Abstract
Invasive pulmonary aspergillosis (IPA) is a severe opportunistic fungal infection that predominantly affects immunocompromised individuals, including those with hematologic malignancies, organ transplants, and, more recently, patients with post-COVID-19 immune dysregulation. Despite advancements in medical mycology, IPA continues to pose significant diagnostic and therapeutic challenges, contributing to high global morbidity and mortality. Diagnostic accuracy remains limited due to nonspecific clinical manifestations and the suboptimal performance of conventional tools such as bronchoalveolar lavage culture and galactomannan testing. However, recent innovations including polymerase chain reaction-based molecular assays, lateral flow devices, and immuno-positron emission tomography/magnetic resonance imaging offer improved sensitivity, specificity, and speed. Therapeutically, triazoles remain the cornerstone of IPA management, complemented by echinocandins and liposomal amphotericin B in refractory cases. The role of combination therapy and antifungal susceptibility testing is growing in response to rising azole resistance. Additionally, novel antifungal agents and immunotherapeutic approaches are currently under clinical investigation. Effective management of IPA requires a timely, multidisciplinary approach that combines advanced diagnostics with personalized antifungal strategies. Continued research is essential to standardize molecular techniques, refine immunotherapy, and expand access to next-generation antifungals to reduce the global burden of this life-threatening infection. This review aims to synthesize current evidence on the diagnosis and treatment of IPA, critically evaluate the strengths and limitations of existing diagnostic and therapeutic approaches, and explore emerging strategies to enhance clinical outcomes in the context of rising antifungal resistance.
Keywords
Introduction
Invasive pulmonary aspergillosis (IPA) is a severe fungal infection that predominantly affects immunocompromised individuals. Despite diagnostic advancements over the past decade, IPA continues to pose significant challenges in clinical practice. Global data from 120 countries estimate that invasive fungal diseases account for approximately 6.5 million cases and 3.8 million deaths annually, with around 2.5 million deaths (68%) directly attributed to fungal infections. Among these, more than 2.1 million cases of IPA occur in individuals with chronic obstructive pulmonary disease (COPD), those in intensive care units, and patients with lung cancer or hematological malignancies—resulting in an estimated annual mortality of 1.8 million (85.2%). Furthermore, chronic pulmonary aspergillosis has an estimated annual incidence of 1,837,272 cases, contributing to 340,000 deaths (18.5%). 1
In the post-COVID-19 era, the virus’s long-term impact on the immune system has raised significant concerns, particularly due to the growing population of immunocompromised individuals,2,3 As a result, the incidence of IPA is expected to increase. The diagnosis of IPA remains challenging due to its nonspecific clinical manifestations and the limited sensitivity and specificity of current diagnostic tools. Treatment is similarly complicated by delayed detection, the emergence of azole-resistant strains, suboptimal antifungal therapies, resource constraints, and the infection’s persistently high mortality rate.
This review aims to provide a comprehensive overview of current diagnostic approaches and treatment strategies for IPA. We also explore emerging diagnostic technologies and novel antifungal therapies based on recent research, and we highlight key challenges that must be addressed to improve patient outcomes in the future.
Invasive pulmonary aspergillosis diagnostic techniques
Currently, no single diagnostic test offers adequate sensitivity, specificity, and reliability to serve as a definitive tool for diagnosing IPA. As a result, diagnosis typically involves a combination of clinical, radiological, and microbiological assessments. In many cases, treatment decisions are made based on clinical judgment and risk assessment rather than waiting for confirmatory results from a diagnostic gold standard.
Clinical and radiological criteria
Clinical and radiological evaluation forms the initial step in assessing patients with suspected IPA. These methods are critical for identifying high-risk individuals and guiding further diagnostic testing.
Clinical manifestations
IPA commonly manifests with symptoms such as fever, cough, dyspnea, and chest pain. These symptoms tend to be progressive and are often unresponsive to conventional antibiotic therapy. In immunocompromised patients, the disease may progress rapidly, underscoring the importance of early detection. Identifying individuals at high risk such as those with neutropenia or those receiving prolonged immunosuppressive therapy is essential. In these patients, the presence of persistent respiratory symptoms and poor response to antibiotics should prompt immediate evaluation for IPA.4,5
The lack of robust clinical criteria for diagnosis and treatment has resulted in many adverse outcomes for patients due to a lack of early treatment, especially for ICU patients. ICU patients, regardless of their immune status, should be considered at risk for IPA. Therefore, many studies have attempted to develop diagnostic algorithms based on clinical criteria, aiming to provide early diagnosis and treatment for patients. Initial evaluations have also shown great promise when applied in clinical practice. However, since most of the algorithms were developed from retrospective database analysis, their reliability and effectiveness need to be validated through prospective studies. 6
Radiological imaging
Routine chest radiography has limited diagnostic utility in IPA. High-resolution computed tomography (HRCT) is considered the most valuable imaging modality, while magnetic resonance imaging (MRI) may serve as an alternative when appropriate. Classical HRCT findings of angioinvasive aspergillosis include macronodules (>1 cm) with a halo sign, pleural-based or alveolar consolidations, masses (especially in organ transplant recipients), internal low attenuation, the reverse halo sign, cavities, air-crescent signs (Figure 1), ground-glass opacities, and pleural effusion. Although no CT finding is completely specific or sensitive for IPA, these imaging characteristics can support early and accurate diagnosis, enabling timely clinical decision-making.7–9
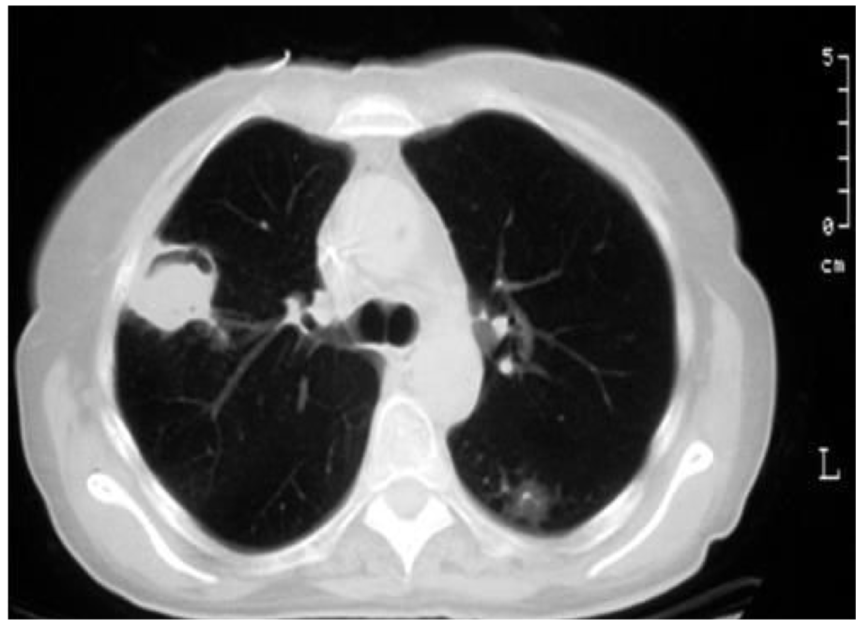
Air crescent sign of IPA on chest CT: a peripheral crescentic collection of air surrounding a mass of inflammatory consolidation, the air crescent sign.
Microbiological diagnostic methods
Testing of lower respiratory tract specimens
Respiratory tract culture remains the gold standard for diagnosing IPA when fungal colonies are identified on culture media (Figure 2). 10 Bronchoalveolar lavage (BAL) fluid is currently the most widely accepted and specific specimen for fungal culture. In situations where bronchoscopy with BAL is not feasible, cultures from sputum or exhaled air samples may serve as alternative diagnostic tools.

Morphological characteristics of
BAL fluid culture
BAL is the preferred method for obtaining lower respiratory tract specimens for fungal culture. However, fungal growth in culture can be slow, and sensitivity is often suboptimal, particularly in severely immunocompromised patients.5,11
Exhaled air sample culture
A novel technique involving the culture of exhaled air from mechanically ventilated patients has shown potential as a bedside diagnostic approach. Early studies indicate strong correlation with conventional diagnostic methods, suggesting its utility in critically ill populations. 12
Sputum testing
Antigen detection assays, such as galactomannan (GM) and lateral flow assay (LFA) tests on sputum samples, are being explored as noninvasive alternatives to BAL-based diagnostics, especially in patients with respiratory failure. These methods have demonstrated sensitivity comparable to BAL-GM testing and are more practical for routine use in clinical settings. However, their diagnostic accuracy requires further validation. 13
Antigen identification techniques
GM assay
GM is a polysaccharide component of the
Serum GM
The sensitivity and specificity of serum GM testing vary by patient population but are particularly useful in those with hematologic malignancies. False positives may occur due to cross-reactivity with other fungi or concurrent use of certain antibiotics.14–16 Detection of GM antigen in serum is a direct method for identifying cell wall components of
Bronchoalveolar lavage-GM
Testing GM levels in BAL fluid yields higher sensitivity than serum testing and is therefore considered more reliable for IPA diagnosis.13,19 GM testing from non-directed bronchial lavage (NBL) provides comparable results to bronchoalveolar lavage fluid for the detection of invasive pulmonary aspergillosis, making it a viable screening option in critically ill patients with high pretest probability for the disease. While bronchoalveolar lavage remains the gold standard for confirming the diagnosis of invasive aspergillosis, the use of NBL can help accelerate diagnosis and treatment initiation, thereby addressing the time sensitivity associated with invasive procedures. 20
(1-3)-β-D-glucan assay
(1-3)-β-D-glucan (BDG) is another component of the fungal cell wall that can be detected in serum. While it offers high sensitivity, it lacks specificity for
The integration of antigen and antibody testing across various specimen types offers promising avenues for the early and accurate diagnosis of IPA. 22
Histopathological examination
Histopathology of lung tissue obtained via biopsy is considered the gold standard for confirming IPA. This method allows direct visualization of
Molecular techniques
Molecular diagnostics, particularly polymerase chain reaction (PCR) based methods, have emerged as promising tools for the early and specific detection of
PCR techniques
Both nested PCR and real-time PCR are widely used to detect
Despite their potential, PCR assays for fungal detection show variability in diagnostic accuracy, depending on the primers used and the type of specimen analyzed. Studies suggest that PCR testing can complement GM and β-D-glucan assays. While GM tests tend to be more sensitive, PCR assays generally provide higher specificity. 16
Due to the non-specific clinical presentation and the limited sensitivity and specificity of radiological and mycological tests, the diagnosis of IPA is based on a graded scale of probability: possible, probable, or proven. The European Organization for Research and Treatment of Cancer and the Mycoses Study Group Education and Research Consortium (EORTC-MSGERC) has established standardized definitions for these diagnostic categories, particularly in immunocompromised patients These definitions incorporate three key components: host factors (underlying immunosuppressive conditions), clinical criteria (including signs and radiological features suggestive of IPA), and mycological evidence (direct or indirect microbiological detection of
The diagnostic classification of invasive pulmonary aspergillosis according to the European Organization for Research and Treatment of Cancer and Mycoses Study Group Education and Research Consortium.26,27

Possible IPA: Requires at least one host factor and one clinical criterion. Probable IPA: Requires at least one host factor, one clinical criterion, and one mycological criterion. Proven IPA: Can be diagnosed independently of host, clinical, or mycological criteria if there is direct histological or microbiological evidence of fungal invasion. (1,3)-beta-D glucan was not considered to provide mycological evidence of any IPA. For details, see Refs. 27 and 28.
Any, May or may not be present; BAL, bronchoalveolar lavage; CT, computed tomography; HSCT, hematopoietic stem cell transplant; IPA, invasive pulmonary aspergillosis; PCR, specific polymerase chain reaction for Aspergillus species;
In summary, the EORTC provides guidance for the following definitions. 26
Possible IPA: Requires at least one host factor and one clinical criterion.
Probable IPA: Requires at least one host factor, one clinical criterion, and one mycological criterion.
Proven IPA: Can be diagnosed independently of host, clinical, or mycological criteria if there is direct histological or microbiological evidence of fungal invasion.
Emerging diagnostic approaches
Advancements in diagnostic technology aim to improve the speed, accuracy, and accessibility of IPA testing.
Lateral flow assay
These point-of-care assays use monoclonal antibodies to rapidly detect
Immuno-PET/MRI imaging
An innovative approach involves labeling humanized monoclonal antibodies with radioactive isotopes for use in positron emission tomography (PET) combined with MRI. This technique enables non-invasive, in vivo visualization of fungal infections with high specificity and may help distinguish IPA from other pulmonary pathologies. While promising, this method is still in the preclinical phase and requires further validation in human studies. 30
Role of AI in supporting imaging diagnosis
A recent study assessed the performance of chest CT-based artificial intelligence (AI) models trained via unannotated supervised training (UST) and annotated supervised training (AST) in differentiating pulmonary mycosis, IPA, and pulmonary tuberculosis. Both models achieved an overall accuracy of 66.1%. The UST model demonstrated sensitivities of 27.3% for pulmonary mycosis, 73.9% for IPA, and 76.0% for pulmonary tuberculosis, whereas the AST model achieved sensitivities of 9.1%, 69.6%, and 88.0% for the same conditions, respectively. These findings suggest that integrating AI with imaging modalities may enhance early diagnostic guidance, particularly in high-risk populations for IPA. 31 However, AI technology still requires substantial development before it can be reliably applied in clinical settings.
Diagnostic challenges
Effective diagnosis and treatment of IPA demand a multidisciplinary approach that integrates clinical assessment, pulmonary imaging, microbiological testing, and molecular diagnostics. 8 While traditional methods such as GM testing and BAL culture are still widely used, emerging technologies like LFDs and immuno-PET/MRI imaging offer promising alternatives for rapid and accurate diagnosis.
However, several challenges remain:
Absence of a definitive diagnostic gold standard: No single test provides sufficient sensitivity and specificity, necessitating a combination of clinical, radiologic, and laboratory criteria.4,5
Lack of PCR assay standardization with high diagnostic accuracy remains a challenge. Multicenter studies with larger sample sizes are needed to establish standardized PCR protocols and improve diagnostic performance.19,21
Rising antifungal resistance: The increasing prevalence of resistant
Approach to invasive pulmonary aspergillosis treatment
IPA is a life-threatening respiratory infection associated with high morbidity and mortality, particularly in immunocompromised individuals. Although a variety of antifungal agents and combination therapies have been developed and implemented in clinical practice, treatment remains challenging due to difficulties in early diagnosis, the emergence of antifungal resistance, and the limitations of current therapeutic options.
Current treatments and their effectiveness
First-line therapy
Based on their mechanisms of action, antifungal agents used in the clinical treatment of IPA can be broadly categorized into three main classes. Azoles inhibit the enzyme 14α-lanosterol demethylase, thereby disrupting the synthesis of ergosterol, an essential component of the fungal cell membrane. The resulting depletion of ergosterol compromises membrane integrity and leads to fungal cell death. Polyenes, exemplified by amphotericin B (AmB), bind selectively to ergosterol in the fungal cell membrane, forming pores that disrupt membrane permeability and lead to cell lysis. Echinocandins inhibit the enzyme 1,3-β-D-glucan synthase, thereby blocking the synthesis of β-D-glucan, a critical component of the fungal cell wall. This disruption weakens the cell wall and leads to osmotic instability. 33
The mainstay of IPA treatment is antifungal therapy. Among available agents, triazoles are the most widely recommended and commonly used as first-line therapy due to their broad antifungal spectrum, favorable safety profiles, and oral bioavailability (Table 2). 27
Antifungal agents for the treatment of invasive pulmonary aspergillosis. 27

Source: © European Respiratory Society. All rights reserved.
ALP, alkaline phosphatase; ALT, alanine aminotransferase; AST, aspartate aminotransferase; CPA, chronic pulmonary aspergillosis; D1, day 1; DDI, drug–drug interaction; GGT, gamma glutamyltranspeptidase; GM, galactomannan; IPA, invasive pulmonary aspergillosis; TDM, therapeutic drug monitoring; W1, week 1.
Azoles
Including voriconazole, posaconazole, and isavuconazole, are currently the most widely recommended first-line antifungal agents for the treatment of IPA. These agents are preferred due to their broad-spectrum antifungal activity, oral bioavailability, and more favorable safety profiles compared to older agents such as amphotericin B.34–36 Among them, voriconazole has demonstrated superior efficacy, with studies showing improved survival rates compared to conventional amphotericin B therapy.37,38
Voriconazole, considered the first-line agent of choice for IPA, has demonstrated superior efficacy and improved survival, and is generally well tolerated compared to amphotericin B. While associated with fewer adverse effects, it can still cause significant toxicities, including hepatotoxicity, visual disturbances, and neurotoxicity, warranting careful monitoring during treatment.34,36,39
Isavuconazole
A newer-generation triazole, isavuconazole offers comparable efficacy to voriconazole but with fewer hepatotoxic effects and a better overall safety profile.35,36,40 Clinical trials have shown that isavuconazole is non-inferior to voriconazole, making it a viable alternative, particularly in patients who are intolerant to the latter. 38
Posaconazole
Often used as salvage therapy or prophylaxis in high-risk patients, posaconazole has been shown to reduce mortality, especially when incorporated into combination regimens.36,41
Echinocandins
Echinocandins such as caspofungin and micafungin, act by inhibiting β-(1,3)-D-glucan synthesis, a key component of the fungal cell wall. While not typically used as first-line monotherapy, echinocandins are valuable as salvage options or as part of combination therapy in refractory or severe IPA cases. These agents are generally well-tolerated and exhibit low toxicity profiles.42,43
Caspofungin
Studies suggest that caspofungin, when used in combination with voriconazole, improves treatment outcomes in high-risk patients with IPA. 42
Micafungin
Experimental studies have demonstrated enhanced efficacy when micafungin is combined with isavuconazole, indicating its potential role in future combination regimens. 43
Amphotericin B
Amphotericin B, particularly in its liposomal formulation (L-AmB), remains an important antifungal agent for IPA treatment. Despite its potent antifungal activity, the drug's use is limited by significant nephrotoxicity and infusion-related adverse effects.36,39
Liposomal amphotericin B
This formulation reduces toxicity and is typically reserved for patients who are intolerant to triazoles or echinocandins. It has demonstrated effectiveness, especially in salvage therapy or when used in combination with other antifungal agents.36,40
Triazole antifungals demonstrate superior efficacy and safety compared to AmB in the primary treatment of IPA. Among these, isavuconazole appears to be the most optimal choice, offering the greatest reduction in all-cause mortality in patients with proven and probable IPA, along with the most favorable safety profile. Triazoles significantly reduced all-cause mortality compared to AmB across broader patient groups, including those with proven, probable, and possible IPA. Although no significant differences were observed in the incidence of overall adverse events among the triazoles, isavuconazole (ISA) demonstrated the most favorable safety profile. Furthermore, the incidence of severe adverse events was significantly lower with triazoles compared to AmB, with ISA again showing the best profile. Notably, renal and urinary disorders were the most common severe adverse events associated with AmB. 34
Combination therapy
Current evidence does not support replacing voriconazole with an echinocandin as first-line monotherapy for the treatment of IPA. However, the potential benefit of combining an echinocandin with voriconazole has been considered for many years. Mechanistically, the combination is plausible, as triazoles inhibit fungal cell membrane synthesis, whereas echinocandins target the fungal cell wall, offering a potential synergistic effect against
Combination antifungal therapy typically involving a triazole and an echinocandin has been explored as a strategy to improve treatment outcomes, particularly in patients with severe or refractory IPA. Evidence suggests that combination regimens may reduce mortality and improve clinical response rates, especially among high-risk individuals.42,43 The addition of an echinocandin to a mold-active triazole in the treatment of IPA may be employed in two distinct clinical settings: primary and salvage combination therapy. Primary combination therapy involves the initial administration of both agents in treatment-naive patients. In contrast, salvage combination therapy refers to the addition of an echinocandin following an inadequate response to initial monotherapy with a mold-active triazole. In current clinical practice, this strategy is most commonly applied when monotherapy with agents such as voriconazole fails to achieve a satisfactory clinical response. 44
Voriconazole and anidulafungin
This combination has demonstrated improved survival in patients with IPA, particularly those with advanced disease. 42
Isavuconazole and micafungin
Experimental models have shown that this combination enhances antifungal efficacy and reduces fungal burden, indicating potential clinical benefit. 43
The treatment of IPA should be continued for a minimum of 6–12 weeks, with the exact duration depending on factors such as the degree and duration of immunosuppression, the anatomical site of infection, and the patient’s clinical and radiological response to therapy. 45
Prophylaxis in high risk individuals
Primary prophylaxis may be indicated for patients at high risk of developing IPA. Several patient populations are recognized as being at high risk for IPA due to immunosuppression and environmental exposures. These include lung transplant recipients, hematopoietic stem cell transplant (HSCT) recipients, individuals with hematologic malignancies, and patients with certain chronic diseases or undergoing specific immunosuppressive therapies. 45
Prophylaxis against IPA with posaconazole or voriconazole is recommended during periods of prolonged neutropenia in individuals at high risk. In the setting of lung transplantation. For patients with chronic immunosuppression due to graft-versus-host disease (GVHD), antifungal prophylaxis should be continued throughout the course of immunosuppressive therapy, particularly in those receiving corticosteroids at doses equivalent to >1 mg/kg/day of prednisone for more than 2 weeks and/or those undergoing treatment with other anti-GVHD agents such as lymphocyte-depleting therapies or tumor necrosis factor-α (TNF-α) inhibitors for refractory GVHD. 45
Among solid organ transplant recipients, lung transplant patients are at the highest risk for IPA. The reported incidence of fungal infection in this group ranges from 9% to 32%, with early-onset infections associated with significantly higher mortality rates.46,47 The most common infectious agent is
Liver transplant recipients, especially those requiring renal replacement therapy, are also vulnerable to IPA. 49 For patients with chronic immunosuppression due to GVHD, antifungal prophylaxis should be continued throughout the course of immunosuppressive therapy, particularly in those receiving corticosteroids at doses equivalent to >1 mg/kg/day of prednisone for more than 2 weeks and/or those undergoing treatment with other anti-GVHD agents such as lymphocyte-depleting therapies or tumor necrosis factor-α (TNF-α) inhibitors for refractory GVHD. 45 Additionally, critically ill patients in intensive care units, particularly those with COPD, prolonged corticosteroid use, or end-stage liver disease are at increased risk. 50 Patients with antineutrophil cytoplasmic antibody-associated vasculitis receiving immunosuppressive treatment may also benefit from prophylactic antifungal strategies. 51 Prophylaxis against IPA with posaconazole or voriconazole is recommended during periods of prolonged neutropenia in individuals at high risk. 45
HSCT recipients, particularly those experiencing graft-versus-host disease, face a high risk of IPA due to prolonged neutropenia and intensive immunosuppression. Prophylaxis with voriconazole has been shown to be effective, with some studies reporting no cases of IPA in HSCT recipients receiving this regimen. 52
Patients with hematologic malignancies undergoing intensive chemotherapy for acute leukemia or those diagnosed with myelodysplastic syndrome are also at significant risk, primarily due to prolonged periods of neutropenia. In such high-risk patients, antifungal prophylaxis is essential. Posaconazole is recommended and has demonstrated efficacy in reducing IPA incidence. 9 For patients with hematologic malignancies, two evidence-based strategies are commonly adopted: (1) primary prophylaxis with posaconazole. (2) Preemptive monitoring without prophylaxis, involving regular surveillance with biomarkers (e.g., serum GM or PCR) at least twice weekly. The choice between these strategies depends on local epidemiology, access to rapid diagnostics, and individual patient risk factors. 9
While antifungal prophylaxis plays a vital role in reducing IPA incidence and improving outcomes in high-risk populations, careful consideration must be given to potential adverse effects, drug interactions, and the development of antifungal resistance. The emergence of azole-resistant
Current challenges in IPA treatment
Despite the availability of effective antifungal agents, the treatment of IPA remains fraught with challenges:
Antifungal resistance
Azole resistance
The rise of azole-resistant
Multidrug resistance
Some strains of
Antifungal susceptibility testing
AFST should be routinely performed in patients with confirmed or suspected IPA, particularly when clinical response is suboptimal or azole resistance is suspected. Minimum inhibitory concentration testing and mutation screening are recommended. In areas with >10% azole resistance, initial combination therapy with voriconazole and an echinocandin or liposomal amphotericin B is advised. 9
Limited efficacy in specific populations
Immunocompromised patients
Those undergoing hematopoietic stem cell transplantation or living with hematologic malignancies are at elevated risk of treatment failure due to impaired immune responses.56,57
Localized infections
Fungal infections in poorly vascularized or protected sites, such as the central nervous system, may limit antifungal drug penetration and lead to poor outcomes. 41
Toxicity and drug interactions
The use of antifungal therapy in IPA is complicated by unpredictable pharmacokinetic profiles, the strong association between drug exposure and both efficacy and toxicity, and numerous drug–drug interactions mediated via the CYP450 system. These interactions are of particular concern during voriconazole therapy, the current gold standard for IPA treatment, but are also observed to a lesser extent, with other triazole agents such as Posaconazole. 57
Hepatotoxicity
Triazoles, particularly voriconazole, are associated with liver toxicity. This may necessitate dose adjustments or treatment discontinuation.56,57
Nephrotoxicity
AmB, especially in conventional formulations, poses a risk of kidney injury, particularly in patients with pre-existing renal impairment.36,39
Delayed diagnosis
Timely diagnosis is crucial for effective IPA management. However, the lack of highly sensitive and specific diagnostic tools often results in delays, which can worsen prognosis. Early initiation of therapy is essential to improve survival and treatment outcomes.57,58
The advent of novel anti-cancer and immune-modulating therapies, together with the recent COVID-19 pandemics, has led to a marked increase in both the incidence and clinical spectrum of pulmonary aspergillosis. The heterogeneity of clinical and radiological manifestations, combined with the limited sensitivity and specificity of available microbiological tests, poses substantial diagnostic challenges. It is therefore essential to educate clinicians to maintain a high index of suspicion and to recognize pulmonary aspergillosis in these emerging or atypical patient populations, in whom the disease is frequently overlooked. 27
New treatments and their prospects
Emerging therapeutic strategies offer hope for improving treatment outcomes and long-term prognosis in patients with IPA. These approaches include the development of novel antifungal agents, innovative drug delivery systems, immunomodulatory therapies, and optimized combination regimens. 59
Novel antifungal agents
Several new antifungal agents currently under investigation show promise in overcoming resistance and improving clinical efficacy.
Olorofim
A dihydroorotate dehydrogenase inhibitor, olorofim has demonstrated potent antifungal activity in preclinical studies and is now in late-stage clinical development.40,59 Olorofim has demonstrated safety and good tolerability in clinical trials in both oral and intravenous formulations. Reported adverse effects include elevated liver transaminases and gastrointestinal symptoms such as nausea and diarrhea. Although clinical data on therapeutic efficacy remain limited, early findings suggest activity against molds, including Aspergillus, and potential value in treating drug-resistant infections such as azole-resistant aspergillosis. To date, combining olorofim with azoles has not shown significant clinical benefit. Further studies are needed to assess the potential advantages of such combinations in mold infections. 60
Fosmanogepix
This Gwt1 enzyme inhibitor exhibits broad-spectrum antifungal activity, including efficacy against
Ibrexafungerp
A triterpenoid antifungal agent, ibrexafungerp, has shown effectiveness against azole-resistant
BAL2062 is a first-in-class antifungal, siderophore-like molecule structurally similar to ferrichrome, but distinguished by three amino acid substitutions and an aluminum rather than iron chelate. It is transported into fungal cells via the Sit1 transporter—absent in humans—conferring fungal specificity. BAL2062 exhibits fungicidal activity against Aspergillus spp., including azole-resistant strains. Its favorable safety profile, consistent pharmacokinetics, and lack of drug accumulation support continued development for the treatment of invasive aspergillosis. 60
Inhaled nanomedication
Inhaled drug delivery systems are being developed to enhance antifungal concentrations directly at the site of pulmonary infection while minimizing systemic toxicity. Nanoparticle-based formulations of agents such as posaconazole and voriconazole are under investigation for their potential to improve efficacy, reduce side effects, and provide targeted delivery within the lungs. 62
An optimized formulation of posaconazole nanocrystals presents a promising strategy for the topical treatment of IPA, offering enhanced drug efficacy, efficient lung deposition, strong antifungal activity, and a lower risk of systemic side effects. 63
Immunotherapy
Immunotherapy represents a novel adjunctive strategy that enhances the host’s immune response rather than directly targeting the pathogen. Potential approaches include monoclonal antibodies, cytokines, cytokine inhibitors, checkpoint inhibitors, cell-based therapies, and vaccination. Initial studies have demonstrated promising results in preclinical models and early-phase clinical trials. However, because at-risk populations exhibit diverse immune profiles—from neutropenia to hyperinflammation personalized immunotherapy strategies are essential for optimizing efficacy and safety. Immunodiagnostic tools may assist in identifying patients likely to benefit from specific immunomodulatory approaches. High-quality clinical trials involving well-characterized patient populations are needed before immunoprophylaxis or immunotherapy can be widely implemented in IPA management.40,59
Conclusion
The diagnosis of IPA continues to necessitate a multidisciplinary approach that integrates clinical evaluation, pulmonary imaging, microbiological testing, and molecular diagnostics. While conventional methods such as BAL culture and GM testing remain widely utilized, emerging technologies, including lateral flow devices and immuno-PET/MRI imaging, offer promising advancements for more rapid and accurate detection. Diagnosing IPA in clinical practice remains challenging and often depends on clinical judgment and experience, particularly in patients belonging to identified high-risk groups. Future research should prioritize enhancing the sensitivity and specificity of diagnostic tools, standardizing molecular diagnostic techniques, and developing innovative technologies to overcome current limitations.
The treatment of IPA remains complex, requiring a comprehensive strategy that includes antifungal therapy, supportive care, and, in select cases, surgical intervention. Although triazoles and echinocandins continue to serve as the foundation of antifungal therapy, the growing threat of drug resistance and limitations in existing treatments underscore the urgent need for continued innovation. The emergence of new antifungal agents, inhaled delivery systems, and immunotherapies provides renewed optimism for improving clinical outcomes and reducing the global burden of this life-threatening infection. Prophylactic treatment of IPA in high-risk populations should be initiated promptly. The decision to implement antifungal prophylaxis should weigh the benefits of preventing fungal infection against the potential adverse effects of antifungal agents, while also taking into account the local epidemiological patterns of
Footnotes
Acknowledgements
The author would like to thank Dr. Nu Nguyet Anh Nguyen from the University of Social Sciences and Humanities, Vietnam National University, Ho Chi Minh City, for her support with English language editing. Sincere thanks are also extended to the reviewers, especially Reviewer 1, whose thorough and insightful comments have greatly contributed to improving the clarity and scientific value of this manuscript.
