Abstract
Antimicrobial resistance (AMR) poses a significant global health threat by diminishing the effectiveness of once-powerful antimicrobial agents, leading to higher rates of illness and death, along with escalating healthcare costs. While bacterial resistance is a primary concern, resistance is also increasing against antifungal, antiparasitic, and antiviral drugs. Many of the last-resort drugs are becoming less effective due to AMR. Projections indicate that by 2050, AMR could cause up to 10 million deaths annually, making it the leading cause of death worldwide, a situation that could result in a post-antibiotic era with substantially increased morbidity and mortality. This review aims to raise awareness about the dangers of AMR and its potential to become a silent global pandemic. It begins by examining antimicrobial drugs, followed by a discussion on AMR, focusing on resistance to antibacterial, antifungal, antimalarial, and antiviral drugs, along with its effects on health, and the economy, and prioritized global pathogens. Finally, it explores preventive measures and innovative strategies to combat AMR.
Keywords
Overview of antimicrobials and antimicrobial resistance as a global threat
Introduction
An antibiotic is a compound produced by, or extracted from, a microorganism that can either kill or prevent the growth of another microorganism. An antimicrobial is a substance derived from any source (microorganisms, plants, animals, synthetic, or semi-synthetic) that acts against any type of microorganism, such as bacteria, fungi, parasites, and viruses. Every antibiotic falls under the category of antimicrobials, but not all antimicrobials are antibiotics. 1 Antimicrobials, by inhibiting cell wall, protein, and nucleic acid synthesis of germs, 2 are used in controlling infections affecting humans and animals. 3 Since the early 20th century, antimicrobials have saved millions of lives 3 and have made hospital procedures like general surgery, organ transplants, renal dialysis, and cancer chemotherapy possible, as their ability to manage associated infections is vital for the success of these treatments.4,5 Hence, without effective antimicrobials, carrying out hospital procedures may lead to severe infections such as sepsis, leading to increased morbidity and mortality.
Antimicrobial resistance (AMR) emerges when microorganisms such as bacteria, fungi, viruses, and parasites adapt after being exposed to antimicrobial agents such as antibacterials, antifungals, antivirals, antimalarials, and anthelmintics, ultimately reducing or eliminating the effectiveness of these medications. 6 Simply put, AMR is when a microorganism can withstand the growth-inhibiting or killing effects of an antimicrobial, going beyond the normal sensitivity of that particular germ.7,8 This review explores various aspects related to antimicrobial agents, including the discovery of antimicrobials, AMR, and an overview of resistance to antibacterial, antifungal, antimalarial, and antiviral drugs. It also covers the prevalence and economic impact of AMR and identifies global priority pathogens (superbugs). Finally, it explains control strategies to contain AMR. Hence, this review aims to awaken the community, healthcare providers, and regulatory bodies to the silent pandemic nature of AMR and its control strategies.
The discovery and use of antimicrobial agents
The modern antimicrobial era began with the discovery of Salvarsan in 1910 by Paul Ehrlich and penicillin in 1928 by Alexander Fleming. Although Salvarsan had a complicated injection process and could cause side effects, it was highly successful when marketed by Hoechst, eventually becoming the most widely prescribed drug. This popularity persisted until penicillin replaced it in the 1940s. 9 Surprisingly, the exact mechanism by which Salvarsan works is still unclear, and the debate regarding its chemical structure was resolved lately. 10 Moreover, Salvarsan has also been shown to extend life expectancy by more than 20 years. 11 Consequently, Salvarsan is considered the first modern antimicrobial that was effectively used to treat bacterial infections during that era.
Paul Ehrlich’s systematic screening approach laid the foundation for drug discovery methods, leading to the identification and development of thousands of drugs used in clinical practice. In the early days of antibiotic research, this method was instrumental in the discovery of sulfa drugs, specifically sulfonamidochrysoidine (KI-730, Prontosil). 12 It turned out to be a prodrug, with its active component, sulfanilamide. 13 In the early 20th century, scientists were driven by a strong desire to discover and identify new therapeutic compounds. Their focused efforts and achievements laid the groundwork for innovating new groups of antimicrobial medications that are still in use today, particularly during the 1940s to the 1970s.
Alexander Fleming serendipitously discovered penicillin on September 3, 1928. 14 Twelve years after penicillin discovery, Howard Florey and Ernest Chain found a method on how to purify penicillin to produce sufficient penicillin to be used clinically. 15 Their method ultimately paved the way for the large-scale production of penicillin, employing deep-tank fermentation pioneered by Pfizer. This process allowed for extensive use of penicillin by the Allied Forces during World War II to treat traumatic and postoperative infections.16,17 Following the war, penicillin became available for widespread prescription to the general public, earning the nickname “wonder drug” due to its remarkable effectiveness. 18 The timeframe from the 1950s to the 1970s is often called the “golden era” of antimicrobial discovery. During this period, many of the antimicrobial classes that we use today were discovered or synthesized. 19
Moreover, following the discovery of penicillin, antimicrobials have transformed medicine and saved many lives, 20 and it was seen as signaling the end of infectious diseases. 21 The use of antimicrobials has significantly lowered mortality rates from infectious diseases, that previously accounted for the highest human mortality. 22 The discovery and use of antimicrobials was significant healthcare breakthrough of the last century. 11 Due to the use of antimicrobials, Sir Frank MacFarlane Burnet, a Nobel Prize laureate in Medicine in 1960, wrote about the reduction in infectious diseases in 1962. 23
Overview of antimicrobial resistance by microorganisms of medical importance
The introduction of sulfonamides to the market was soon followed by the emergence of sulfa drug resistance. 13 Even before penicillin was widely used, there were indications that bacteria could break it down through enzymatic degradation. 21 When Alexander Fleming received the Nobel Prize in 1945 for discovering penicillin, he used his Nobel lecture to caution that bacteria could become “easily resistant to penicillin” 24 Penicillin resistance emerged in the 1940s, shortly after it began to be used widely. 25 Resistant strains to different antimicrobials, such as methicillin and vancomycin, were reported just a few years after their introduction. The emergence of penicillin-resistant bacteria by 1960 raised serious concerns. In response, new β-lactam antibiotics were developed and incorporated into medical practices to tackle the issue. 26 The rapid emergence of resistance shortly after the use of existing antimicrobials causes people to despair, casting doubt on the possibility of a future free from infections.
But during this time, bacteria began to become resistant to the new antibiotics by secreting β-lactamases capable of inactivating such drugs. Methicillin-resistant Staphylococcus aureus (MRSA) was discovered in the UK in 1961, which was a pivotal point for the occurrence of AMR. 27 Since then, MRSA has spread extensively around the world. 28 Moreover, due to their overuse globally, resistance to antimicrobials has become a significant issue, with some strains developing resistance within a year of an antimicrobial’s introduction, and many others developing resistance within 5 years. 2 Timeline of selected antimicrobials’ first use and resistance reported has been shown in Table 1.
Timeline of antimicrobial drug resistance for selected antimicrobials.

Each time a new class of antimicrobials is introduced, resistance to it and other classes soon follow. Even the latest antimicrobial classes, despite their broad-spectrum activity, are already showing signs of reduced susceptibility. 37 Without timely intervention, the prospect of reverting to the pre-antibiotic era is increasingly likely in numerous regions around the world. 38 AMR has been linked to higher rates of illness and death, longer hospital stays, and rising healthcare costs.39,40 Drug-resistant fungal strains, such as Candida auris, gram-positive bacteria like Staphylococcus aureus, and anaerobic species including Clostridium difficile, all pose significant health risks to the population. 41 Almost all microorganisms of medical importance, such as bacteria, fungi, parasites, and viruses, have developed resistance to most of the available antimicrobial agents.
Antibacterial drug resistance
Currently, AMR has been found in microorganisms of medical importance, including viruses, parasites, fungi, and bacteria. 42 When compared to the other germs resistance, antibacterial drug resistance is the main factor behind disease burden due to their nature of rapid development of resistance to different new antibacterials across all WHO regions, posing significant challenges.39,42 It’s important to highlight those treating diseases caused by resistant bacteria is generally more challenging than those caused by non-resistant strains. 4 The challenge becomes even more severe as the number of new antibacterials in the drug development pipeline continues to decline. 43
Various studies have thoroughly described the mechanisms by which bacteria resist antimicrobial drugs.44,45 Bacteria can acquire resistance to antibacterial agents through four main mechanisms. 46 These mechanisms include enzymatic degradation of antibacterials, the activation of efflux pumps to maintain subinhibitory intracellular drug levels, alterations in the target site of the antibacterial, and modifications in cell membrane permeability. 47 An additional bacterial characteristic, aside from the selection of antibacterial resistance genes, that contributes to AMR is biofilm formation. 48 A biofilm is a stable, three-dimensional structure created by microorganisms that cluster on a surface to establish a colony. 49
Resistance traits may be passed down genetically from one generation to another. In addition, bacteria can acquire AMR genes through horizontal gene transfer (HGT), which occurs via processes like conjugation, transformation, or transduction. 8 Among the mechanisms of HGT, conjugation is particularly important. This process necessitates direct physical contact between the cells of the bacteria, facilitating the transfer of genetic material. A sex pilus forms during conjugation, allowing a plasmid to be transferred to the recipient bacterium. Through a single conjugation event, multiple antibiotic resistance genes encoded on the plasmid can be transmitted, potentially resulting in the emergence of multidrug-resistant strains. 50 Transformation is a DNA recombination process in which a microorganism absorbs external DNA segments, also known as naked DNA, from the environment, incorporating them into its genetic structure, and making them inheritable traits. 51 Transduction is a bacteriophage-mediated process in which genetic material is transferred between a virus (bacteriophage) and a bacterial cell it infects. 44
Multidrug-resistant (MDR) bacteria, defined as bacterial strains having resistance to one or more antimicrobial agents from three or more different classes of antibiotics, pose a major public health threat in developing countries. These bacteria lead to higher morbidity and mortality rates and make treatment more difficult. 52 The growing frequency of bacterial strains that are resistant and multi-resistant has turned into a major challenge for healthcare systems worldwide. 53 The majority of pathogens are becoming MDR, posing a heightened risk of treatment failure with conventional therapies. This leads to an elevated burden of disease, longer hospitalizations, and increased healthcare expenses.54,55
Antifungal drug resistance
The rise of antifungal drug resistance has alarmed the scientific community, signaling the occurrence of a new health concern alongside antibacterial and antiviral resistance. 56 Moreover, the prevalence of diseases due to invasive fungal pathogens is on the rise globally. 57 Recent epidemiological data indicate a global surge in serious mycoses, with over 150 million cases recorded annually and more than 1.7 million resulting in death each year.58,59 In total, such projections suggest that over 6.55 million people are afflicted annually by a life-threatening fungal disease, causing more than 3.75 million deaths, with about 2.55 million of these deaths directly attributable to the fungal disease itself. 60 The WHO released the first-ever list of fungi that pose a risk to public health to guide research and development efforts. The purpose of this list is to improve the worldwide response to mycoses and antifungal resistance (Table 2). 61
WHO priority fungal pathogens. 61
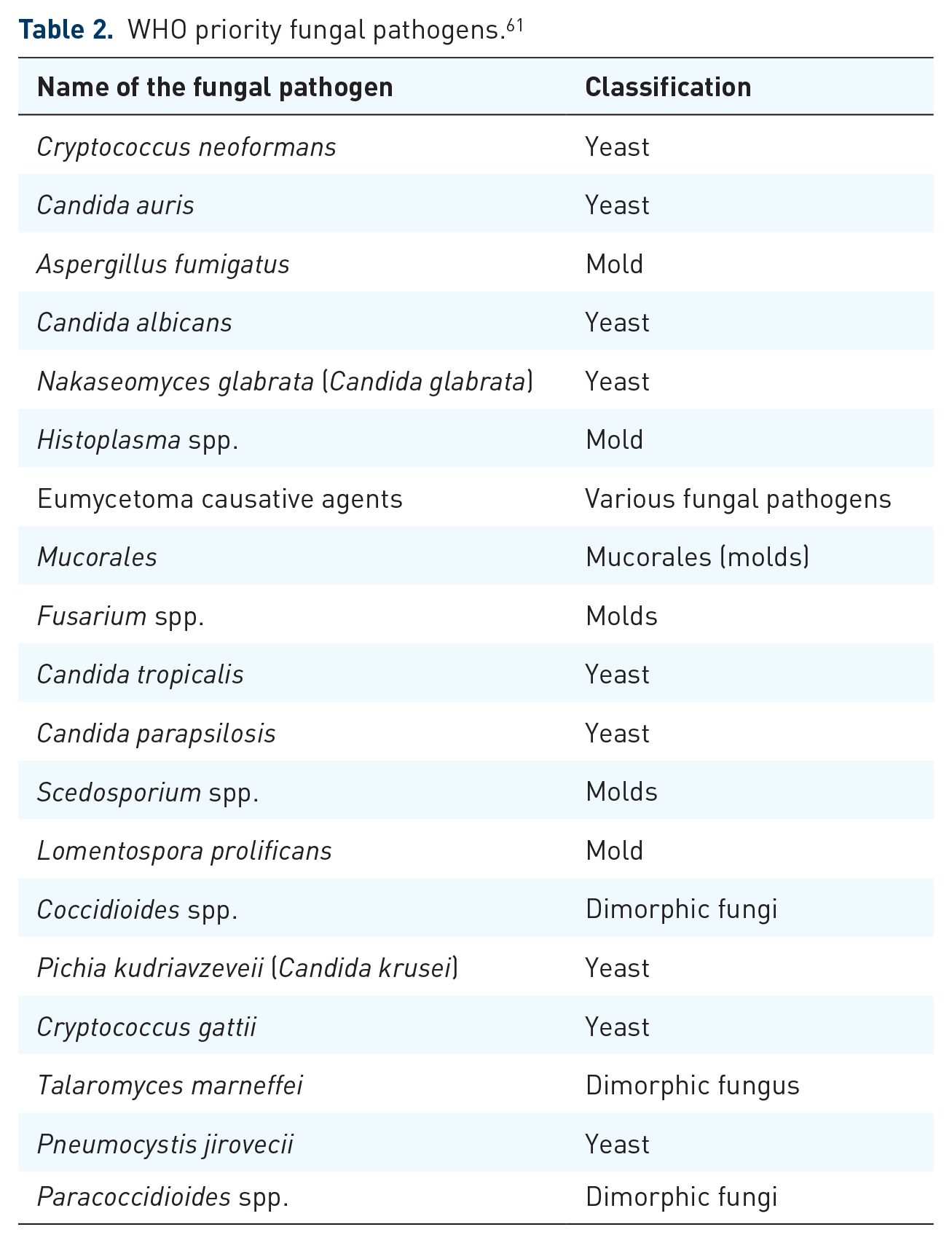
Numerous adaptive mechanisms of resistance to antifungal drugs have been recognized, including the alteration or excessive expression of drug targets, the upregulation of multidrug efflux pumps, and the activation of cellular stress pathways. 62 Substitutions of amino acids within cytochrome P450 14 α-demethylase, encoded by the ERG11 (CYP51) gene, have been identified as a resistance mechanism to azole antifungals due to changes in its affinity for azoles.63,64 Azole resistance may be driven by energy-dependent efflux pumps, like those known as ATP-binding cassette (ABC) multidrug transporters. 65 Amino acid substitutions in the target site of the 1,3-β-d-glucan synthase (GS) Fks1p subunit are enough to cause reduced susceptibility to echinocandins. 66 Polyene resistance is typically associated with a reduction in the target ergosterol, which is caused by mutations leading to the loss of function in ergosterol synthesis genes. 64
Growing azole resistance in isolates of non-Candida albicans, azole resistance in Aspergillus fumigatus, and echinocandin resistance in Candida glabrata are examples of acquired antifungal resistance.67,68 In addition, new species are appearing to resist multiple classes of currently available antifungal agents, such as Candida auris. 69 Currently, the majority of antifungal drugs become ineffective due to antifungal resistance. This scenario is severe, especially in immunocompromised patients such as cancer patients (leukemia or lymphoma), autoimmune diseases (such as lupus or rheumatoid arthritis), patients with Human Immunodeficiency Virus/Acquired Immunodeficiency Syndrome (HIV/AIDS), and patients taking other immune-suppressing treatments.
Antimalarial drug resistance
Worldwide in 2023, there were approximately 263 million cases of malaria, leading to 597,000 deaths. 70 Malaria elimination efforts face challenges due to antimalarial drug resistance, such as artemisinins and their partner drugs in Artemisinin Combination Therapy (ACT).71,72 Currently, this problem has been noted in Plasmodium falciparum, Plasmodium vivax, and Plasmodium malariae. 71
Resistance mechanisms in malaria parasites include drug efflux, changes to the drug target, and enzymatic degradation or modification of the drug. In addition, there are fewer common mechanisms linked to the parasite’s lifecycle and metabolism. 73 For example, mutations in pfk13 contribute to artemisinin resistance by prolonging the parasite’s stay in the earlier, less drug-sensitive ring stage. This also enhances the unfolded protein response, which halts development as artemisinin is rapidly cleared and the damaged peptides are removed. 72 Hence P. falciparum artemisinin resistance, which emerged in the Mekong Subregion in 2008, resulting from mutations in the pfkelch13 gene. 74
The K76T mutation in the chloroquine resistance transporter gene (pfcrt) is strongly related to chloroquine resistance in P. falciparum. 75 Mechanistic studies show that mutations in PfCRT enable the transporter to pump chloroquine out of the digestive vacuole membrane into the cytosol, preventing it from reaching its target, heme/hemozoin.76,77 Although PfCRT is the primary driver of chloroquine resistance, sequence variants of PfMDR1, an ABC transporter located on the digestive vacuole membrane, can influence the extent of resistance. 78 Moreover, chloroquine resistance in P. vivax appears to be driven by an unidentified mechanism, involving transcriptional alterations in the orthologous pvcrt-o gene. This points to interspecies variations in the native function of the transporter, which is still not fully understood. 79
Early reports suggesting quinine resistance surfaced as far back as 1910. 80 During the WHO Global Malaria Eradication Program (1955–1969), the first instances of chloroquine resistance in P. falciparum were reported independently in South America and Southeast Asia.81,82 A comprehensive review of antimalarial drugs resistance was conducted by Matthew M. Ippolito et al. 83 and Didier Menard et al. 84 Resistance to artemisinin and its derivatives in P. falciparum is a growing concern in Southeast Asia, leading to high failure rates with ACT in several countries within the Greater Mekong subregion. This resistance threatens the region’s malaria elimination goals. If these hard-to-treat parasite strains reach sub-Saharan Africa, it could significantly increase malaria-related morbidity and mortality, indicating a need for intensive surveillance. 84 Every antimalarial drug has reports of resistance. This includes artemisinin, its derivatives, and ACT, which are the last resort for managing infections caused by P. falciparum.
Antiviral drug resistance
Antiviral drug resistance is becoming a growing concern, particularly among immunocompromised patients. 85 Acyclovir-resistant herpes simplex virus is recognized to cause conditions such as meningitis, esophagitis, and pneumonia in immunocompromised patients.86,87 When HIV replicates while the infected person is on antiretroviral (ARV) drugs, it can lead to the selection of drug-resistant strains. This HIV drug resistance can undermine the effectiveness of ARV drugs for both HIV prevention and treatment, potentially increasing HIV incidence, morbidity, and mortality 88 Zidovudine resistance was initially detected in clinical isolates in 1989. 34 The consequences of resistance to non-nucleoside reverse transcriptase inhibitors (NNRTIs) are generally more apparent than those of zidovudine resistance. As observed in in vitro studies, NNRTI-resistant strains can rapidly develop in real-life settings. 89 Nevertheless, resistance to HIV medications can undermine the effectiveness of antiretroviral (ARV) drugs in lowering HIV incidence as well as reducing HIV-associated morbidity and mortality. 90 Thus, WHO recommends consistent and periodic surveillance of HIV drug resistance to guide public health recommendations at local, national, and global levels. 91 Although various viruses can develop resistance, ARV drug resistance is particularly prominent and could potentially undermine the progress achieved in ARV treatment.
As the use of antiviral agents becomes more widespread in-patient care, antiviral resistance has transitioned from being primarily a laboratory concern to a clinical issue. Currently, resistant viruses are most commonly found in immunocompromised individuals who have been taking antiviral drugs for extended periods. Therefore, the development of antiviral resistance is likely to become more frequent and significant in the long term. 92 Developing antiviral drugs is very challenging due to the significant genetic diversity of viruses showing rapid mutation rates, which is aggravated by the development of resistance to antiviral drugs.
Acyclovir resistance in herpes simplex virus (HSV) is associated with mutations in the viral thymidine kinase (TK) or DNA polymerase. 93 Mutations in TK can result in either a total loss of TK activity (TK deleted or deficient virus) or, less commonly, a change in the substrate specificity of TK (TK altered virus). 94 In clinical practice, suspected or confirmed acyclovir-resistant HSV is typically treated with foscarnet, or less commonly, cidofovir. Treatment is often initiated empirically based on the prevalence of TK mutations, but cross-resistance can occur due to DNA polymerase mutations. There have also been reports of resistance to both foscarnet and cidofovir developing during therapy.95,96 Similar to HSV, varicella zoster virus (VZV) also produces a TK, and most cases of VZV drug resistance are due to mutations in the TK, 97 which often leads to a premature stop codon, rendering the virus TK-deficient, and they tend to cluster at specific loci within the VZV TK gene. 98 Foscarnet resistance has emerged in a small number of patients undergoing treatment with the drug for acyclovir-resistant VZV, 99 and linked to a mutation in the viral DNA polymerase. 99 In hepatitis B virus (HBV), high-level lamivudine resistance is mainly driven by M204I/V mutations, which occur in the YMDD (tyrosine-methionine-aspartate-aspartate) motif within the C domain of the polymerase gene, 100 and, less commonly, by A181V/T mutations. 101
There are two biochemical mechanisms by which resistance to nucleoside reverse transcriptase inhibitors (NRTIs) occurs. The first mechanism involves base changes that enable the reverse transcriptase (RT) enzyme to selectively exclude NRTIs during synthesis, inhibiting their incorporation into the DNA chain. 102 The second mechanism involves mutations that increase the rate of phosphorolytic removal of the chain-terminating NRTI residue from the 3’ end of the elongating viral DNA, allowing DNA synthesis to continue.102,103 The K70R mutation at codon 70 was initially identified in patients undergoing AZT monotherapy. 104 The mutations associated with NNRTI resistance are found near the hydrophobic pocket of the RT. 105 K103N is the most common resistance mutation that develops during the failure of an NNRTI-based regimen106,107 Resistance to protease inhibitors is linked to mutations in the protease gene, where amino acid changes decrease the ability of the inhibitor to bind to the protease. 108
AMR as a global threat
The United Nations General Assembly Resolutions in December 2015 109 and September 2016 110 acknowledged AMR as a global threat 111 and declared it to be among the top 10 global health threats. 112 AMR endangers our capacity to effectively treat infectious diseases.34,113 The lack of new antimicrobials in the development pipeline and infections caused by MDR pathogens becoming resistant make AMR to be a global threat.42,114 AMR is often called the “Silent Pandemic,” emphasizing the urgent need for immediate and more effective action to address the problem. 115
Antimicrobials that are effective against common bacterial infections such as sepsis, urinary tract infections, and diarrhea are becoming increasingly rare worldwide. 116 Consider the implications if all existing antimicrobials were to become ineffective in preventing and treating common infections. Such a scenario would be dire, signaling the arrival of the dreaded “post-antibiotic era.” Although we hope to avoid this grim future, there are already signs indicating that it could become a reality.
Prior research suggests that underdeveloped nations are probably the ones most negatively impacted by antibiotic resistance, despite the paucity of comprehensive data. This susceptibility results from ongoing developmental obstacles, such as specific problems with their healthcare systems.117,118 In developing countries, the risk that AMR could lead to higher rates of morbidity and mortality is elevated. This is due to a greater burden of bacterial illnesses, limited access to diagnostic tools, particularly microbiology, and fewer second-line antibiotics. 119 To put it another way, the lack of community awareness about AMR, limited access to high-quality and effective antimicrobials, inadequate antimicrobial stewardship programs, and weak drug regulatory activities in these countries all contribute to the rise of AMR.
The growing resistance rates among community-acquired infections, such as upper and lower respiratory tract infections, bacterial diarrhea, and typhoid fever, are not being met with the development of newer antibiotics. 120 This reduced effectiveness has led to greater difficulties in treating various illnesses, such as gonorrhea, tuberculosis, septicemia, and pneumonia.121,122 AMR can be transmitted and spread across large geographical areas through the environment. 123 Wastewater contamination, which frequently results from hospital or intensive farming operations, is a typical mechanism for antibiotics and antibiotic-resistant microorganisms to enter the environment. 124 The ongoing selective pressure has driven the evolution and spread of resistant strains, making many antimicrobials decrease their effectiveness. 3 When AMR is combined with inadequate infection control practices, resistant bacteria can readily transmit to other patients and into the environment. 125 Resistant bacteria can spread between individuals via direct contact, airborne particles, contaminated surfaces, food, and water. 126
Prevalence of AMR
Global estimates indicated that the number of deaths directly associated to AMR was 1.27 million in 2019.39,127 In addition, the Institute for Health Metrics and the University of Washington estimated that AMR might have contributed to nearly 5 million deaths worldwide in 2019. 39 If no decisive measures are implemented to address AMR, the projected number of deaths from infections caused by multidrug-resistant pathogens could rise to 10 million per year by 2050. 128 Approximately 50,000 lives are estimated to be lost annually in the USA and Europe due to infections related to AMR. 129 Notably, AMR-related mortality has risen to become the third leading cause of death globally. 39 Estimated number of deaths by 2014, 2019, and 2050 is shown in Figure 1.

The African continent experiences a significant level of resistance to widely used antimicrobials. 130 A study titled “Antimicrobial resistance in human and animal pathogens in Zambia, Democratic Republic of Congo, Mozambique and Tanzania” highlighted a rising trend in resistance to commonly used antimicrobials such as third-generation cephalosporins. 131 The occurrence of penicillin-resistant pneumococci, initially noted in the 1960s, has increased significantly over the past decade.132,133 A research carried out in 1997 indicated that approximately 33% of pneumococci exhibited resistance to penicillin. 133 A study conducted in Nigeria showed that 82% of Klebsiella isolates from blood showed resistance to ceftriaxone. 134 A different study from Nigeria conducted in 1996 found that 82% of Klebsiella isolates were resistant to ceftazidime and 71% were resistant to ceftriaxone. 135 AMR to various antimicrobials used in human medicine has been observed in bacteria isolated from food animals. Approximately 54% of Klebsiella and Escherichia coli species demonstrate strong resistance to third-generation cephalosporins. 136 Nowadays, there is no safe zone around the world free of AMR.
Economic impact of AMR
AMR could potentially reduce the global gross domestic product (GDP) by up to 3% by 2030, leading to an additional US$ 700 billion in global healthcare costs that year, with low-income countries bearing the brunt of this impact.128,137,138 The World Bank has estimated that up to 3.8% of the global gross domestic product could be lost due to AMR by 2050. 137 Another study reported that the Global GDP decline by 2050 has been estimated to be 2−3.5%, incurring a global cost of US$ 100 trillion by 2050. 128
The European Center for Disease Prevention and Control (ECDC) reported that approximately €1.5 billion is spent on additional patient care expenses due to infections related to AMR. 139 Worries about the financial impact of using antimicrobials to treat infections that are resistant to several drugs are growing. Furthermore, various studies highlight the significant additional costs these infections impose on the healthcare system. 140 The management of drug-resistant infections through longer hospital stays, the use of costly additional medications, heightened morbidity, and reduced workforce productivity results in greater expenses. This escalation in costs can lead to increased poverty and pose obstacles to achieving the Sustainable Development Goals.
Global priority pathogens/superbugs
Microorganisms that possess resistance mechanisms to at least one agent in a minimum of three different classes of antibiotics are commonly classified as MDR organisms.52,141 Superbugs are microbes that are resistant to the antimicrobial drugs. In many cases, infections caused by superbugs have limited treatment options. 142 Infections caused by superbugs lead to increased morbidity and mortality rates because treatment options for these resistant bacteria are significantly compromised. In addition, they incur hefty treatment expenses and frequently necessitate lengthy hospital stays. 143 Superbugs include hospital-acquired infections due to gram-positive bacteria like Staphylococcus epidermidis, Clostridioides difficile, and Streptococcus pneumoniae and gram-negative bacteria like Burkholderia cepacia, Stenotrophomonas maltophilia, Campylobacter jejuni, Citrobacter freundii, Enterobacter spp., Haemophilus influenzae, Proteus mirabilis, Salmonella spp., and Serratia species. 144 Without proper intervention to combat AMR, superbugs could turn into biological threats that claim millions of lives annually.
WHO, in 2017, identified ESKAPE pathogens Enterococcus faecium, Staphylococcus aureus, Klebsiella pneumoniae, Acinetobacter baumannii, Pseudomonas aeruginosa, and Enterobacter species as critical-priority bacteria, which play a significant role in the development of deadly AMR.43,145 Mycobacterium tuberculosis, the bacteria responsible for tuberculosis, should also be included in this list. Moreover, a global priority pathogen of a list of drug-resistant bacteria has been published (Table 3). 146 Bacterial pathogens are ranked as critical, high, or medium based on various factors like mortality rates, resistance prevalence, treatability, or the current development pipeline. 147 The high and medium categories encompass bacteria with growing drug resistance, which necessitates enhanced monitoring and prevention efforts. 148 Drug research and development, along with antimicrobial stewardship programs, should prioritize efforts to combat superbugs and the ESKAPE pathogens.
Priority pathogens for R&D of new antibiotics. 146
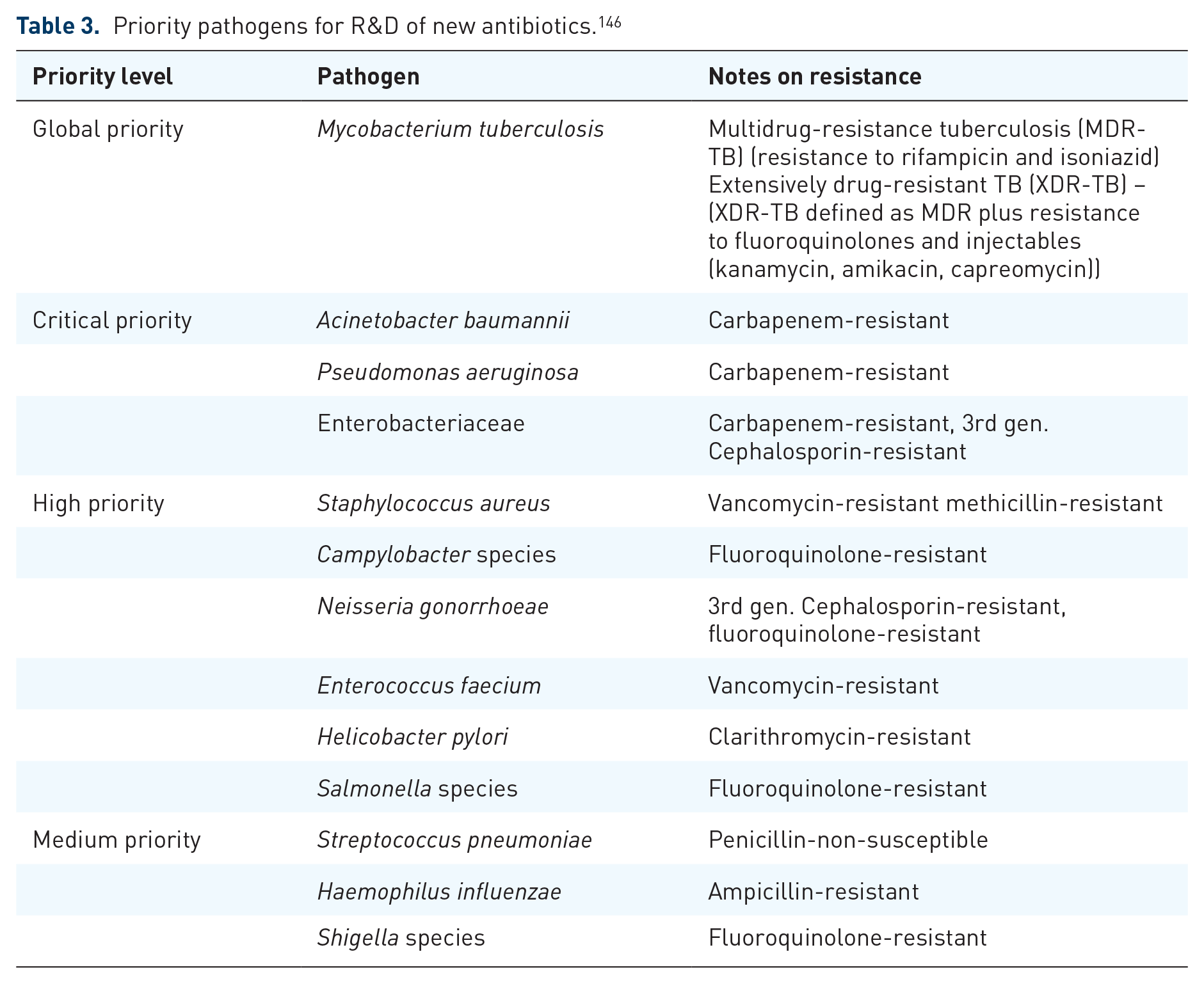
The primary causes of nosocomial infections worldwide include Enterococcus faecium, Staphylococcus aureus, Klebsiella pneumoniae, Acinetobacter baumannii, Pseudomonas aeruginosa, Enterobacter spp., and Escherichia coli, collectively known as the ESKAPE-E pathogens. 149 Noteworthy innate resistance and a broad capacity to develop resistance to several drugs are features of the ESKAPE-E infections. These pathogens have been identified by the WHO as a global health threat that urgently requires intervention. 43
In other words, critical-priority bacteria comprise Acinetobacter baumannii and Pseudomonas aeruginosa, which are resistant to carbapenem, and Enterobacteriaceae, which are resistant to both carbapenem and third-generation cephalosporins. 43 Whereas, high-priority bacteria include Enterococcus faecium, resistant to vancomycin; Helicobacter pylori, resistant to clarithromycin; and Staphylococcus aureus, resistant to methicillin. Other high-priority bacteria are Shigella spp., resistant to fluoroquinolones; Campylobacter spp., also resistant to fluoroquinolones; Haemophilus influenzae, resistant to ampicillin; and Streptococcus pneumoniae, resistant to penicillin. 150 In addition, the CDC grouped disease-causing germs into categories of urgent, serious, and concerning, threats and a watch list, depending on their AMR threat level 8 (Table 4). In addition, prioritizing disease-causing microorganisms allows stakeholders to take focused and effective action to control AMR.
Threat level of germs in causing AMR. 8

The growing trend of AMR has rendered even last-resort drugs less effective. For instance, vancomycin, a drug for MRSA, is facing diminished efficacy due to increasing resistance.151,152 Isolates of vancomycin-intermediate Staphylococcus aureus (VISA), linked to hospital stays, persistent infections, and the extension or failure of vancomycin therapy, have been found in various regions around the world. 153 The first vancomycin-resistant Staphylococcus aureus (VRSA) strain, with a minimum inhibitory concentration (MIC) for vancomycin of 16 μg/mL or less, was documented in the United States in 2002. This came decades after reports of similar European isolates that exhibited reduced susceptibility to teicoplanin, a glycopeptide antibiotic from the same class as vancomycin.154,155 Currently, treatment options for infections due to resistant germs are very limited.
Prevention and control strategies for antimicrobial drug resistance
Global actions/measures taken to tackle AMR
The World Health Organization and its global action plan on AMR and global antimicrobial resistance and use surveillance system
In May 2015, the 68th session of the World Health Assembly approved a global action plan, emphasizing the urgent need to enhance the knowledge and evidence base on AMR through surveillance and research to address the growing AMR threat. 156 World Health Organization (WHO) and its Global Action Plan summarize five pivotal strategic goals to address AMR: (1) Improve knowledge and awareness of AMR; (2) enhance knowledge via surveillance and research to strengthen infection control practices; (3) Enforce proper sanitation, hygiene, and infection prevention measures; (4) optimize the use of antimicrobials in human and animal healthcare; and (5) Encourage long-term investment in the development of new drugs, diagnostic tools, and vaccines. 156
During the same year, the WHO launched the Global Antimicrobial Resistance and Use Surveillance System (GLASS). Since then, substantial investment has been made to enhance AMR surveillance, aiming to generate high-quality evidence regarding its scale, distribution, and diversity worldwide. 157 Notably, GLASS is the first system to facilitate the harmonized global reporting of official national data on AMR and antimicrobial consumption (AMC). 157
One Health
The One Health approach, developed in 2003, is a collaborative effort between various sectors and is now supported by the joint quadripartite consortium, which includes the Food and Agriculture Organization of the United Nations (FAO), WHO, the World Organization for Animal Health (WOAH, formerly OIE), and the United Nations Environment Program (UNEP). 158 According to the One Health Institute at the University of California, Davis, One Health is a strategy aimed at ensuring the health and welfare of humans, animals, and the environment through collaborative problem-solving at local, national, and international levels. 159 AMR is a One Health issue, highlighting the connection between the health of humans, animals, and the environment. 8
It addresses a wide range of sustainable development goals to design, execute, and oversee programs, policies, and research on AMR surveillance. The goal is to enhance evidence and promote robust intersectoral collaboration among humans, animals, plants, and their common environment. 160 The One Health approach is built on three core principles: (1) communication, (2) coordination, and (3) collaboration, bringing together professionals from human, animal, and environmental sectors to share their expertise and work together. 161 A global evaluation of the One Health approach would promote AMR prevention through awareness initiatives, education on proper antibiotic usage, advocacy for political support, and antimicrobial stewardship. Moreover, cutting-edge computational and sequencing technologies, such as whole-genome sequencing (WGS) and next-generation sequencing (NGS), are valuable tools for investigating AMR across different sectors of the One Health framework. 162
Antimicrobial Stewardship Program
An Antimicrobial Stewardship Program (ASP) is an organized effort designed to ensure the proper use of antimicrobials. Its main objectives are to enhance to improve patient outcomes, reduce AMR, and limit the transmission of infections caused by MDR pathogens. 163 Originally, ASP was established to coordinate interventions that optimize antimicrobial use by selecting the most appropriate agent, determining the correct dose, route of administration, and duration of treatment, with a primary focus on ensuring the best possible outcomes for patients. 164
Reduced AMR can be achieved through the careful and responsible use of antimicrobials, following the principles outlined in the ASP. 165 Strategies to manage resistance involve educating both the general public and healthcare professionals about antimicrobial resistance, promoting the rational use of antimicrobials, and employing preventive measures to minimize infections and prevent resistance from spreading, such as vaccination and effective infection control protocols. 166
A key document in the area of AMR is the O’Neill report, which addresses all measures aimed at curbing its likely future implications. This includes fostering awareness and better hygiene practices to reduce the risk of the spread of infections, reducing unnecessary antimicrobial use, enhancing surveillance of AMR, and promoting the development of novel diagnostic tools. 128 One of the most effective measures proposed for ASP is to raise public awareness about AMR its preventive strategies, which necessitates efficient engagement with all relevant stakeholders. 167 The figure below depicts the global actions and measures implemented to combat AMR (Figure 2).

Global actions/measures taken to control AMR.
Novel strategies to control AMR
To control the emergence of AMR, alongside efforts such as the GAP-AMR, GLASS, One Health initiative, and AMS programs, it is essential to utilize novel antimicrobials or strategies with proven effectiveness. These include antimicrobial peptides, efflux pump inhibitors, antimicrobial combinations, phage therapy, and optimizing drug delivery systems, which are discussed below.
Antimicrobial peptides have gained significant interest as a new category of antibiotics. These peptide-based antimicrobials possess an amphipathic structure, which arises owing to their positive charges and hydrophobic amino acid components. 168 These peptides typically consist of fewer than 100 amino acids and often feature residues with a positive charge, including lysine, arginine, and histidine, along with a significant fraction of hydrophobic residues (exceeding 50%). 169 Antimicrobial peptides mainly exert their effect by interacting directly with the bacterial cell membrane. 170 In addition to compromising membrane integrity, antimicrobial peptides disrupt the production of proteins and DNA and suppress essential cellular processes such as folding of proteins, synthesis of the cell wall, and metabolic turnover.171,172 Owing to their multiple modes of action, AMPs exhibit potent antimicrobial effects on a broad spectrum of microorganisms, such as Gram-positive and Gram-negative bacteria, fungi, and viruses.173,174 Furthermore, these peptides show efficacy against disease-causing organisms that exhibit resistance to commonly used medications. 175
As traditional antimicrobial agents, antimicrobial peptides have their own pros and cons. One notable advantage of antimicrobial peptides is their ability to target biological sites different from those of traditional antibiotics. 176 Moreover, a key characteristic of many antimicrobial peptides is their capacity to utilize various mechanisms of action, which boosts their overall antimicrobial efficacy. For example, human cathelicidin LL-37 demonstrates direct antimicrobial effects, immune modulation, and antibiofilm activity. 177 While LL-37 is predominantly recognized for its action on the bacterial cell membrane, it also modulates both pro-inflammatory and anti-inflammatory immune responses. 178 Moreover, LL-37 demonstrates antibiofilm activity at concentrations that are physiologically relevant, which are significantly lower than its in vitro MIC.179,180 In contrast, some antimicrobial peptides have demonstrated notable nephrotoxicity, primarily as a result of the high doses required for effective treatment. 181 Even antibiotics that are clinically in use, like colistin, are reserved as a last resort due to their nephrotoxic effects. 182 Novel drug delivery systems can help minimize the systemic adverse effects associated with antimicrobial peptide therapy. Some commercially available peptide-based antibacterials include dalbavancin, daptomycin, colistin, oritavancin, polymyxin B, teicoplanin, vancomycin, and telavancin. 183
Another strategy to control AMR is the use of efflux pump inhibitors (EPIs). EPIs are compounds that obstruct the function of efflux pumps, which are key targets in drug resistance. By integrating these inhibitors into combination therapies, the effectiveness of antimicrobials can be significantly improved.184,185 EPIs are typically simple, stable, and affordable compounds that are generally safe for humans. 186 Certain EPIs also have the capability to prevent bacterial biofilm formation. Compounds such as thioridazine, Phe-Arg β-naphthylamide (PAβN), and arylpiperazine NMP are categorized as EPIs. When introduced, these compounds have been demonstrated to considerably decrease biofilm formation in different bacteria, including E. coli, K. pneumoniae, S. aureus, and Pseudomonas putida. 187
Restoring bacterial susceptibility has been successfully achieved by the use of combination antimicrobials. A synergistic effect can improve treatment efficacy when two or more medicines are combined according to the pathogenic microorganisms’ susceptibility patterns. This strategy inhibits the same target through different mechanisms (e.g., streptogramins), targets in different pathways (e.g., antibiotics used in antituberculosis therapy), or targets at different points within the same pathway (e.g., trimethoprim and sulfamethoxazole).188,189 Combination antibiotic therapy is often chosen to increase treatment efficacy for a number of infectious disorders, including HIV/AIDS, malaria, and tuberculosis. 188
Phage therapy targets and eradicates harmful bacteria by using bacterial viruses, or bacteriophages. Often referred to as “bacteria eaters,” phages have a number of benefits over conventional antibiotics, including ease of availability, diversity, natural replication, low toxicity, host-specificity, lack of antibiotic cross-resistance, and minimum environmental impact. Phage therapy has demonstrated efficacy in treating topical infections where antibiotics have failed, although it is unlikely to completely replace antibiotics. 190 Combining phages with antibiotics has demonstrated a synergistic effect, especially in addressing multidrug-resistant biofilms. 191
Another promising strategy to combat AMR is the use of novel drug delivery systems. These systems have been developed to overcome the limited cell permeability of antibiotics, enhancing the drug’s ability to penetrate cells. 192 Among the drug delivery systems, nanoparticles (NPs) can operate as both active antibacterial agents and drug delivery systems (DDS); their application is showing promise in the fight against AMR. Hence, by preventing biological degradation of the loaded antibiotics and blocking efflux pumps, NPs can operate as drug nanocarriers and aid in the fight against bacterial resistance. 193 In addition, NPs allow for controlled and sustained drug release, ensuring that therapeutic agents remain active for extended periods. This minimizes adverse effects on healthy cells and tissues by requiring lower dosages of the antibacterial agent to produce a therapeutic effect. 194 In addition, reactive nitric oxide (NO) and reactive oxygen species (ROS) produced by metallic NPs harm bacterial cell components. 195 Furthermore, liposome-encapsulated antimicrobials have demonstrated the capacity to circumvent a number of microorganism resistance mechanisms, such as enzymatic degradation, efflux mechanisms, and impermeable outer membranes. 196 Consequently, the effectiveness of antimicrobials in the fight against AMR can be increased by employing innovative DDS that enable targeted delivery, sustained release, and prevention of enzymatic degradation of antimicrobials.
Conclusion
AMR is an increasingly serious global health issue that endangers the effectiveness of antibacterials, antifungals, antiparasites, and antivirals. As bacteria, viruses, fungi, and parasites develop ways to resist these medications, the impact on human health and agriculture is significant. AMR results in prolonged illnesses, greater mortality rates, and higher medical expenses as a result of the requirement for more complex and intensive treatments. AMR is declared to be a global threat and is estimated to claim about ten million lives per year by 2050. More notably, the ESKAPE pathogens are responsible for a higher number of nosocomial infections and pose a serious threat to public health. Therefore, to control AMR, in addition to collaborative efforts through the One Health initiative and antimicrobial stewardship programs, the development and use of next-generation antimicrobials like antimicrobial peptides, bacteriophages, EPIs, and the use of novel antimicrobial delivery systems are crucial.
