Abstract
Background:
While isavuconazole (ISA) has demonstrated non-inferiority to voriconazole (VCZ) for invasive aspergillosis (IA) in clinical trials, real-world comparisons are limited.
Objectives:
To compare treatment completion, adverse events, hospitalizations, and healthcare costs in patients treated with VCZ versus ISA for IA.
Design:
Retrospective cohort study using Merative MarketScan claims data (2017–2020).
Methods:
Adults (⩾18 years) diagnosed with IA (International Classification of Diseases, Tenth Revision, Clinical Modification (ICD-10-CM) codes) who received VCZ or ISA monotherapy were included. Treatment completion was defined as ⩾42 days of therapy. Descriptive statistics and logistic regression were used to assess outcomes and predictors of antifungal selection, adverse events, hospitalizations, and treatment completion.
Results:
Among 335 patients, 84% (n = 282) received VCZ and 16% (n = 53) received ISA. Baseline characteristics were comparable, although the VCZ group had higher Medicaid enrollment, and the ISA group had more patients with malignancy. Treatment completion rates were comparable (92% each, p = 1), as were median treatment durations (VCZ: 120 days, ISA: 112 days, p = 0.95). Adverse event rates were not significantly different (VCZ: 49%, ISA: 60%, p = 0.18), but CNS-related events occurred more frequently with ISA (16% vs 9%, p = 0.32). VCZ was associated with lower outpatient pharmacy costs (median $1,596.68 vs $11,000.66, p < 0.001) and total hospitalization costs (median $40,681.89 vs $121,545.89, p = 0.01). Malignancy was associated with lower odds of receiving VCZ (OR 0.30, p = 0.001), and younger age predicted higher odds of treatment incompletion (OR 0.97, p = 0.035). Female sex was associated with increased adverse event risk. Notably, VCZ use was not associated with increased adverse events or treatment incompletion.
Conclusion:
VCZ was prescribed five times more frequently than ISA for IA, despite similar treatment durations and completion rates. VCZ was associated with lower costs and did not increase the risk of adverse events or treatment discontinuation. These findings suggest that VCZ remains commonly used and potentially more cost-effective treatment option for IA.
Plain language summary
Isavuconazole (ISA) is as effective as voriconazole (VCZ) for treating invasive aspergillosis (IA), but real-world comparisons are limited. This study used insurance claims data from 2017–2020 to compare treatment outcomes, side effects, hospitalizations, and costs in 335 adults treated with either VCZ (84%) or ISA (16%). Both VCZ and ISA had similar treatment durations and high completion rates (92%). Side effects occurred at similar rates, although patients prescribed VCZ were more likely to be on other medications that could cause harmful drug interactions. VCZ was significantly less expensive, with lower pharmacy ($1,597 vs $11,001) and hospitalization costs ($40,682 vs $121,546). Patients with cancer were less likely to receive VCZ, and younger patients were more likely to stop treatment early. Female patients were more likely to experience side effects. However, VCZ itself was not linked to a higher risk of treatment failure or side effects. Overall, VCZ was prescribed more often, performed as well as ISA, and was more cost-effective, but requires careful monitoring for drug interactions.
Introduction
Invasive aspergillosis (IA) is a life-threatening fungal infection predominantly affecting individuals with immunodeficiencies and is characterized by high mortality rates.1,2 IA often involves the respiratory tract and sinuses, but may disseminate, causing infective endocarditis, central nervous system (CNS) infections, or ocular infections. Antifungal therapy typically lasts for 6–12 weeks, although the duration may vary based on factors like the severity and duration of immunosuppression, extent of infection, and individual patient response.1,3 In some cases, surgical intervention may be necessary.
Voriconazole (VCZ) is currently the preferred antifungal agent for IA, as recommended by the Infectious Diseases Society of America (IDSA) guidelines. 1 However, this recommendation predates the 2016 SECURE trial, which demonstrated that isavuconazole (ISA) was non-inferior to VCZ. 4 Subsequently, the 2017 European Society for Clinical Microbiology and Infectious Diseases, the European Confederation of Medical Mycology and the European Respiratory Society (ESCMID-ECMM-ERS) joint guideline 5 and anticipated future updates to the IDSA guidelines consider ISA and VCZ as equally effective options. Despite its efficacy, VCZ is associated with numerous adverse events, and its variable metabolism, influenced by cytochrome P450 (CYP) 2C19 polymorphisms and drug interactions, often results in fluctuating plasma concentrations, necessitating therapeutic drug monitoring (TDM).6–8
In contrast, the SECURE trial revealed a more favorable safety profile for ISA, with significantly lower rates of hepatobiliary disorders (9% vs 16%), eye disorders (15% vs 27%), and skin/subcutaneous disorders (33% vs 42%) compared to VCZ. 4 Furthermore, ISA was associated with lower discontinuation rates (8% vs 14%). ISA also offers predictable linear pharmacokinetics and does not require routine TDM, as its pharmacokinetic properties are not linked to improved efficacy or safety.9,10 Additionally, a cost-effectiveness analysis suggests that ISA may reduce overall healthcare costs by an average of U.S. dollars (USD) 7,418 per patient compared to VCZ. 11 However, real-world data comparing the safety, tolerability, and treatment completion rates of ISA and VCZ in patients with IA outside of controlled clinical trials remain limited.
Methods
The objectives of this study were to compare treatment completion rates, adverse events, hospitalizations, and healthcare utilization costs between patients diagnosed with IA treated with VCZ and those treated with ISA.
Study design and population
This retrospective cohort study used data from the Merative MarketScan Database from 2017 to 2020. Adult patients (⩾18 years) with a diagnosis of IA were identified based on International Classification of Diseases, Tenth Revision, Clinical Modification (ICD-10-CM) codes (Supplemental Table 1). Patients were required to have filled a prescription for either VCZ or ISA monotherapy, with a minimum 7-day supply within 30 days before or 90 days after their diagnosis of IA (Supplemental Table 2).1,12 The index date was defined as the date of the first pharmacy claim. Continuous health plan enrollment was required for at least 90 days before the index date, throughout the treatment period, and for 28 days following treatment cessation (e.g., follow-up period). Patients with prior antifungal therapy or a history of liver transplantation or hepatic dysfunction (e.g., hepatic failure, viral hepatitis, or cirrhosis) were excluded to avoid potential confounding factors that could influence prescribing practices, consistent with the criteria used in the SECURE trial (Supplemental Table 3). 4
Patients meeting the inclusion criteria were categorized into two cohorts based on their initial prescription for either VCZ or ISA. They were followed from the start of treatment through the follow-up period (28 days posttreatment), consistent with prior clinical trial designs. 4
Data collection
Baseline characteristics, including age, sex, place of service (outpatient and inpatient settings), insurance type, and comorbidities, were collected before the index date (Supplemental Tables 4 and 5). Comorbidities such as type 2 diabetes, malignancies, and respiratory disorders were identified using ICD-10-CM codes (Supplemental Table 5). 4 Medications contraindicated with either VCZ or ISA were identified through National Drug Codes within 90 days before, during, or after treatment (Supplemental Tables 6 and 7).6,13
Treatment duration was calculated based on the total days of VCZ or ISA dispensed, and any switches to alternative antifungal therapies were documented. Refill gaps were categorized as 0–10 days, 11–20 days, or 21–30 days. The end of treatment was defined as no additional refills within 30 days of the last prescription supply.14,15 Patients were considered to have completed treatment if they received at least 42 days of antifungal therapy, in accordance with clinical guidelines. 1
All-cause adverse events, including cardiovascular, gastrointestinal, CNS, hepatobiliary, and skin disorders, as well as pyrexia, chills, and eye disorders, were defined based on established associations with VCZ or ISA, as reported in package inserts and published clinical trials.4,6,13,16 These events were identified using ICD-10-CM codes during both the treatment and follow-up periods (Supplemental Table 8).
All-cause hospitalizations were evaluated during both treatment and follow-up periods. Outpatient expenses, including costs associated with laboratory tests (e.g., basic metabolic panels, liver function tests, TDM based on Current Procedural Terminology (CPT) codes) and pharmacy claims, were analyzed and reported in USD. Inpatient costs were evaluated using data from the MarketScan database, which reports total gross hospital payments for admissions occurring during the treatment and follow-up periods. This encompasses all charges billed by the hospital for services rendered during the inpatient’s stay. To mitigate potential data errors, services associated with negative total paid amounts were excluded. 17
Outcomes
The primary outcome was the proportion of patients with IA who completed treatment (⩾42 days) with VCZ compared to ISA. Secondary outcomes included adverse event rates, all-cause hospitalization rates, and healthcare costs during the treatment and follow-up periods.
Statistical analysis
Descriptive statistics were employed to describe demographic and baseline characteristics, contraindicated medications for VCZ or ISA, and clinical outcomes. Differences between categorical variables in the treatment groups were assessed using Chi-squared or Fisher’s exact tests, as appropriate. Continuous variables were compared between the two treatment groups using independent samples t-tests for normally distributed data and Wilcoxon rank-sum tests for non-normally distributed data. Logistic regression analyses were conducted to examine factors associated with initial antifungal therapy selection (VCZ or ISA), adverse events, all-cause hospitalizations, and treatment completion. Covariates included age, sex, insurance provider, insurance type, malignancy status, and initial antifungal therapy (included only in models for adverse events, hospitalizations, and treatment completion). The statistical analyses were performed using R software, version 4.2.1 (Vienna, Austria). A p-value of <0.05 was considered statistically significant.
Ethics statement
The Merative MarketScan Database is fully de-identified and lacks any data identifiers prohibited by the Health Insurance Portability and Accountability Act (HIPAA) Privacy Rule. Accordingly, the University of Georgia Institutional Review Board (IRB) determined that this research did not involve human subjects as defined by the U.S. Department of Health and Human Services and FDA regulations, and IRB approval was not required (PROJECT00007315).
This study follows the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines (Supplemental File). 18
Results
Patient demographics and baseline characteristics
A total of 335 patients were included in the study, with 84% (n = 282) receiving VCZ and 16% (n = 53) receiving ISA as initial therapy. IA was diagnosed in an inpatient setting for 50% of patients in the VCZ group and 53% in the ISA group (p = 0.78). Most cases were invasive pulmonary aspergillosis, accounting for 88% in the VCZ group and 92% in the ISA group, while disseminated aspergillosis occurred in 2% and 4%, respectively (p = 0.63 overall).
Baseline characteristics, including age and sex, were comparable between groups (Table 1). However, Medicaid enrollment was more frequent among patients treated with VCZ. Chronic obstructive pulmonary disease (COPD) was the most prevalent comorbidity in both the VCZ and ISA groups (42% vs 28%, respectively) (p = 0.07). Notably, rates of acute myeloid leukemia and multiple myeloma were significantly lower in the VCZ group (3% for both) compared to the ISA group (13% and 9%, respectively) (p = 0.01 and p < 0.001, respectively).
Baseline characteristics of patients with IA treated with VCZ or ISA.

Data are presented as n (%) unless otherwise noted.
IA, invasive aspergillosis; IQR, interquartile range; ISA, isavuconazole; VCZ, voriconazole.
Contraindicated medications prior to antifungal therapy
Patients in the VCZ group were more likely to be prescribed medications with potential contraindications before initiating antifungal therapy, including salmeterol/fluticasone (13% vs 0%, p = 0.01) and alprazolam (11% vs 0%, p = 0.01). Conversely, rifampin use was higher in the ISA group (9% vs 2%, p = 0.03).
Treatment completion and duration
Treatment completion rates were high and comparable between the groups, with 92% completing therapy in both the VCZ and ISA groups (p = 1) (Figure 1). Among those who completed therapy, the median duration was 120 days (IQR 74–180, range 42–1350) for VCZ and 112 days (IQR 60–180, range 45–756) for ISA (p = 0.95). For patients who did not complete treatment, the median duration was 30 days in both groups (p = 0.40).

Clinical outcomes between patients with IA treated with VCZ or ISA.
The majority of patients initiated antifungal therapy after their IA diagnosis, with 76% in the VCZ group and 83% in the ISA group (p = 0.32). The median time from IA diagnosis to the first antifungal prescription was 5 days (IQR 0–13) for VCZ and 8 days (IQR 1.25–19) for ISA (p = 0.32). Among patients who completed treatment, the median time to the first fill was 5 days (IQR 0–13) for VCZ and 8 days (IQR 1–19) for ISA (p = 0.31). For those who did not complete treatment, the median time was 7.5 days (IQR −1 to 14.2) for VCZ and 6 days (IQR 4–17) for ISA (p = 0.88).
Pharmacy utilization and refill gaps
During treatment and follow-up, there were 1746 pharmacy claims for VCZ and 352 for ISA. The average number of claims per patient was similar between groups (VCZ: 6.19 vs ISA: 6.64, p = 0.57). A single pharmacy claim was significantly more common in the VCZ group (15 patients, 1.3%) compared to the ISA group (2 patients, 0.9%) (p = 0.75). Most refill gaps were short, with 87% of patients in the VCZ group and 81% in the ISA group experiencing gaps of 0–10 days (p = 0.02). Refill gaps of 11–20 days were more common among the ISA group (15%) compared to the VCZ group (9%) (p = 0.01), while gaps of 21–30 days occurred at similar rates in both groups (3% vs 3%, p = 0.88).
Patients treated with VCZ were significantly more likely to be prescribed simvastatin (32% vs 0%, p < 0.001), alprazolam (22% vs 0%, p < 0.001), and tamsulosin (9% vs 0%, p = 0.02) compared to those treated with ISA.
Adverse events
Adverse events were documented in 139 individuals in the VCZ group and 32 in the ISA group, with a comparable proportion of patients experiencing at least one adverse event (49% vs 60%, p = 0.18) (Table 2). The most common adverse events were gastrointestinal disorders (47% vs 50%, p = 0.95), pyrexia (24% vs 16%, p = 0.45), hepatobiliary disorders (12% vs 6%, p = 0.53), and skin and subcutaneous tissue disorders (10% vs 16%, p = 0.36). CNS-related events occurred in 9% of the VCZ group and 16% of the ISA group (p = 0.32).
All-cause adverse events documented in patients with IA treated with VCZ or ISA, regardless of treatment completion.
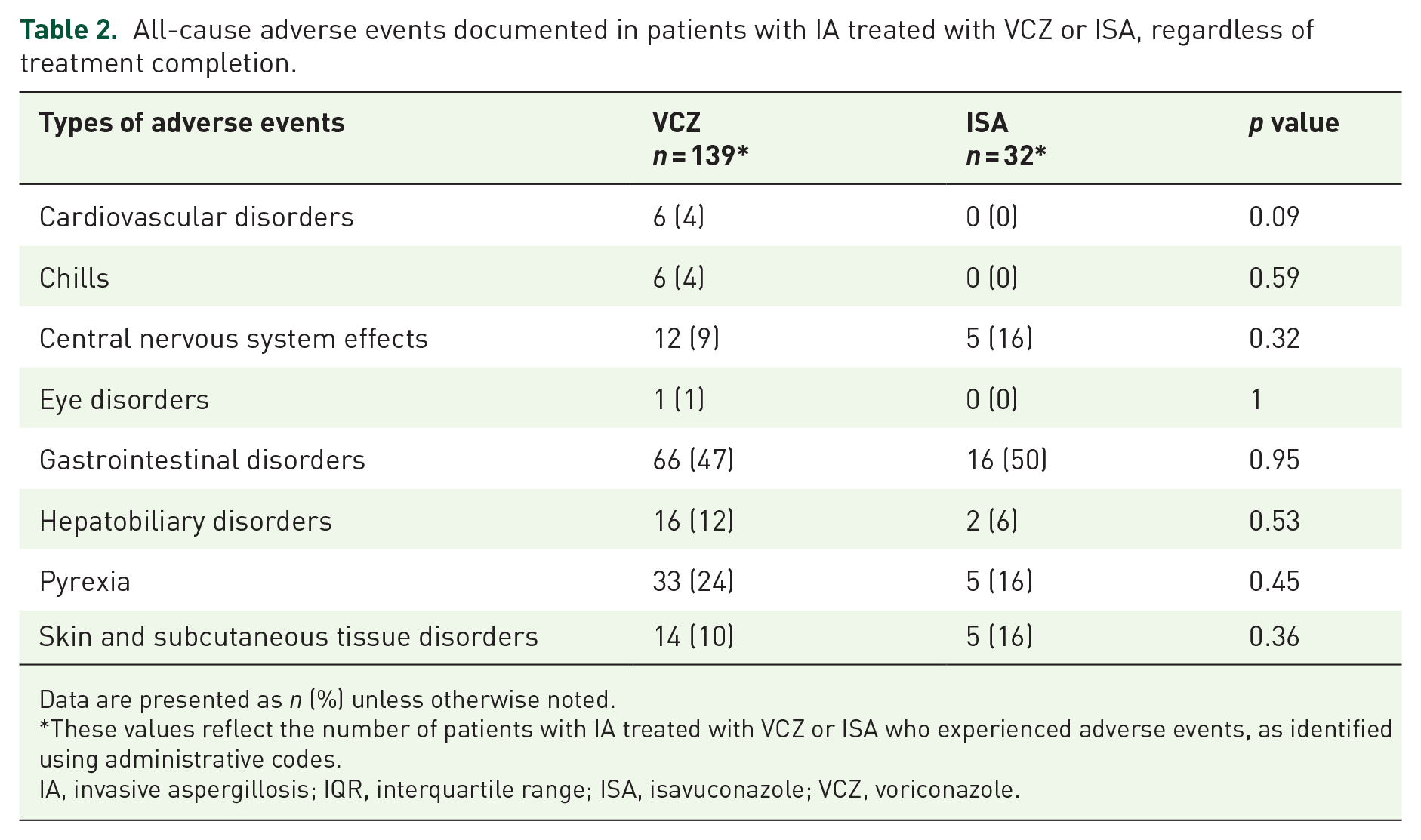
Data are presented as n (%) unless otherwise noted.
These values reflect the number of patients with IA treated with VCZ or ISA who experienced adverse events, as identified using administrative codes.
IA, invasive aspergillosis; IQR, interquartile range; ISA, isavuconazole; VCZ, voriconazole.
Among patients who completed treatment, 143 adverse events were documented in the VCZ group and 32 in the ISA group. The proportion of patients experiencing adverse events was again comparable between groups (50% vs 63%, respectively) (p = 0.13). The most frequent adverse events were gastrointestinal disorders (49% vs 52%, p = 0.97), pyrexia (22% vs 16%, p = 0.63), and skin and subcutaneous tissue disorders (9% vs 13%, p = 0.51). CNS-related events remained comparable (9% VCZ vs 16% ISA, p = 0.33).
In patients who did not complete treatment, 11 adverse events were recorded in the VCZ group and 1 in the ISA group. The proportion of patients experiencing adverse events was similar (39% vs 25%, respectively) (p = 1). The most frequent events in the VCZ group were pyrexia (44%), gastrointestinal disorders (22%), skin/subcutaneous tissue disorders (22%), and hepatotoxicity (11%). The sole documented adverse event in the ISA group was a skin and subcutaneous tissue disorder.
Two patients in the VCZ group, including one with documented skin and subcutaneous tissue disorder, were switched to alternative antifungal therapies (ISA or posaconazole). In the ISA group, one patient was switched to VCZ and later back to ISA before being diagnosed with transaminitis.
Outpatient pharmacy costs
Pharmacy costs for VCZ were significantly lower than those for ISA (Table 3). Among patients with nonzero charges, the median pharmacy costs were $1,535.68 for VCZ compared to $11,000.66 for ISA (p < 0.001). For patients who completed therapy, the median costs were $1,569.00 for VCZ and $10,909.46 for ISA.
Outpatient pharmacy and hospital costs for patients with IA treated with VCZ or ISA.
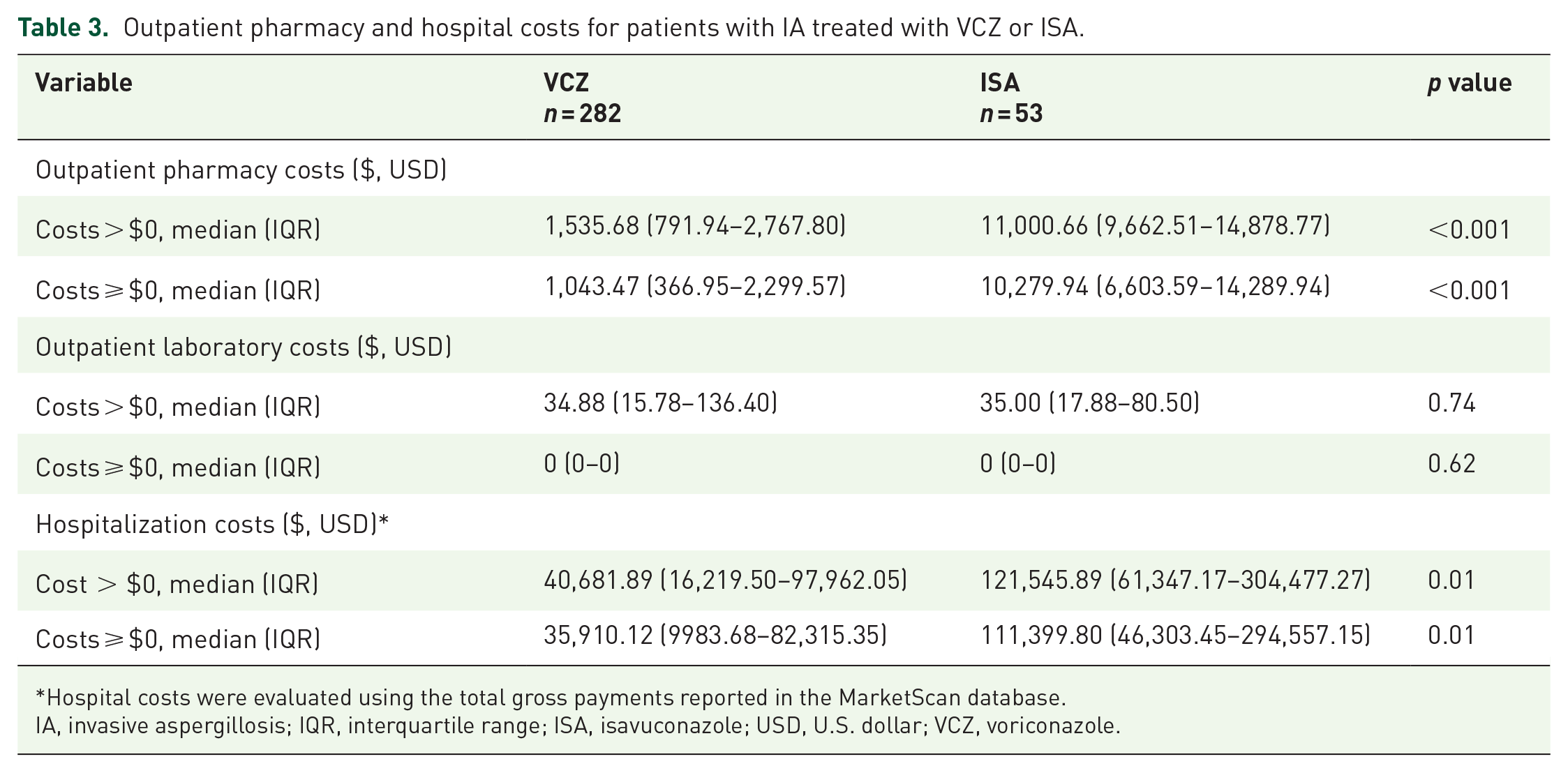
Hospital costs were evaluated using the total gross payments reported in the MarketScan database.
IA, invasive aspergillosis; IQR, interquartile range; ISA, isavuconazole; USD, U.S. dollar; VCZ, voriconazole.
When zero charges were included, median pharmacy costs were $1,043.47 for VCZ and $10,279.94 for ISA (p < 0.001). Among patients who completed therapy, the median costs were $1,065.91 for VCZ and $10,279.94 for ISA.
Laboratory monitoring and costs
Outpatient laboratory monitoring was infrequent in both groups (21% vs 17%, p = 0.64). Basic metabolic panels were performed in 2% of patients in the VCZ group compared to 6% in the ISA group, with similar proportions observed among those who completed therapy (2% vs 6%). Liver function tests were obtained in 4% of the VCZ group and 8% of the ISA group, again with comparable rates among patients who completed therapy (4% vs 8%). TDM was conducted in only three patients (1%) in the VCZ group, all of whom completed therapy, while no patients in the ISA group underwent TDM.
Laboratory costs were comparable between groups, with a median of $34.88 for VCZ and $35.00 for ISA (p = 0.74) when including charges greater than zero.
Hospitalizations and costs
Hospitalization rates during treatment or follow-up were similar between the VCZ and ISA groups (21% vs 17%, p = 0.64). Total gross hospital costs were substantially lower for patients treated with VCZ at $40,681.89 compared to $121,545.89 for ISA (p = 0.01).
Multivariable logistic regression analyses
In logistic regression analyses evaluating predictors of initial antifungal therapy selection, patients with malignancy had significantly lower odds of receiving VCZ compared to those without malignancy (OR 0.30, 95% CI 0.15–0.64, p = 0.001) (Table 4). In the adverse events model, female sex was associated with increased risk (OR 1.69, 95% CI 1.07–2.67, p = 0.024), while comprehensive insurance was associated with reduced odds of adverse events (OR 0.22, 95% CI 0.07–0.63, p = 0.005; Table 5). VCZ use was not significantly associated with adverse events. For all-cause hospitalizations, malignancy was the only significant predictor (OR 2.84, 95% CI 1.38–5.76, p = 0.004), and VCZ treatment was not associated with increased odds of hospitalization (Table 6). In the model assessing treatment incompletion, younger age was significantly associated with higher odds of incompletion (OR 0.97, 95% CI 0.94–0.99, p = 0.035; Table 7). VCZ use was also not significantly associated with treatment incompletion.
Logistic regression analysis of factors associated with initial voriconazole use.

EPO/HMO, exclusive provider organization/health maintenance organization; POS, point of service; PPO, preferred provider organization.
Logistic regression analysis of predictors of adverse events.

EPO/HMO, exclusive provider organization/health maintenance organization; POS, point of service; PPO, preferred provider organization; VCZ, voriconazole.
Logistic regression analysis of predictors of all-cause hospitalization.

EPO/HMO, exclusive provider organization/health maintenance organization; POS, point of service; PPO, preferred provider organization; VCZ, voriconazole.
Logistic regression analysis of predictors of treatment incompletion.

EPO/HMO, exclusive provider organization/health maintenance organization; POS, point of service; PPO, preferred provider organization; VCZ, voriconazole.
Discussion
This study evaluated treatment completion rates, safety, and costs associated with VCZ and ISA for IA. Both VCZ and ISA demonstrated high treatment completion rates (>90%) with no significant difference observed between them, aligning with findings from a large clinical trial and reinforcing the real-world relevance of our results. 4
Bivariate analyses of baseline characteristics revealed that younger age was significantly associated with treatment incompletion. Patients who did not complete therapy were, on average, younger (50.6 ± 13.2 years) than those who completed it (54.6 ± 12.8 years). Insurance type was also modestly associated with treatment completion. A greater proportion of patients who did not complete therapy were enrolled in a comprehensive plan (35% vs 12%), while fewer were enrolled in a preferred provider organization (PPO) plan (27% vs 38%). In contrast, commercial insurance status and the presence of malignancy were not significantly associated with treatment completion.
Use of VCZ was not significantly associated with incomplete treatment, suggesting comparable tolerability or adherence relative to ISA in real-world settings. This contrasts with a meta-analysis of six clinical studies including 803 patients, which reported significantly lower treatment discontinuation due to drug-related adverse events with ISA (9.31%) compared to VCZ (16.46%) (RR = 0.56, p = 0.003). 19 However, trial sequential analysis from that meta-analysis suggested that the evidence remains inconclusive due to limited sample size, demonstrating the need for further high-quality studies to confirm these differences. Due to limitations in our dataset, we were unable to determine the specific reasons for treatment discontinuation, such as adverse events, patient preference, or other clinical factors.
Despite comparable efficacy, VCZ was prescribed five times more frequently than ISA, a pattern consistent with previous analyses of real-world treatment trends for invasive pulmonary aspergillosis using TriNetX global research network data. 20 While patients with Medicare or commercial insurance were prescribed VCZ or ISA at similar rates, Medicaid beneficiaries were more frequently prescribed VCZ. This prescribing disparity may reflect differences in drug cost, formulary restrictions, or provider familiarity with VCZ. However, due to the lack of geographic data, we were unable to assess state-specific formulary preferences that might further elucidate these differences. 21 Logistic regression analysis showed that most demographic and insurance-related factors were not significantly associated with antifungal selection. However, patients with malignancy were significantly less likely to receive VCZ, suggesting a clinical preference for ISA in this population—potentially driven by prior reports of its more favorable safety profile, reduced drug interaction potential, and lack of routine TDM recommendations.4,19
Although most patients in both the VCZ and ISA groups began antifungal therapy after their IA diagnosis, the median time to treatment initiation and first prescription fill, 5 days for VCZ and 8 days for ISA, raises concern given the time-sensitive nature of IA management. While these timelines may reflect standard outpatient practice, previous studies have demonstrated that delays in initiating antifungal therapy, even as short as 6 days, can nearly double mortality rates, underscoring the importance of timely treatment. 22 The observed delay in therapy initiation may reflect diagnostic uncertainty, challenges in securing insurance authorization, or delays in outpatient prescription fulfillment. Unlike controlled clinical trials, where treatment is typically initiated promptly under protocol-driven timelines, real-world settings introduce logistical and systemic barriers that may compromise the timeliness of care. Further studies are needed to explore barriers to timely antifungal therapy initiation in real-world settings.
Compared to the SECURE trial cohort and other studies that predominantly included patients with hematologic malignancies, our population had a higher burden of chronic comorbidities (e.g., COPD and type 2 diabetes) and fewer cases of disseminated aspergillosis.4,23,24 This difference likely reflects the predominance of invasive pulmonary aspergillosis in our cohort and may have influenced prescribing patterns, particularly the lower VCZ use in patients with malignancy. Treatment durations were also notably longer than those reported in the SECURE trial (median 120 vs 47 days for VCZ; 112 vs 45 days for ISA), 4 in line with prior real-world data. 25 Extended treatment durations in IA are often guided by clinical, microbiological, and radiographic responses, typically ranging from 6 weeks to over 12 weeks, with secondary prophylaxis recommended for patients with ongoing immunosuppression.1,5,26 Although we could not distinguish between antifungal use for treatment versus prophylaxis, the fact that most patients initiated therapy after their IA diagnosis suggests predominantly therapeutic use. Additionally, we were unable to assess key clinical factors such as laboratory data and radiographic findings that influence treatment duration.
Antifungal therapy switching was rare and consistent with prior studies. 20 Switches are often driven by adverse effects, QTc prolongation, or drug interactions. Although more patients in the VCZ group were prescribed contraindicated medications, overall switch rates remained low. Despite increasing clinical experience with ISA for treatment of IA, its use as initial therapy or as a switch option from VCZ remains limited, potentially influenced by factors such as cost, availability, perceived risks, or concerns regarding clinical efficacy. 24
Adverse events were documented in 51% of patients in our cohort, with no significant difference between the VCZ and ISA groups. Although this rate is lower than those reported in prior clinical trials,4,25,27 it is consistent with findings from two retrospective analyses.25,28 The lower incidence likely reflects limitations of administrative claims data, which may underestimate adverse events compared to direct clinical assessments in prospective trials. For comparison, a pooled analysis of four studies (n = 703) reported drug-related adverse events in 59% of patients treated with VCZ and 43% of those receiving ISA, 19 highlighting the higher event capture in trial settings and suggesting potential differences in reporting sensitivity. Gastrointestinal events were most common, consistent with SECURE trial findings. 4 Unlike the SECURE trial, which found significantly fewer eye, hepatobiliary, and skin-related adverse events with ISA, we observed similar rates across groups.4,25
Notably, female patients in our study had significantly higher odds of experiencing adverse events, which may reflect sex-based differences in pharmacokinetics, adverse event susceptibility, or reporting behaviors. Interestingly, a real-world pharmacovigilance study using the FDA Adverse Event Reporting System (FAERS) found VCZ-associated adverse events to be significantly more frequent in male patients. 29 However, that study did not control for important confounding factors such as concomitant medications or underlying comorbidities, limiting its conclusions regarding sex differences. Another FAERS-based study suggested that both sex and age can influence the occurrence of adverse reactions, particularly when VCZ is used in combination with immunosuppressants. 30 Although our analysis only captured the presence of potentially contraindicated medications, these findings are consistent with data indicating that VCZ exposure and toxicity may be affected by age, sex, and body weight-based dosing, all of which may contribute to variability in adverse event risk.30,31
Although not statistically significant, our observation of a numerically higher rate of CNS-related events in patients treated with ISA compared to VCZ (16% vs 9%) warrants careful attention, especially considering existing pharmacovigilance data. Notably, reports from the French Pharmacovigilance Database indicate a substantial association between VCZ and visual disturbances (18%) and nervous system disorders (14%). 32 Furthermore, analysis of the FAERS database revealed significantly elevated reporting odds ratios for VCZ relating to psychiatric, nervous system, and eye disorders with a rapid median onset of CNS events within 1 day. 33 These external data strongly corroborate the established CNS toxicity profile of VCZ, attributed to its significant penetration across the blood-brain and blood-eye barriers, achieving substantial cerebrospinal fluid concentrations 34 and leading to known adverse effects like visual disturbances and altered mental status.
Conversely, while preclinical studies suggest ISA can penetrate the blood-brain barrier, clinical evidence of significant CNS adverse events with ISA remains limited. 34 The numerically higher rate in our ISA group might reflect the greater prevalence of disseminated aspergillosis, due to potential CNS involvement, rather than a direct drug effect. However, this observed difference necessitates further investigation to elucidate a potential distinct CNS adverse event profile associated with ISA. Our findings suggest the importance of vigilant monitoring for neurological symptoms in patients receiving ISA, particularly those with disseminated disease or other risk factors for CNS involvement.
Cardiovascular adverse events were infrequent in our study. However, the use of administrative data likely led to underreporting. Given that ISA is known to shorten the QTc interval, while VCZ carries a risk of QTc prolongation and torsades de pointes (TdP),6,13 the scarcity of documented cardiac events in our cohort may reflect a genuinely low incidence or limitations in detection via administrative codes. Future research incorporating electrocardiographic monitoring is crucial for a more comprehensive understanding of the cardiac safety profiles of both agents.
Despite recommendations for routine monitoring, 35 laboratory testing was infrequent in both treatment groups. Furthermore, despite the known pharmacokinetic variability of VCZ and guidelines recommending TDM to optimize its use,5,36 few patients underwent TDM in the VCZ group in our study, demonstrating a potential disconnect between guidelines and real-world practice. While the SECURE trial employed standard VCZ dosing without TDM and recent research suggests TDM may not improve short-term outcomes in adult patients with IA,4,37 TDM for ISA is generally not recommended, although certain patients subgroups, such as those with higher body weight, adverse reactions, poor response, critical illness, or those undergoing extracorporeal membrane oxygenation (ECMO) or continuous renal replacement therapy, might benefit despite limited target concentration data.25,38–41 Although TDM could have potentially impacted therapy duration and adverse events in our cohort, our analysis was limited to cost data for laboratory tests. Notably, excluding the small subset of patients who underwent TDM from our logistic regression analysis did not significantly alter the original findings, with treatment type remaining nonsignificant and only minor changes in coefficient estimates observed.
Our study revealed significantly lower outpatient pharmacy costs for VCZ compared to ISA, a finding that contrasts with some prior cost-effectiveness simulations favoring ISA. While studies in the United States, Sweden, and Spain have reported higher overall costs for ISA alongside favorable cost-effectiveness ratios, analyses from Brazil suggest substantial cost savings with ISA.11,22,42,43 Our analysis did not include formal cost-effectiveness or cost-utility evaluations, nor did it account for costs related to adverse event management or TDM. Nonetheless, the observed difference in outpatient pharmacy costs emphasizes the significant economic considerations in selecting antifungal therapy for IA.
Limitations
This study, utilizing the Merative MarketScan Database to compare outcomes between VCZ and ISA for IA, is subject to several limitations inherent to its retrospective design and reliance on administrative claims data. The 90-day continuous enrollment requirement prior to the index date may have introduced selection bias, and the study period was constrained by the institutional user agreement. Although the database encompasses a diverse population across the United States, the lack of detailed geographic variables may limit the generalizability of our findings. Furthermore, the unequal sample sizes between treatment groups could impact the reliability of comparisons.
The use of administrative codes for identifying comorbidities, adverse events, and IA diagnoses carries the risk of undercoding and misclassification, potentially underestimating recurrent infections, relapses, and broader patient outcomes beyond those captured in claims. Notably, inpatient mortality during the treatment and follow-up periods could not be determined, as the MarketScan Database has obscured specific hospital discharge statuses, including inpatient deaths, since 2016 to protect patient privacy. 44 While a time frame was used to identify adverse events, causality with the antifungal therapy cannot be definitively established due to the potential influence of comorbidities and concurrent treatments. The reliance on ICD-10-CM codes for adverse event identification also introduces measurement bias, and less severe events were likely underreported. Despite these limitations, our study aimed to assess real-world VCZ and ISA use by capturing a broad spectrum of all-cause adverse events during the study period, a necessary approach given the lack of direct medication-adverse event linkage in the database.
The absence of crucial diagnostic confirmation data (histopathology, serology, microbiology) is another limitation, although prior research suggests moderate sensitivity of administrative codes for IA classification. 45 While we analyzed IA subgroups, differentiating between Aspergillus species, which exhibit varying presentations, susceptibility patterns, and outcomes, was not feasible. Our adherence measure, based on prescription fills and refill gaps, provides an imperfect proxy for actual medication intake. Similarly, TDM rate evaluation using CPT codes lacks precision. Finally, the unavailability of data on in-hospital antifungal administration limits a complete understanding of treatment details. 21
Conclusion
Despite comparable high treatment completion rates and durations, VCZ was prescribed five times more often than ISA for IA. VCZ was associated with significantly lower outpatient pharmacy and total hospitalization costs but a higher likelihood of baseline contraindicated medication use, demonstrating the need for vigilant drug interaction management. While overall adverse event rates were comparable, CNS events were numerically more frequent with ISA. Malignancy was associated with lower VCZ use, and younger age predicted treatment incompletion, but VCZ use itself was not significantly associated with treatment incompletion or adverse events. Notably, laboratory monitoring, including VCZ TDM, was infrequent, suggesting the need for clearer clinical guidance and improved adherence to existing recommendations. These findings emphasize the importance of individualized antifungal selection that balances safety, tolerability, and cost. Future studies with larger, more clinically detailed cohorts are needed to clarify the comparative safety profiles of VCZ and ISA, evaluate the clinical utility of routine TDM, and define cost-effective treatment strategies for IA in real-world practice.
Supplemental Material
sj-docx-1-tai-10.1177_20499361251347778 – Supplemental material for Voriconazole versus isavuconazole for invasive aspergillosis: a retrospective analysis in a medically insured U.S. population (2017–2020)
Supplemental material, sj-docx-1-tai-10.1177_20499361251347778 for Voriconazole versus isavuconazole for invasive aspergillosis: a retrospective analysis in a medically insured U.S. population (2017–2020) by Sophea Chan, Laura Leigh Stoudenmire, Xianyan Chen, Duna Zhan, Andrés F. Henao-Martínez and Daniel B. Chastain in Therapeutic Advances in Infectious Disease
Supplemental Material
sj-docx-2-tai-10.1177_20499361251347778 – Supplemental material for Voriconazole versus isavuconazole for invasive aspergillosis: a retrospective analysis in a medically insured U.S. population (2017–2020)
Supplemental material, sj-docx-2-tai-10.1177_20499361251347778 for Voriconazole versus isavuconazole for invasive aspergillosis: a retrospective analysis in a medically insured U.S. population (2017–2020) by Sophea Chan, Laura Leigh Stoudenmire, Xianyan Chen, Duna Zhan, Andrés F. Henao-Martínez and Daniel B. Chastain in Therapeutic Advances in Infectious Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
