Abstract
Ankaferd blood stopper is a standardized mixture of the plants Thymus vulgaris, Glycyrrhiza glabra, Vitis vinifera, Alpinia officinarum, and Urtica dioica and has been used as a topical hemostatic agent and with its clinical application established in randomized controlled trials and case reports. Ankaferd has been successfully used in gastrointestinal endobronchial mucosal and cutaneous bleedings and also in abdominal, thoracic, dental and oropharyngeal, and pelvic surgeries. Ankaferd’s hemostatic action is thought to form a protein complex with coagulation factors that facilitate adhesion of blood components. Besides its hemostatic action, Ankaferd has demonstrated pleiotropic effects, including anti-neoplastic and anti-microbial activities and tissue-healing properties; the underlying mechanisms for these have not been well studied. Ankaferd’s individual components were determined by proteomic and chemical analyses. Ankaferd also augments transcription of some transcription factors which is shown with transcriptomic analysis. The independent effects of these ingredients and augmented transcription factors are not known precisely. Here, we review what is known of Ankaferd blood stopper components from chemical, proteomic, and transcriptomic analyses and propose that individual components can explain some pleiotropic effects of Ankaferd. Certainly more research is needed focusing on individual ingredients of Ankaferd to elucidate their precise and effects.
Ankaferd Blood Stopper is a standardized mixture of plants Thymus vulgaris, Glycyrrhiza glabra, Vitis vinifera, Alpinia offacinarum and Urticora dioica plants and have been used as a topical hemostatic agent. As a traditional medicine, Ankaferd is shown to have pleiotropic effects. ABS demonstrated anti-neoplastic, anti-microbial actions, also promoted tissue healing. In this study, literature about ingredients of ABS formula with respect to previously performed chemical, transcriptomic and genomic analysis is presented to explain these pleiotropic effects.
Introduction and effects of Ankaferd
Ankaferd is a novel hemostatic agent that was formulated from a traditional extract used in Anatolia and is a standardized mixture of the plants Thymus vulgaris, Glycyrrhiza glabra, Vitis vinifera, Alpinia officinarum, and Urtica dioica. Ankaferd’s hemostatic action and clinical application has been established in randomized controlled trials and case reports. However, Ankaferd has pleiotropic effects including anti-neoplastic, anti-microbial, anti-mutagenic, and antioxidant as well as tissue-healing properties, 1 although the underlying mechanisms have not been well studied. Here, we review the literature on Ankaferd to clarify the mechanistic basis of this pleiotropism. The procoagulant function of Ankaferd blood stopper (ABS) has been extensively discussed elsewhere. 2
Ankaferd has been successfully used as a topical hemostatic agent, for example, in the treatment of digestive tract ulcers as well as various types of bleeding including variceal/non-variceal, gastrointestinal,3–5 endobronchial, 6 mucosal (including in patients with hemorrhagic diathesis7–9), and cutaneous. 10 Ankaferd has also been employed in abdominal, thoracic, dental and oropharyngeal, and pelvic surgeries (Table 1).11–14 The precise mechanism of Ankaferd’s hemostatic action is not known, but it is thought to form a protein complex with intrinsic coagulation factors that facilitate erythrocyte and thrombocyte adhesion. The hemostatic mechanism of Ankaferd will not be further addressed here; instead, we refer readers to a recent review of this subject. 2
Clinical trials on Ankareferd’s hemostatic action.

ACB: autogenous cortical bone greft, ABS: Ankaferd blood stopper; AOT: Oral anticoagulant therapy; CABG: coronary artery bypass grafting; ASA: acetylsalicylic acid; INR: international normalized ratio; GIS: gastrointestinal system; GR: gingival recession; HCT : hemostasis by conventional technique; PCNL: percutaneous nephrolithotomy; SS: saline solution; VEGF: vascular endothelial growth factor.
Ankaferd has long been known to promote the repair of bone, periodontal, muscle, skin, gastrointestinal, and oropharyngeal tissues and urogenital mucosae after surgery or injury, although some contradictory findings have been reported.27–30
The antimicrobial activity of Ankaferd is thought to derive from its oxygen-enhancing capacity through erythrocyte aggregation. The antimicrobial spectrum is broad and includes microorganisms such as methicillin-resistant Staphylococcus aureus, Pseudomonas aeruginosa, Klebsiella, Acinetobacter baumannii, Mycobacterium tuberculosis, and Candida albicans.31,32 Ankaferd has also been used to treat hydatid cysts via injection.33,34
Less is known about the anti-neoplastic activity of Ankaferd, although it was shown to inhibit cell proliferation, promote apoptosis, and prevent blast cell transformation of B-cell chronic lymphocytic leukemia cells in vitro. 35 It has also demonstrated toxicity toward multiple myeloma and plasmacytoma cells both in vitro and in vivo, while decreasing M protein production.36,37 Other solid malignancies such as sarcoma and colon cancer are also targets of the anti-neoplastic action of Ankaferd.
Method
The constituents of Ankaferd have been identified by proteomic and transcriptomic analyses.38,39 Proteins were searched in the Uniprot Protein Knowledge Database by accession number, whereas genes were searched in the GeneCards database. Both proteins and genes were searched in National Library for Health and Institute for Scientific Information databases. References and related literature were reviewed by three authors. Based on this information, we hypothesized that the constituents of Ankaferd contribute to each of its three major pleiotropic actions, which are discussed below.
Antimicrobial effects
Ankaferd has antibacterial, germicidal, and antimycobacterial effects. In vitro studies have shown that ABS is effective against both Gram-positive and Gram-negative bacteria; 40 foodborne pathogens such as Escherichia coli and Salmonella typhimurium; 41 resistant nosocomial pathogens such as Pseudomonas, Klebsiella, Acinetobacter, Enterococcus, and Staphylococcus species; 42 Echinococcus granulosus; 33 and resistant Tuberculosis strains.
The antimicrobial effects of Ankaferd may be attributable to its chemical components. A time-of-flight mass spectroscopy analysis of ABS revealed the presence of the antioxidants tocotrienol, tryptophan, thymol, lycophene, enoxolone, tomatine, tertiary butylhydroquinone, vitamin E derivatives, and galangin. 43
Anti-neoplastic effects
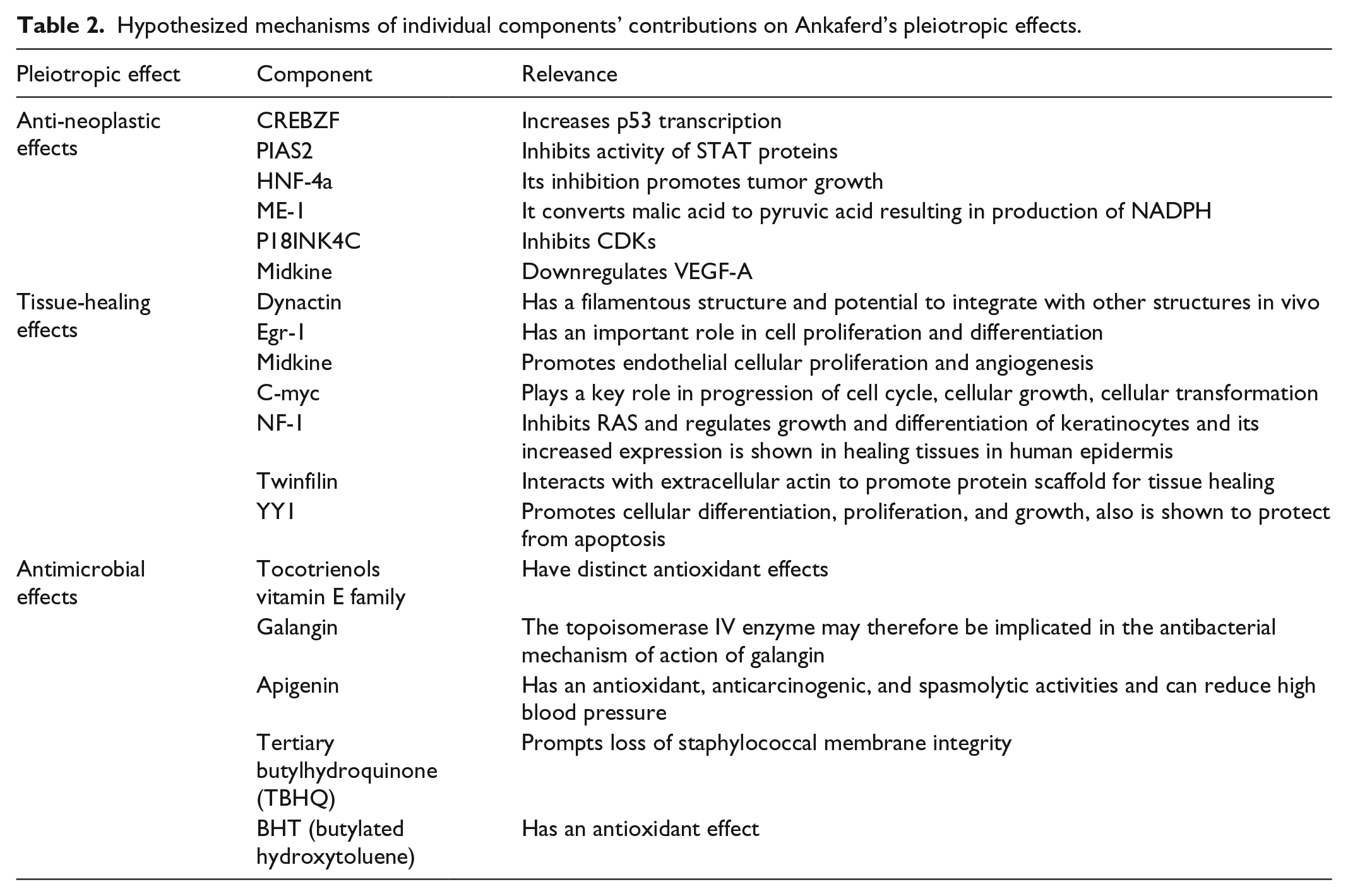
Another property of Ankaferd is the capacity to inhibit neoplasia. ABS has demonstrated cytotoxicity against human erythrocytes at some concentrations, as well as against tumoral cells in hematologic malignancies such as multiple myeloma, chronic myelogenous leukemia, and lymphoma.35,37,44 Possible related ingredients of Ankaferd are CREBZF, PIAS-2, HNF-4a, ME-1, P18INK4C, and Midkine and addressed below in alphabetical order.
In transcriptome analyses, ABS has been found to increase the expression of cyclic AMP response element-binding protein (CREB)/ATF BZIP transcription factor (CREBZF), a member of the mammalian CREB family of transcription factors; this increased the level of the anti-neoplastic protein p53, thereby enhancing gene transcription. 45 CREBZF also regulates the unfolded protein response to protect against excessive protein synthesis during endoplasmic reticulum stress. 46
Hepatocyte nuclear factor (HNF)-4a is a nuclear receptor found not only in the liver but also in other tissues, which is involved in embryonic development, cellular differentiation, and hepatocyte-specific protein synthesis. Recent studies have shown that HNF-4a also has anti-neoplastic activity, with its inhibition leading to tumor growth. 47 HNF-4a is a component of ABS extract and may be partly responsible for its anti-tumorigenic effects.
Malic enzyme (ME)-1 is an intracellular cytosolic protein and a component of ABS that converts malic acid to pyruvic acid, yielding nicotinamide adenine dinucleotide phosphate (NAPDH). ME-1 plays an important role in cancer metabolism, since NADPH is required for anaerobic respiration; indeed, ME-1 level is upregulated in some cancers.48,49 However, the role of ME-1 in ABS remains unclear given that it remains outside of cells, and its in vivo function has not been investigated.
Midkine is a heparin-binding protein that is involved in cellular growth, survival, migration, and differentiation. Midkine was found to suppress vascular endothelial growth factor A, which plays a key role in tumoral angiogenesis; its inhibition may thus inhibit tumor growth.50,51 Although a more likely role for midkine is the promotion of tissue healing (discussed below), we speculate that it also has anti-tumoral effects.
Protein inhibitor of activated signal transducer and activator of transcription (PIAS)-2 is another component of ABS extract. This protein belongs to PIAS family, whose members suppress the activity of STAT proteins, 52 which are critical components of the Janus kinase (JAK) cascade that acts downstream of many growth factor receptors. JAK-STAT signaling is a major pathway involved in human carcinogenesis. 53 Thus, the anti-neoplastic action of ABS may be exerted via inhibition of STAT protein via PIAS-2.
The cyclin-dependent kinase (CDK) inhibitor P18INK4C 54 inhibits tumorigenesis, and its deficiency promotes tumor growth.55,56 CDKs are serine/threonine kinases that regulate the cell cycle and thus play a vital role in human cancers. P18INK4C may participate in the tumor-suppressor activity of ABS by inhibiting CDKs.
Tissue-healing effects
In addition to being a hemostatic agent, ABS has an established role in promoting tissue repair after radiation damage and colitis; 57 bone, cartilage, muscle, and tendon remodeling;27,58,59 and repair of dermal and epidermal tissue, 29 gastrointestinal mucosa, and full-thickness injuries in the abdominal viscera.9,60 We hypothesize that Dynactin, Egr-1, Midkine, NF-1, Twinfilin, V-myc, and Yin Yang 1 can contribute to this effect thus mentioned in following paragraphs with possible mechanisms.
Dynactin along with its partner motor protein dynein is an integral component of cytoskeletal machinery that controls organelle movement during cell division. Dynactin has a central alpha helix with adjacent peptides that interact with dynein and other proteins. Dynactin is an ABS constituent; given its filamentous structure and capacity for integrating with other structures in vivo, it may contribute to the stimulatory effect of ABS on tissue healing.61,62
Inflammation is an integral aspect of tissue repair. Early growth factor (Egr)-1 is a transcription factor and ubiquitous growth factor present in ABS that has an important role in cell proliferation and differentiation. Increasing Egf-1 transcriptional activity may promote tissue regeneration.63,64
Midkine, the heparin-binding protein described above, is known to stimulate the growth of and provide protection to tissue, 65 but also regulates immunity and inflammation. Midkine promotes endothelial cell proliferation and angiogenesis and has been implicated in the pathogenesis of various diseases. 66
Neurofibromin (NF)-1 is a tumor suppressor whose mutation is linked to neurofibromatosis type 1 and juvenile myelomonocytic leukemia. It is a Ras GTPase-activating protein that inhibits Ras and regulates the growth and differentiation of keratinocytes; it is overexpressed in the human epidermis during tissue repair.67,68
Twinfilin is an actin monomer-binding protein that stabilizes and facilitates the function of actin fibrils. 69 It is highly conserved between yeasts, humans, and other mammals. Twinfilin in ABS may interact with extracellular actin to form a protein scaffold that facilitates tissue repair. 70
V-myc avian myelocytomatosis viral oncogene homolog (c-myc) and its product nuclear c-Myc phosphoprotein play a key role in cell cycle progression and cell growth and transformation. Although c-myc is a known oncogenic protein, its transcription is upregulated during wound healing; 71 as such, it may be another ABS component that contributes to tissue repair.
Yin Yang 1 is a ubiquitous and highly conserved transcription factor enriched in ABS that is known to promote cell differentiation, proliferation, and growth, especially in the central nervous system, and protect against apoptosis. 72
Conclusion
The aim of this review is to constitute a framework for future research about pleiotropic effects of ABS. It is composed of hypotheses of Ankaferd ingredients’ individual contributions to pleiotropic effects. There are some limitations of this review. The hypotheses are constructed regarding the molecules’ actions in literature. First, these effects may not be reproduced in vivo and within the environment that the drug acts. Discrete studies on each ingredient is needed to confirm isolated effects of these molecules. Second, there may be some alternative explanations for these effects including contribution of other molecules or divergent effects of known molecules. Third, the process for each pleiotropic action needs to be explained more precisely with more studies to enable better assumptions and a more straightforward research tract about underlying molecular processes.
Ankaferd is an effective hemostatic agent that has been shown to be effective in suppressing gastrointestinal, dental, urologic, oropharyngeal, thoracic, and dermal bleeding in clinical trials. Animal studies and case reports have demonstrated the pleiotropic effects of Ankaferd that may have therapeutic benefits, although there have been no controlled clinical trials evaluating this in humans. We hypothesized that specific factors in ABS contribute to this pleiotropism (Table 2). Additional studies investigating these individual components can broaden the therapeutic potential and applicability of ABS.
Hypothesized mechanisms of individual components’ contributions on Ankaferd’s pleiotropic effects.
Footnotes
Acknowledgements
All the mentioned work that was used in this review is referenced with the permission of the authors.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
