Abstract
Objective:
Prescription dispensing services are not digitalized in the majority of governmental hospitals and private clinics in Saudi Arabia, which makes them prone to error. We aimed to evaluate handwritten prescriptions in a region in Saudi Arabia for consistency and accuracy according to international guidelines.
Methods:
A sample of handwritten prescriptions (dated 2016 or 2017) from selected pharmacies in the Al-Qassim region (n = 556) were evaluated for missing information on items related to prescriber, patient, and medication according to the World Health Organization and Food and Drug Administration guidelines.
Results:
We found that the prescriber’s name and contact information were missing in two-thirds of the prescriptions (66%). Patients’ addresses were always missing (100%). No prescription contained medication warnings; a majority lacked information on mode of administration (68%) and methods to avoid refilling (66%).
Conclusion:
Saudi Arabia should take a multipronged approach, including digitalization of prescription dispensing services, in both public and private health care facilities in order to reduce prescription errors.
Keywords
Introduction
Saudi Arabia belongs to the world’s high-income countries. 1 Most of the services it provides to its citizens, except health care, are digitalized (e.g. banking, education, and transport). A recent survey of 185 governmental hospitals showed that the majority did not digitalize medical records or pharmacy services. 2 Eighty percentage (80%) of its health services are owned by the government and provided free of cost to the citizens and the expatriates employed in governmental sectors; the remaining 20% are private healthcare facilities whose services are available through insurance or out-of-pocket payments. 3
Medication errors that can be harmful to patient safety and wellbeing are of two broad types: (1) prescription errors that occur from the act of writing a prescription and (2) faults that occur from erroneous medical judgment. 4 Saudi studies show that the prescription error rate is much higher with handwritten (35.7%) than with electronic (2.5%) prescriptions.5,6 These studies also report a wide variation of errors from handwritten prescriptions (range: 7%–94%).7–9 Convenience sampling, a difference in sources (e.g. hospital and primary care center), or use of disparate assessment tools for evaluating prescriptions could potentially explain the difference in results.
In this article, we assessed a sample of handwritten prescriptions prescribed by physicians working in the Al-Qassim region of Saudi Arabia against the items that the World Health Organization (WHO) and the United States Food and Drug Administration (FDA) guidelines say an ideal prescription should contain.10,11
Materials and methods
We conducted a cross-sectional survey of medical prescriptions by means of a convenience selection of pharmacies (total = 06, primary care center = 05, and private = 01) from four major cities (i.e. Buraidah, Unaizah, Al Bukayriyah, and Ar-Rass) in Al-Qassim, Saudi Arabia, in order to obtain drug prescriptions. We considered a handwritten prescription eligible if the prescription date was 2016 or 2017. The research assistants (fifth-year medical students) collected around 600 prescriptions (scanned them as the pharmacies did not allow hard copies to be taken) between March and May 2017, of which 556 met the eligibility criteria. We needed to evaluate at least 381 prescriptions, assuming 50% of prescriptions would have missing items, a 95% significance level, and a design effect of 1.0. 12
The research assistants received training on prescription items and guidelines (i.e. WHO and FDA). Two assistants assessed each eligible prescription according to a codebook that contained the WHO and FDA items of an ideal prescription,10,11 and each item was recorded as being either present or absent. A senior author (pharmacologist) adjudicated any discrepancies in the merged data. A majority of the items were common between the guidelines, with some items unique to each (Appendices A and B of Supplemental material). These items could be broadly grouped as (a) prescriber (name, full address, telephone number, department, signature, date of the prescription, prescriber’s stamp, and dispensed stamp), (b) patient (name, full address, and age), and (c) medication (brand name, strength/potency, total quantity, dosage form, medications’ instructions/warnings labeling, and written method to avoid refilling of the medications).
The ethical committee at the Ministry of Health for Al-Qassim reviewed and approved the study protocol (Reference No. 20170204). The committee did not require us to have formal written consent as there was no patient contact or abstraction of personal information.
Statistical analysis
We ran the items’ frequencies, tabulated them, and graphed the frequency of missing items (1–5, 6–8, and ⩾9 items) for each guideline using SPSS (version 25).
Results
None of the included prescriptions (n = 556) contained all items in the WHO and FDA guidelines; the frequency of missing items according to WHO was 1–5 = 243, 6–8 = 305, and ⩾9 items = 08 and according to FDA was 1–5 = 195, 6–8 = 345, and ⩾9 items = 16 (Figure 1).
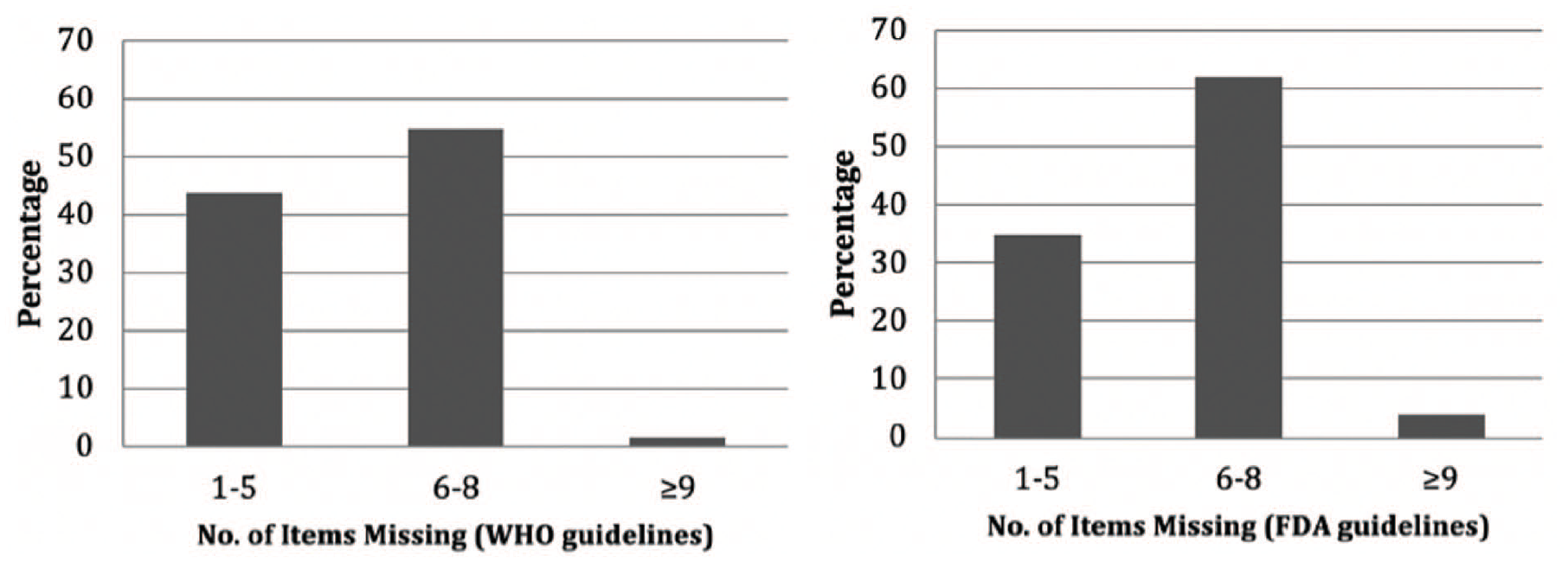
Percentage of missing items in a sample of handwritten prescriptions from Al-Qassim, Saudi Arabia, according to guidelines from the World Health Organization and the US Food and Drug Administration (n = 556).
Almost all prescriptions included the date (99.3%) and signature (92.1%) of the prescriber (Table 1). They had information on full name (34%), address (33.6%), telephone number (34%), and department (32%) of the prescriber (Table 1). None of the prescriptions contained warnings about the prescribed medications, while 32.2% contained instructions about their administration, and 34% included instructions to avoid refilling those medications (Table 1).
Characteristics of the prescriptions (n = 556).

WHO: World Health Organization, FDA: US Food and Drug Administration.
Nearly all prescriptions had the patient’s name (99.6%) and age (79.1%); none, however, had the patient’s full address (Table 1). All prescriptions contained the medication’s name, whereas a respective 76.3%, 67.6%, and 86.9% of prescriptions contained information on strength, total quantity, and dosage (Table 1).
Discussion
Our study showed that crucial information, such as the prescriber’s name, address, and telephone number, was absent in approximately two-thirds of the prescriptions (66%, 66.4%, and 66%, respectively). These results were at odds with results of previously published studies that ranged from no absence (0%) of prescriber’s name8,13 to absence in only a minority of the cases (16.7%), 14 and total absence (100%) of prescriber’s address or telephone number 13 or absence in a majority of cases (90%). 14
Our findings on patient’s name and age (present: 99.6% and 79.1%, respectively) and patient’s address (absent: 100%) were supported by the literature.13–15 Similarly, our results regarding medication warnings being absent from all prescriptions or missing instructions in two-thirds of the cases as to how to administer the medications and how to avoid refilling (67.8% and 66%, respectively) were supported by one study 14 but not another. 15
Our findings about medication strength (23.7%), dosage (13.1%), and quantity (32.4%) being absent from the prescriptions differed from previous reports that found an absence range of 40%–73% for strength,13,14 4%–43% for dosage,7,8,13–15 and 24%–94% for quantity.13,14 However, the proclivity to prescribe generic drugs, as witnessed among physicians in our study, was similar to other studies.13,14
Although there have been many studies on medication errors from Saudi Arabia and the greater Middle East, they greatly differ in what they defined as error; some were interested in the mere presence/absence of prescription items, some looked at the accuracy of information, and others examined adverse drug effects.2,3,6–9 Use of different standards to quantify errors (e.g. WHO, FDA, and the British National Formulary) further complicated the picture as the items were somewhat dissimilar among them.7–9
In 2018, Saudi Arabia successfully began to enforce a law prohibiting dispensation of antibiotics without a prescription, 16 which is a significant step in the right direction to reduce adverse medication-related events. The country should also lay out a concrete, time-bound plan to digitalize its public and private healthcare facilities, including prescription dispensation services. Apart from electronic prescription services, it should consider other proven means of reducing prescription errors, such as formulating guidelines, policies, and procedures for e-prescriptions, instituting error-detection tools, providing staff education, and strengthening clinical pharmacy services. 17 Without these additional safety/quality control measures in place, mere digitalization of prescription services may not bring the intended beneficial effects. 18
We acknowledge several limitations of this study. We did not abstract certain information, such as diagnosis, gender of the patient, or the prescriber’s specialty. We used a non-random sampling method to obtain the prescriptions, limited our assessment to handwritten prescriptions only, and targeted only one province of Saudi Arabia for data collection, all of which should be taken into account when interpreting the data.
Conclusion
Saudi Arabia should take a multipronged approach, including digitalization of prescription dispensing services, in both public and private healthcare facilities in order to reduce prescription errors.
Supplemental Material
sj-docx-1-smo-10.1177_2050312120977153 – Supplemental material for Assessment of handwritten prescriptions from Saudi Arabia according to international guidelines
Supplemental material, sj-docx-1-smo-10.1177_2050312120977153 for Assessment of handwritten prescriptions from Saudi Arabia according to international guidelines by Souhib Mohammed Youssef, Mohamed Saddik Zaghloul, Mohammed Fayez Ahmed, Abdul Nasser Ahmed Barmo, Asghar Mehdi Muhammed Mehdi and Nazmus Saquib in SAGE Open Medicine
Footnotes
Acknowledgements
The authors would like to thank the research assistants at Sulaiman Al Rajhi University for their help with data collection and Ms Erin Strotheide for her editorial contributions to this article.
Author contributions
S.M.Y. conceived of the study. N.S. and A.M.M.M. helped with study design and protocol development. M.F.A. and A.N.A.B. collected the data with the assistance of other student research assistants. M.S.Z. and N.S. performed data analysis and drafted the article. All authors approved its final version.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a grant from the annual budget of the research unit of Sulaiman Al Rajhi University, Saudi Arabia, but it had no role in the study design, collection, analysis or interpretation of data, in the writing of the report, or in the decision to submit the article for publication.
Ethical approval
This study was approved by the Research Ethics Committee of Al-Qassim (study approval number 20170204 and dated 14 February 2017). The ethics committee at the Ministry of Health for Al-Qassim reviewed and approved the study protocol (Reference No. 20170204).
Informed consent
The Ministry of Health ethics committee did not require us to have formal written consent as there was no patient contact or abstraction of personal information.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
