant difference was observed in serum glucose by manual and AID in amikacin and buprenorphine (p = 0.496 and p = 0.298, respectively), bilirubin by manually and by AID in amikacin and buprenorphine (p = 0.702 and p = 0.481, respectively) and albumin by manually and by AID in amikacin and buprenorphine (p = 0.315 and p = 0.425, respectively). The serum protein by manually and by AID in amikacin did not show significance (p = 0.183), but in buprenorphine, a significant decrease was observed (p = 0.018). The AID group showed significant decrease. A significant decrease was observed in serum cholesterol by manually and by AID in amikacin and buprenorphine (p = 0.018 and p = 0.003, respectively). The decrease in cholesterol was significant by manual amikacin compared to AID amikacin, but not in other groups.
Biochemical parameters after the administration of amikacin and buprenorphine by manually and by AID. Mean ± SE (n = 6 each).
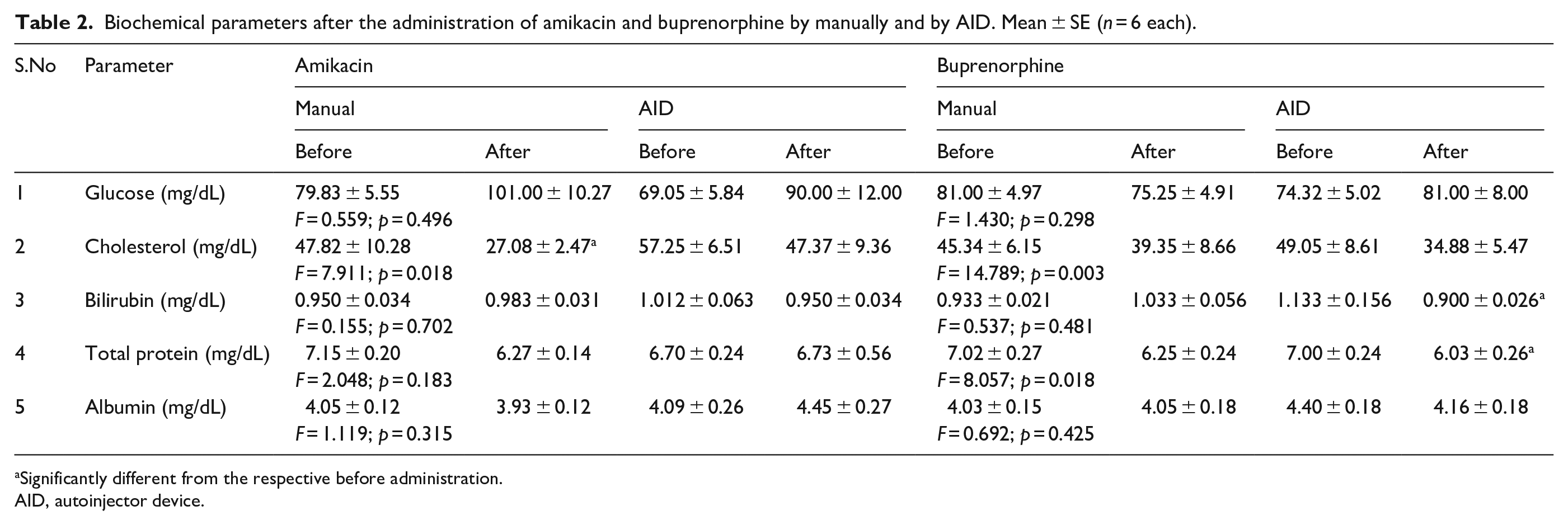
Significantly different from the respective before administration.
AID, autoinjector device.
AID, autoinjector device.
Table 3 shows the mean and SEM of serum parameters of urea, creatinine and uric acid of manual and AID administration in amikacin and buprenorphine, before and after injection. No significant difference was observed in serum urea by manual and AID in amikacin and buprenorphine (p = 0.275 and p = 0.255, respectively), creatinine by manual and AID in amikacin and buprenorphine (p = 0.572 and p = 0.871, respectively) and uric acid by manual and AID in amikacin and buprenorphine (p = 0.133 and p = 0.966, respectively).
Urea, creatinine and uric acid after the administration of amikacin and buprenorphine by manually and by AID.

Mean ± SE (n = 6 each).
AID, autoinjector device; SE, standard error.
Table 4 shows the mean and SEM of serum enzymes of AST, ALT, ALP and GGT by manual and AID administration in amikacin and buprenorphine, before and after injection. No significant difference was observed in serum AST by manual and AID in amikacin and buprenorphine (p = 0.588 and p = 0.237, respectively), ALT by manual and AID in buprenorphine (p = 0.150), ALP by manual and AID in amikacin (p = 0.634) and GGT by manual and AID in amikacin and buprenorphine (p = 0.547 and p = 0.752, respectively). Significant difference was observed in ALT by manual and AID in amikacin (p = 0.039) and ALP by manual and AID in buprenorphine (p = 0.024). However, none of the enzyme parameters showed significant difference from the respective manual injection.
Enzyme levels after the administration of amikacin and buprenorphine by manually and by AID.
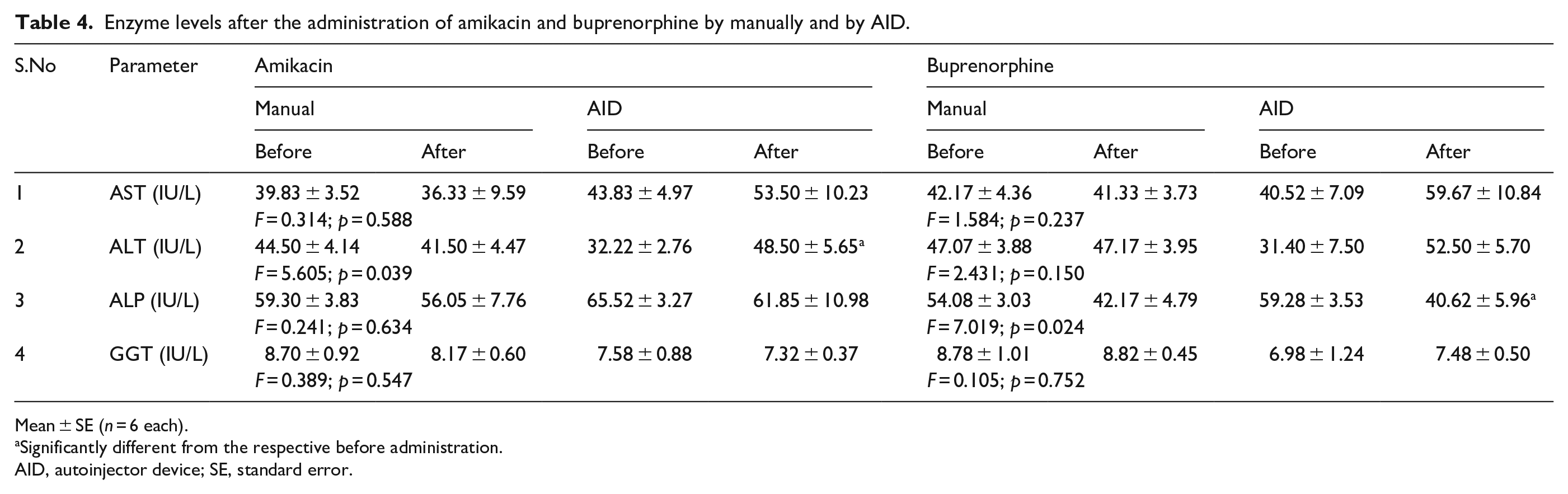
Mean ± SE (n = 6 each).
Significantly different from the respective before administration.
AID, autoinjector device; SE, standard error.
The overall results revealed that there was no difference in the manual and AID injections. The changes that were observed by the manual and the AID were also within the clinical limits.
Discussion
Drug discovery, formulation and delivery systems are necessary for prevention and treatment of human ailments, and for improving the quality of life and survival. Prevention and treatment of infection and pain are the most important requirements throughout the life. Developments in medical field are evaluated critically by a variety of biological assays. In pharmacological research, animal experimentation is a major part in the evaluation of the developed drug, device or the technique. Various animals ranging from zebra fish to non-human primates are used for the research purpose. Although rodents are used initially, higher animals including rabbits are used as the next stage. 12 The amikacin and buprenorphine AIDs are new developments and are tested for its efficacy and safety in rat. 2 Although the AID was developed for i.m. injection, it was evaluated in rat by i.p. injection, as i.m. is not feasible.10,13 The developed AID for human has an option to select full or partial drug delivery (2.0 and 1.2 mL, respectively) equivalent to full or partial needle length of injection (25 and 15 mm, respectively). 11 Hence, for i.m. injection in rabbit the partial delivery was selected. Another animal model where the drug can be injected through i.m. by AID, similar to humans was preferred and rabbits was found to be appropriate due to pain identification by the normal behaviour. Usually, the rabbit would be active, curious and vigilant. Pain can be studied or visualised by change in gait, licking, rubbing, abnormal posture or decreased food and water intake.14,15
AID is used for injecting a drug by the individual or by a caretaker, and also in mass casualty situations, where the healthcare systems are affected. Many AIDs are available for emergency conditions and disease for i.m. and s.c. administration viz., nerve gas poisoning, allergies. anaphylaxis, seizure, migraine, opioid overdose, diabetes, rheumatoid arthritis, multiple sclerosis, psoriasis, systematic lupus erythematosus, hepatitis and infertility treatment.2,3,16 The rapid effect produced by AID has been utilized for its use during emergency conditions. Amikacin and buprenorphine AIDs are new developments for infection and pain management, respectively. The positive result of the previous studies, like antibacterial or analgesic action, unaltered changes in haematological and biochemical parameters showing its safety in rodent models, necessitated the use of a non-rodent model with i.m. injection for the tolerability.9,13,17 Hence, the present study was carried out in rabbit model.
In the present study, the rabbits were assessed for safety and tolerance to the drug injections by manually and by AID with general health, and liver and kidney function tests. For rapid and detectable variation particularly during stress, evaluation of haematological parameters is carried out. 18 Hb, RBC, PCV levels decrease due to haemorrhage, excessive haemolysis or deficiency in erythropoietin. Erythropoietin is necessary for the production of RBC. 19 In chronic renal diseases, the production of erythropoietin may be affected which would indirectly affect RBC production. 20 Some of the pesticides may affect Hb, RBC and PCV. 21 In the present study, a decrease in Hb was observed in amikacin and buprenorphine, both in manual and in AID administration. This could be due to the repeated injections of the drugs and also due to withdrawal of blood for the analyses (from each rabbit, four times blood was withdrawn). The decrease in Hb was more in AID than the manual injection, since the AID intended for humans was used in the present study for the rabbits. This could have caused haemorrhage, but the animals were active with normal movements and no sign of injection-related changes were observed. The serum proteins like albumin are produced by the parenchymal cells of the liver and utilized as an indicator for the hepatic function. Albumin concentration in the serum can be altered due to infections, trauma, liver dysfunction and kidney disease. 22 Bilirubin, a product of Hb degradation from the RBC, can be elevated due to liver diseases and also may be due to haemolysis. Many toxic chemicals and during stress, glucose and cholesterol may altered. 23
Biochemical changes are utilised in assessing systemic effect particularly for hepatic and renal functions. In the present study, urea and creatinine levels were not changed in manual and by AID, both in amikacin and buprenorphine. An increase in serum enzymes indicates liver damage. Usually, when an injury is produced in the liver, there is an increase in the concentration of serum enzymes. AST and ALT are concentrated in larger proportion in liver. AST is found in liver, heart, skeletal muscles, kidney, brain and RBC. In the case of liver damage depending upon the injury, several fold increases in serum AST and ALT are reported. 24 In the case of an ischemic injury, AST level will be increased than ALT level. 25 Liver damage can occur by drugs, chemicals, radiation, and also by herbal- and plant-derived products in experimental animals.26 –29 The other enzymes such as ALP and GGT, and also bilirubin is indicative of liver injury. 25 ALP is also present in placenta, kidneys, intestine or leucocytes. An increase in ALP occurs in cirrhosis, hepatitis, congestive cardiac failure and bone diseases. ALP levels can decrease hypothyroidism, pernicious anaemia and zinc deficiency. 24 GGT is present in hepatocytes, biliary epithelial cells, renal tubules, pancreas and intestine, and can be used as an early marker of oxidative stress. They transport peptides into the cell across the cell membrane and also in glutathione metabolism.25,30 In the present study, both the AIDs, amikacin and buprenorphine did not cause any serious alteration in the haematological, biochemical and enzyme parameters. The few changes observed were also with in the reported clinical limits. The dose of amikacin was approximately 60 mg/kg and that of buprenorphine was 0.07 mg/kg. The reported lethal dose (LD)50 for amikacin is more than 400 mg/kg (i.v. rabbit) and for buprenorphine is 140 mg/kg (i.v. rat).31,32 The dose used in the present study was much less than the LDs and would be unlikely to cause any toxic effects either by manually or by AID. Both amikacin and buprenorphine are effective and safe antibiotic and analgesic.33,34
Limitations
All animal studies are generally carried out with six animals per group for biochemical, pharmacological and toxicological parameters. This is adequate for an expected difference of 50% among the groups with 20% SD, 90% power and 5% significance level. For the animal safety and toxicological studies, limitations are not generally expected as it is a planned study. However, pharmacokinetics study can be carried out to prove the effectiveness of the AID over manual injection.
Conclusion
The present study of i.m. injection of amikacin and buprenorphine in rabbits by the AID indicates the safety and tolerability of the device and is recommended for further experimentation. Pharmacokinetics studies are required to differentiate the absorption, distribution and excretion, compared with manual injection. These AIDs are intended for self-administration during emergency and mass causality situation and are suitable for adults and children, as well as farm and pet animals.
Footnotes
Acknowledgements
The authors are grateful to the Honourable Chancellor, Saveetha Institute of Medical and Technical Sciences (Chennai, India) for the support and encouragement. The authors also thank M/s Neon Laboratories (Mumbai, India) for providing water-filled drug cartridges and M/s Sigma Engineering (Hyderabad, India) for providing the autoinjectors.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethics approval
Ethical approval for this study was obtained from Institutional Animal Ethical Committee of Saveetha Medical College (SU/BRULAC/RD/001/2013).
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
Animal welfare
The care and maintenance of the animals were as per the guidelines of Committee for the Purpose of Control and Supervision of Experiments on Animals (CPCSEA, India).
