Abstract
Genomic medicine is an emerging practice that followed the completion of the Human Genome Project and that considers genomic information about an individual in the provision of their clinical care. Large and start-up direct-to-consumer genetic testing companies like Ancestry, 23andMe, Luna DNA, and Nebula Genomics have capitalized on findings from the Human Genome Project by offering genetic health testing services to consumers without a clinical intermediary. Genomic medicine is thus further propelled by unprecedented supply and demand market forces driven by direct-to-consumer genetic testing companies. As government entities like the National Human Genome Research Institute question how genomics can be implemented into routine medical practice to prevent disease and improve the health of all members of a diverse community, we believe that stakeholders must first examine how and scenarios in which stakeholders can become motivated to share or receive genomic information. In this commentary, we discuss consumers three scenarios: satisfying personal curiosity, providing a social good, and receiving a financial return. We examine these motivations based on recent events and current avenues through which have engaged or can engage in genomic data sharing via private, secure (e.g. centralized genomic databases and de-centralized platforms like blockchain) and public, unsecure platforms (e.g. open platforms that are publicly available online). By examining these scenarios, we can likely determine how various stakeholders, such as consumers, might prefer to extract value from genomic information and how privacy preferences among those stakeholders might vary depending on how they seek to use or share genomic information. From there, one can recommend best practices to promote transparency and uphold privacy standards and expectations among stakeholders engaged in genomic medicine.
The National Human Genome Research Institute (NHGRI) defines genomic medicine as a “medical discipline that involves using genomic information (also referred to as genomic data) about an individual as part of their clinical care (e.g. for diagnostic or therapeutic decision-making) and the health outcomes and policy implications of that clinical use.” 1 Genomic medicine emerged following the completion of the Human Genome Project, given the project’s overarching objective to “advance our understanding of biology and disease and to improve health.” 2 Several studies have been completed or are underway to determine specific gene variants that cause or increase the likelihood of developing various diseases in demographically diverse populations.
Capitalizing on these research findings are direct-to-consumer genetic testing (DTC-GT) companies that, without a clinical intermediary, offer consumers raw genomic information and summary reports that describe how the consumers’ DNA might influence their health. As leading DTC-GT companies like Ancestry and 23andMe continue to create and introduce new health products, sometimes with regulatory authorization, genomic medicine is further accelerated by supply and demand market forces.3–5 Furthermore, consumers can download and share their genomic information and reports directly with their health care providers and families and other interested third parties (e.g. researchers and health product developers). The clinical validity and utility of these tests, however, remain debated by the scientific and medical community. In addition, DTC-GT for health-related purposes contributes to increased burdens placed on health care systems and providers, as DTC-GT consumers often seek medical advice for interpreting and acting on the data findings. 6
In their latest vision statement, the NHGRI questioned how genomics can be implemented into routine medical practice to prevent disease and improve the health of all members of a diverse community. 7 Specifically, the NHGRI asked, “how do we make sure genomic information is available and useful for all?” Given that DTC-GT testing companies are making genomic information available and useful to individuals and entities at unprecedented levels, we examine three scenarios in which individuals can become motivated to garner and exchange their genomic information beyond their initial interface with DTC-GT companies and across various platforms that offer various incentives. Understanding motivations to share genomic information within and across these three scenarios is important to elucidate privacy expectations, preferences, and trade-offs within those scenarios. Having this understanding would be especially useful for policymakers and other stakeholders or data optimists who are responsible for upholding consumer privacy and have an interest in maximizing the social benefits of sharing DTC-GT data. Moreover, these scenarios can be used as reference points to create practical guidance or tools that can be used to uphold consumer privacy needs and expectations.
Why is genomic information shared and used?
Consumers are increasingly engaged in genomic testing to learn about or predict their health, ancestry, and other personal health factors (e.g. amenability to weight loss through specific dieting) and can access genomic testing without a healthcare intermediary and at any time via DTC-GT companies and newly emerging preventive genomics clinics. 8 Growing levels of DTC-GT consumer engagement combined with the rapid growth of genomic medicine suggest that consumers are either motivated or inspired to generate, use, and share their raw genomic information when the information can be leveraged in ways that serve consumers’ health needs and goals, values, curiosity, and/or beliefs (e.g. determine genetic health risks, whether or not based on geographic ancestry, or identifying biological family members). Industry, public health practitioners, and health care providers are also motivated to use and share genomic information, particularly when genomic information can be aggregated or combined with phenotypic data, health survey data, and data generated in “real-world” settings (e.g. geographic/location data, consumer wearable data, etc.) to reveal meaningful insights about the combined effects of genetics, behavior, and the environment. Insights gleaned based on evidence generated from such data might be valuable for patients, consumers, and other stakeholders who might ultimately benefit from such discoveries.
Here, we discuss three factors that motivate stakeholders to generate and share genomic information: to satisfy personal curiosity, to provide a social good, and to receive a return on investing or contributing their genomic data. We examine these motivations based on recent events and current avenues through which individuals have engaged or can engage in genomic data sharing via private, secure (e.g. centralized genomic databases and de-centralized platforms like blockchain) and public, unsecure platforms (e.g. open platforms that are publicly available online). By examining the extent to which stakeholders have developed or engaged in these platforms, we can examine and discuss how stakeholders become motivated to engage in genomic information sharing activities.
Satisfy personal curiosity
Millions of individuals engage in DTC-GT to satisfy their personal curiosity about their health and geographic ancestry. In April 2019, it was estimated, based on information taken from websites of three leading DTC-GT companies (23&Me, Ancestry, and FamilyTreeDNA), that over 26 million genomic records exist for DTC-GT consumers across those three companies. This is despite public concerns about how DTC-GT companies use, sell, and/or share consumers’ genomic information and despite the fact that most, but not all, DTC-GT companies have privacy policies that incorporate elements of transparency around their data practices.9–11 Beyond finding out about their genetic ancestry and health-related predispositions, DTC-GT consumers also share their genomic information directly in public databases like GEDMatch to find biological relatives. 9 The privacy risks to openly sharing genomic information in public forums continue to reveal themselves over time, affecting both individuals who share such information and their biological family members, as seen in the recent Golden State Killer cases. 9 Thus, it is possible that some DTC-GT consumers either lack salient concerns about privacy or believe the benefits to learning about their geographic ancestry and genomic health risks outweigh privacy risks. 10
Contribute to social good
Thousands of individuals are motivated to share or donate their genomic information, de-identified or otherwise, to serve the greater or social good. Publicly available platforms like Open Humans and the Harvard Personal Genome Project (PGP) appeal to individuals who wish to openly share their identifiable genomic information for the purpose of advancing science and medicine (the “greater good”) and under the notion that genomic information privacy and the true de-identification of genomic information are either impossible or an illusion.12,13 For instance, the PGP has nearly 6,000 project participants who shared their personal genomic and trait data in a free and open manner to serve the “greater good.” 13 Open Humans has over 3,000 participants who have uploaded their genomic data to participate in various research projects like the Genevieve Genome Report, which helps individuals match their genomes against public variant data housed in the public ClinVar database to possibly determine genomic health risks.14–16 Genomic information can be shared as a social good but not openly, with the information protected by security standards that uphold participant privacy. One example in the United States is the National Institutes of Health (NIH) All of Us Research Program Research Hub, which is a national program that endeavors to recruit ethnically diverse individuals to help the NIH discover new ways to understand health and disease. 17
Haeusermann et al. conducted one of the first global surveys (n = 550) to determine individual impetus to openly share their DTC-GT results on a public platform called openSNP and without any institutional oversight and determine individual attitudes on issues of privacy. On openSNP, users can upload their genomic data using either pseudonyms or their real names and sometimes link their social media profiles to their openSNP profiles. Most of the survey respondents came from the United States (60.33%), followed by Canada (5.17%) and the United Kingdom (4.61%), with the remaining coming from other countries that include Australia (3.32%), France (2.21%), Switzerland (2.21%), Russia (2.03%), and Italy (1.48%). At least 79% of the respondents (with variation between men and women up to 89%) felt it relevant or very relevant to share their genomic information to not only learn about themselves, but also contribute to the advancement of medical research. Since then, other studies have found similar results. 10
Receive a return on data investment
The PGP seemingly refers to the “greater good” as “unimpeded research and other scientific, patient care and commercial purposes worldwide.” Thus, one can assume that the greater good also involves engaging individuals or entities that seek to leverage genomic and trait data in the economy or market. Recent events also show that individuals and entities are being incentivized to generate and exchange genetic information for economic returns like shares in a company or legal tender via distributed ledger or blockchain platforms that are appraised as highly secure, de-centralized, and private. 18 This newly emerging practice, however, is accompanied by the need for nondiscrimination protections related to determining the value of consumer-generated data.
The California Attorney General acknowledged this occurrence in a recent notice of proposed rulemaking action regarding the California Consumer Privacy Act 19 of 2018 (CCPA) and described what business practices constitute as discriminatory when offering financial incentives in exchange for consumer data and provide guidance on how to calculate the value of consumer data when designing financial incentives. To promote transparency around this practice, the CCPA requires that businesses publicly disclose the estimated value of a consumer’s data and the method used to calculate and determine the value of the data. As an example of transparency, in their Security Exchange Commission (SEC) Offering Circular, LunaDNA publicly disclosed their valuation methods to determine the cash value of different types of “Member Data” and the number of shares issued in exchange for the contribution of such Member Data (see Figure 1). 20 LunaDNA is the first blockchain enterprise to offer company shares in exchange for genomic information with SEC approval. Their valuation methods for genomic information exchanged via blockchain are likely adopted by other genomics companies that leverage blockchain and promote consumer ownership over DTC-GT data in the marketplace like Nebula Genomics, EncrypGen, and Zenome. 21
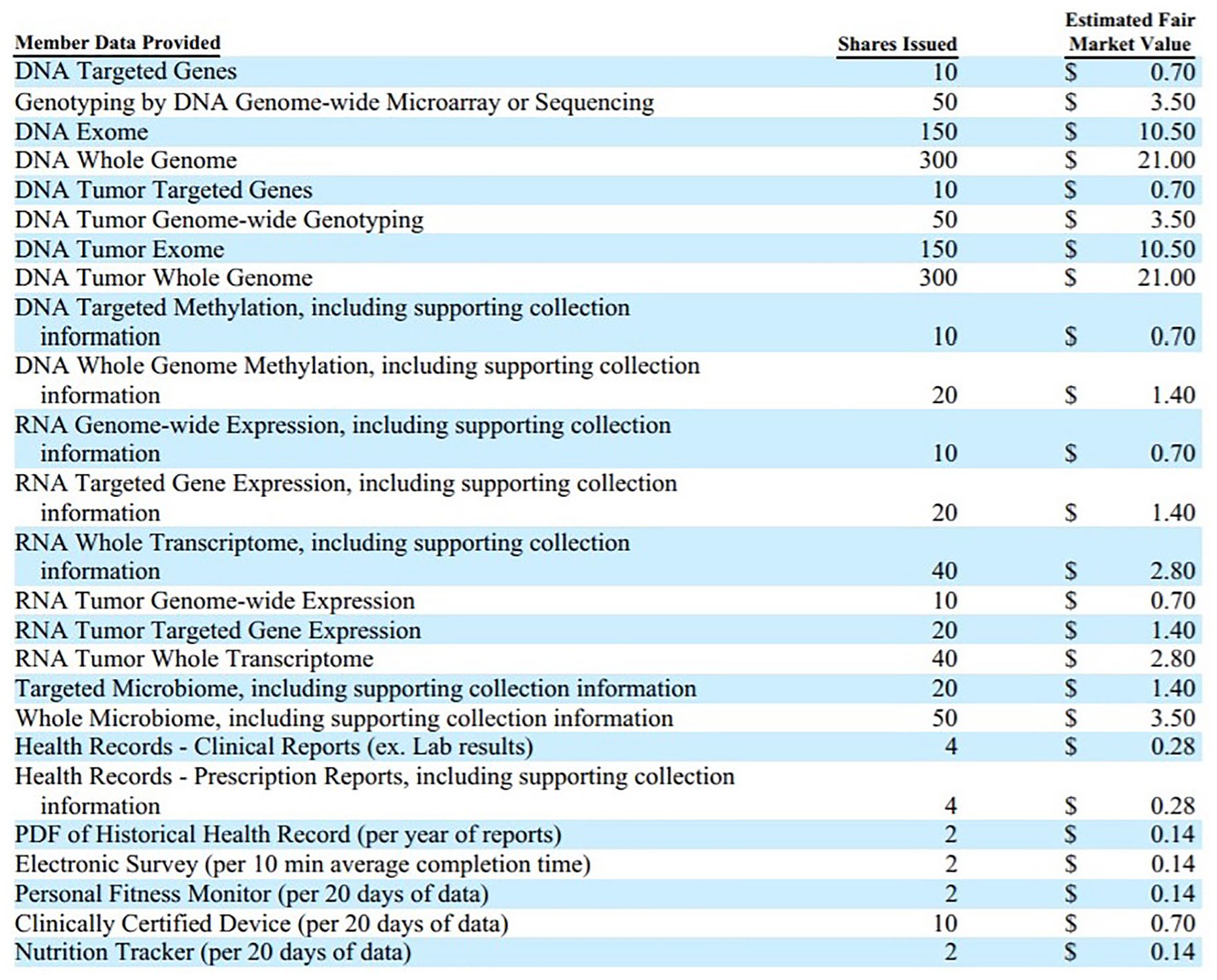
Aggregate offering price or aggregate sales for Luna DNA, LLC, shares based on the fair value of LunaDNA member data.
Understanding stakeholder preferences and expectations
DTC-GT consumers pay to gain insights into what their genomic information means and, upon this, have an option to consent or opt-in to participation in research conducted internally and commercially by DTC-GT companies. Consumers may also consent to DTC-GT companies sharing insights gleaned from consumers’ genetic data with third parties. This consent is likely given, however, after consumers skim terms and conditions that might be lengthy or difficult to understand. 9 Also, there is much variation in transparency or clarity around DTC-GT companies’ data sharing activities internally and with third parties.10,22
Many questions arise about what DTC-GT consumers might expect to learn from their genetics, if they fully consider the potential impact or consequences to their engagement in DTC-GT, and if they consider genomic data sharing as a personal or social investment. For instance, how do DTC-GT genetic test consumers perceive the value of test results that suggest a high risk for a disease with no known cure or treatment? Furthermore, do consumers care about how DTC-GT companies share or leverage consumers’ information when the companies establish business partnerships? Finally, when DTC-GT companies leverage consumers’ genetic data to pursue partnerships with third parties and develop genomics-based products (e.g. pharmacogenomic testing and drugs), what is the likelihood that consumers are able to afford such products? These are important questions to consider when determining DTC-GT consumer preferences and expectations, which are grounded in how those stakeholders perceive the utility of the genomic information they obtain, share, or permit for use.
Illustrating this point is one case (Greenberg v. Miami Childrens Research Institute) in which a physician-researcher isolated and patented the Canavan gene sequence and developed a genetic testing process to detect the gene in patients. 23 Parents of children with Canavan disease, who shared their children’s tissue to support the research, contended that if they had known upfront of the physician-researcher’s intent to commercialize the genomic information through patenting and restrictive licensing, then they would not have shared, or would have kept private, their children’s genomic information. Thus, it is important to better understand variations in privacy preferences or expectations (see Figure 2) and their influences on how stakeholders obtain, share, or permit for use of genomic information. This case also illustrates the role of transparency, as a lack of transparency among key stakeholders can have detrimental consequences long-term.

Underpinnings to variation in stakeholder privacy preferences and expectations when sharing genomic information.
A systematic literature review conducted by Clayton et al. 24 sought to determine concerns about genetic privacy and how these concerns affect individuals’ willingness to accept genetic testing in clinical care and to participate in genomics research. After reviewing 53 studies involving the perspectives of 47,974 participants, they concluded that there is variation in how much individuals want control over the use of their data. They also explained how little attention has focused on understanding how contextual factors like information salience and social forces (e.g. the presence or absence of privacy and nondiscrimination laws and regulations) can influence individual opinions, perceptions, and decisions to share and use genomic information. Thus, more research is needed to understand how the identified, and possibly unidentified, personal, social, and economic factors can intrinsically motivate individuals to engage in genomic information sharing.
Looking forward
Here we highlight several circumstances under which individuals are or can become motivated to share their genomic information: to satisfy personal curiosity, to provide a social good, and to receive financial return. As stated, clearly understanding stakeholders’ motivations across these three scenarios is important in order to elucidate privacy expectations, preferences, and trade-offs within those scenarios. A clear understanding could contribute to the establishment of practical guidance to uphold consumer needs and expectations across these scenarios. Clayton et al. emphasized the importance in determining (1) social practices that make the collection and use of genomic data more trustworthy and (2) circumstances that cause individuals to set aside their concerns about genomic information sharing and engage in genomic information sharing activities. Thus, there is opportunity to engage and promote further research that can closely examine how various stakeholders perceive or value genomic information within and across these three contexts. As examples, further research could inform the development of protective policy and legal frameworks, predictive modeling informed by behavioral economic and/or ethical theories, and the development of new or adapted frameworks that can be used for intervention development and analyses. New or adapted models and frameworks within this scope are would be timely and are needed to inform policymaking at various levels and promote transparency and best practices that could effectively and realistically uphold privacy standards and consumer expectations.
Footnotes
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr. Rachele Hendricks-Sturrup is employed by the Future of Privacy Forum.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Dr. Christine Lu is supported in part by an Ebert Career Development Award at Harvard Pilgrim Health Care Institute and Harvard Medical School, and reports contract with the Center for Genomic Medicine, Massachusetts General Hospital outside the submitted work.
