Abstract
Regular and robust audit of conscious sedation practice is key to the delivery of safe and effective patient care in all clinical settings. Analysis of outcomes will help to highlight any necessary modifications to procedures and techniques. The Intercollegiate Advisory Committee for Sedation in Dentistry (IACSD) states that conscious sedation procedures must be the subject of robust and regular audit in which every member within the team takes part.1 This completed audit cycle highlighted the efficacy of intravenous (IV) midazolam in anxious patients undergoing oral surgical procedures and the suitability for its use in the primary care setting. No patient safety concerns or complications were recorded in this audit and midazolam was administered within the recommended dose range. By the conclusion of two audit cycles, all clinicians providing intravenous sedation within the service were aware of the missing elements in sedation record-keeping and were able to complete accurate contemporaneous records before, during and after conscious sedation, ensuring the continuous delivery of high-quality, safe, and patient-centred care.
Learning Objectives
To identify patient referrals that can be accepted for sedation treatment within primary care based on an assessment of ASA status
To identify the mean dose of midazolam administered to patients undergoing treatment under IV sedation in a primary care setting
To appreciate the importance of contemporaneous clinical record keeping in IV sedation
Introduction
The 2020 Report of the Intercollegiate Advisory Committee for Sedation in Dentistry (IACSD) states that, “Conscious sedation procedures must be the subject of robust and regular audit”, and that, “The focus must be an ongoing review of procedures and processes with analysis of outcomes and modifications made to procedures and techniques as necessary”. 1
Titration of midazolam to achieve an optimum level of sedation is an important aspect of the sedation technique in terms of achieving the best possible outcome for patients while maintaining patient safety. A number of factors including age, physical status, co-morbidities, and medications can affect a patient’s response to midazolam sedation and, as such, careful titration is essential to ensure the appropriate endpoint is met and over-sedation is avoided.2,3,4 The Scottish Dental Clinical Effectiveness Programme (SDCEP) Conscious Sedation in Dentistry guidance defines conscious sedation as:
“a technique in which the use of a drug or drugs produces a state of depression of the central nervous system enabling treatment to be carried out, but during which verbal contact with the patient is maintained throughout the period of sedation. The drugs and techniques used to provide conscious sedation for dental treatment should carry a margin of safety wide enough to render loss of consciousness unlikely.”
5
The appropriate dose of sedation drugs should therefore aim to achieve this state and avoid oversedation which could lead to serious adverse events including, in rare circumstances, death.
The main side-effect of conscious sedation with midazolam is hypoxaemia as a result of respiratory depression. 2 Oxygen saturations dropping below what is normal for the patient can be a sign that they are becoming over-sedated. This could be due to incorrect titration or bolus injection. 6 In some cases, supplemental oxygen is required to improve the patient’s oxygen saturation. 7
The intravenous (IV) sedation agents that have been advocated for use in dentistry are midazolam, propofol, ketamine, and fentanyl. 8 Remimazolam is a new benzodiazepine that has recently been found to provide good outcomes in conscious sedation in dentistry due to its ultrashort-acting properties. 9 Remimazolam, as its name suggests, combines the properties of midazolam and remifentanil and it acts on the gamma aminobutyric acid (GABA) receptors, but its ester-linked side chain to the diazepine ring makes it an ultrashort-acting sedation drug that is metabolised rapidly to inactive metabolites by liver tissue esterases. 9 Also, the hepatic drug-metabolising enzyme cytochrome P450 (CYP) is not involved in the metabolism, similar to that of remifentanil. 10 These combined properties therefore make remimazolam a drug of fast onset, high clearance, short half-life, and few side-effects. 9 Due to remimazolam’s novelty, more studies are warranted to further evaluate and establish its safety in dental sedation.
Additional IV sedation drugs are utilised by dental sedationists in secondary care settings when midazolam alone does not produce the desired sedative effect in a minority of patients. 11 The use of multiple sedation drugs in anaesthetic practice is common but dental sedationists wishing to utilise multiple drugs for sedation must complete extensive enhanced, advanced sedation training and are required to work in a specific clinical environment suited to the technique.1,4,5 Concomitant use of multiple IV sedation drugs can result in unpredictable or detrimental outcomes if used inappropriately due to less predictable effects and narrower therapeutic indices, resulting in reduced margins of safety.1,12
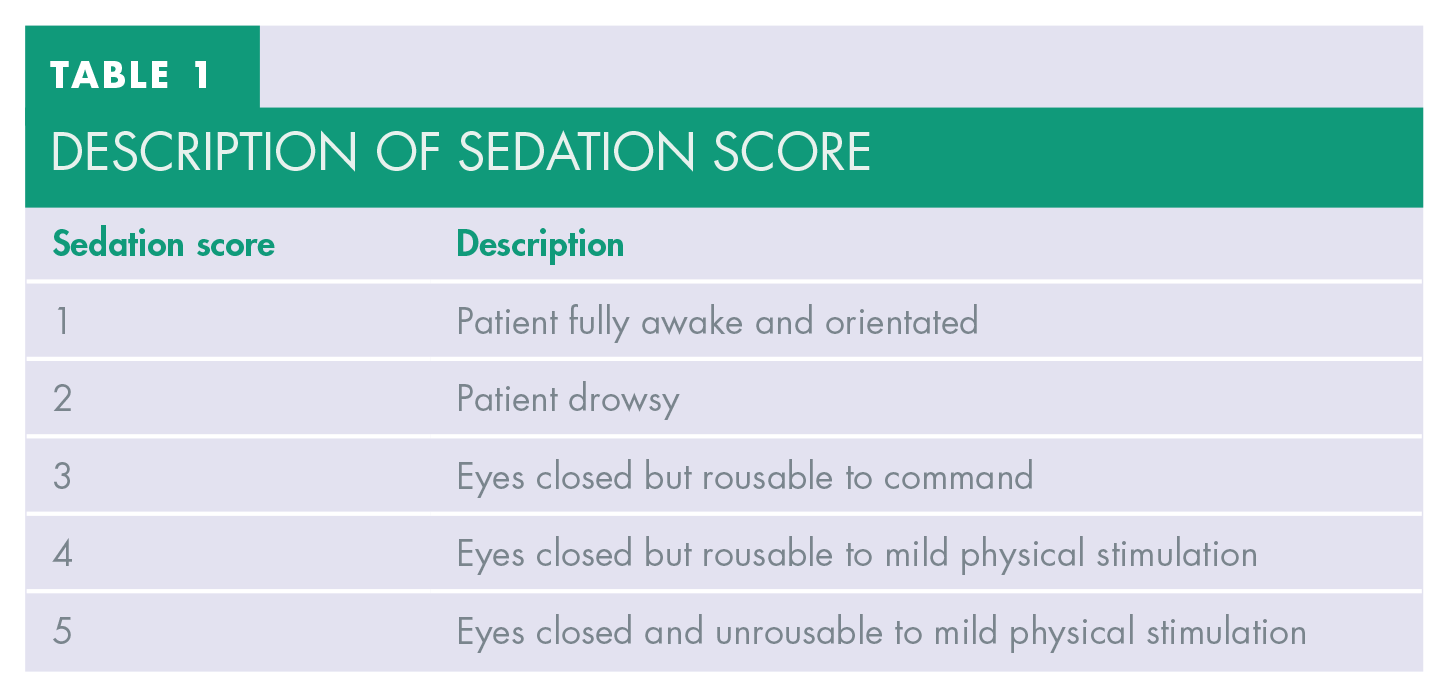
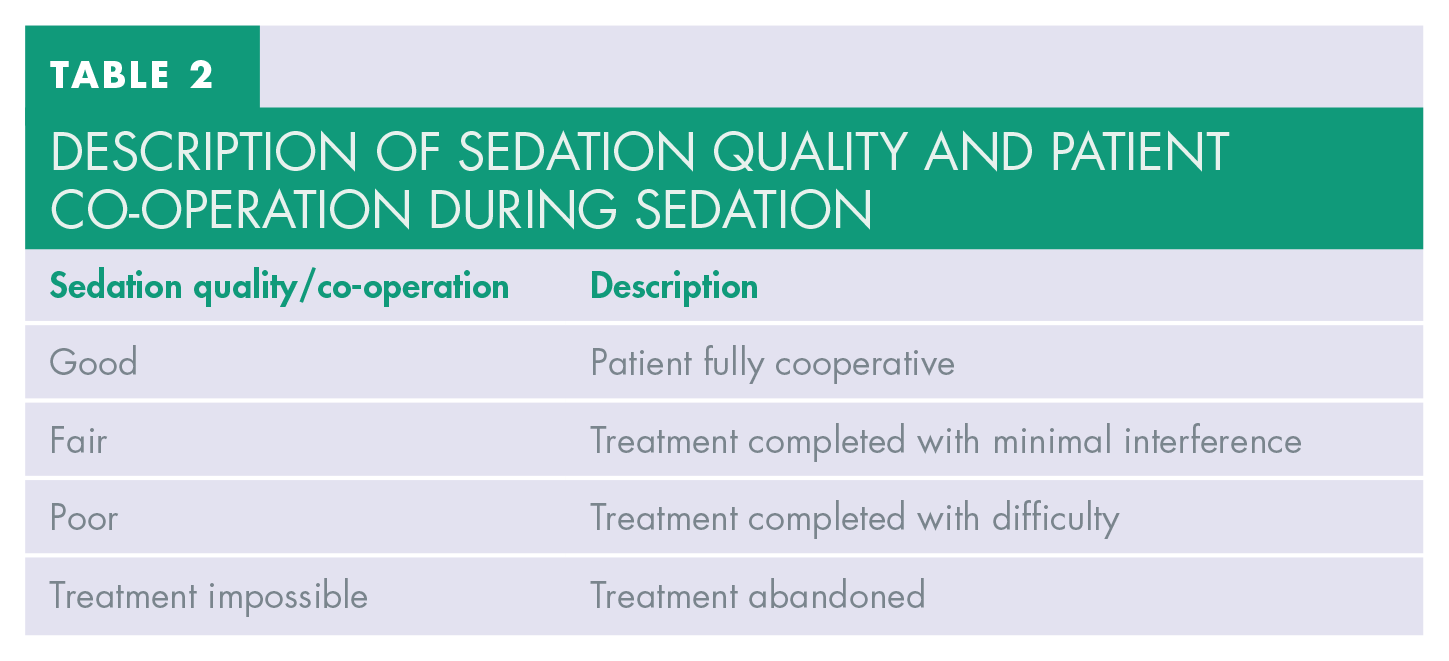
Midazolam remains the most common IV sedation agents for adult dental procedures in the UK. 13 IV midazolam is the drug of choice in the primary care setting due to its wide margin of safety, excellent sedative properties and minimal cardiovascular and respiratory side-effects, rendering it the safest, most fit-for-purpose and effective IV sedation drug for dental patients.6,14,15 All sedative drugs produce a state of depression of the central nervous system. Both IACSD and SDCEP guidelines state that the level of consciousness of the sedated patient should be closely monitored perioperatively.1,5 The SDCEP guideline also suggests that “the patient’s reaction, sedation score and success of sedation” should be recorded and maintained in the patient’s clinical records as evidence of such monitoring (see Tables 1 and 2). 5
Description of sedation score
DESCRIPTION OF SEDATION QUALITY and patient co-operation during sedation
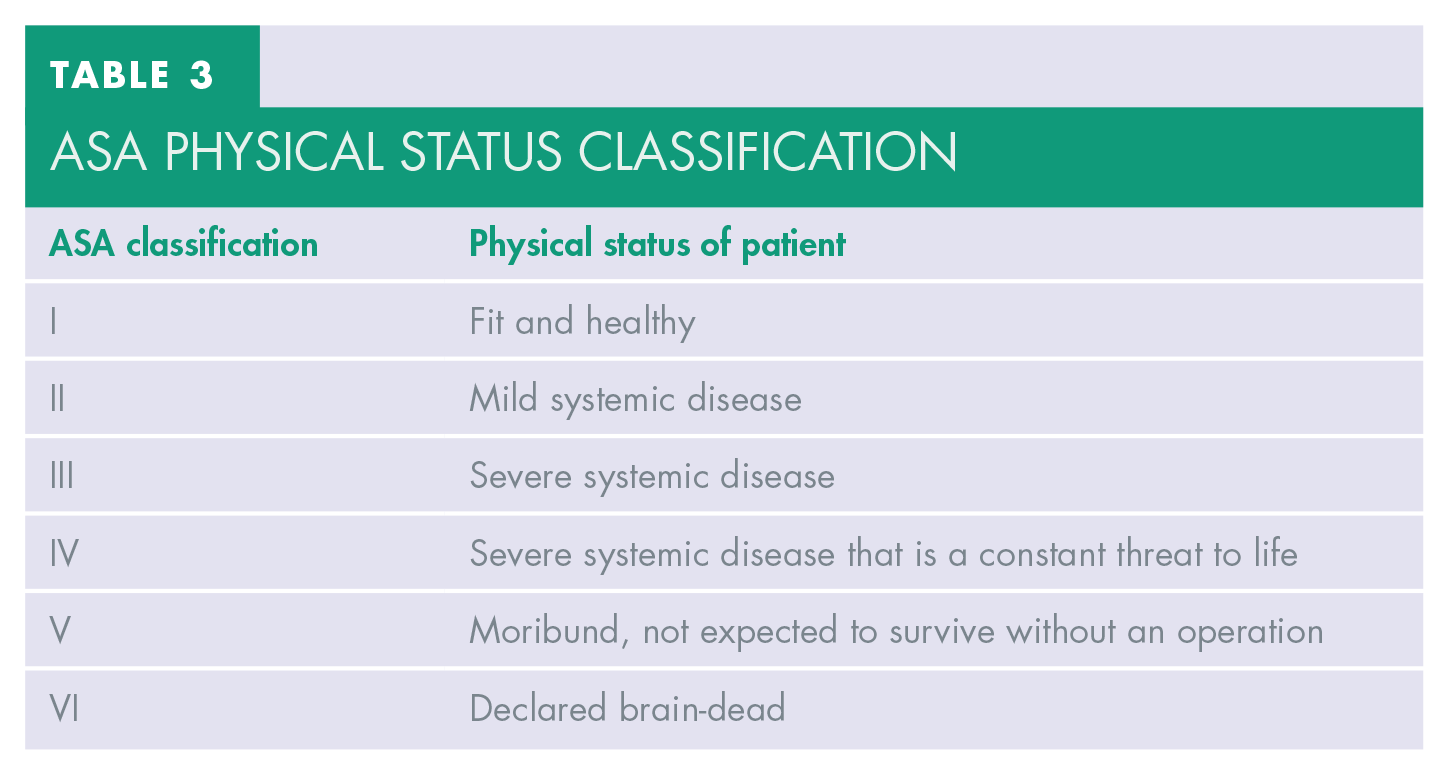
Prior to provision of dental sedation, all patients were assessed at a separate appointment. At this visit, a comprehensive dental, social, and medical history was obtained. The patient’s vital signs including blood pressure, heart rate, oxygen saturation, height, weight, and body mass index (BMI) were also recorded. As part of this assessment process, an American Society of Anaesthesiologists (ASA) classification was assigned to the patient to aid in determining the most appropriate sedation management modality (Table 3).5,16 It is recommended that only patients with ASA classification I and II are suitable for conscious sedation treatment in the primary care setting.2,4 Patients with ASA III or above requiring IV sedation should be referred on to a secondary care unit where appropriate facilities exist for their management. 4
Asa physical status classification
In view of the importance of collation of vital information before, during, and after the provision of IV sedation, this audit was undertaken to assess the dose of midazolam used for each patient and the standard of IV sedation record-keeping, ensuring compliance with sedation policies and enhancement of patient safety.
Methods
Sample/population
Adult patients (16 years and over) referred by their general dental practitioner to the primary care oral surgery service for treatment under IV sedation between September and November 2019 and between January and March 2020 were included in this audit. The patients whose treatment was completed had their records stored in the dental software Carestream R4+® (Carestream Dental, Atlanta, USA) database. Patients who had midazolam IV sedation were included. Patients with complex medical histories (ASA III or above) that were referred for treatment under IV sedation were triaged, assessed and then referred on to a secondary care setting more appropriately suited to their medical status. 4
Data collection
The digital sedation records of 22 patients were randomly selected and retrospectively assessed between September and November 2019 (first cycle), a further 22 randomly selected sedation records were assessed between January and March 2020 (second cycle). The details of the sedation records that were assessed included:
patient details
gender
age
record of ASA classification at pre-assessment
dose of midazolam used for sedation
whether midazolam was titrated to achieve a predetermined endpoint
the use of flumazenil as reversal agent
record of sedation score
record of sedation quality/patient’s co-operation
whether supplemental oxygen was required
whether any adverse events/complications arose throughout sedation treatment
The data derived from the clinical records were entered into a spreadsheet (Microsoft Excel) for further analysis.
Standards
The standards of the audit were set based on the IACSD and SDCEP guidance.1,5 The standards were:
The dose of midazolam sedation given should never render the patient unconscious or unresponsive
All patients should have midazolam titrated to a predetermined endpoint. A bolus or fixed dose technique should not be used. The titrated dose is recorded in the patient notes
All patient records should include a record of any sedation-related peri- or post-operative significant events or complications
All patient records should include a record of the sedation score and sedation quality
All patient records should include ASA classification obtained at the pre-operative assessment appointment
Audit compliance parameters
The parameters were set to be compliant to the set standards when compliance was above 90%; whereas compliance below 90% were considered non-compliant to the set standards within this audit study.
Clinical audit action plan
An action plan was drawn up after the first audit cycle to ensure changes could be made or implemented to improve the compliance to set standards. The key actions were:
The authors disseminated findings of first audit cycle to all oral surgery clinicians via email, arranged a group meeting and discussed the appropriate actions and changes required to improve compliancy in December 2019
The authors conducted a ‘lunch and learn’ meeting to stress and reinforce the importance of ‘ASA status’ recording at pre-operative appointments to all sedation clinicians to ensure the case load suitability in primary care setting in December 2019
The authors standardised the sedation proforma by the addition of ‘sedation score’ to the existing paperwork in January 2020, after analysing the results obtained in first audit cycle
Results
First audit cycle
The first audit cycle was carried out between September and November 2019, the summary of the audit cycle is shown in Table 4.
Summary of first audit cycle
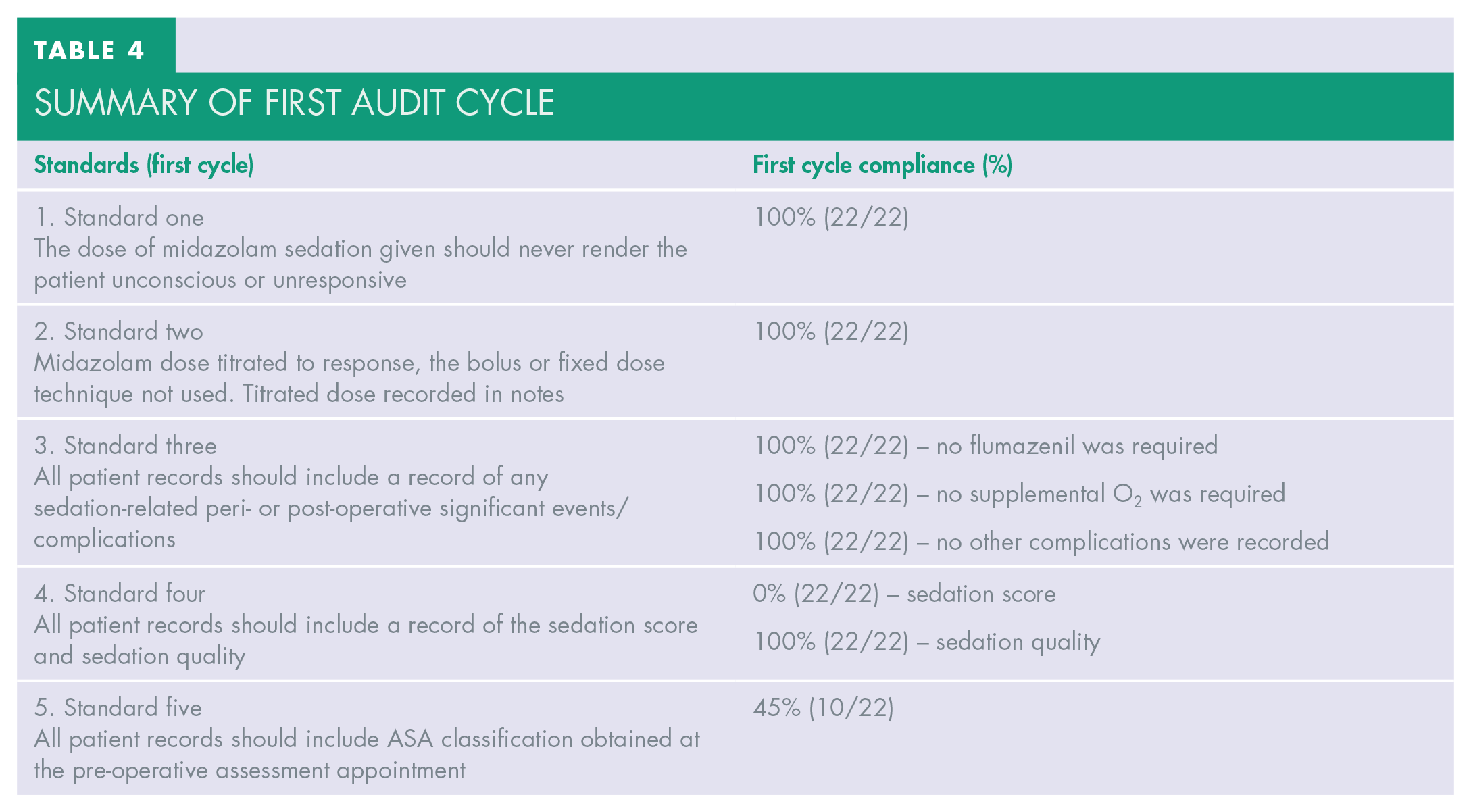
In the first audit cycle, the female to male ratio of patients receiving treatment under IV midazolam sedation was 8:3. The doses of midazolam titrated were all within the ‘standard’ limit. All patients remained responsive. Of the 22 patients, the most frequent titrated dose to achieve the predetermined sedation endpoint was 3mg (59%, 13 out of 22 patients), whereas 4mg and 5mg were jointly the second most frequent dose titrated, with equal numbers of patients in this first audit cycle (18%, four out of 22 patients each). Only one patient (5%) achieved the predetermined sedation endpoint after a dose of 2mg (Figure 1). The mean dose to achieve sedation endpoint was 3.5mg.
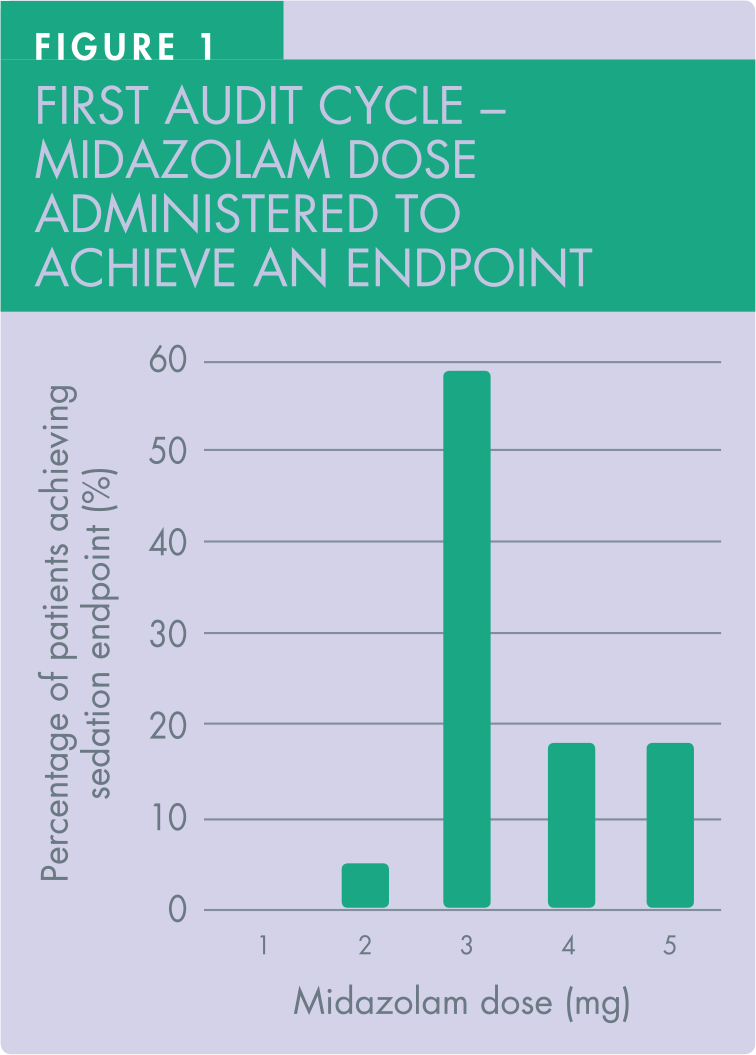
All patients received midazolam titrated to a predetermined sedation response. No bolus or fixed dose technique was employed as evidenced by detailed, contemporaneous record-keeping in 22 patients’ case notes of the first audit cycle.
There was no recorded evidence of sedation-related peri- or post-operative significant events or complications. Flumazenil was not used, and no supplemental oxygen was required in any of the 22 patients.
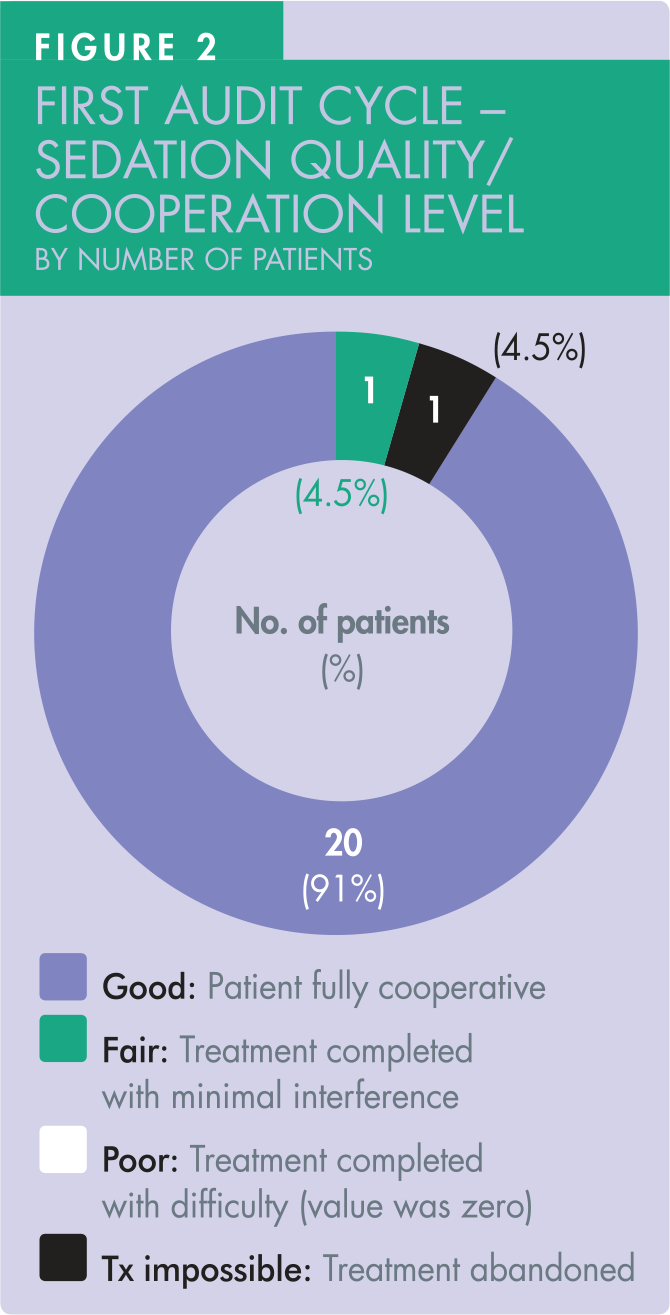
In all 22 patients, no sedation score was recorded. However, both the quality of sedation and the level of patient co-operation was recorded in those 22 patients, achieving 100% compliance (Figure 2). Twenty patients (91%) achieved ‘good’ sedation quality and level of co-operation. Of the 22 patients, one patient achieved ‘fair’ sedation quality, but the treatment was successfully completed. Another patient’s treatment was not completed because of poor co-operation, resulting in an onward referral to secondary care.
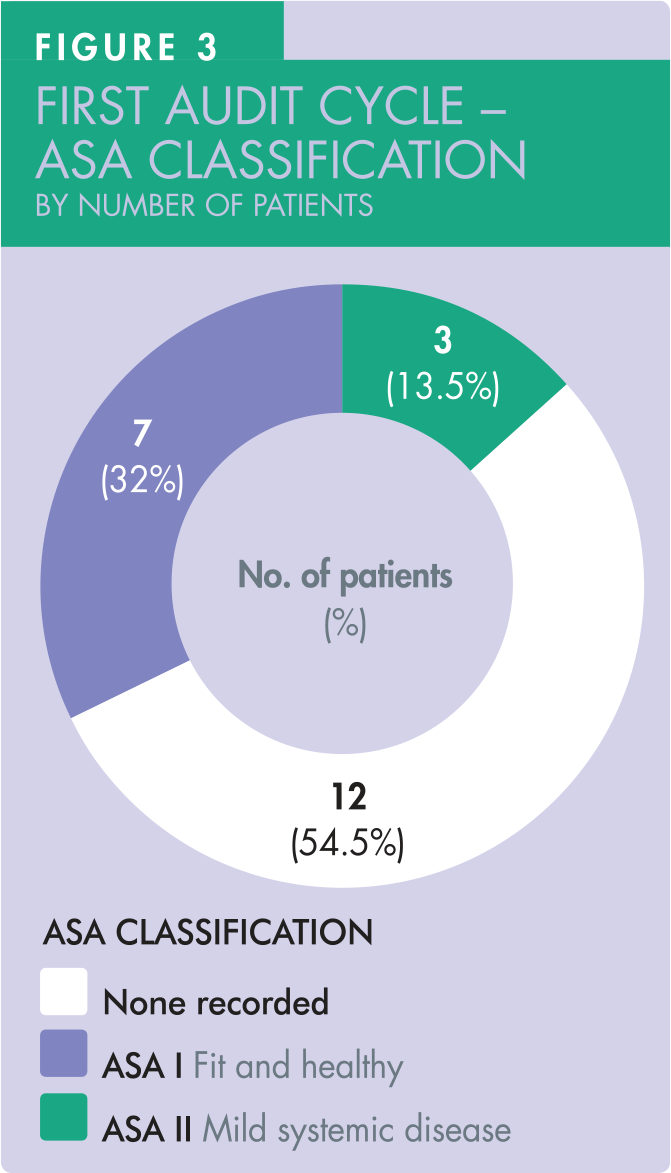
Only 45.5% of the patients (10 out of 22) had their ASA status recorded in their clinical records. Of these 10 patients, seven were noted as ASA I, three patients as ASA II (Figure 3).
Second audit cycle
This was carried out between September and November 2020; the summary of the audit cycle is shown in Table 5.
Summary of second audit cycle
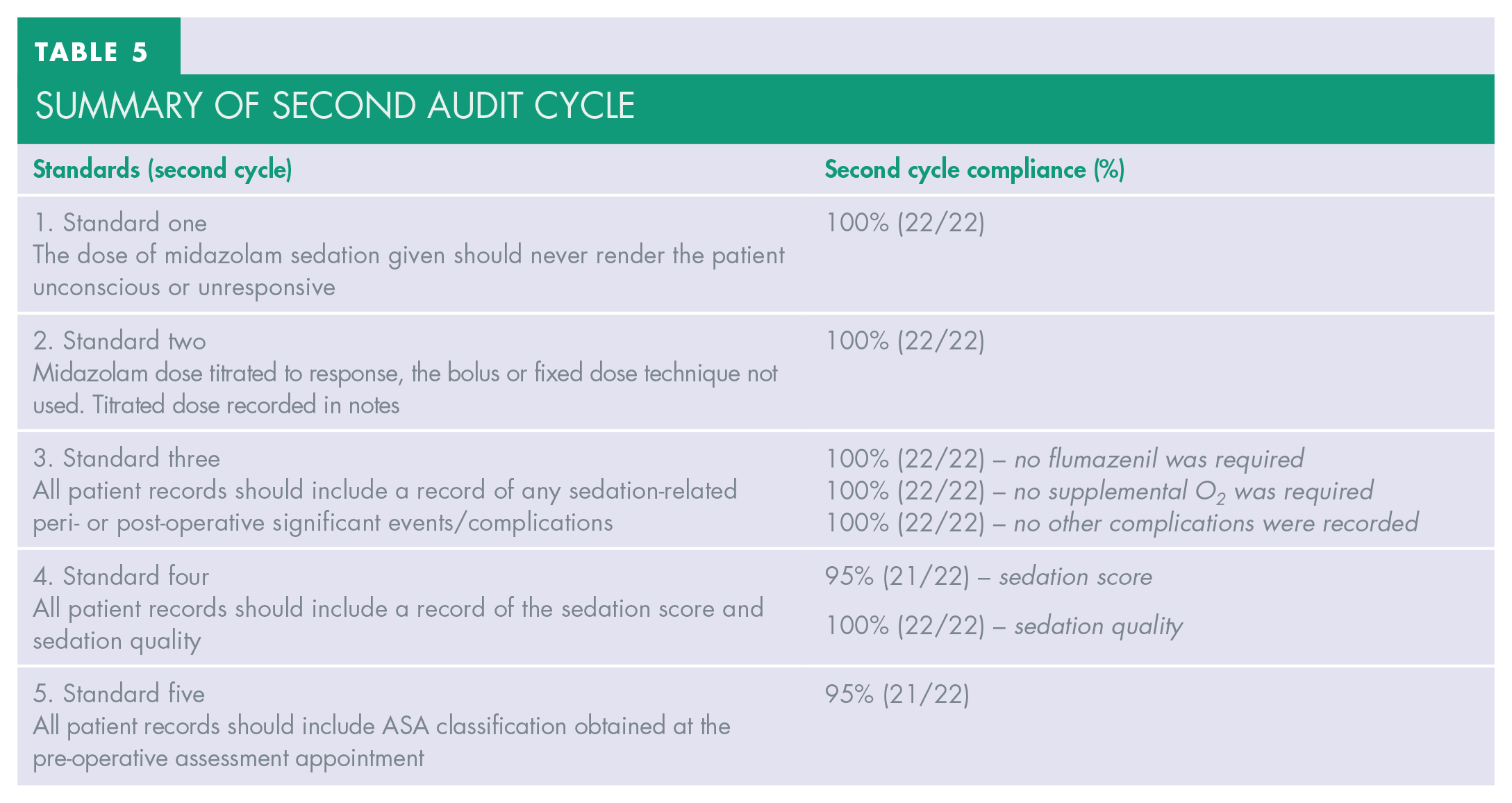
In the second audit cycle, the female to male ratio in receiving treatment under IV midazolam sedation was 15:7. The doses of midazolam administered were, without exception, within the ‘standard’ limit and no patient became unresponsive. Of the 22 patients, the most commonly administered dose to achieve the pre-determined sedation endpoint was 4mg (45.5%, 10 out of 22 patients).
The second most administered dose was 3mg (27.2%, six out of 22 patients). Of the 22 patients in the cycle, four patients achieved endpoint with 5mg midazolam (18.2%) and two patients achieved endpoint with 6mg (9.1%) (Figure 4). The mean dose of midazolam titrated was 4mg.
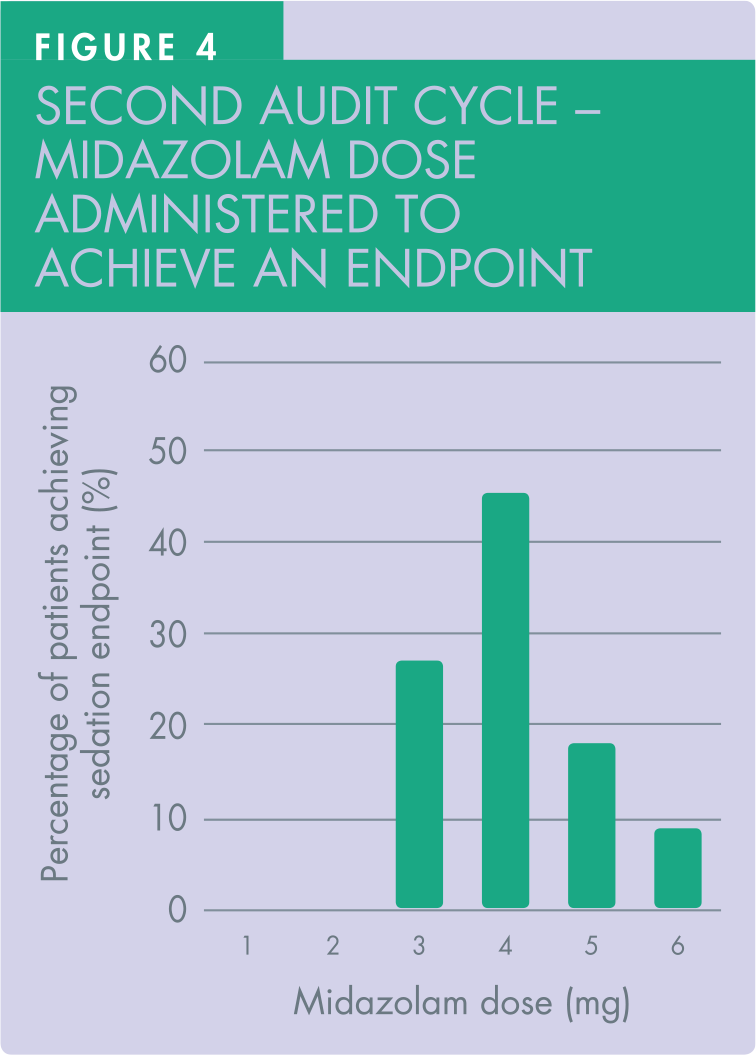
All patients received the midazolam titrated to response. This was evidenced by detailed, contemporaneous record-keeping in the 22 patients’ digital case notes in the second audit cycle.
There was no evidence of any sedation-related peri- or post-operative significant events or complications recorded. Flumazenil was not required, and no supplemental oxygen was required in any of the 22 patients.
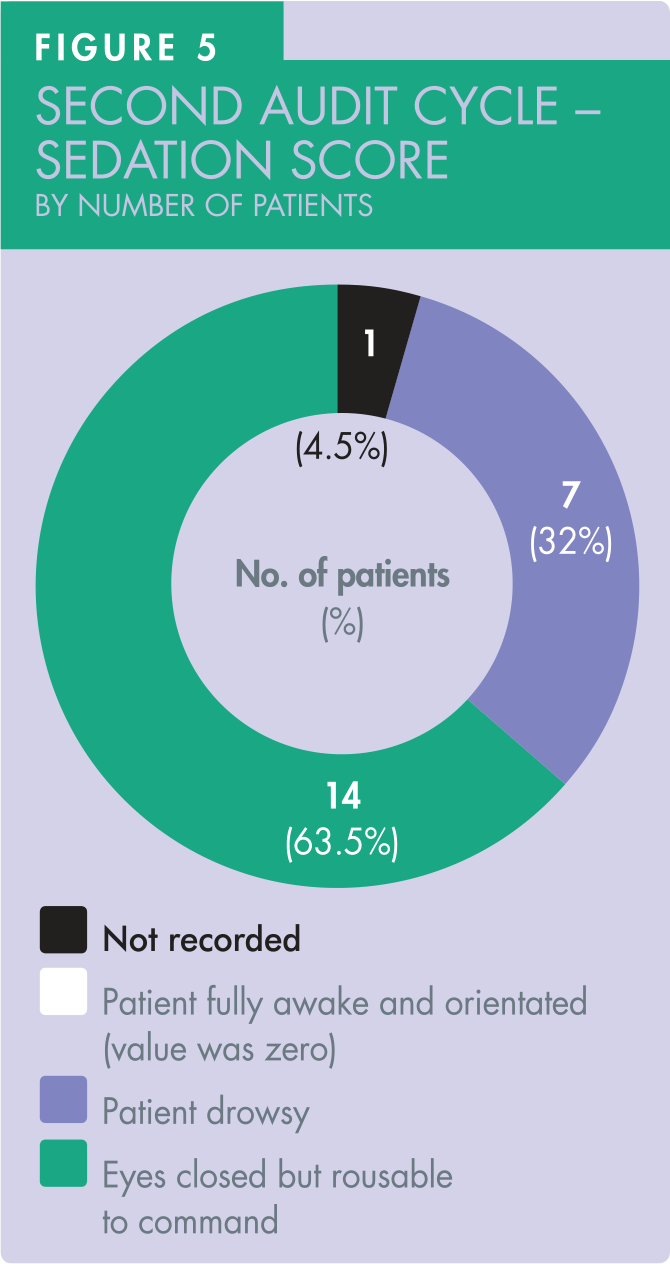
There were 21 patients with a recorded sedation score, achieving 95% compliance against the set standard. None of the patients scored ‘1’ in this second audit cycle. Seven patients (32%) scored ‘2’. The most common sedation score recorded was ‘3’ in 14 of the 22 patients (63.5%) (Figure 5).
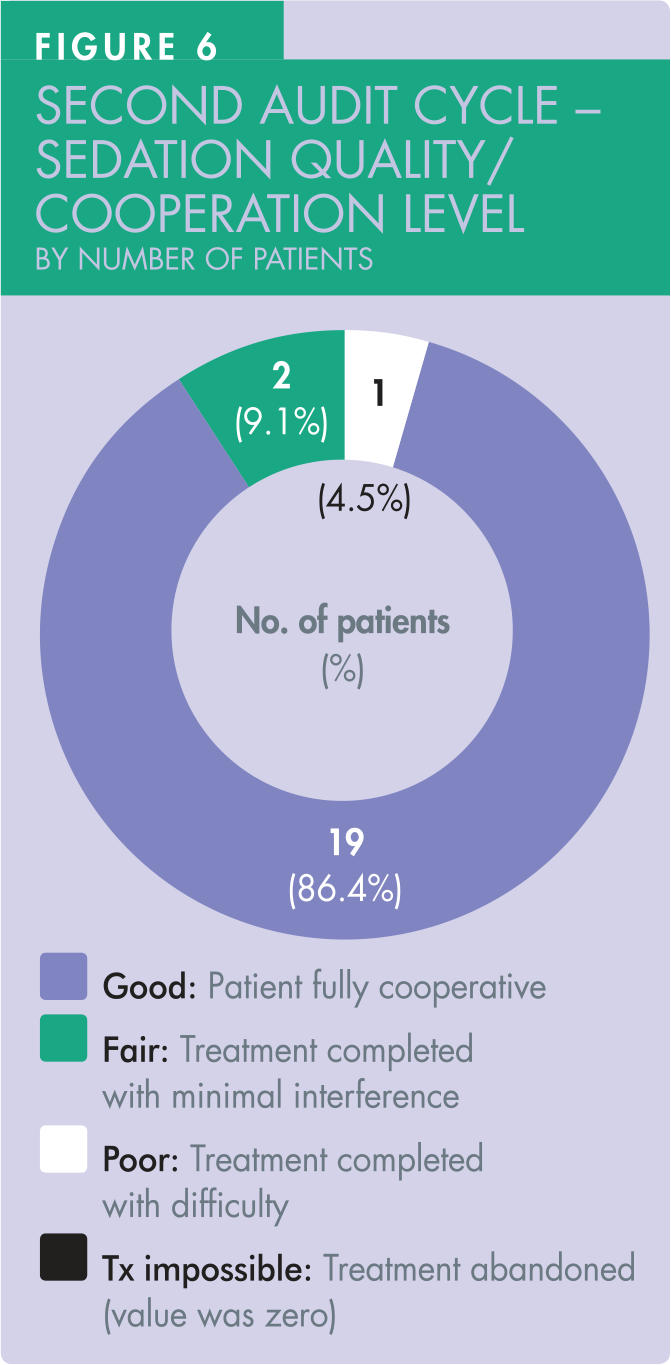
The sedation quality and the level of patient co-operation were recorded in 22 patients, achieving 100% compliance (Figure 6). Nineteen patients (86.4%) achieved a ‘good’ sedation quality/level of co-operation. Of the total 22 patients, two patients (9.1%) achieved ‘fair’ sedation quality, one patient (4.5%) achieved ‘poor’ sedation quality. All treatments were successfully completed under sedation.
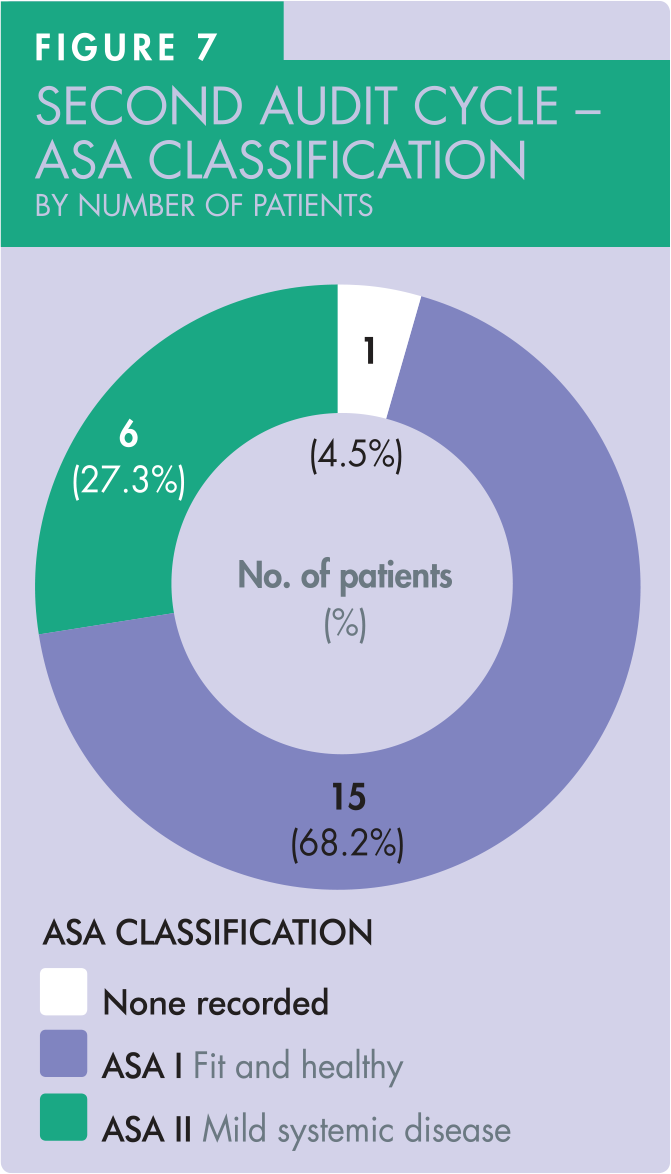
In the second audit cycle, 21 ASA assessments were recorded in the clinical records, achieving 95% compliance against the set standard. Of these, 15 patients with ASA I status and 6 with ASA II status were treated. One patient did not have ASA status recorded in clinical record (Figure 7).
Discussion
This audit was conducted to identify the dose of midazolam administered to patients, assess compliance in midazolam IV sedation practice and the standard of IV sedation record-keeping against set parameters in a primary care sedation and oral surgery service. The two cycles of audit confirmed that there were no safety concerns within the service. 16
All midazolam doses administered intravenously in both audit cycles were within the “recommended dose limit” with no outliers. Although there are no identified ‘safe’ dose limits for IV midazolam, the British National Formulary suggests a maximum dose of 7.5mg per course of treatment in adults. 17 Careful titration of midazolam dose to the individual patient’s response in achieving a predetermined endpoint is crucial in the prevention of oversedation.2,3 Both cycles of the audit revealed the most common dose of midazolam administered was between 3mg and 4mg, to achieve the desired sedative, anxiolytic, and amnesic effects. Interestingly, this reflects findings highlighted in a study which postulated 3mg of midazolam administered intravenously as the optimal dose for maximising patient experience and the dentist’s operative satisfaction. 18 This study also demonstrated the amnesic effect and the absence of unintentional patient movement despite the relatively light level of sedation from 3mg of midazolam. 18
The first audit cycle revealed that a “sedation score” was not recorded for every patient treated under IV sedation. The failure to record a sedation score in the first audit cycle could have been due to the design of the original proforma, which was subsequently rectified to include “sedation score”. Additionally, all clinicians were reminded to ensure that a sedation score was recorded for each patient at the completion of IV sedation treatment through meetings and emails. The second audit cycle revealed a consistent record of sedation score, with the exception of one case only.
The majority of the sedation scores recorded were ‘3’: eyes closed but rousable to command. This could be because many of the patients referred for surgical treatment under IV sedation receive their routine dental care under local anaesthetic but are too anxious to have surgical procedures carried out this way. Midazolam IV sedation remains the most appropriate treatment modality to overcome the anxiety in these patients, keeping them calm during unpleasant surgical procedures.4,19 A sedation score of ‘3’ demonstrates the anxiolytic effect of midazolam in daunting and potentially distressing oral surgical procedures 13,15
Sedation quality and the level of patient co-operation achieved were generally good with midazolam. 20 Treatments were also successfully completed in patients exhibiting ‘fair’ or ‘poor’ co-operation. 20 Only one patient in the first audit cycle failed to complete treatment, subsequently necessitating an onward referral to secondary care for further assessment and treatment under general anaesthesia.
Midazolam is an effective sedation agent: 2 this reflects the practice of clinicians in reliably assessing anxious patients’ suitability for pharmacological sedation alongside non-pharmacological patient management techniques. It is good practice to record both the quality of the sedation achieved and the operating conditions for the purpose of reflective practice and for future reference as a patient may subsequently be re-referred for treatment under IV sedation. In such a case the assessing sedationist can refer to the sedation record and thereby assist the assessment of the patient’s suitability for IV sedation. This again reinforces the importance of detailed, contemporaneous sedation record-keeping.
All clinicians consistently titrated midazolam to response, hence avoiding over-sedation. No fixed dose or bolus technique was used, and no reversal agent was necessary. There were no complications nor adverse events recorded either peri- or post-operatively in this study.
Completion of the second audit cycle confirmed that all clinicians recorded the ASA classification at the pre-operative assessment appointment. One patient’s ASA status in the second audit cycle was determined after discussion between two clinicians. This showed the subjectivity of the ASA classification system where there can be inconsistent assignment of ASA physical status between clinicians, resulting it being subject to criticism.21,22 Due to its subjective definitions, 23 it has been postulated that ASA classification has poor inter-rater reliability leading to variable interpretation of a patient’s physical status by different clinicians, which can lead to confusion.
All patients treated within our service for IV sedation fell within the recommended ASA I and ASA II categories (for treatment in a primary care setting).2,4 This confirms that the clinicians are aware of the importance of recording ASA status as part of the assessment of patient suitability for treatment under IV sedation. The record of ASA status enhances the patient selection process and helps to provide a better grading outcome for predicting the incidence of peri- and post-operative complications. 24
Conclusions
This completed audit highlighted the effectiveness of IV midazolam in anxious patients undergoing oral surgical procedures, and the suitability for its use in the primary care setting. The dose of midazolam administered intravenously to each patient in the study was within the recommended dose range. No patient safety concerns or complications were recorded, and all administered midazolam was titrated to a predetermined endpoint. The second cycle of audit highlighted the importance of recording the sedation score in monitoring the quality of sedation administered by a clinician. The significance of ASA classification recording cannot be overemphasised in the assessment of a patient’s physical status and suitability for sedation treatment, especially in a primary care setting. Completion of the second audit cycle records confirmed that all clinicians providing IV sedation within the service were aware of the missing elements in sedation recording. By maintaining the good practice of recording accurate contemporaneous records before, during, and after conscious sedation, clinicians help to ensure delivery of high-quality, safe and effective care.
