Abstract
A 79-year-old female diagnosed with myelodysplastic syndrome (MDS) and germline GATA2 mutation, on compassionate cobimetinib, was admitted with subacute cough and dyspnea. Chest imaging demonstrated a new, large, left hilar mass and consolidation with scattered diffuse mediastinal, supraclavicular, and hilar lymphadenopathy. A core biopsy of the right supraclavicular lymph node was performed. Acid-fast bacilli (AFB) cultures from both the lymph node and blood were positive, while all fungal cultures were negative. Two distinct AFB colonies were observed on solid media and identified as Mycobacterium avium complex and Mycobacterium kansasii. Disseminated non-tuberculous mycobacterial infections involving two distinct species are rare, pose treatment challenges, and may correlate with cobimetinib administration for MDS, as well as GATA2 germline mutations.
Introduction
Over the past decade, non-tuberculous mycobacterial (NTM) disease has become increasingly prevalent, particularly among immunocompromised individuals. This rise is attributed to improved diagnostics, an aging population, and a growing number of immunosuppressed patients. 1 NTM infections disproportionately affect the elderly and are more common in warm, humid regions, correlating with population shifts to these regions and climate change. 2 Risk factors include structural lung disease (e.g., chronic obstructive pulmonary disease (COPD), bronchiectasis, and malignancy), smoking, aging, and immunosuppression. A study of 2846 patients with hematologic malignancies reported an NTM incidence of 1.2% (34 cases), with Mycobacterium avium complex (MAC) being the predominant species (38%). Of the 34 cases, 28 had pulmonary symptoms, while 8 presented with disseminated disease, notably linked to neutropenia. 3
MAC is a slow-growing NTM infection, which can take up to 20 days to grow on culture. 1 While Mycobacterium kansasii has been isolated frequently in tap water, MAC has often been isolated in soil and natural water sources. 3 MAC typically present as a pulmonary disease but can also present with lymphadenopathy in immunocompromised hosts (such as HIV/AIDS). Most pulmonary NTM infections are not spread person-to-person but rather acquired due to inhalation of infected aerosolized droplets. 1
M. kansasii is another slow-growing NTM that can cause pulmonary disease, lymphadenitis, and infections of the skin, soft tissue, musculoskeletal system, and indwelling catheters. Disseminated disease occurs in uncontrolled HIV or heavily immunosuppressed hosts. 1 Pulmonary disease is the most frequent presentation and mimics M. tuberculosis, with symptoms including weight loss, hemoptysis, chest pain, night sweats, and fever. Radiographic findings include pulmonary cavitation, pleural scarring, and fibrosis.
Co-infection with multiple NTMs is rare, reported mostly in individuals with profound immunosuppression, such as HIV/AIDS, anti-interferon-γ autoantibodies, or idiopathic CD4 lymphopenia.4–7 One case documented a 59-year-old healthy Thai man who succumbed to disseminated MAC and M. kansasii lymphadenitis after developing refractory lymphopenia despite treatment. This underscores NTM’s lethality even in immunocompetent hosts. 4
A retrospective study of 213 patients with non-MAC NTM infections found M. gordonae (usually a colonizer) and M. kansasii as the most common isolates. COPD and malignancy were frequent comorbidities, and co-infection with multiple NTMs occurred in 55 patients (26%). 7 It is pertinent to identify co-infections with more than one NTM due to variations in antibiotic susceptibility, significantly influencing treatment regimen decisions. 8
Case
A 79-year-old female patient with myelodysplastic syndrome (MDS) was admitted with a 2-week history of intermittent low-grade fevers, non-productive cough, and progressive exertional dyspnea. She denied hemoptysis and weight loss, and the rest of the review of systems was unremarkable. She relocated from Kansas City, MO (Midwest US) to Utah a few weeks before the onset of symptoms. She denied any prior international travel, known contact with tuberculosis (TB) patients, homelessness, incarceration, substance use, and exposure to animals.
Oncologic history was notable for low-risk SF3B1-mutant MDS diagnosed in 2016, monitored without treatment, which evolved by 2019 into a combined myeloproliferative/myelodysplastic neoplasm with new ASXL1, GATA2, and NRAS mutations. Initial treatment with hydroxyurea and decitabine was discontinued due to drug-related adverse effects, and compassionate-use cobimetinib (targeting RAS mutation) was initiated 1 month prior to presentation.
On admission, she was hypoxic, requiring 3 L/min of oxygen via nasal cannula, and had sinus tachycardia with a heart rate of 100–110 bpm. Physical exam at admission was notable for frailty, generalized weakness without focal neurological deficits, pallor, and oral thrush. Respiratory exam revealed crackles in bilateral mid and lower zones of her lungs. To evaluate the cause of dyspnea and hypoxia, a CT chest was performed, revealing a large left perihilar mass, left lower lobe consolidation suggestive of post-obstructive pneumonia, and hilar, supraclavicular, and mediastinal lymphadenopathy (Figure 1). The differential diagnoses for her acute on chronic cough, constitutional symptoms, and lymphadenopathy on imaging included both infectious etiologies-particularly fungal/mycobacterial causes and non-infectious etiologies including MDS conversion to lymphoma.

CT Chest with new left hilar soft tissue conglomerate and left-sided consolidations (red arrow indicating L hilar mass).
The patient had scant sputum, and bronchoscopy was considered but deferred due to her frailty and high procedural risk. Hence, a core biopsy of her right supraclavicular lymph node was performed, and the tissue was sent for bacterial, fungal, acid-fast bacilli (AFB) cultures, and histopathology. The lymph node biopsy was smear positive for 1+ AFB, an Xpert® (Cepheid (Sunnyvale, CA)) MTB/RIF Polymerase chain reaction (PCR) on the tissue was negative for M. tuberculosis. Fungal cultures remained negative. While awaiting AFB culture confirmation, the patient developed new symptoms including headaches, worsening dyspnea, and a large right-sided pleural effusion. A head CT was unremarkable. Cerebrospinal and pleural fluids were obtained via lumbar puncture and thoracentesis, respectively, and the results are summarized in Table 1. Additionally, tissue from the lymph node biopsy was sent to the University of Washington for Nontuberculous Mycobacteria DNA Detection by PCR and plasma was submitted for metagenomic Next-Generation Sequencing (mNGS) (Karius) for expedited results. Table 2 summarizes all positive culture results and time to positivity.
Lab results.

ADA, Adenosine Deaminase; CSF, Cerebrospinal Fluid; PMN, polymorphonuclear
Time to culture positivity for each specimen collected.

AFB, Acid-fast bacilli; MAC, Mycobacterium avium complex.
The plasma mNGS Karius test yielded a positive result for M. kansasii (>316,000 molecules/μL

Growth of M. kansasii (larger, smooth yellow colonies) and MAC (smaller, cream colonies).
Cultures from bone marrow aspirate and three blood samples inoculated into BD BACTEC™ Becton, Dickinson and Company (Sparks, Maryland) Myco F/lytic bottles flagged positive at 19 days (bone marrow) and 10 days (blood) for both MAC and M. kansasii. MAC was detected first by 16S rRNA Sanger sequencing, with M. kansasii colonies appearing on LJ medium a mean of 6.5 days later. Blood isolates required 16S rRNA sequencing for identification due to poor MALDI-TOF scores. Pleural fluid, inoculated into MGIT™, flagged positive on day 14, and only M. kansasii complex was recovered.
During admission, her condition worsened with increasing oxygen requirements. Empiric treatment with moxifloxacin, azithromycin, and ethambutol was initiated based on broad-range PCR and Karius results showing M. kansasii; rifamycins were avoided due to chemotherapy interactions. After MAC was also recovered in cultures, the regimen was continued as it covered both NTM species. She showed mild clinical improvement and was discharged on the same regimen. Drug susceptibility results are shown in Table 3. Histopathology images can be seen in Figures 3 and 4. Despite appropriate NTM treatment, factors like her advanced age, frailty, heavy infection burden, poor medication tolerance, underlying hematologic malignancy likely compounded her decline; two weeks after discharge, she worsened with profound fatigue, transitioned to hospice, and died shortly thereafter.
Antimicrobial susceptibility testing results for MAC and M. kansasii isolates with minimum inhibitory concentration (µg/mL) and interpretations.

MAC, Mycobacterium avium complex.

H&E stain of the lymph node biopsy demonstrating acute necrotizing lymphadenitis.
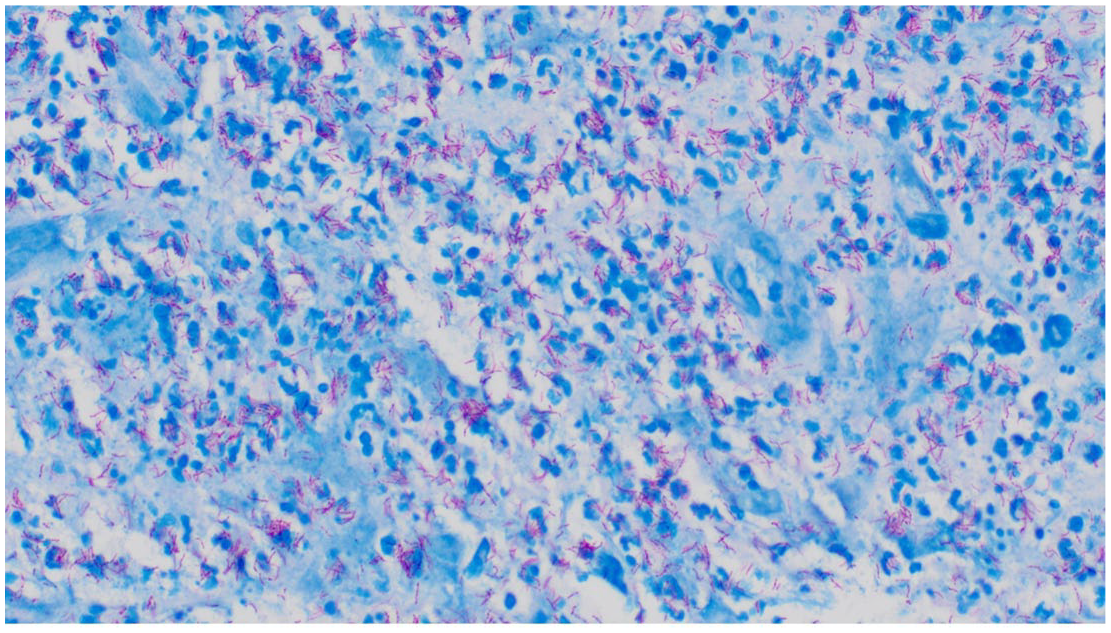
AFB stain of the lymph node biopsy.
Discussion
Our patient had multiple risk factors for developing NTM infections including her age, underlying malignancy with progressive MDS, cobimetinib use, and GATA2 mutation.
MDS patients are at increased risk of infections due to neutrophil dysfunction. Additionally, immunomodulatory therapy may also increase susceptibility to NTM infections. 9
Our patient also had ASXL1 and GATA2 mutations. ASXL1-mutated AML typically has a worse prognosis.10,11 GATA2 mutations, whether germline or somatic, predispose individuals to NTM and fungal infections, likely due to a depletion of monocytes and NK cells. This results in increased vulnerability to opportunistic infections like disseminated NTM, and prophylactic azithromycin is sometimes used to reduce this risk. 12 GATA2 mutations at different loci lead to various syndromes, including MonoMAC syndrome, characterized by monocytopenia and MAC infection. 13 Mortality in MonoMAC is around 28%, and allogeneic stem cell transplant is a treatment option if diagnosed early. 14 Unfortunately, in this case, the patient was diagnosed too late for therapeutic benefit due to severe NTM infection.
Co-infection with multiple NTM species is rare but seen in severe immunosuppression, such as advanced HIV, chronic immunosuppressant use (e.g., steroids), hepatitis, and hematologic malignancy. 8 Mortality rates are elevated in individuals with substantial immunosuppression, particularly those with malignancy or advanced HIV, primarily due to immune dysfunction and failure to recover, rather than disease recurrence.8,15
Next-generation sequencing (NGS) has expedited diagnosis, especially since mycobacteria grow slowly in cultures. NGS also aids in screening high-risk individuals for GATA2 mutations, as bronchoscopy may not be feasible in thrombocytopenic hematologic malignancy patients. 14
In this case, the NTM PCR and the Karius test helped initiate appropriate antimicrobial therapy before traditional culture results were available. However, these tests are expensive and may detect non-pathogenic microorganisms or fail to identify the pathogen. For example, Karius did not detect both NTM species in our case. Furthermore, a recent study showed that cell-free DNA testing often lacks substantial clinical impact. 16
The patient’s chest imaging and lymphadenopathy raised suspicion for atypical infections, leading to a lymph node biopsy and additional samples for AFB cultures to enhance diagnostic yield and confirm disseminated infection. This case highlights the importance of combining cultures with histopathology and molecular testing to avoid missed diagnoses. Traditional AFB cultures can take weeks, making faster diagnostic tools like Karius or PCR-based tests valuable. MAC was isolated within 9 days, followed by M. kansasii on day 11. Both organisms are slowly growing mycobacteria and may appear as indistinguishable pinpoint colonies early on solid media. It is likely that MAC, with a doubling time of 10–16 h, compared to M. kansasii (19–20 h) had a growth advantage in mixed cultures, resulting in its earlier identification. M. kansasii was observed on solid media only after continued incubation and exposure to light as photochromogenic colonies, which takes some time to appear and hence likely explains a delay in identification in the mixed cultures.
Although MAC grew earlier in culture, only M. kansasii was identified via mNGS (Karius testing) or NTM PCR. This discrepancy may reflect differences in microbial DNA shedding and host-pathogen interactions. The Karius test detects microbial cfDNA released into circulation, which may not directly correlate with viable organism burden detected by culture. M. kansasii may exhibit greater systemic DNA release due to differences in tissue tropism, susceptibility to host immune-mediated lysis (via immune mechanisms such as IL-12), or necrotizing pathology, even in the context of lower viable bacterial load.17,18 MAC, which often persists within macrophages and forms organized granulomas, may shed less detectable DNA into plasma. 19
It is currently unclear why MAC was not detected on Karius or NTM PCR but we theorize that it might be due to low organism burden, uneven distribution of pathogens in tissues or body fluids, specimen sampling, and/or differences in the limit of detection of the molecular assays. MAC grew better in culture despite there being a higher DNA quantity of M. kansasii in blood. Although Karius and the NTM PCR detected only M. kansasii, the traditional AFB cultures from biopsies corroborated the genomic findings by recovering M. kansasii and providing instrumental information by growing MAC, an organism that neither molecular test detected. Thus, these results highlight that culture remains the gold standard due to its ability to grow and differentiate multiple organisms in co-infections as it was this case for M. kansasii and MAC. This role of culture was not only crucial for guiding appropriate empiric treatment but also provided organisms to perform susceptibility testing.
Conclusion
This case highlights the importance of considering NTM infections in immunocompromised patients, especially in those with an acute-on-chronic exacerbation of symptoms, and in individuals with GATA2 mutations or receiving immunomodulatory therapy like cobimetinib. This heightened awareness ensures the timely initiation of appropriate diagnostic testing, resulting in expedited diagnosis and treatment planning. Additionally, this case highlights the limitations of the current diagnostic tools for mycobacterial infections available in clinical laboratories.
Supplemental Material
sj-pdf-1-tai-10.1177_20499361251359825 – Supplemental material for Disseminated Mycobacterium kansasii and Mycobacterium avium complex co-infection in GATA2 mutated myelodysplastic syndrome: a case report
Supplemental material, sj-pdf-1-tai-10.1177_20499361251359825 for Disseminated Mycobacterium kansasii and Mycobacterium avium complex co-infection in GATA2 mutated myelodysplastic syndrome: a case report by Sabrina Newstead, Daniel Montelongo, Salika Shakir and Neeraja Swaminathan in Therapeutic Advances in Infectious Disease
Footnotes
Acknowledgements
We would like to thank the AFB lab at ARUP Laboratories in their help with the microbiologic workup and for the photos provided. We would also like to thank Dr Benjamin Bradley, MD, PhD, for providing the histopathology images.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
