Abstract
Background:
Enterococci are significant contributors to healthcare-associated infections in Ethiopia.
Objectives:
This systematic review and meta-analysis synthesize data on the prevalence, antimicrobial susceptibility patterns, and associated risk factors for Enterococcus infections in Ethiopia
Design:
Systematic review and meta-analysis
Data sources and methods:
A comprehensive search was conducted in Scopus, PubMed, Web of Science, Cochrane Library, and Google Scholar, covering studies published in English over the past 5 years, with the last search on October 30, 2024. Inclusion criteria targeted original studies on Enterococcus prevalence, resistance, and risk factors in Ethiopian healthcare and community settings. Risk of bias was evaluated using the ROBINS-I tool. Meta-analytic techniques calculated pooled prevalence, log-adjusted odds ratios (log-AORs), and p-values, accompanied by heterogeneity and subgroup analyses.
Results:
Thirteen studies encompassing 3598 participants (mean age: 29.26 ± 6.6 years) reported a pooled prevalence of 6.67% (95% CI: 5.50–8.85) for Enterococcus species, with substantial regional variation (0.03–55.88). Major risk factors included prolonged hospital stays (OR = 6), catheterization (OR = 3.5), and diabetes (OR = 3.92). The pooled log-AOR was 0.986 (95% CI: 0.214–1.759; p = 0.01). Antimicrobial susceptibility tests identified cephalexin and co-trimoxazole as the most effective antibiotics (100% sensitivity each), while oxacillin, trimethoprim-sulfamethoxazole, and norfloxacin exhibited the highest resistance rates (85.71%, 80.00%, and 80.00%, respectively). Considerable heterogeneity was observed (I2 = 92.31%, τ2 = 5882.35).
Conclusion:
This systematic review and meta-analysis found a 6.67% pooled prevalence of Enterococcus infections in Ethiopia, with high variability in antibiotic resistance and key risk factors such as prolonged hospital stays, catheterization, and diabetes. Effective antibiotics included cephalexin and co-trimoxazole, while high resistance was noted for oxacillin and norfloxacin. The findings emphasize the need for targeted infection control and antimicrobial stewardship.
Plain language summary
Background: Enterococci are bacteria that can cause serious infections, in community and hospital settings. In Ethiopia, they are an emerging concern in both healthcare and community environments. Purpose of the Study: This study brings together and analyzes recent research to understand how common Enterococcus infections are in Ethiopia, how resistant they are to antibiotics, and what risk factors increase the likelihood of infection. How the Study Was Done: Researchers searched five major scientific databases for English-language studies published within the past five years, up to October 30, 2024. They included original studies that reported the prevalence of Enterococcus infections, antibiotic resistance patterns, and associated risk factors. Each study’s quality was assessed, and the data were combined and analyzed using statistical methods. What the Study Found: Thirteen studies involving 3,598 participants were included. On average, 6.67% of individuals had Enterococcus infections, although prevalence varied significantly by region, from less than 1% to over 55%. People with extended hospital stays, catheter use, or diabetes were more likely to become infected. Some antibiotics, such as cephalexin and co-trimoxazole, were found to be effective against Enterococcus. However, others, like oxacillin and norfloxacin, showed high resistance rates and were less effective. Conclusion: Enterococcus infections are present across Ethiopia, with prevalence rates varying by region. While certain antibiotics remain effective, high resistance to others is a growing concern. The study underscores the urgent need for improved infection control measures and more judicious use of antibiotics to limit the spread of resistant infections.
Introduction
Enterococci species, a group of Gram-positive bacteria, are significant contributors to healthcare-associated infections (HAIs) globally. The two most clinically relevant species, Enterococcus faecalis and E. faecium, exhibit markedly different resistance profiles. While E. faecalis is more commonly associated with community-acquired infections 1 and remains relatively susceptible to many antibiotics, E. faecium frequently displays multidrug resistance and is the primary species linked to vancomycin-resistant enterococci (VRE) in hospital settings.1,2 They predominantly infect immunocompromised patients and individuals with prolonged hospital stays or those exposed to invasive procedures, manifesting primarily as urinary tract infections (UTIs), bacteremia, endocarditis, and wound infections. 3 Among these, UTIs are particularly common.3,4 The emergence of vancomycin-resistant enterococci (VRE) has intensified concerns worldwide, presenting treatment challenges due to the limited antibiotic options available. 5 Vancomycin resistance in enterococci is primarily mediated by the acquisition of vanA and vanB gene clusters, which alter the target site of vancomycin by modifying the terminal dipeptides of peptidoglycan precursors from D-Ala-D-Ala to D-Ala-D-Lac or, less commonly, D-Ala-D-Ser, thereby reducing the drug’s binding affinity. 6 These resistance mechanisms are critical for interpreting antimicrobial susceptibility testing (AST) results and guiding appropriate therapeutic strategies.6,7 Global studies report a high prevalence of VRE in clinical settings, with primary risk factors including long hospitalizations, prior antibiotic exposure, and invasive procedures. 3 In Ethiopia, however, research on the prevalence and antimicrobial susceptibility patterns (AST) of enterococci, especially VRE, remains limited. Existing data indicate a notable presence of VRE, particularly in hospital settings,3,5 where resistance complicates treatment and increases morbidity and mortality risks. 8
Given the global burden and rising antimicrobial resistance, understanding the epidemiology of VRE in low-resource settings such as Ethiopia is crucial, where limited healthcare infrastructure may exacerbate these challenges. 9 In Ethiopia, AST data reveal alarming resistance trends in enterococcal isolates. Studies highlight high resistance levels to essential antibiotics such as ciprofloxacin, with a resistance rate of 98%, and penicillin, with an 86% resistance rate. 3 Additionally, enterococci exhibit substantial resistance to tetracycline, chloramphenicol, and amoxicillin/clavulanate, posing treatment challenges in low-resource settings. 5 Urine samples carry the highest rate of enterococcal infection at 54.5%, reflecting the prevalence of UTIs among these isolates, particularly among diabetic patients, who are more susceptible due to weakened immune defenses. 10 Other immunocompromising conditions, such as advanced age, malignancies, organ transplantation, chronic kidney disease, HIV/AIDS, and the use of immunosuppressive therapies, have also been associated with an increased risk of Enterococcus infections. 11 Associated risk factors for these infections include prolonged hospital stays (AOR: 5.23, p-value: 0.035), previous catheterization (AOR: 3.5, p-value: 0.042), and prior antibiotic use, all of which substantially increase the risk of VRE and multidrug-resistant (MDR) enterococcal infections.12,13 The increase in multidrug-resistant (MDR) microorganisms triggering infections is growing worldwide and becoming more serious in developing countries.14 –16 Patients with chronic illness also face heightened vulnerability, with an odds ratio of 16.21 (p-value: 0.001), underscoring the complex interplay between patient health status and infection risk. 3
The limited data on region-specific prevalence and AST patterns of enterococci in Ethiopia present significant knowledge gaps that hinder the development of effective infection control strategies. Considerable variability has been reported in infection rates across Ethiopian regions, likely influenced by geographic and healthcare system differences across the regions. 12 These gaps highlight the need for systematic research to capture the burden of antimicrobial resistance in Enterococci across healthcare and community settings. This systematic review and meta-analysis therefore seek to address the main research questions: (1) What is the current prevalence of enterococcal infections in healthcare and community settings in Ethiopia? (2) What are the AST patterns for multiple antibiotics against enterococcal infections? (3) Which specific risk factors are associated with enterococcal infections? Addressing these questions will increase our understanding of the enterococcal burden in Ethiopia, providing evidence-based recommendations to inform targeted infection control and antimicrobial stewardship strategies.
This systematic review and meta-analysis are justified by the urgent need to synthesize the fragmented data on enterococcal infections in Ethiopia, enabling a comprehensive understanding of prevalence, AST patterns, and associated risk factors. By providing an evidence-based assessment of enterococcal prevalence and resistance, this study helps inform targeted infection control measures and optimize antimicrobial stewardship in Ethiopia. Addressing these gaps is significant for public health and clinical practice, offering perceptions to policymakers and healthcare providers to curb the spread of multidrug-resistant enterococci (MDR) and enhance patient outcomes in Ethiopian healthcare and community settings.
The objectives of this study were to determine the prevalence of Enterococcus species infections in both healthcare and community settings across various regions in Ethiopia, evaluate the antimicrobial susceptibility patterns of Enterococcus species isolated in these settings, and identify and analyze the associated risk factors for Enterococcus infections in the country.
Methods
Eligibility
Only original, peer-reviewed studies, both observational (cross-sectional, cohort, case-control) and experimental, that investigated the prevalence, antimicrobial susceptibility testing (AST) patterns, and associated risk factors of Enterococcus species in healthcare and community settings in Ethiopia within the last 5 years and published in English and all age groups were included. Studies were excluded if they were reviews, books, book chapters, literature reviews, short communications, conference papers, non-peer-reviewed sources, or unpublished/gray literature. In addition, original articles that did not report data on Enterococcus prevalence or AST patterns were excluded. All studies not published in English were also excluded.
Search strategy, study selection process, and information sources
The databases searched to identify pertinent evidence included Scopus, PubMed, Web of Science, Cochrane Library, and the search engine Google Scholar. Each source was last searched on October 30, 2024. The complete search strategies applied to each database are shown in Supplemental File 1. We used a combination of Medical Subject Heading (MeSH) terms, free text words, Boolean operators, and specific filters to retrieve the most relevant, recent, and original studies on Enterococcus species in healthcare and community settings in Ethiopia.
An independent systematic approach was followed to ensure transparency and minimize bias in the study selection process. In this review, the process for deciding whether a study met the inclusion criteria was meticulously structured and involved a systematic approach. The search results from multiple sources were merged using the EndNote 20 reference management software, and duplicates were removed. Titles and abstracts were then screened to remove obviously irrelevant studies, with the authors being intentionally overinclusive during this stage. Afterward, the full texts of the potentially relevant studies were retrieved for further assessment https://training.cochrane.org/handbook/current/chapter-04#section-4-6-3. Eight reviewers: ZH, WY, BAT, AD, AA, HB, YL, and ST independently screened each record and report retrieved, ensuring a thorough evaluation of study eligibility. To maintain objectivity, all reviewers worked independently throughout the process. In cases where there were multiple reports for the same study, these were linked together to avoid double-counting. The full texts were carefully examined to ensure compliance with the predetermined eligibility criteria. For studies with incomplete or unclear data, the team reached out to the study investigators for clarification. Disagreements over eligibility for inclusion were settled through discussion. When there was a debate about interpretation, three reviewers: BMS, AY, and AM acted as arbitrators. Following this process, the final inclusion decisions were made, and data collection proceeded. Additionally, completed studies were added to the included studies table, depending on eligibility criteria https://training.cochrane.org/handbook/current/chapter-04#section-4-6-4. The PRISMA 2020 guidelines (https://www.prisma-statement.org/prisma-2020-flow-diagram) were strictly followed starting from evidence identification to inclusion (Figure 1).

PRISMA flow diagram.
Data extraction and management
In the systematic review process, eight independent reviewers, GAA, DA, YA, MJ, BA, MG, TB, and MK, were involved in collecting data from various reports. Each reviewer worked independently to extract relevant data, which was systematically recorded in an Excel spreadsheet. This structured approach ensured that all pertinent information was captured consistently across the different reports. In cases where discrepancies arose between the data collected by the reviewers, arbitration was conducted by three designated individuals, MTA, TH, and KE, who reviewed the conflicting data to reach a resolution. To confirm or obtain additional data, reviewers contacted study investigators directly, utilizing email or phone communications to clarify uncertainties or request further information that was not adequately covered in the reports. All communications with study investigators were meticulously documented, providing a transparent record of inquiries and responses, which contributed to the reliability of the data collection process. Finally, the collected data, including the Excel spreadsheets and correspondence with investigators, were archived to allow for future access and sharing, ensuring that the data could be revisited for further analysis or verification by other researchers (https://training.cochrane.org/handbook/current/chapter-05).
Data analysis
This systematic review and meta-analysis were designed with a rigorous data extraction approach, aiming to capture a comprehensive set of outcome domains across all included studies. The extraction process involved meticulous planning to ensure all relevant information (from study characteristics to outcome data) was systematically gathered, in alignment with the best practices established in the Methodological Expectations of Cochrane Intervention Reviews (MECIR) and the PRISMA guidelines (https://www.prisma-statement.org/prisma-2020-statement) (Supplemental File 5). By adhering to these standards, the review sought to achieve both transparency and reproducibility, enabling findings that are robust, reliable, and valuable for healthcare stakeholders and review users.
To provide a solid foundation for analysis, key study characteristics were extracted, including author(s), year of publication, geographic region, study design, sampling technique, and sample size. These details establish the context of each study, crucial for evaluating methodological quality and comparability. Sample type and associated risk factors, such as catheterization history, comorbid conditions, and pre/postoperative drainage, were recorded to identify population susceptibility to Enterococcus infections.
For statistical analysis, data on adjusted odds ratios (AORs) and p-values were collected when reported, providing insight into the strength and significance of associations between risk factors and prevalence or resistance patterns of Enterococcus species. Additionally, counts of positive and negative samples were recorded, allowing for the calculation of prevalence rates across studies. Antimicrobial susceptibility testing results for multiple antibiotics, including ciprofloxacin, penicillin, chloramphenicol, erythromycin, tetracycline, vancomycin, and more, were gathered, with resistance, intermediate, and sensitivity rates documented.
All outcomes compatible with the specified domains were collected from each study, covering relevant measures, time points, and analyses. In cases where multiple measures or time points existed, preference was given to those reflecting the primary or most comprehensive findings, supporting the depth and accuracy of the analysis. Additional variables, such as funding sources (if reported) and participant demographics, were noted to assess potential biases and contextualize resistance patterns across subgroups within the population.
The review’s approach to missing or unclear data followed guidelines from Chapter 5 of the Cochrane Handbook (https://training.cochrane.org/handbook/current/chapter-05). When AORs or p-values were absent, they were noted as missing without imputation, ensuring the integrity of the data synthesis. For missing AST data on specific antimicrobials, it was assumed that these elements were outside the scope of the study rather than reflecting resistance absence, preserving the primary intent of each study. The comprehensive planning and adherence to established reporting standards facilitated the construction of structured tables and figures, bias assessments, and both qualitative and quantitative syntheses, including meta-analyses.
Quality assessment
To assess the risk of bias in cross-sectional studies for this systematic review and meta-analysis, four independent reviewers (ZH, AA, HB, and TH) evaluated each study separately using the Risk Of Bias In Non-randomized Studies-I (ROBINS-I) tool to ensure transparency, consistency, and minimize subjective bias. The assessment focused on several key domains. First, confounding bias was evaluated by examining whether studies controlled for essential confounders, such as socioeconomic status and health conditions. Studies lacking adequate control received higher risk ratings. Selection bias was considered next, with reviewers looking at how participant selection might introduce biases that affect exposure and outcome, potentially limiting generalizability. Studies with selection issues were rated moderate to high risk. In the measurement (information) bias domain, reviewers assessed the accuracy and consistency of exposure and outcome measurements, with studies using validated tools rated as low risk, and those with unvalidated measures rated higher. Bias due to deviations was reviewed to identify any unplanned changes in methodology or co-interventions, with studies following original protocols rated low risk and those with significant deviations rated higher. Bias due to missing data was also considered, with studies minimizing or properly handling missing data receiving low risk, while those with considerable missing data were rated higher. Outcome measurement bias (detection bias) was examined by determining whether outcomes were measured consistently across groups and whether exposure knowledge influenced assessments, with studies using standardized, blinded methods rated low risk. Finally, reporting bias was evaluated to check whether outcomes and analyses were reported as planned or selectively, with studies providing comprehensive reports receiving low risk. After independently assessing each domain, an overall risk-of-bias judgment for each study was derived by synthesizing the ratings across domains. The final judgment, categorized as Low, Moderate, Serious, Critical, or No information if details were missing, was based on the highest risk level across domains, ensuring a clear and justified summary of the overall risk. (https://training.cochrane.org/handbook/current/chapter-25#section-25-3).
In assessing the risk of bias due to missing results in a systematic review and meta-analysis of cross-sectional studies, funnel plots were used to detect potential reporting biases. These plots can highlight asymmetry, which might indicate selective non-reporting or underreporting of results, often due to p-values or the direction of the findings. However, asymmetry could also stem from other factors, such as study design issues. To address this, sensitivity analyses were performed, and efforts to locate unpublished studies or missing data from various sources were conducted to quantify the impact of these biases (https://training.cochrane.org/handbook/current/chapter-13).
To assess the certainty (or confidence) in the body of evidence for an outcome in a systematic review and meta-analysis from cross-sectional studies, the Grades of Recommendation, Assessment, Development and Evaluation (GRADE) approach is used. This method evaluates five key domains: risk of bias, consistency of effect, imprecision, indirectness, and publication bias.
The first domain, risk of bias, assesses how the design, execution, and methodological quality of studies impact the reliability of the results. Cross-sectional studies, being nonrandomized, are often prone to biases such as selection bias, measurement bias, and confounding, which may lower the certainty of evidence. The second domain, consistency of effect, examines whether the results across different studies are consistent or show significant variability (heterogeneity). If there is substantial variability in the results, this suggests inconsistency, which leads to downgrading the certainty of the evidence.
Imprecision, the third domain, addresses uncertainty in the effect estimates, especially when studies have small sample sizes or wide confidence intervals. In these cases, the estimates may be less precise, leading to a potential downgrade in the certainty of evidence. The fourth domain, indirectness, considers whether the evidence directly applies to the population, intervention, or outcome of interest. If the studies do not align well with the review’s target population or context, the evidence may be downgraded due to indirectness.
Finally, publication bias examines whether selective reporting or non-publication of studies has distorted the evidence. If there is concern that only positive results are published, or if negative studies are missing, the certainty of the evidence may be reduced. The GRADE approach typically begins with low certainty for nonrandomized studies, such as cross-sectional studies, due to their inherent biases. However, the certainty rating can be upgraded in certain cases, such as when studies show a large effect size, a clear dose–response relationship, or minimal confounding. (https://training.cochrane.org/handbook/current/chapter-14#section-14-2).
Statistical analysis
Effect measures were calculated to quantify the magnitude and direction of prevalence, AST patterns, and associated risk factors of Enterococci species across multiple studies in healthcare and community settings in Ethiopia. They were used to synthesize findings and estimate an overall effect regarding the objectives of this review. The effect measures like standard error, odds ratio (OR), AOR, SE of the pooled log-adjusted odds ratio (log-AOR), pooled log-AOR at 95%, p-value, Egger’s regression test, pooled p-value, pooled standard deviation, pooled standard error, mean age of participants along with standard deviation, total sample size, pooled prevalence, and overall prevalence were computed. Moreover, considerations for effect measures (interpretability, study heterogeneity, study design, reporting, subgroup analyses, and sensitivity) were sought.
In meta-analysis, the choice of synthesis model (random-effects or fixed-effect) was determined by the level of statistical heterogeneity across studies. Statistical heterogeneity was assessed using Cochran’s Q test and the I2 statistic. The I2 statistic quantifies inconsistency, with values of 0%–40% indicating low heterogeneity, 30%–60% moderate, 50%–90% substantial, and 75%–100% considerable heterogeneity. High heterogeneity supports using a random-effects model, while low heterogeneity favors a fixed-effects model to obtain reliable conclusions (https://handbook-5-1.cochrane.org/chapter_9/9_5_2_identifying_and_measuring_heterogeneity.htm). In light of the considerable heterogeneity observed (I2 = 92.31%), a random-effects model was used for the meta-analysis to account for variability across studies and provide more generalizable summary estimates. Moreover, to explore heterogeneity in meta-analysis, several strategies were employed. These include subgroup analysis to examine the influence of specific study characteristics, and meta-regression to assess the impact of continuous variables. Other methods include changing the effect measure, excluding outlying studies, or conducting sensitivity analyses to assess the robustness of findings (https://handbook-5-1.cochrane.org/chapter_9/9_5_3_strategies_for_addressing_heterogeneity.htm).
Operational definition
Healthcare settings refer to hospitals, clinics, health centers, and other formal medical institutions where patients receive care and where clinical specimens are collected.
Community settings refer to nonhospital environments, including households, schools, and other public or communal environments where enterococcal infections may be studied or reported outside of institutional healthcare facilities.
Incomplete data refer to studies in which most of the pertinent information related to our research question or objectives was missing, such as missing data on antimicrobial susceptibility or prevalence.
Inappropriate data refer to studies in which the findings were interpreted in a way that did not align with our inclusion criteria where results were not applicable to our context.
Results
Study population and characteristics
The study selection process began with identifying 197 records from various databases: Scopus (85), PubMed (35), Web of Science (49), Cochrane Libraries (11), and Google Scholar (17). No additional records were found from other sources. After removing duplicates, 120 unique records were screened. During screening, 113 records were excluded as they did not meet the inclusion criteria, which excluded reviews, books, book chapters, literature reviews, short communications, and conference papers. The remaining 17 full-text articles were assessed for eligibility, and four articles were excluded after full-text reading with reason, that is, out of scope (n = 1), incomplete data (n = 1), and inappropriate (n = 2). Eventually, 13 studies met all criteria and were included in the review.
In the review process, several studies initially appeared to meet the inclusion criteria but were ultimately excluded for specific reasons. Studies that might appear to meet the inclusion criteria, but which were excluded, and why they were excluded are explained in Supplemental File 2.
The study characteristics in a systematic review and meta-analysis of cross-sectional studies include several key components for clear reference and comparability. Each study is assigned a unique study ID for easy reference throughout the review. The Citation includes the author(s), publication year, and a reference number to ensure straightforward retrieval. The Setting column indicates where each study was conducted and the specific environment, such as a community or healthcare facility. Details on the Study Population cover characteristics such as age range and specific groups (e.g., hospitalized patients or general community members). Sample Size records the number of participants in each study, and study design usually identifies cross-sectional designs, noting any additional design elements if present. Key Findings summarize the primary results relevant to the review’s objectives, while the column listing risk factors evaluated lists specific factors examined in each study. The prevalence (%) column shows the reported prevalence of the outcome of interest, and other remarks provide additional context, including bias assessment, limitations, or special considerations. This structured approach allows for a thorough, concise comparison of each study’s main attributes and findings (see Supplemental File 3). Supplemental File 4 offers a clear and organized overview of the risk of bias assessments for each study, facilitating the visualization of potential biases across studies in the systematic review and meta-analysis.
Meta-analysis
The 13 cross-sectional studies used varied sampling methods (random, systematic, consecutive, convenience) with sample sizes ranging from 170 to 403, totaling 3598 participants from diverse Ethiopian regions (mean age 29.3 ± 6.6 years; 1782 males, and 1816 females). While most studies demonstrate methodological rigor, risk of bias arises from the use of convenient sampling in some cases, potentially limiting representativeness. Variability in sample sizes and differences between hospital and community settings may contribute to heterogeneity, influencing study outcomes (I2 = 92.31%, τ2 = 5882.35; Q = 156.00, p = 0.01).
The pooled prevalence of Enterococcus species across all studies was 6.67. Main associated risk factors identified include prolonged hospital stays, catheterization history, chronic illnesses, diabetes, and prior antibiotic usage. Statistical analyses reveal significant associations, with a pooled log-AOR of 0.986 and a pooled p-value of 1.31 × 10–2. This log-AOR of 0.986 was derived from a multivariable meta-analysis of AORs reported across studies. This value corresponds to an AOR of approximately 2.68 (e^0.986), indicating that the exposure was associated with 2.68 times higher odds of the outcome, after controlling for confounding variables. Notable odds ratio (OR) observed were 3.6 (95% CI: 1.8–7.1) for catheterization history, and 3.92 (95% CI: 2.1–7.3) for alcohol consumption. These findings indicate that catheterization, and alcohol use are significantly associated with an increased risk of Enterococci infections, underscoring the multifactorial nature of risk in both healthcare and community settings.
Antimicrobial susceptibility test patterns revealed variable sensitivity and resistance trends. Cephalexin and co-trimoxazole stand out with 100% sensitivity, suggesting they are highly effective against the Enterococci strains tested. Similarly, ceftriaxone (85%) and meropenem (85.71%) show high sensitivity with minimal resistance, indicating strong effectiveness. Vancomycin (69.94%) and nitrofurantoin (75.19%) also display relatively high sensitivity, making them favorable choices for many cases.
Conversely, some antibiotics are associated with high microbial resistance, indicating potential limitations. Ampicilin (67.63%), penicillin (64.24%), and erythromycin (54.36%) are associated with high microbial resistance, suggesting reduced effectiveness against several strains of Enterococci. Notably, norfloxacin and trimethoprim-sulfamethoxazole, each have 80% resistance, while oxacillin reaches 85.71% resistance with very low sensitivity, indicating these drugs may be largely ineffective.
A few antibiotics show mixed patterns. Doxycycline (53.24% sensitivity and 45.32% resistance) has a balanced profile, while ciprofloxacin and chloramphenicol have close to a 50% split between sensitivity and resistance, reflecting inconsistent effectiveness. Gentamycin also has a near-even response, with 44.44% sensitivity and 55.56% resistance. Amikacin demonstrates high sensitivity (71.43%) but has notable resistance (28.57%), suggesting it may be effective in most but not all cases. Azithromycin presents moderate sensitivity (40%) alongside a high resistance rate (60%), implying limited utility. Clindamycin has an even split, making it variably effective depending on the strain.
In summary, cephalexin and co-trimoxazole are particularly promising due to their 100% sensitivity, whereas Norfloxacin, trimethoprim-sulfamethoxazole, and oxacillin show high resistance, suggesting these may be less viable treatment options. Table 1 shows AST patterns of Enterococci species against different antimicrobials.
Antibiotic sensitivity, intermediate sensitivity, and resistance patterns (counts and percentages) of Enterococcus species.

The systematic review and meta-analysis on the prevalence, antimicrobial susceptibility patterns, and associated risk factors of Enterococci species in healthcare and community settings in Ethiopia reveal insightful findings. The overall pooled prevalence of Enterococci across the studies was noted at 5.50%, with individual study prevalence ranging widely from 0.03% to 55.88%, indicating substantial variation across different settings and sample populations. Sample sizes varied between 170 and 403 individuals, with age ranges spanning from 1 to 83 years, allowing for broad representation across age demographics. Certain studies focused on specific age groups, such as preschool children with a mean age of 6 years, secondary school students, and hospitalized individuals with an average age of 40.9 years. Gender distribution was nearly balanced across studies, with males and females each well represented; for instance, a sample of 323 participants included 186 males and 137 females.
In examining associated risk factors, several health conditions and procedures were significantly linked with Enterococci presence. Notably, a history of catheterization showed a strong association, with an OR of 3.5, and extended hospital stays (over 48 h) were also significant, with an OR of 6. Diabetes mellitus, particularly in cases of prolonged duration, further indicated a considerable risk. Behavioral and demographic factors, such as invasive procedure history (OR = 21.2), alcohol consumption (OR = 3.92), and consuming uncooked food (OR = 8.6), were also associated with elevated infection risks. Lower education levels (secondary school and below) were linked to a higher prevalence, with an OR of 12.8. Additionally, age-based trends indicated higher susceptibility among preschool children and elderly patients, who showed increased infection rates.
The analysis of associated risk factors included pooled log-AORs with a consistent pooled log-AOR of 0.394 across studies, highlighting the overall association of certain characteristics against Enterococci strains infection. This finding was supported by a pooled p-value of 0.001, confirming the statistical significance of these characters, which signals potential concerns over antimicrobial resistance against Enterococci species in the Ethiopian healthcare context. Overall, this analysis underscores the importance of targeted interventions in high-risk groups and settings, considering the identified prevalence, associated risk factors, and evolving resistance patterns of Enterococci species.
Summary estimates and its precision
The meta-analysis included 13 studies with a total sample size of 3598 participants. The pooled prevalence of Enterococcus species infections was 6.67% (95% CI: 0.50–8.85). The pooled log-AOR for associated risk factors was 0.986 (95% CI: 0.214–1.759), indicating a statistically significant association between the risk factors and the prevalence of Enterococcus infections. The pooled p-value of less than 0.001, confirming the significance of these findings.
However, statistical heterogeneity among the included studies was substantial, as indicated by an I2 statistic of 92.31% and a significant Q-test result (Q = 156). The tau-squared (τ2) value was 5882.35, reflecting considerable variability in the true effect sizes. This heterogeneity highlights the diversity in study populations, methodologies, and regional factors, which should be carefully considered when interpreting the results. The meta-analysis results in a summary estimate, with a precision indicated by a (95% confidence interval), the measures of statistical heterogeneity (I2), the Q-test, and the τ2 value are shown in Table 2.
Each of the summary estimate and its precision and measures of statistical heterogeneity.
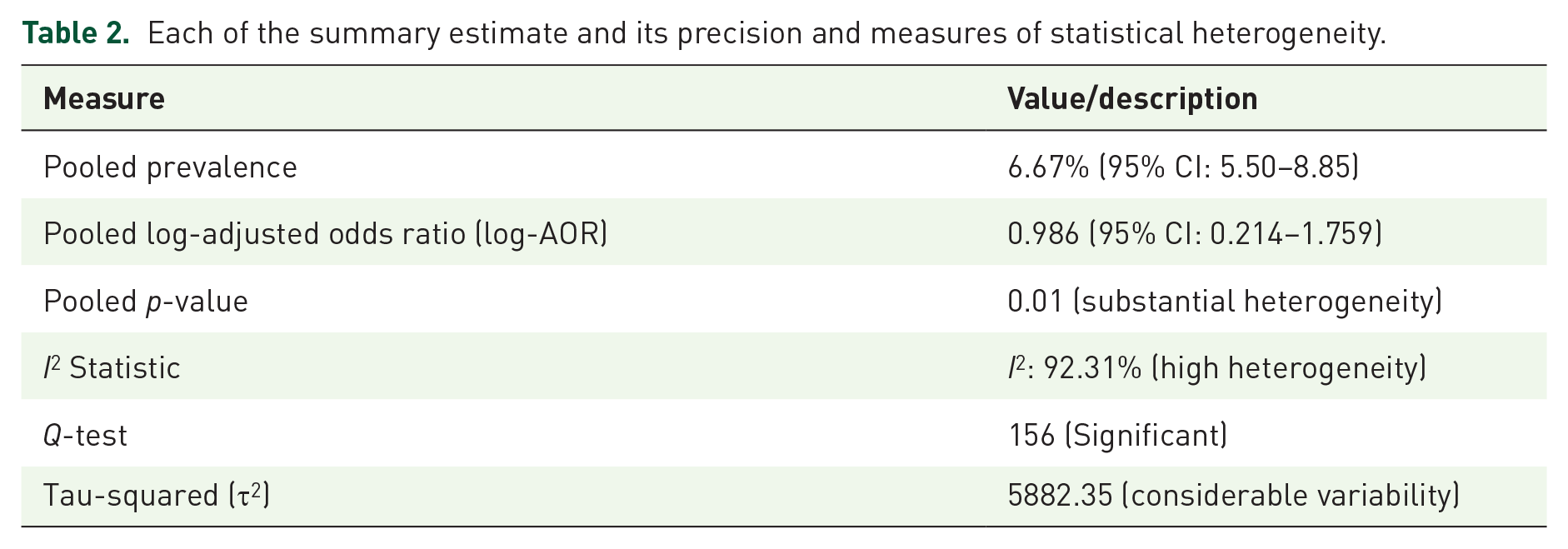
The heterogeneity in this analysis suggests that 92.31% of the variability in effect sizes across studies is due to heterogeneity rather than random sampling error, as indicated by the I2 statistic and p-value. The τ2 value reflects the estimated variance of the true effect sizes, providing an estimate of the degree of variation among the studies’ true effects. Additionally, the significant Q-test result, with a value of 156, confirms the presence of substantial heterogeneity, indicating that the studies differ from one another in ways that go beyond what would be expected by chance alone.
Measures of statistical heterogeneity and publication bias
Figure 2 presents a forest plot summarizing the effect sizes and 95% confidence intervals (CIs) for 13 individual studies. Each study’s effect size is represented by a blue square, with the size of the square reflecting its relative weight in the meta-analysis (approximately 7.69% for each study). Horizontal lines indicate the 95% CIs, and studies with wider lines have less precise estimates. The pooled effect size is displayed as a green diamond at the bottom of the plot, with a value of 276.77 (95% CI: 233.37–320.16), indicating a statistically significant overall effect.

Forest plot of effect sizes across studies using a random-effects model.
Heterogeneity statistics reveal considerable variability between studies (I2 = 92.31%, τ2 = 5882.35), suggesting that the observed differences in effect sizes are not solely due to random chance. Cochran’s Q-test (Q = 156.00, p = 0.01) further confirms significant heterogeneity. Despite this variability, the test of the overall effect (z = 12.50, p < 0.01) demonstrates strong evidence of an effect, with the overall estimate being statistically significant. These findings highlight the importance of considering underlying differences between studies when interpreting the results.
The Galbraith plot (Figure 3) provides a visual representation of the relationship between individual study effect sizes and their precision, offering insights into heterogeneity and potential outliers. The central line in the plot represents the overall effect size (pooled estimate) derived from the meta-analysis, with studies closer to this line demonstrating alignment with the pooled effect. Confidence bands, typically shown as dashed lines, represent the 95% confidence limits, within which most studies should ideally fall if there is low heterogeneity. The plot reflects the high heterogeneity reported in the meta-analysis (I2 = 92.31%). A noticeable spread of points indicates variability in effect sizes across studies, which may arise from differences in study design, populations, or settings. There are no studies that appear to deviate significantly from the central trend line and fall outside the confidence bands, suggesting the absence of outliers.

Galbraith plot.
The Galbraith plot presented in Figure 3 illustrates the relationship between study effect sizes and their precision, aiding in the detection of potential outliers and assessment of heterogeneity. In this analysis, the majority of the studies remained within the confidence bands, indicating no significant deviations or potential outliers. Despite the high heterogeneity reported in the meta-analysis (I2 = 92.31%), the absence of studies falling outside the confidence limits in the Galbraith plot suggests that the variability observed across studies is not driven by individual outlier characteristics. This lack of apparent outliers eliminates the need for sensitivity analyses to exclude such studies. The observed heterogeneity is likely attributable to genuine differences in study populations, settings, or methodologies rather than extreme or aberrant results from specific studies. This finding supports the integrity of the pooled estimates and reinforces the robustness of the meta-analysis conclusions despite the high level of heterogeneity.
Subgroup analysis
The subgroup analysis examines the antimicrobial susceptibility testing (AST) patterns of Enterococcus species by categorizing them into three subgroups: sensitive, intermediate, and resistant. Each subgroup represents the AST outcomes for various antibiotics tested across included studies, with the numbers indicating the total count of Enterococcus species in each category.
Statistical evaluation of subgroup differences highlights significant variability between the sensitive, intermediate, and resistant subgroups, with a test of subgroup differences yielding a p-value < 0.05 (Figure 4). This indicates that the observed variations in AST patterns are unlikely to be due to chance. The heterogeneity within each subgroup, as indicated by I2 values, was lower than the overall heterogeneity reported in the meta-analysis (I2 = 92.31%), suggesting that categorizing by AST patterns helps explain some of the variability among studies.
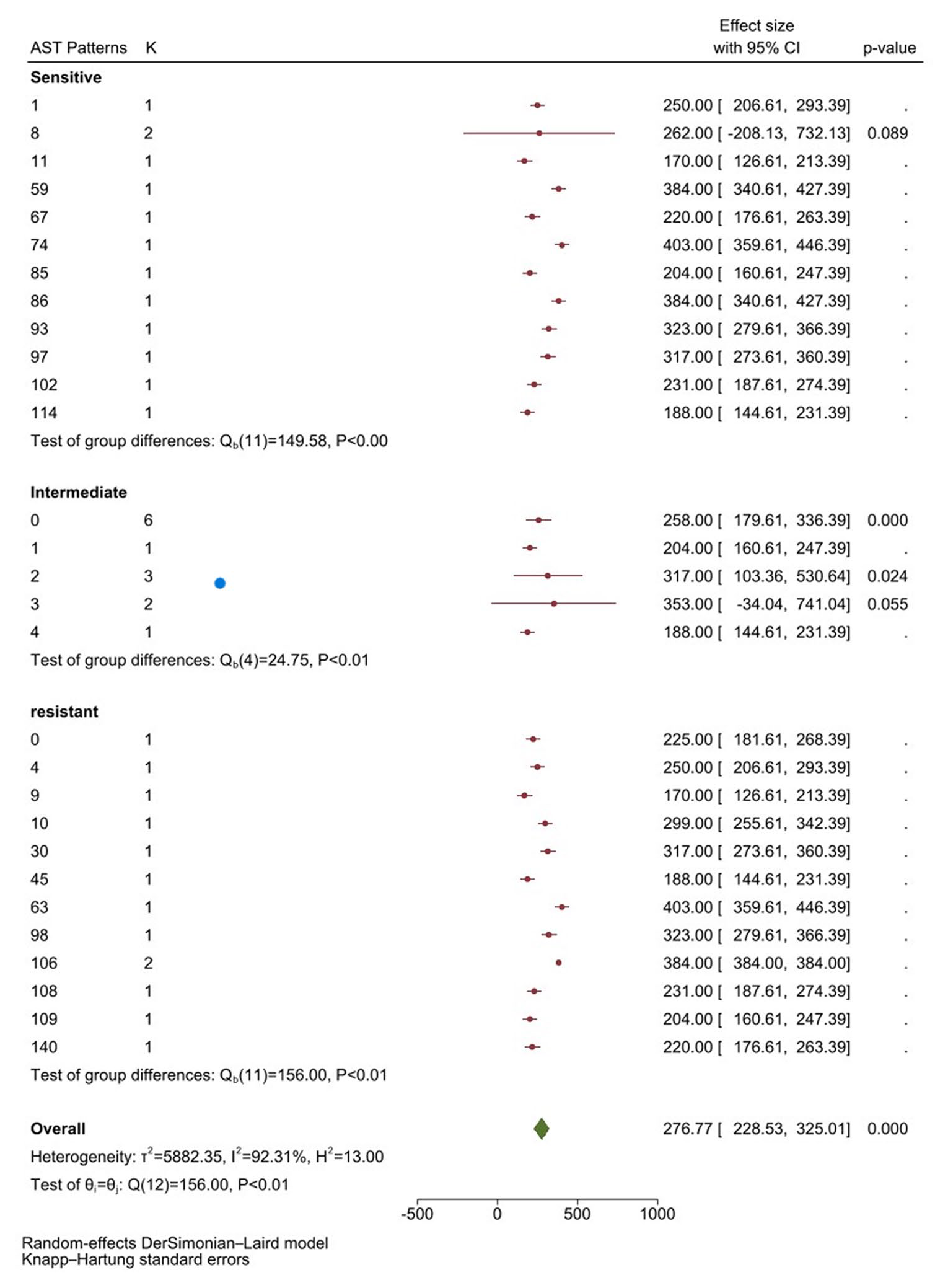
Subgroup analysis by AST.
Reporting biases
The funnel plot (Figure 5) shows slight asymmetry, with more studies appearing on one side of the pooled effect size line, which could indicate potential publication bias. This asymmetry reflects true heterogeneity across the studies rather than bias alone. The plot also displays greater dispersion at the bottom, representing smaller studies with larger standard errors, which is typical in meta-analyses and points to potential small-study effects. These small studies often show more variability in effect sizes due to methodological differences or biases. To address this, a trim-and-fill analysis was conducted, identifying two potentially missing studies. When these studies were added, the pooled effect size shifted slightly, but the overall conclusions of the meta-analysis were not significantly altered, suggesting that while some bias may exist, it has minimal impact on the results. Additionally, the wide spread at the bottom of the plot corresponds with the high heterogeneity (I2 = 92.31%) observed in the analysis, reflecting variability in study characteristics such as sample size, populations, or methodologies, rather than random variation. In conclusion, while the funnel plot suggests the presence of some publication bias or small-study effects, the results of the trim-and-fill analysis indicate that these issues do not critically undermine the validity of the meta-analysis. The observed asymmetry reflects genuine differences across studies rather than systematic reporting bias, supporting the robustness of the meta-analysis findings.

Funnel plot.
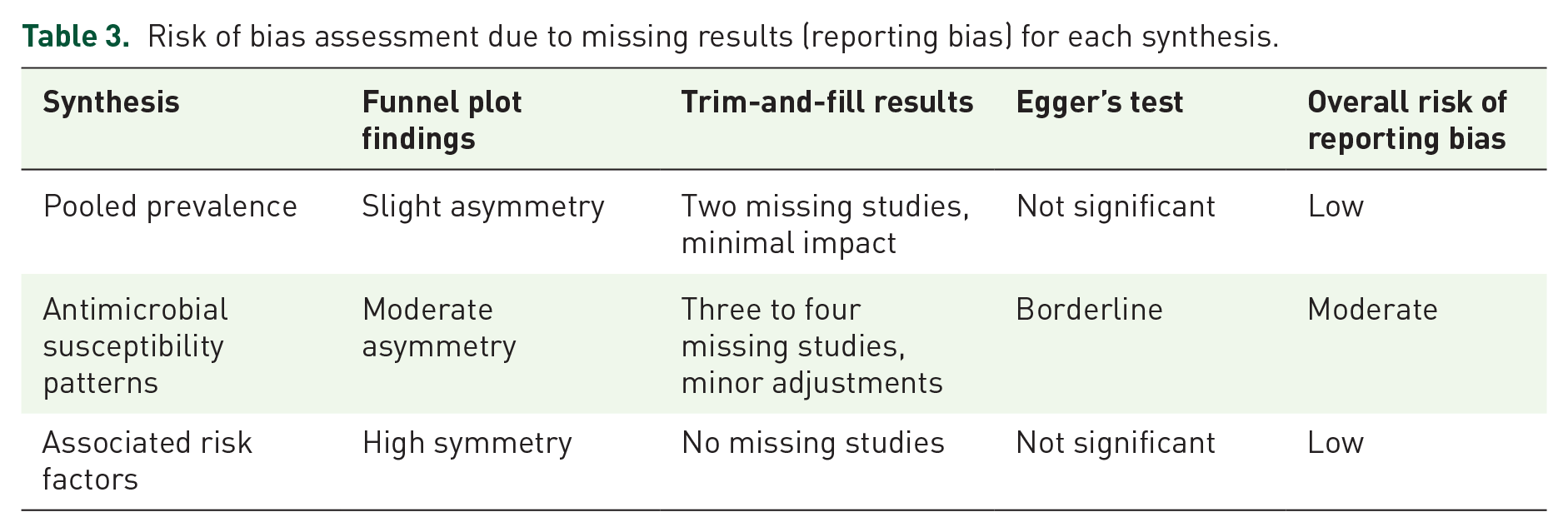
Risk of bias assessment due to missing results (reporting bias) for each synthesis
The risk of bias due to missing results (reporting bias) in each synthesis is evaluated by assessing whether all relevant outcomes are reported. Studies that selectively report only positive or significant findings increase the risk of bias. Incomplete reporting can lead to an overestimation of treatment effectiveness or skew results. To minimize reporting bias, comprehensive outcome reporting and examining any potential discrepancies between registered protocols and published results were ensured. Table 3 shows the risk of bias assessment due to missing results (reporting bias) for each synthesis.
Risk of bias assessment due to missing results (reporting bias) for each synthesis.
Certainty of evidence
The certainty in the body of evidence for outcomes assessed in the systematic review and meta-analysis was evaluated using the GRADE approach, focusing on risk of bias, consistency of effect, imprecision, indirectness, and publication bias. The evidence for the prevalence of Enterococci species was rated as moderate, with low risk of bias and minimal indirectness, though substantial variability in prevalence rates (0.03–55.88) suggested inconsistency, and some imprecision arose from wide confidence intervals. Antimicrobial susceptibility patterns also had moderate certainty due to consistent methodology and low publication bias, but mixed resistance trends and variable precision affected the rating. The associated risk factors, including catheterization, chronic illness, and prolonged hospital stays, showed moderate certainty, with clear methodologies and direct applicability to the Ethiopian healthcare context, though imprecision was noted in the variability of odds ratios. For pooled AORs, the evidence ranged from moderate to high certainty, with low bias, high consistency across studies, and significant p-values providing robust precision and confidence. Overall, the evidence is reliable but constrained by some variability and imprecision, underscoring the need for further targeted research.
Discussion
This systematic review and meta-analysis on Enterococci species in Ethiopia highlight the significant findings. The pooled prevalence of Enterococci infections was 6.67% (95% CI: 5.85–7.49). This was in line to studies conducted by Ashagrie et al. (6.77%). 17 The prevalence in this study was higher than previously reported studies by Woreta et al. (1.5%), 18 Kiros T et al. (2.7%), 19 Oumer et al. (2.2%), 13 Abera et al. (2.7%), 5 Kebede T et al. (1.5%), 20 Melaku TM et al. (2.7%), 21 Weldu Y et al. (0.3%), 22 Worku S et al. (1.2%), 23 G/Medhin (0.9%),10,23,24 and Girma et al. (0.7%). 25 However, it was lower than reported by Tilahun et al. (8.9%) 3 and Zike et al. (55.9%). 12 The observed variability in the prevalence of Enterococci infections across different studies in Ethiopia may be attributed to sampling methods (such as random or systematic approaches versus convenient sampling that might affect representativeness), geographic differences (with urban areas like Addis Ababa having better healthcare infrastructure), and hospital-based studies show higher infection rates due to invasive procedures and prolonged stays, while community settings often report lower prevalence. Antimicrobial resistance patterns contribute to treatment challenges, and socioeconomic factors, including living conditions, hygiene, and healthcare accessibility, further affect the prevalence of Enterococci species in Ethiopia.
We applied a pooled log-AOR, a statistical measure that summarizes the overall strength of association between risk factors and an outcome by combining data from multiple studies. It accounts for the adjusted effects reported in individual studies and balances out extremely high and low odds ratios, providing a more stable, averaged estimate of risk across diverse settings and populations. The log-AOR of associated risk factors of this review was 0.986 (95% CI: 0.214–1.759), reflecting the combined effect of risk factors across studies while accounting for variability and heterogeneity. This value aligns closely with the AOR for the risk factor of a previous history of urinary tract infection (UTI), which was 1.04. 19 The pooled log-AOR was notably higher than the AORs observed for specific risk factors, such as preschool children (AOR: 0.014), elementary school children (AOR: 0.190), 18 circumcision (AOR: 0.063), 25 and previous antibiotic use (AOR: 0.1), 26 in association with Enterococcal infections. However, it was remarkably lower than the AORs observed for specific risk factors like catheterization (AOR = 56), 17 invasive procedures (AOR = 26.91), chronic illness (AOR = 16.12) and admission (AOR = 13.73), 5 previous history of hospitalization (AOR = 11.9), 12 prolonged hospital stay (AOR = 5.23), 3 and diabetes mellitus (AOR = 5.3). 22 The differences in AORs reflect the varying strengths of association between specific risk factors and Enterococcal infections. High AORs, such as those for catheterization, invasive procedures, and chronic illness, indicate strong associations likely due to these factors directly compromising physical barriers or immunity, thereby increasing susceptibility. Conversely, low AORs for factors like preschool children or circumcision suggest weaker associations, possibly because these factors contribute indirectly or less significantly to infection risk. The pooled log-AOR, representing the combined effect of risk factors across studies, balances these extremes, accounting for variability and heterogeneity while emphasizing the cumulative impact of multiple, sometimes interrelated, risk factors.
The analysis of antibiotic effectiveness against Enterococcus species revealed that the most effective antibiotics were cephalexin and co-trimoxazole, both with 100% sensitivity. This was identical with a report by Woreta et al. 18 In this study, Vancomycin (27.61% resistant) contrasts with a report showing 100% sensitivity.18,24,27,28 The study highlights that the least effective antibiotics against Enterococcus species, characterized by the highest resistance percentages, are oxacillin (85.71% resistant), trimethoprim-sulfamethoxazole (80.00% resistant), and norfloxacin (80.00% resistant). This was in line to a report by Woreta et al. 18 while Gashaw et al. reported the least effective antibiotics were penicillin (80% resistance in Enterococcus spp.) and ampicillin (60% resistance in Enterococcus spp.) provided Enterococcus spp. was 100% resistance to penicillin in the study by Amsalu et al. 27 Antimicrobial susceptibility testing variations against antibiotics arise from multiple factors, including genetic differences among bacterial strains, the mechanism of resistance, and local antibiotic use patterns. Additionally, environmental factors such as selective pressure misuse of specific antibiotics in a given region can promote the dominance of resistant strains. Variations in testing methods, laboratory protocols, and interpretative criteria can also influence AST results.
Implications of this research in a global context: While this review focuses on the Ethiopian context, the findings reflect broader global concerns regarding the prevalence of Enterococcus infections, associated risk factors, rising antimicrobial resistance, and the need for strengthened infection control and antimicrobial stewardship strategies worldwide.
Limitations of the evidence included in the review: The evidence in this review faced significant limitations, including substantial heterogeneity across studies, as indicated by a high I2 statistic (92.31%). This reflects variability in study designs, populations, and regional healthcare practices, which complicates comparisons. Additionally, imprecise effect estimates, caused by small sample sizes or wide confidence intervals in individual studies, reduce the reliability of pooled results. Some studies relied on convenience sampling, potentially limiting representativeness, and publication bias was suggested by asymmetry in the funnel plot, although its impact was assessed as minimal.
Limitations of the review processes used: The review processes were rigorous but not without limitations. The reliance on secondary data limited the ability to validate results directly, and inconsistent data reporting across studies were challenges to address gaps. While the ROBINS-I tool ensured systematic bias assessment, the lack of registered protocols for some studies may have led to deviations in reporting or outcomes. The review also excluded studies published in languages other than English, which might have introduced selection bias.
Implications for practice, policy, and future research: The findings highlight a pressing need for tailored infection control and antimicrobial stewardship strategies, particularly in high-risk groups such as those with prolonged hospital stays, diabetes, catheterization, invasive procedures chronic illness, admission history and previous history of hospitalization. For policy, the results advocate for stricter regulation on antibiotic use to mitigate resistance and emphasize resource allocation toward improving laboratory capacities for AST in low-resource settings. Future research should focus on reducing heterogeneity by standardizing methodologies and incorporating larger, more representative samples. Efforts to address gaps, such as evaluating resistance mechanisms and conducting region-specific studies, will further enhance the evidence base for informed decision-making.
Conclusion
This systematic review and meta-analysis revealed a pooled prevalence of Enterococcus infections of 6.67% across Ethiopian healthcare and community settings, with significant variability in antimicrobial susceptibility patterns and associated risk factors. Key risk factors included prolonged hospital stays, catheterization, and diabetes, underscoring the multifactorial nature of Enterococcal infections. Cephalexin and co-trimoxazole were the most effective antibiotics followed by ceftriaxone and meropenem, whereas high resistance rates were noted for oxacillin, trimethoprim-sulfamethoxazole, and norfloxacin. The findings highlight the urgent need for targeted infection control measures and antimicrobial stewardship strategies to mitigate the impact of antimicrobial resistance, particularly in high-risk populations and healthcare environments.
Future directions
Future research should prioritize region-specific studies to address heterogeneity and gaps in the prevalence and resistance patterns of Enterococcus species. Standardized methodologies, larger representative samples, and longitudinal designs are critical to understanding temporal trends and resistance mechanisms. Additionally, exploring the genetic basis of resistance and evaluating the effectiveness of policy interventions aimed at antimicrobial stewardship will provide actionable insights to curb the spread of multidrug-resistant Enterococci in Ethiopia. Enhanced laboratory capacity and community-based studies are essential to bridging the knowledge gap between healthcare and community settings.
Supplemental Material
sj-docx-1-tai-10.1177_20499361251354905 – Supplemental material for Prevalence, antimicrobial susceptibility patterns, and associated risk factors of Enterococci species in healthcare and community settings in Ethiopia: a systematic review and meta-analysis
Supplemental material, sj-docx-1-tai-10.1177_20499361251354905 for Prevalence, antimicrobial susceptibility patterns, and associated risk factors of Enterococci species in healthcare and community settings in Ethiopia: a systematic review and meta-analysis by Zigale Hibstu Teffera, Wubetu Yihunie Belay, Bantayehu Addis Tegegne, Abebe Dagne, Adane Adugna, Habtamu Belew, Yasabe Leykun, Samrawit Tefera, Bewket Mesganaw Shtie, Abebe Yenesew, Abateneh Melkamu, Gashaw Azanaw Amare, Desalegn Abebaw, Yibeltal Akelew, Mohammed Jemal, Baye Ashenef, Mamaru Getinet, Temesgen Baylie, Mihiretie Kiber, Mamaru Tilahun Afework, Tiruzer Hibistu and Kassaye Enchalew in Therapeutic Advances in Infectious Disease
Supplemental Material
sj-docx-2-tai-10.1177_20499361251354905 – Supplemental material for Prevalence, antimicrobial susceptibility patterns, and associated risk factors of Enterococci species in healthcare and community settings in Ethiopia: a systematic review and meta-analysis
Supplemental material, sj-docx-2-tai-10.1177_20499361251354905 for Prevalence, antimicrobial susceptibility patterns, and associated risk factors of Enterococci species in healthcare and community settings in Ethiopia: a systematic review and meta-analysis by Zigale Hibstu Teffera, Wubetu Yihunie Belay, Bantayehu Addis Tegegne, Abebe Dagne, Adane Adugna, Habtamu Belew, Yasabe Leykun, Samrawit Tefera, Bewket Mesganaw Shtie, Abebe Yenesew, Abateneh Melkamu, Gashaw Azanaw Amare, Desalegn Abebaw, Yibeltal Akelew, Mohammed Jemal, Baye Ashenef, Mamaru Getinet, Temesgen Baylie, Mihiretie Kiber, Mamaru Tilahun Afework, Tiruzer Hibistu and Kassaye Enchalew in Therapeutic Advances in Infectious Disease
Supplemental Material
sj-docx-3-tai-10.1177_20499361251354905 – Supplemental material for Prevalence, antimicrobial susceptibility patterns, and associated risk factors of Enterococci species in healthcare and community settings in Ethiopia: a systematic review and meta-analysis
Supplemental material, sj-docx-3-tai-10.1177_20499361251354905 for Prevalence, antimicrobial susceptibility patterns, and associated risk factors of Enterococci species in healthcare and community settings in Ethiopia: a systematic review and meta-analysis by Zigale Hibstu Teffera, Wubetu Yihunie Belay, Bantayehu Addis Tegegne, Abebe Dagne, Adane Adugna, Habtamu Belew, Yasabe Leykun, Samrawit Tefera, Bewket Mesganaw Shtie, Abebe Yenesew, Abateneh Melkamu, Gashaw Azanaw Amare, Desalegn Abebaw, Yibeltal Akelew, Mohammed Jemal, Baye Ashenef, Mamaru Getinet, Temesgen Baylie, Mihiretie Kiber, Mamaru Tilahun Afework, Tiruzer Hibistu and Kassaye Enchalew in Therapeutic Advances in Infectious Disease
Supplemental Material
sj-docx-4-tai-10.1177_20499361251354905 – Supplemental material for Prevalence, antimicrobial susceptibility patterns, and associated risk factors of Enterococci species in healthcare and community settings in Ethiopia: a systematic review and meta-analysis
Supplemental material, sj-docx-4-tai-10.1177_20499361251354905 for Prevalence, antimicrobial susceptibility patterns, and associated risk factors of Enterococci species in healthcare and community settings in Ethiopia: a systematic review and meta-analysis by Zigale Hibstu Teffera, Wubetu Yihunie Belay, Bantayehu Addis Tegegne, Abebe Dagne, Adane Adugna, Habtamu Belew, Yasabe Leykun, Samrawit Tefera, Bewket Mesganaw Shtie, Abebe Yenesew, Abateneh Melkamu, Gashaw Azanaw Amare, Desalegn Abebaw, Yibeltal Akelew, Mohammed Jemal, Baye Ashenef, Mamaru Getinet, Temesgen Baylie, Mihiretie Kiber, Mamaru Tilahun Afework, Tiruzer Hibistu and Kassaye Enchalew in Therapeutic Advances in Infectious Disease
Supplemental Material
sj-docx-5-tai-10.1177_20499361251354905 – Supplemental material for Prevalence, antimicrobial susceptibility patterns, and associated risk factors of Enterococci species in healthcare and community settings in Ethiopia: a systematic review and meta-analysis
Supplemental material, sj-docx-5-tai-10.1177_20499361251354905 for Prevalence, antimicrobial susceptibility patterns, and associated risk factors of Enterococci species in healthcare and community settings in Ethiopia: a systematic review and meta-analysis by Zigale Hibstu Teffera, Wubetu Yihunie Belay, Bantayehu Addis Tegegne, Abebe Dagne, Adane Adugna, Habtamu Belew, Yasabe Leykun, Samrawit Tefera, Bewket Mesganaw Shtie, Abebe Yenesew, Abateneh Melkamu, Gashaw Azanaw Amare, Desalegn Abebaw, Yibeltal Akelew, Mohammed Jemal, Baye Ashenef, Mamaru Getinet, Temesgen Baylie, Mihiretie Kiber, Mamaru Tilahun Afework, Tiruzer Hibistu and Kassaye Enchalew in Therapeutic Advances in Infectious Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
