Abstract
Background:
This work attempts to investigate the standards of physical premises of pharmacies in terms of cleanliness, ventilation, protection from physical environment, spaciousness and storage facilities of two selected districts of Nepal: Kathmandu and Kaski.
Methods:
A cross-sectional, observational study of 100 randomly selected retail and wholesale pharmacies (55 from Kathmandu and 45 from Kaski) was conducted for their compliance to provisions of physical premises: cleanliness, ventilation, protection from physical environment, spaciousness and storage facilities. Preliminarily, scoring was done by converting 5-point Likert scale to a 100-point scale as follows: 1 = 0–20, 2 = 21–40, 3 = 41–60, 4 = 61–80 and 5 = 81–100. These scores were then reported as poor (score ⩽ 40), below average (score of 41–49), average (score of 50), above average (score of 51–60) and good (score ⩾ 61). Cronbach’s alpha for retails and wholesales obtained was .720 and .757, respectively.
Results:
In wholesales, protection from physical environment was least scored among other components, with the mean score 34.7 ± 8.3 for Kathmandu and 37.3 ± 13.3 for Kaski. Ventilation was scored 35.3 ± 6.3 for Kathmandu and 39.3 ± 11.6 for Kaski. All other components were obtained below average. However, in retails, the storage facilities were rated ‘poor’ with mean score 36.3 ± 13.9 for Kathmandu and 36.0 ± 11.9 for Kaski. Most other components were scored above average except for ventilation which was scored ‘good’. It was found that there was no significant difference between physical premises of the pharmacies of Kathmandu and Kaski in terms of all the indicators. The findings suggest that physical premise of many pharmacies was not found appropriate as provisioned in the good pharmacy practice requirements of Codes on Sales and Distribution of Drugs.
Conclusion:
Most pharmacies were found non-compliant to the aforementioned pharmacy premise components. Therefore, key partners should come up with commitment for increasing compliance to the legislative provisions of physical premises for ensuring quality and safety of medicines.
Introduction
Private health care is growing in low- and middle-income countries (LMIC) 1 and as such pharmacies are often the first and only source of health care and access to medicines. 2 These pharmacies are popular for their proximity, convenience, inexpensiveness, flexibility in operating hours and responsiveness. They are governed within the national practice standards stipulating the minimum physical premise/medicine protection facility for entry into the business and quality of care. However, the quality of pharmacy services may be poor due to lack of regulatory intervention and poor knowledge on pharmacy service as in other developing countries.2,3
In Nepal, two main categories of pharmacies can be identified; wholesales that sell medicines to retails and retails that sell medicines to consumers, both of them are operated by pharmacists, pharmacy assistants or professionals (vyabashi in Nepali language).4–6 The monitoring of pharmaceutical quality and the physical premises of these pharmacies is the responsibility of the Department of Drug Administration (DDA) 7 and is governed by the provisions of the Drugs Act, 1978 and Good Pharmacy Practice (GPP) requirements as mentioned in Codes on Sales and Distribution of Drugs, 2014. 8
Previous research has pointed out the availability of poor quality medicines in Nepal 9 and there are reports of non-compliances to regulatory provisions by pharmacies 10 as in other developing countries.3,11,12 Pharmacy practice standards with proper physical premises are key determinants of pharmacy services. However, there is lack of documented information and awareness on such practices in Nepal. Therefore, it is attempted to identify status of five physical premise components which are important for safety/quality of medicines and suggest technical and institutional reforms among the stakeholders.
Research problem
Pharmacies are not compliant to physical premise requirements of the legislative guidelines. The major problem is in five critical aspects: cleanliness, ventilation, protection from physical environment, spaciousness and storage facilities.
Operational definition of terms
Cleanliness. Pharmacy is considered clean if freed from dirt, dust, pests and rodents (cleaned properly and has dustbin).4,8
Ventilation. Pharmacy is well-ventilated if there is an adequate passage of ambient air required for aeration.
Protection from physical environment. Pharmacy is considered protected from physical environment if protected from direct sunlight or heat and humidity.
Spaciousness. Pharmacy is considered spacious if there is adequate space for holding shelves of medicines and proper movement for clients (for dispensing and counselling in retails while for storage, packing and clearance for wholesales).
Storage facilities. Pharmacy is considered having good storage facility if there is provision of adequate space for different groups of drugs and expired medicines to be stored at defined temperature range.
Research questions
What is the score/level of cleanliness, ventilation, protection from physical environment, spaciousness and storage facilities of the selected pharmacies?
Objectives
To score and rate level of cleanliness, ventilation, protection from physical environment, spaciousness and storage facilities taking consideration of the operational definitions.
Hypothesis
Pharmacies are scored maximum with ‘good’ score if they are clean, ventilated, protected from physical environment, spacious and with good storage facilities.
Methods
A cross-sectional observational study of pharmacies of Kathmandu and Kaski were conducted from October 2015 to March 2016. Kathmandu and Kaski covered 22.7% of total registered pharmacies of Nepal: 32.5% of the total registered wholesales and 21.1% of the total registered retails. 13 The total registered allopathic retails in these districts was 25.2% and allopathic wholesales was 33.7%. 13
Prior to the start of data collection, 125 pharmacies were randomly selected from the printed list of registered pharmacies obtained from Pharmacy Registration Section of DDA and verbal consent was taken from each of the pharmacies after explaining the purpose and importance of the study. The pharmacies were selected without considering their size, the number of people, the location and the category of people working in them. Participation in the study was on voluntary basis. All types of hospital pharmacies, veterinary pharmacies, ayurvedic pharmacies and homeopathic pharmacies were excluded in this study.
Study design
The semi-structured questionnaire was developed having taken into consideration the physical premise requirements that have a crucial bearing on quality and safety and circulated among pre-selected people (three pharmacists, three drug regulators and three professionals) working in the field. They assessed pharmacy practice indicators of physical premise mentioned in the official standard provisions for pharmacy practices4,8 and suggested five study components: cleanliness, ventilation, protection from physical environment, spaciousness and storage facilities. 8
Finally, a data-collection sheet was prepared through consensus (Table 1) and pre-tested on a sample of 15 pharmacies; the data of which were not used in final analysis. The final collection of data was done by the principal investigator on location.
Data collection sheet.
Name of the pharmacy: DDA No.
Location: Authorized personnel:
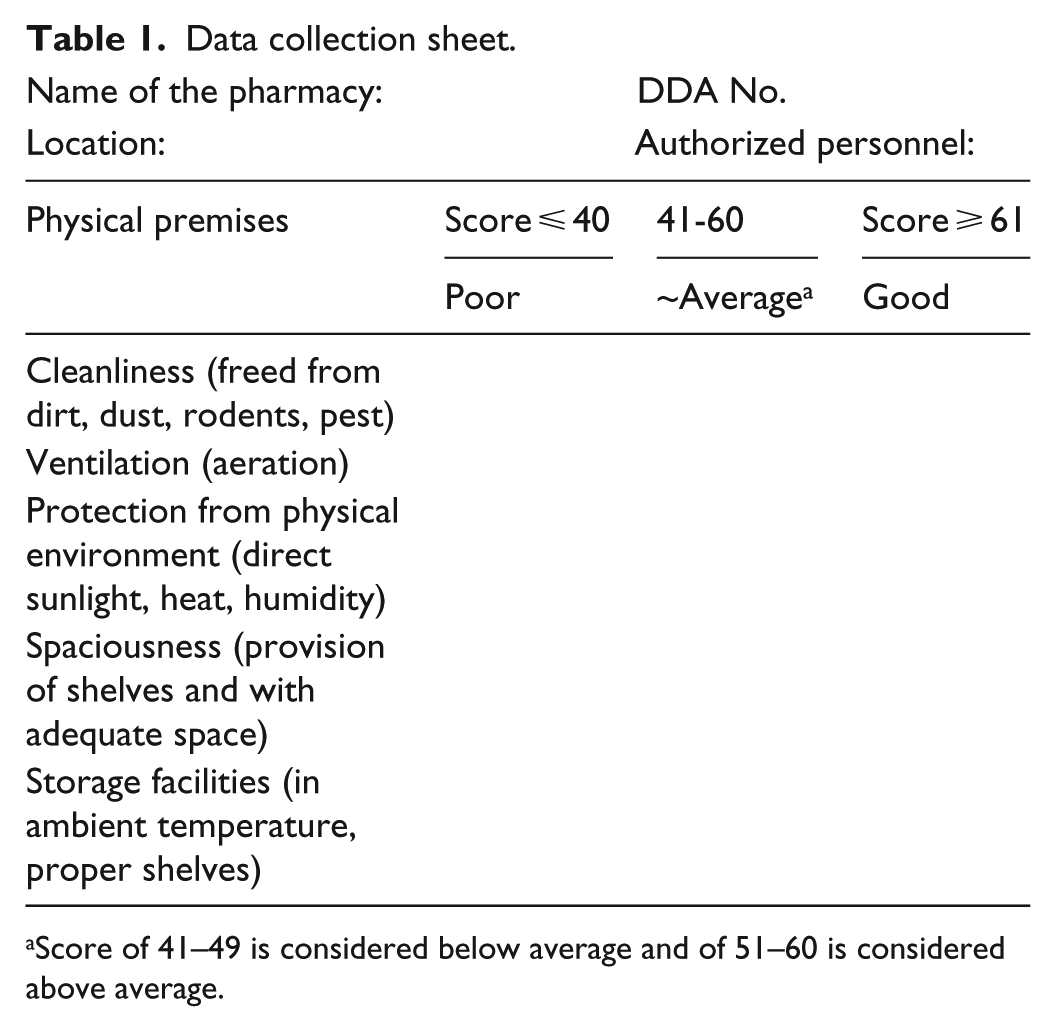
Score of 41–49 is considered below average and of 51–60 is considered above average.
We scored each scale by first converting the 5-point Likert scale to a 100-point scale as follows: 1 = 0–20, 2 = 21–40, 3 = 41–60, 4 = 61–80 and 5 = 81–100. 14 Responses to each item in a scale were summed and then divided by the number of items in that scale to create a scale score that ranged from 0 to 100. For clear understanding of physical premise standards, these scores were then reported as poor (score ⩽ 40), below average (score of 41–49), average (score of 50), above average (score of 51–60) and good (score ⩾ 61).
Statistical analysis
For validation of questionnaires, internal consistency, a type of reliability of items within each of the two groups, wholesales and retails, was measured using Cronbach’s alpha. 15 The mean values and standard deviations of scale scores were calculated for the five types of issues. Paired sample test was used to evaluate significant difference in the percentage scores among the different issues. p value <.05 was considered to be significant. All hypothesis tests were based on a two-sided alpha of .05. 15 The spidograph was designed such that all five areas are given equal weight with up to hundred as maximum score, independent of the number of questions contributing to the assessment. All data analyses were conducted using SPSS version 17 (SPSS Inc., Chicago, IL, USA).
Results
Survey responses
Based on the proportionate number of pharmacies of Kathmandu and Kaski district, 70 pharmacies from Kathmandu and 55 from Kaski were randomly selected for study. 14 Of them, 100 valid questionnaires were returned (25 pharmacies withdrew from study), representing 88.3% (40/48) retails of Kathmandu, 68.2% (15/22) wholesales of Kathmandu, and 78.9% (30/38) retails of Kaski and 88.2% (15/17) wholesales of Kaski. The overall response rate was 80%.
Cronbach’s alpha of the selected physical premise components (cleanliness, ventilation, protection from physical environment, spaciousness and storage facilities) for wholesales was .757 (.666 for Kathmandu and .885 for Kaski) and that for retails was .720 (.859 for Kathmandu and .844 for Kaski) indicating good consistency of the selected components.
Tables 2 and 3 demonstrate physical premises and the respective modified Likert test scoring. The overall physical facilities’ mean score was scored 47.8 ± 10.4 (42.3 ± 1.4 for wholesales and 54.4 ± 16.0 for retails). Therefore, score for physical facilities was 42.3 for wholesales below what is required by the regulations mean, while retails scoredslight above average (54.4). From the study, protection of wholesales from physical environment was found poor with the lowest mean score 34.7 ± 8.3 for Kathmandu and 37.3 ± 13.3 for Kaski (Table 2). Similarly, storage facilities received least scoring in wholesales and retails of both districts (Tables 2 and 3).
Scoring of physical premises of wholesales.
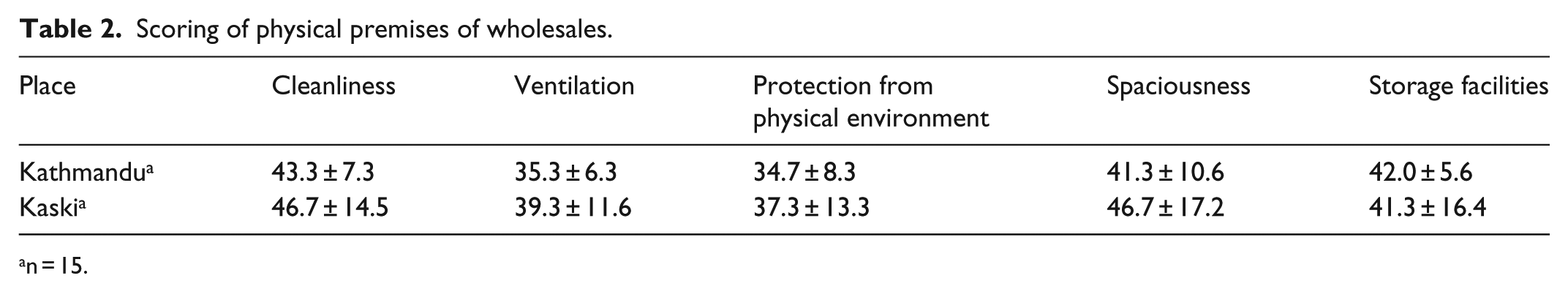
n = 15.
Scoring of physical premises of retails.
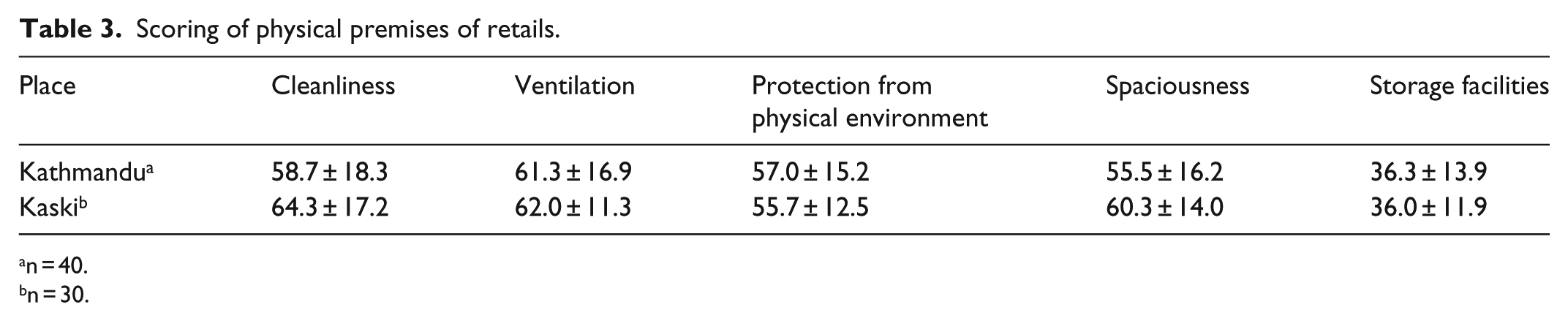
n = 40.
n = 30.
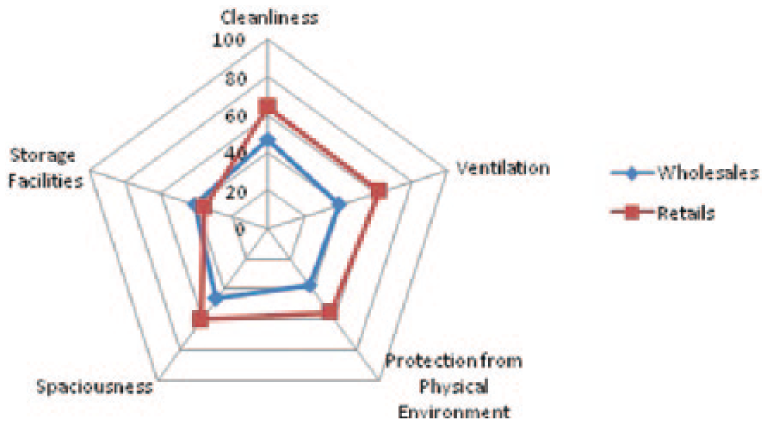
Figures 1 and 2 show that the retails of both districts were better than wholesales except for storage facilities.

Spidograph depicting pharmacy practice assessment score of five components in Kathmandu.
Spidograph depicting pharmacy practice assessment score of five components in Pokhara.
As shown, there was no significant difference between the physical premises of pharmacies (wholesales and retails) of both districts (Tables 4 and 5).
Differences in physical premises of wholesales of two districts among different components.
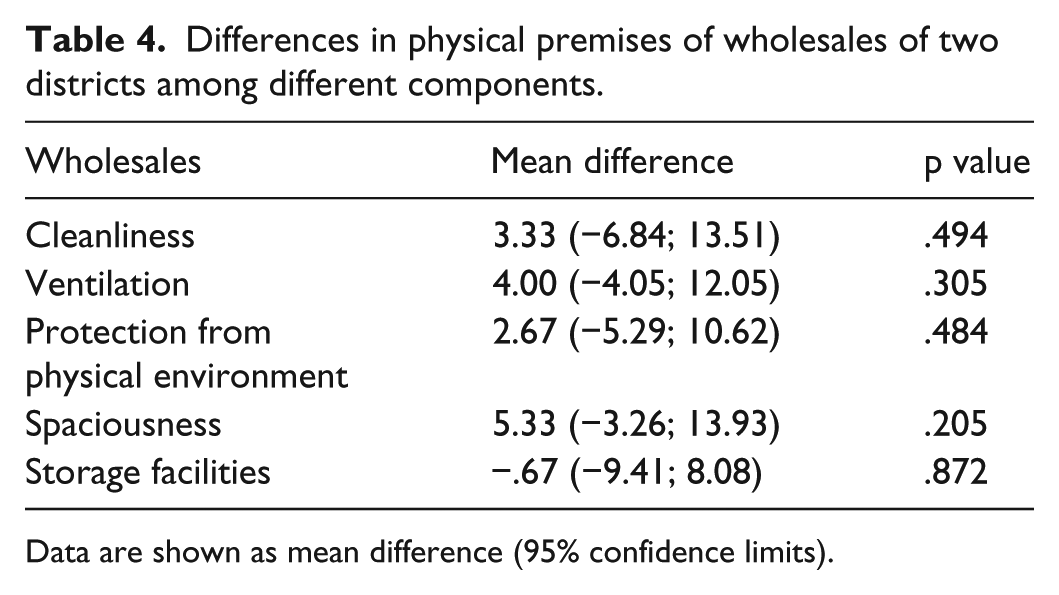
Data are shown as mean difference (95% confidence limits).
Differences in physical premises of retails of two districts among different components.

Data are shown as mean difference (95% confidence limits).
Discussion
This study highlights the overall physical premises of pharmacies that are quality and safety domains of medicines available in Nepalese market. In this study, the physical premises of majority of wholesales were rated below average or poor, while those of retails were comparatively provisioned better (Tables 2 and 3). However, storage facilities of pharmacies were rated below average or poor in both the districts (Tables 2 and 3). Similar substandard physical environment and storage practice among other non-compliances were reported in Srilanka and Pakistan.11,12 Retails in Kathmandu were rated slightly above average in cleanliness, protection from physical environment and spaciousness although was rated good for ventilation. Pharmacies of Kaski were observed better in terms of physical premises than those of Kathmandu (Tables 2 and 3, Figures 1 and 2).
Inappropriate storage of medicines by professionals in developing countries, 11 high incidence of low-quality drugs3,9 and non-compliance to pharmacy practice standards warrants the need of stringent drug policy programme in developing countries.3,10 Furthermore, poor knowledge and practice,16,17 lack of funding, lack of recognition of pharmacists, 18 undermining of pharmacy practice trainings may be subsidiary factors for such rating. Continuing Pharmacy Education (CPE), indicator-based assessment of pharmacies and strong regulatory intervention could be an alternative for improving pharmacy practice standards.
Therefore, the development of pharmacy practice indicators is important for improving standards of premise, including quality of care; and stringent regulatory practice and follow-up inspections are advisable. Furthermore, with strong financial support and proper training of employees, above issues can be minimized.
The limitation of the study is that the results may be biased as they were drawn from study conducted in selected pharmacies of two districts only. Since the study design is focused on surveying the physical premise, constraints of identifying dependent and independent variables exist. Furthermore, research is also needed to develop and validate additional indicators, especially those measuring physical premise and quality of services.
Conclusion
Although the physical premise of retails was better than wholesales of Kathmandu and Kaski districts, many pharmacies were not provisioned as mentioned in the legislative requirements. Increasing advocacy programme and bringing stringent regulatory action may increase their compliance to the national requirements. Therefore, this study is expected to improve pharmacy premise standards that will lead to improved quality of services by the pharmacies.
Footnotes
Acknowledgements
The authors would like to thank Department of Drug Administration (DDA), Kathmandu. B.K.P. carried out the study and applied the statistics, and B.K.P. and I.I. prepared the final article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethics approval
Ethical approval for this study was waived as the study does not involve data of patients or use of human subjects. Hence, Department of Drug Administration (DDA) do not provide ethical approval for such study.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
Informed consent
Verbal informed consent was obtained from all subjects before the study.
