Abstract
Objective:
The global professional risk assessment applied to the central unit of antineoplastic agent preparations is part of a mandatory approach required by the European legislation for workers. This study identified the hazardous situations related to the staff activity and then enabled the preparation of a formal plan of occupational prevention.
Methods:
The nature of study approved by a working group constituted by experts was the global risk analysis. After identifying the hazardous situations, the global risk analysis estimated the risk level of each hazardous situation based on a criticality score, including severity and frequency. The global risk analysis highlighted the initial and residual risks after establishing a plan to reduce the high criticality risks.
Results:
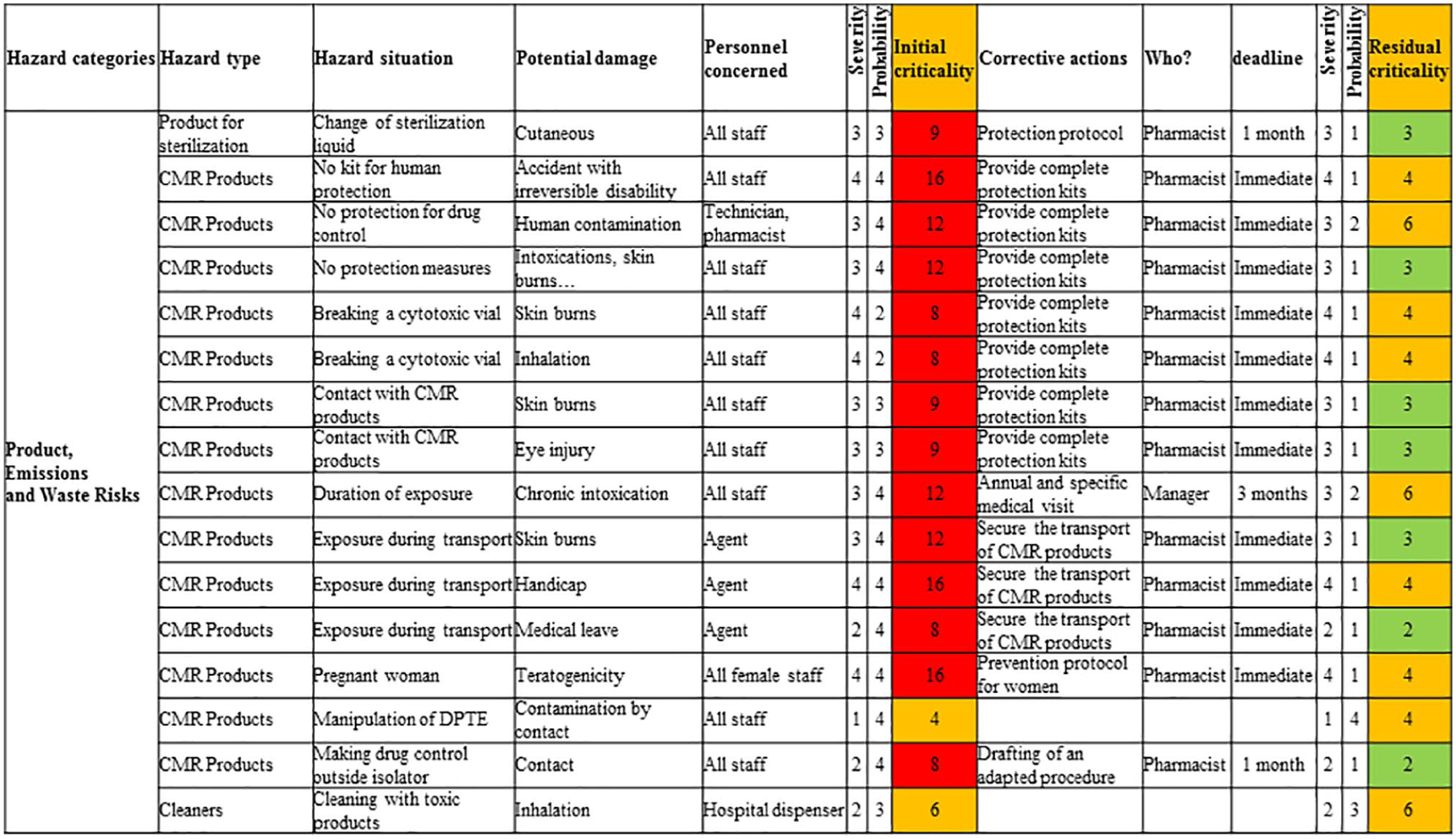
Hence, 33 unacceptable hazardous situations were identified. The critical categories of professional risks were “Product, emissions, and waste risks” with 17 (55%) hazardous situations; “Psychosocial risk factors” with 8 (24%) hazardous situations; and “Risks related to work equipment” with 6 (18%) hazardous situations. Once the risk reduction plan was in place, all hazardous situations were considered under control. The corrective actions led to a reorganization of human resources, the update of protection protocols, and optimization of ergonomic work tools. Staff-specific medical monitoring and regular surface contamination tests have been scheduled annually. In addition, initial and continuous training, specific to product and waste risks, has been updated.
Conclusion:
The global professional risk assessment related to centralized antineoplastic agent preparation unit generated failure in our system and enabled corrective actions for staff safety.
Introduction
Because numerous antineoplastic drugs have been found to be carcinogenic or possibly carcinogenic character for humans by the International Agency for Research on Cancer (IARC), handling and preparation of these products expose the health care personnel to occupational contamination. Therefore, in order to avoid contact with these products by health care workers, government institutions have promoted the centralization of the antineoplastic agent preparations in the hospital pharmacy unit. They developed safety guidelines for employees who could be exposed to antineoplastic drugs and provided information on statutory requirements. 1 In addition, hospital managers are required by law to identify hazards and conduct a comprehensive occupational risks assessment of the clinical activity. The processes of hazard identification and risk assessment will define the actions required to ensure the safety and protection of the physical and mental health of the workers. They involve in the first place actions for the prevention of occupational hazards, information and training for workers, and the implementation of the resources and organization necessary to achieve the objectives. The prevention measures in the risk management plan cover the intelligent deployment of techniques, working organization and conditions, social relations, and the impact of work-related issues. In our hospital, it emerged that data on occupational risks were not available for personnel working in the centralized antineoplastic agent preparation unit.
In this context, the objectives of this study were to evaluate potential occupational risks associated with the centralized antineoplastic agent preparation unit; to identify hazardous situations for each danger; to develop a work safety plan; to limit the costs of work-related accidents, occupational illnesses, and absenteeism; and to improve work safety, standards, and conditions.
Materials and methods
Selected method
The nature of the selected method is a global risk analysis (GRA) used in multiple fields covering industrial, military, financial, environmental, and health care activities. The GRA is a systemic, rigorous, and inductive approach performed in two steps: the GRA system and the GRA scenario. 2 The GRA system determines the general and specific hazardous situations and constructs the risk map. The GRA scenario associates each hazardous situation with a scale of severity, frequency, effort, and acceptance of risk level. For each hazardous situation, the occurrence of an accident can be dreaded depending on the association of possible causes that may trigger it (Figure 1). The severity scale is built at five levels corresponding to consequences on the system performance and on the system safety. The frequency scale is associated to recurrence periods. The effort scale is classified in three levels attributed to each risk management action. Then a criticality matrix derived from these scales is built to score the hazardous situations in acceptable, under control, or unacceptable risk. The map highlights the initial and residual risks after establishing a plan to reduce these risks.

Accident scenario description.
The GRA was applied to draft the global risk document–related global process (Figure 2).

Steps to draft the global risk assessment.
Prepare the approach
The occupational risks were in part directly detected in the workplace in real situation. Furthermore, a working group was created to conduct an a priori occupational risk assessment, composed of PhD, PharmD, pharmacy dispenser, agent, and hospital technician. The working group assigned a project leader who was trained in the risk analysis methods. A participative process on knowledge, understanding, and thinking about work is necessary to evaluate the occupational risks in each work unit. The sample size selected in the study was justified by the knowledge of the staff working in the unit about potential occupational risks with antineoplastic agents.
Evaluate the risks
Once the working group was formed, the occupational risks were identified. Information on occupational risks faced by employees was gathered based on ad hoc meetings. Each risk was determined using assessment parameters approved by the working group in terms of severity and frequency. For each identified risk, a least two observations were made to incorporate this risk in the study. As defined in Table 1, a value range from 1 to 4 was attributed to each hazardous situation to quantify severity and frequency. The severity scale was built by taking into account discomfort, degree of physical harm, possibility of hospitalization, and psychological integrity (Table 1). All the scales were established by the working group according to the methodology described previously by Desroches. 2 The occupational risk was then rated by combining severity and frequency to provide a criticality matrix and prioritized (Table 2). To visualize initial and residual risks for hazard categories, radar charts, also called Kiviat diagrams, were built from the data values as represented in Figure 3 and plotted from criticality scale. The radar charts were developed using the software StatCart (V2.0, MAD-Environnement, Nailloux, France).
Severity and frequency scales.

Criticality matrix.

1 to 3: the risk was considered as acceptable.
4 to 7: the risk was considered as tolerable under control.
8 to 16: the risk was considered as unacceptable.
Part of global risk assessment.
Elaborate a program of corrective actions
Preventive actions have been classified by priority based on risk level and the expected effort to apply corrective actions. The working group prioritized unacceptable hazardous situations. The residual risks were re-rated by the criticality of hazardous situations following corrective actions. If new criticality indices were unacceptable, then additional corrective actions were proposed.
Results
Global analysis
The working group was constituted of four hospital dispensers, three hospital pharmacists, one hospital agent, and one technician. All of them were regularly operated in the centralized antineoplastic agent preparation unit. The working group drew up the global document on occupational risks based on the identified occupational hazards and their assessment (Figure 3). Thus, 33 hazardous situations have been identified and considered unacceptable. The critical categories of professional risks were “Product, emissions, and waste risks” with 17 (55%) hazardous situations; “Psychosocial risk factors” with 8 (24%) hazardous situations; and “Risks related to work equipment” with 6 (18%) hazardous situations. To visualize initial and residual professional hazards, radar charts (i.e. Kiviat diagrams) were built (Figures 4 and 5). After the risk reduction plan was designed, all hazardous situations were under control.

Radar chart for initial occupational hazards.
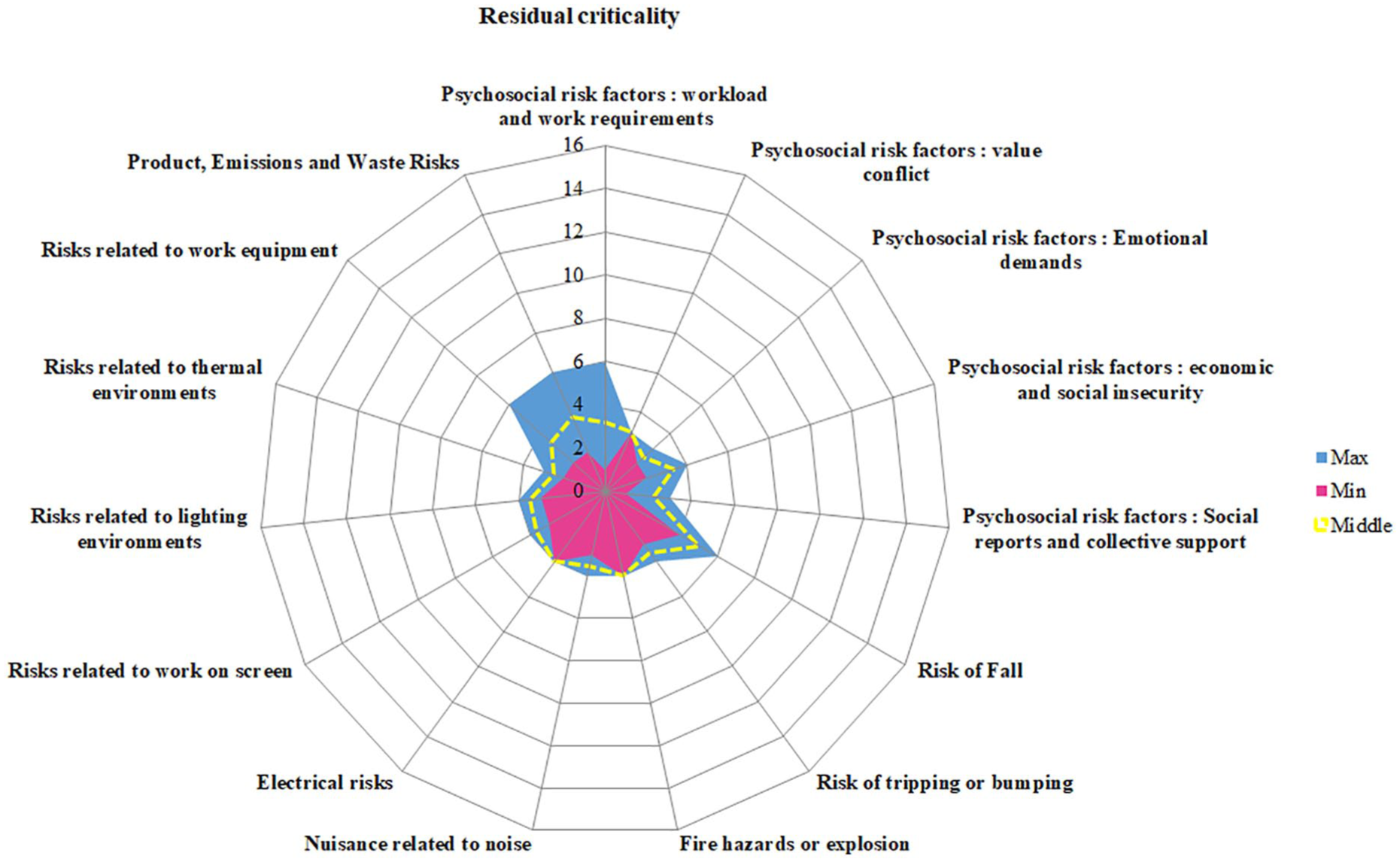
Radar chart for residual occupational hazards.
Psychological risk factors
The working group identified some critical psychological risks such as workload, repetitive movements, and organization. Hazardous situations included stress, fatigue, lack of medical supervision, staff turnover, and absence of training in occupational risk prevention. This may lead to sick leave, needle stick, toxic contamination, and a perception of ineptitude. To reduce these occupational risks, the working group suggested four corrective actions: adjust the number of staff to the activity, provide a break for every 2 h, plan ongoing training on occupational risks for each worker, and request an annual health checkup for each activity. The estimated time to implement corrective actions ranged from 1 to 6 months.
Risks related to work on screen
In the centralized preparation unit, the pharmacists who validate prescriptions and edit production sheets worked all day on the computer screen. The working group noted ocular signs of irritation, dryness, eye fatigue, headache, and visual pain. To reduce incidence of eye symptoms among computer users, eye pauses, appropriate workstation adjustments, lighting control, and dazzle reduction have been instituted.
Risks related to work equipment
The centralized antineoplastic agent preparation unit is a confinement area where staff was fully isolated from the rest of the pharmacy and particularly from the fire alarm relay. To prevent any fire risk, a fire alarm report was installed in the centralized preparation unit. Moreover, the risk of back pain due to extend sitting time during the preparation process was minimized by purchasing ergonomic chairs. The staff was also impacted by the information technology (IT) system due to a bad network connection that caused staff irritation. A general analysis of the IT system identified the slowness of the process. A further hazardous situation reported by personnel was the potential exposure to the toxic vapors emanating from the plastic of the automatic welding machine. A filtering facepiece particles type 2 (FFP2) mask was subsequently requested by the working group to prevent this kind of inhalation toxicity.
Products, emissions, and waste risks
This hazard category is the most risky part of the global occupational risk assessment covering 17 hazardous situations. Exposure to the antineoplastic drug may be due to ocular, inhalation, or skin contact. The use of the isolator mitigates the inhalation risk and is a highly effective device for reducing surface contamination. The main route for exposure is mainly the skin route via possible contamination of the external surface.
Indicators of occupational health and safety
The management of occupational risks related to the centralized antineoplastic agent preparation unit involves the establishment of specific occupational health and safety indicators. The indicators will quantify specific issues related to working conditions. A dashboard was constructed with some relevant indicators (Table 3). In a recent study, safety of occupational exposure to residual contamination with antineoplastic drugs was investigated, and the main corrective action was to strengthen the training program. 3
Relevant indicators of occupational health and safety.

Discussion
The drafting of the global occupational risk document for the centralized antineoplastic agent preparation unit in our hospital made it possible to identify corrective actions in the field of personnel health and safety. This report highlighted the main gaps in our workplace health system. The psychological pressures identified by personnel can have a negative impact on the mental or physical health of the worker. Data showed that psychological stress at work was associated to depression, emotional distress, anxiety, burnout, and psychoactive drug use.4–8 The psychosocial work environment is critical to mental health, especially for employees operating in stress areas such as centralized antineoplastic drug preparation units. 6 The study focused on implementing corrective measures for work stressors. A key approach to preventing work-related stress consists of professional training. 9 The time spent on the computer screen was reported as high by the working group. Because eye symptoms were noted (irritation, dryness, eye strain, headache, and pain), working time on screen should be reduced to a minimum, if possible.10,11
To assess the risks of antineoplastic agents to workers, some publications report an investigation of potential occupational exposure survey by measuring concentrations of antineoplastic drugs on surfaces. In Sugiura et al.’s 12 study, surface contamination by cyclophosphamide was measured at the nursing station and in the biological safety cabinet (BSC). The results showed contamination on surface telephone table (4.45 ng/cm2), floor in front preparation table (1.12 ng/cm2), and floor below IV stand (4.82 ng/cm2). Near BSC, lower levels of contamination were noted on floor in front of BSC (0.11 ng/cm2). In a recent study, skin contact monitoring of antineoplastic drugs was performed using wipe samples from exposed surfaces. 13 The analysis in the preparation room ranged from < limit of detection to 276 ng/cm2 with contamination on computer and telephone (14%), door (5%), cupboard (35%), table (10%), and floor (18%). Further studies have shown that surfaces of commercial vials, pre-filled syringes, and infusion bags can be contaminated by cytotoxic drugs.14–16 In the study of Turci et al., 16 the determination of antineoplastic drug contamination on the surfaces showed high levels of 5-fluorouracil (FU) on drug vials (156 mg) and IV tubing set (22.2 mg). Because no occupational exposure limits have been established for antineoplastic drug concentrations, safe exposure levels should be at zero contamination. The cleaning protocol must be effective depending on the cleaning solution used, volume, exposure time, and application mode. 17 In addition, the efficacy of the decontamination process would include both the surface materials and the physicochemical properties of antineoplastic drugs. 17 Standardization, evaluation, and validation of the cleaning procedures are required to guarantee an optimal washing process. While safety precautions were applied in the centralized preparation unit, potential hazards were identified by the working group such as skin or inhalation exposure, unsafe needle punctures, and accidental contamination due to a lack of occupational hazard knowledge. To remove as much as possible these occupational risks, the exposure time was reduced to a minimum and personal respiratory equipment was supplied. Cytotoxic contamination tests were performed on technical areas. To manage antineoplastic drug exposure in emergencies, safety protocols and protective kits accessible to staff have been established. Moreover, a health program was suggested by the working group for the individual monitoring of personnel health status based on their activities in the centralized preparation unit. The pharmacy manager in charge of this unit assessed for each employee the risks of occupational exposures that could affect them. To support this initiative, the risk manager has planned information and training workshops for staff. Since dermal or inhalation exposures are not excluded, women of childbearing age who wish to have a child were immediately exempted from the centralized preparation unit. Likewise, pregnant women or women who are breastfeeding were inevitably prohibited to work in any activity related to antineoplastic agents. To assess chemical contamination of surfaces when handling antineoplastic drug preparation, antineoplastic agent levels were determined by an external organism. The cartography of most contaminated areas could allow remedial action for cleanup. In the study by Kiffmeyer et al., 18 it was suggested that repeated monitoring of contaminated areas should be scheduled. Decontamination and cleaning procedures for all devices in contact with antineoplastic drugs have been set up, independently of the mandatory use of gloves. Moreover, potential toxicity in humans was assessed by determining concentrations of antineoplastic substances in the urine. Contact exposure to antineoplastic drugs and technical risk assessment tools should be considered by the director of risk management.
In the present work, the global occupational risk document identified hazardous situations in a centralized antineoplastic agent preparation unit and defined occupational risks for each worker depending on their activities and measures to control them. The fields of corrective action concerned information on occupational risks for workers in contact with antineoplastic agents, updating emergency procedures, setting up regular health monitoring and continuous training. For each antineoplastic substance, a safety data sheet containing instructions for its safe use should be readily available to staff. In addition, accurate information on the potential risks of exposure to antineoplastic substances was also vital for a risk management strategy. The work tasks that could expose workers to toxic agents were identified and allowed the risk to be reassessed for each worker. The limitations of the analysis were the subjective appreciation of the experts, the need for sustained analysis, and the sample size of participants.
Conclusion
The drafting of the global occupational risk document for the centralized cytotoxic preparation unit revealed failures in our system and enabled corrective actions for personnel safety.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Ethical approval was not sought for the present study because all participants were informed the study was entirely anonymous. Our institutional board has considered that the category of the study may be reviewed by an exempt procedure. The information obtained is recorded by the investigator in such a manner that the identity of the human subjects cannot readily be ascertained, directly or through identifiers linked to the subjects. All participants were employees of the same department as hospital pharmacy unit. The ethical approval number is not applicable here.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Verbal informed consent was obtained from all subjects before the study and approved by our institutional board. All participants were in the same hospital pharmacy department. The Ethics Committee has waived the requirement to obtain written informed consent from all subjects.
