Abstract
Background:
Simplification of antiretroviral regimens has the potential to improve both patient-reported outcomes (PROs) and therapeutic adherence in people living with HIV (PLWH). Dual therapy with dolutegravir (DTG) plus lamivudine (3TC) demonstrated good safety and efficacy, but its impact on PROs remains to be documented.
Objectives:
To evaluate PROs among adult PLWH switching from standard multi-drug therapy to the DTG/3TC combined therapy Dovato®.
Design:
A non-comparative, 6-month observational study in 25 French medical centers.
Methods:
Sociodemographic and biomedical data were collected from medical records and PROs from self-administered questionnaires at treatment switch (Day, D0) and at months (M) 1 and 6. Primary endpoints included changes between D0 and M6 in perceived toxicity, treatment acceptability, PLWH’s preferences, and the score value of the treatment impact dimension of health-related quality of life (HRQL) (PROQOL-HIV) during follow-up. Secondary endpoints encompassed scores in other HRQL dimensions and self-reported symptoms. Multivariable standard and mixed-effects linear regression models were used to identify factors associated with PRO values and changes over time. Additionally, binary logistic regression was used to identify factors associated with the discontinuation of combined DTG/3TC regimen.
Results:
In the study population (260 PLWH, 64.6% male, mean ± SD age: 51 ± 12 years, 16 ± 10 years since HIV diagnosis), 20 individuals stopped treatment during follow-up, without resumption. Men, individuals previously receiving abacavir/3TC/DTG, and those with better daily comfort and perceived treatment efficacy were less likely to stop treatment. Treatment impact-related HRQL, acceptability of treatment, and number of self-reported symptoms significantly improved at M1 and M6. Mental and cognitive HRQL improved at M6.
Conclusion:
Dolutegravir and lamivudine dual oral therapy improved several dimensions in HRQL and delivered a simplified treatment regimen designed for eligible patients, supporting a patient-centered approach to managing HIV care. Attention should be maintained on the reasons for treatment discontinuation, especially among women.
Trial registration:
Patient-Reported Outcomes HV BItherapy (PROBI) was registered as number NCT04788784 on https://clinicaltrials.gov/study/NCT04788784?term=PROBI&rank=2.
Plain language summary
Background:
Changing the usual HIV treatments to simpler ones could make a big difference for people living with HIV. A therapy using two drugs, dolutegravir (DTG) and lamivudine (3TC), looks promising. It is safe and works well, but we need to learn more about how it affects people's lives and their satisfaction with treatment.
Study goals:
Our study aimed to see how adults living with HIV felt after switching from their usual treatments to a two-drug combo, known as Dovato® (dolutegravir and lamivudine; DTG/3TC). We wanted to understand their experience, including any changes in their quality of life, how well they stuck to their treatment plan, and how satisfied they were with this new approach.
How we did it:
We followed 260 people at 25 different health centers in France for six months after they switched to DTG/3TC. We asked them to fill out questionnaires right after they switched and again after one and six months. These questionnaires helped us understand their quality of life, how easy it was for them to stick to their treatment, any symptoms they felt, and their overall thoughts about the new treatment.
What we found:
Most individuals did well with the switch. Only 20 individuals stopped taking the new treatment. Men, individuals on triple therapy involving the same molecules (DTG/3TC and another molecule), those who felt positive about their daily lives, and those who experienced immediate improvement after switching treatment were more likely to adhere to it. Individuals generally felt better about their health, were happier with their treatment, and reported fewer symptoms after switching. Particularly, they noticed improvements in their mental and cognitive health after six months.
Conclusion:
Switching to the two-drug DTG/3TC treatment improved several dimensions of HRQL and delivered a simplified treatment regimen designed for eligible patients, supporting a patient-centred approach to managing HIV care. It’s important to keep.
Keywords
Introduction
As HIV infection has become a manageable chronic condition, a renewed and increased interest has developed in challenging traditional three-drug antiretroviral regimens and moving toward two-drug regimens for initial or maintenance treatment in people living with HIV (PLWH). As they reduce the number of medications required to manage HIV while maintaining durable virological efficacy, two-drug regimens are now considered a viable alternative to three-drug ones 1 and are part of the standard of care.2,3
Their excellent efficacy and safety profile observed in clinical trials led to the use of dolutegravir (DTG) plus lamivudine (3TC) combination in international guidelines as a therapeutic option for the treatment of naive PLWH.3,4 DTG has a high genetic barrier to drug resistance, is generally well-tolerated, and is considered to have a more favorable metabolic profile compared with drugs from other classes such as protease inhibitors (PIs). 5 In addition, the DTG/3TC combination virological efficacy, both as a first line and as a maintenance therapy, was found to be non-inferior to that of standard three-drug regimens. DTG/3TC dual therapy regimen is recommended in patients without known or suspected resistance to the class of integrase inhibitors, or to lamivudine. 6
Lastly, in an era where the efficacy of HIV therapies is increasingly comparable, Patient-Reported Outcomes (PROs)—that is, self-reported measures of a patient’s health perception—have become the main criteria for selecting the best treatment option. 7 However, while the safety and efficacy of DTG-based two-drug regimens are well established, studies evaluating their impact on PROs are still lacking. The PROBI (Patient-Reported Outcomes HIV BItherapy) mixed-method study aimed to evaluate treatment acceptability, perceived toxicity, patients’ preferences, and health-related quality of life (HRQL) among PLWH switching from standard multi-drug therapy to the dolutegravir and lamivudine (DTG/3TC) combined therapy Dovato® (one tablet per day). The qualitative component of the study explored patients’ and healthcare professionals’ perceptions, knowledge, and representations of both standard and two-drug regimens using semi-structured interviews. The results obtained in this first component of the study are presented elsewhere. 8 The quantitative component, which results are presented here, aimed to assess the dolutegravir and lamivudine regimen’s impact on PROs using self-administered questionnaires. Patients completed these questionnaires at the time of the treatment switch, as well as at 1 month and 6 months afterward.
Materials and methods
Study design and objectives
The multicenter, longitudinal, non-comparative observational cohort study PROBI was conducted from April 2021 to May 2023, under real-life conditions. The study included a 6-month follow-up of PLWH who switched from a three- or four-drug therapy with a single tablet to the two-drug therapy dolutegravir and lamivudine (DTG/3TC) with a single tablet across 25 medical centers in France (10 located in the Paris area and 15 in other French regions). All patients received a single daily tablet of dolutegravir and lamivudine during follow-up. The primary objective of the study was to assess changes with time after the treatment switch in perceived toxicity, acceptability of treatment, PLWH’s preferences, and treatment impact dimension of HRQL. Secondary objectives included a longitudinal assessment of other dimensions of HRQL, self-reported symptoms, adherence to treatment, as well as CD4-T cell count and plasma HIV viral load.
Selection of participants
Outpatients enrolled in the quantitative part of the study fulfilled the following inclusion criteria: (i) being aged older than 18 years, (ii) having been prescribed dolutegravir and lamivudine by a clinician in routine care, (iii) living with HIV-1, (iv) receiving a three- or four-antiretroviral drug regimen, (v) having an undetectable plasma HIV viral load (HIV-RNA ⩽ 50 copies (cp)/mL) for at least 3 months.
Exclusion criteria were as follows: (i) being a pregnant or breastfeeding woman; (ii) having a known hypersensitivity to DTG or 3TC, their metabolites, or formulation excipient; (iii) having a documented resistance to DTG or 3TC; (iv) presenting an active, serious infection (other than HIV-1) requiring parenteral antibiotic or antifungal therapy within 30 days before inclusion; (v) having any other clinical condition or prior therapy that would render one’s unable to comply with protocol requirements; (vi) having an active Centers for Disease Control and Prevention (CDC) stage C HIV disease; (vii) having received radiation therapy or cytotoxic chemotherapeutic agents during the 30 days prior to inclusion and (viii) being co-infected with hepatitis B virus (HBV). All participants were required to have the cognitive and linguistic ability to complete several self-administered questionnaires.
Ethical considerations
Ethical approval was obtained from an Independent Ethics Committee (Comité de Protection des Personnes Sud-Méditerranée, France, study approval 2020-A03514-35). All participants provided oral consent to participate, which was documented in their medical records and further confirmed by them in the self-reported questionnaire. The PROBI study was conducted and reported in accordance with the STROBE statement. 9
Data collection
Sociodemographic, clinical, biological, and therapeutic data were collected from medical records using electronic case report forms. Data concerning psychoactive substance use, perceived financial situation, and PROs were collected using electronic or paper questionnaires. Participants were offered the possibility to answer the questionnaire at hospital. Alcohol consumption was assessed using the Alcohol Use Disorders Identification Test-consumption (AUDIT-c). 10 Unhealthy alcohol use was defined as an AUDIT-c score above 3 (for women) or 4 (for men). Tobacco use was assessed using the Fagerström test for nicotine dependence. 11 Collection of PROs was scheduled at baseline (Day 0, D0), which corresponded to the day of the switch to dolutegravir and lamivudine then at Month 1 (M1) and Month 6 (M6) of follow-up.
Assessment of PROs
HRQL was assessed using the PROQOL-HIV questionnaire, 12 a validated instrument specifically designed to assess the HRQL of PLWH. The PROQOL-HIV covers a wide range of physical, mental, and social issues related to HRQL and is sensitive to sociocultural context, disease stage, and antiretroviral treatment. It consists of 43 items divided into eight dimensions that can be scored into four global dimensions and four additional individual items. The recall period is 2 weeks, and the scores range from 0 to 100 (100 denoting the best HRQL). Overall treatment evaluation (OTE) was assessed at M1 and M6 using a single item evaluating an impact on daily life, since the beginning of the dolutegravir and lamivudine treatment. Positive changes were rated from 1 (“slight”) to 4 (“huge”), and negative ones from −4 (“huge”) to −1 (“slight”), leading to an individual score ranging from −4 to 4. Treatment acceptability (four items, one dimension), perceived toxicity, and preference for oral dual therapy (six items, two dimensions: preference for treatment, lifestyle convenience, and perceived efficacy) were assessed using the perceived toxicity and preference scale (PTP). The OTE and PTP scales were developed within the qualitative component of the PROBI study. Self-reported symptoms were measured using 24 items derived from the HIV Symptom Index. 13 Each item in the symptom questionnaire was coded from 0 to 4 (0: no symptom; 1: symptom without bother; 2: bothers a little; 3: bothers; 4: bothers a lot). A global score (standardized from 0 to 100) was calculated for all item scores. The number of self-reported symptoms and the number of bothersome symptoms (at least a little) were also described. Adherence to antiretroviral treatment was assessed using three items derived from the French ANRS Maladies infectieuses émergentes adherence questionnaire 14 evaluating missed doses during the previous 4 days and during the last weekend, as well as overall adherence assessed using a 10-point visual analog scale (0: never adhered; 10: always adhered).
Study endpoints
Primary PROs endpoints of the PROBI study included changes between Day 0 and Month 6 in scores evaluating the acceptability, perceived toxicity, and preference for dual therapy; and the score associated with the treatment impact (TI, 10 items) dimension of the PROQOL-HIV questionnaire. The other dimensions of the PROQOL-HIV were analyzed as secondary PROs endpoints, along with self-reported symptoms, OTE, and adherence scores. The other secondary endpoints included the percentage of participants with HIV-1 RNA < 50 cp/mL through M6, the number of participants with detectable HIV-1 RNA at M6; the change from baseline (Day 0) in CD4 T cell counts over time, and the number and percentage of discontinuations of the dolutegravir and lamivudine regimen.
Statistical analyses
Analyses were restricted to participants in the PROBI study who started the dolutegravir and lamivudine therapy and filled in the self-administered questionnaires for PROs collection (study population). Characteristics of participants were described at D0 using numbers and percentages. The percentage of questionnaires completed at the hospital was described at D0, M1, M6, then compared according to participants’ educational level (chi-square tests). The distribution of PROs scores was described at D0, M1, and M6 using means and standard deviations (SD). Standard regression models were used to identify factors associated with (i) discontinuation of dolutegravir and lamivudine during follow-up (binary logistic regression) and, (ii) treatment acceptability, preference for treatment, self-reported symptoms, and treatment impact-related HRQL scores at M6 (linear regression). Variables with a p-value lower than 0.20 in the univariable analyses were considered eligible for the multivariable analyses. The final multivariable models were built using a step-by-step method guided by the Akaike Information Criterion. PRO changes from D0 to M6 were analyzed using mixed-effects linear regression models, with individual as the random effect. The significance threshold was fixed at alpha = 0.05 in all analyses. Analyses were performed using R version 4.2.3 15 (R Foundation for Statistical Computing, 2021).
Results
Selection of the study population and questionnaire administration
Among the 266 PLWH initially enrolled in the PROBI study, one individual was excluded due to HBV co-infection. Additionally, five individuals were removed from the study population, including two who did not start treatment with dolutegravir and lamivudine, two who were lost of follow-up, and one who could not fill in the questionnaires. Two individuals did not meet inclusion criteria and were included despite a detectable viral load. This protocol deviation was authorized by the scientific committee, as HRQL is the primary endpoint, efficacy is the secondary endpoint, and the decision was based on a consideration of the patients’ overall clinical characteristics.
The study population thus comprised a total of 260 PLWH (Figure 1).

Flowchart of patient selection in the PROBI study.
Participants completed the questionnaires between April 12, 2021, and May 02, 2023, mostly online (79% at D0, 78% at M1, and 76% at M6) and, for the majority of them, in hospital (55% at D0, 52% at M1, and 53% at M6). Of the participants who completed the questionnaire in the hospital, around half were assisted by a member of the research team (55% at D0, 50% at M1, and 43% at M6). This percentage was significantly higher among participants with no formal education or only primary education compared with participants with a higher educational level (77% vs 50% at M0, p = 0.008; 77% vs 42% at M1, p = 0.002; 77% vs 34% at M6, p < 0.001).
Main characteristics of the study population
The majority of PLWH in the study population were men (64.6%), with a mean age of 51 ± 12 years, and 65.8% were born in Europe. Nearly half (45.6%) were overweight or obese (body mass index, BMI, greater than 25). The most frequent comorbidities recorded in the study population included dyslipidemia (28.5% of individuals), cardiovascular complications (24.2%), and current or past depression (21.5%). The use of antidepressant treatment or anxiolytics concerned 8.8% and 7.7% of the study population, respectively (Table 1). Regarding psychoactive substance use (n = 253), 61 individuals (24%) were daily tobacco smokers. Of these, 17 (27%) exhibited moderate to high nicotine dependence. Additionally, 35% reported unhealthy alcohol use, among whom three had a risk for alcohol dependence. Concerning HIV-related characteristics, the transmission modes most represented in the study population were heterosexual relationships (48.5%) and homo-bisexual relationships (45%). The mean ± SD time since HIV diagnosis was 16 ± 10 years, the mean duration of antiretroviral therapy was 14 ± 8 years, and the mean CD4 nadir value was 282 ± 197 cells/mm3. History of resistance to either lamivudine or emtricitabine was documented in 12 individuals (5.0%). Before the switch to dolutegravir and lamivudine, the vast majority of individuals (99%) had undetectable viral load. Two individuals did not meet inclusion criteria and had a detectable viral load (93 and 2490 cp/mL, respectively). In the study population, 72.4% of individuals were receiving a combination of one integrase inhibitor (INI) and two nucleoside reverse transcriptase inhibitors (NRTI), and within this subset, 36% had been administered a combination of TRIUMEQ® (dolutegravir, abacavir, and lamivudine; DTG/ABC/3TC). The predominant reasons for switching to dolutegravir and lamivudine reported by clinicians included a desire for treatment simplification (86.9%), prevention of long-term toxicity, (19.2%), and circumventing side effects (22%) (Table 1).
Sociodemographic and clinical characteristics of PLWH in the study population before switching to Dovato® (n = 260, PROBI).

Mean (SD).
Other HIV transmission groups: Injection drug use n = 9, Transfusion, or medical procedure n = 3, Mother-to-child transmission n = 2, Unknown n = 3.
NI, integrase inhibitor; NNRTI, non-nucleoside reverse transcriptase inhibitor; NRTI, nucleoside reverse transcriptase inhibitor; PI, protease inhibitor; PLWH, people living with HIV.
During the study period, 20 individuals discontinued dolutegravir and lamivudine (12 women and 8 men), without subsequent resumption. The underlying reasons for these discontinuations included various neuropsychiatric disorders (9 individuals) combined with muscular pain (2 individuals), and other symptoms such as loss of appetite or nephritic colic (6 individuals), digestive disturbances (5 individuals) or osteoarticular disorders (4 individuals). Fatigue and weight gain were reported by three individuals as a reason to stop treatment, and one individual reported adherence difficulties.
Post-discontinuation treatment regimens for these 20 individuals were as follows: 12 switched to 1 INI + 2 NRTI therapy, with one of them receiving dolutegravir, abacavir and lamivudine (DTG/ABC/3TC), seven received a combination of 1 NNRTI + 2 NRTI, and one patient was prescribed a PI + 2 NRTI regimen. Among the three patients who stopped dolutegravir and lamivudine (DTG/3TC) due to weight gain, two were on RPV + FTC + TAF and one on TDF + FTC + EVG.
Temporary treatment cessation due to travel-related difficulties was reported by two individuals. Factors associated with discontinuation of dolutegravir and lamivudine in the multivariable binary logistic regression model are detailed in Figure 2. After adjustment for age and time since HIV diagnosis, women were more likely to discontinue treatment than men, with an adjusted odds ratio (aOR) of 3.09 (95% CI: 1.05–9.57). Conversely, PLWH who were receiving dolutegravir, abacavir, and lamivudine (DTG/ABC/3TC) before the study (0.05 (0.00–0.30)) and those who reported higher scores in the lifestyle convenience and perceived efficacy dimensions of the Toxicity and Treatment Preference scale (0.95 (0.91–0.98)) were less likely to discontinue treatment. Antidepressant use in the previous 6 months (18 individuals) tended to be associated with higher odds of treatment discontinuation (p = 0.05) (Figure 2).
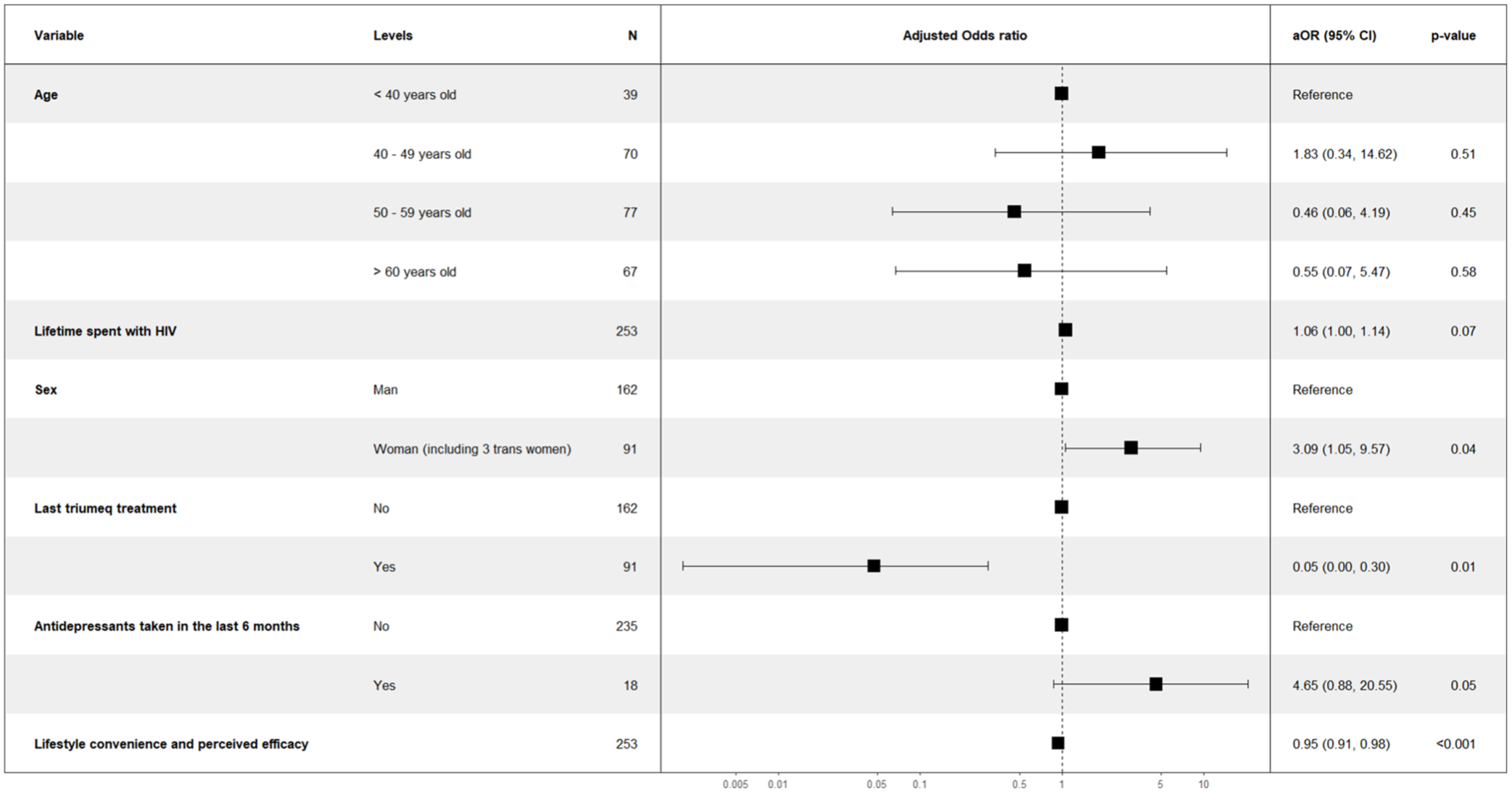
Results of the multivariable logistic regression model on factors associated with Dovato® treatment discontinuation (PROBI, n = 253).
Health-related quality of life
The distribution of HRQL scores at each visit is presented in Table 2. At D0, mean scores for the different dimensions of the PROQOL-HIV were as follows: 82 ± 18 for physical HRQL and symptoms, 83 ± 20 for relationships with others (including social relationships, sexual relationships, and self-image), 83 ± 15 for treatment impact, and 69 ± 24 for mental and cognitive HRQL. Mixed-effects models showed a significant improvement of the treatment impact dimension at M1 and M6 with values for the β model coefficients (95% CI) of 4.9 (3.4–6.5) and 5.1 (3.5–6.7), respectively. The Mental and Cognitive dimension of HRQL also significantly improved at M6 (β: 4.3 (2.1–6.4) (Table 3).
Distribution of PRO scores during follow-up in the PROBI study.

Score between 0 (worse HRQL) and 100 (best HRQL).
Score between 0 (lowest symptom burden) and 100 (highest symptom burden).
HRQL, health-related quality of life; PRO, patient-reported outcome; SD, standard deviation.
Changes over time in PROs during the PROBI study (mixed-effects linear regression models at M1 and M6).

Significativity based on 95% confidence interval.
p<0.05.
Beta, model coefficient; CI, confidence interval; HRQL, health-related quality of life; M1, Month 1; M6, Month 6; N, number of patients; PRO, patient-reported outcome.
In the multivariable model with the treatment impact dimension of the PROQOL HIV scale as the outcome, individuals reporting financial difficulties had lower scores at M6 (β = −5.3, 95% CI: −9.6 to −1.0, p = 0.016).
Acceptability of treatment, perceived toxicity, and preference for dual therapy
Mean scores for acceptability of treatment (Table 2) significantly increased at both M1 (β: 5.2 (2.2–8.3)) and M6 (6.0 (2.9–9.1)) (Table 3). Mean scores for toxicity and treatment preference tended to decline at both M1 and M6 (Table 2), but changes from D0 were not statistically significant (Table 3).
In the multivariable regression model, not working (β = −16.9, 95% CI: −29.3 to −4.6, p = 0.007), living alone (β = −4.9, 95% CI: −10.1 to −0.2, p = 0.061) and taking antiretroviral treatment 4 to 5 days a week (rather than once a day) (β = −17.9, 95% CI: −33.5 to −2.3, p = 0.025) were independently associated with lower treatment acceptability at M6 (Figure 3). These findings are echoed in the lifestyle convenience and perceived efficacy dimension of the Toxicity and Treatment Preference scale (Figure 4). Not working (β = −16.0, 95% CI: −26.0 to −6.1, p = 0.002), moderate to severe nicotine dependence (β = −9.4, 95% CI: −18.0 to −1.2, p = 0.02), and facing financial difficulties (β = −6.4, 95% CI: −12.0 to −0.5, p = 0.034) were significantly associated with lower treatment preference scores at M6. Individuals previously receiving dolutegravir, abacavir, and lamivudine (DTG/ABC/3TC), one more active component treatment than dolutegravir and lamivudine regimen also reported a lower preference at M6 (β = −4.3, 95% CI: −8.6 to −0.7, p = 0.046).

Results of the multivariable linear regression model on treatment acceptability score (PROBI, n = 222).

Results of the multivariable linear regression model on lifestyle convenience and perceived efficacy score (PROBI, n = 223).
Self-reported symptoms
At D0, study participants reported, in mean ± SD, 6 ± 5 symptoms, including 2 ± 3 bothersome symptoms, and the mean ± SD global score on the HIV Index Symptom scale was 14 ± 14 (Table 2). The total number of self-reported symptoms, the number of bothersome symptoms, and the global score on the HIV Symptom index all significantly decreased throughout follow-up (Table 3).
In the multivariable model, the following factors were independently associated with the global HIV symptom score at M6: moderate to severe nicotine dependence (β = 6.8, 95% CI: 0.4 to 13.1, p = 0.036), financial difficulties (β = 5.8, 95% CI: 1.4 to 10.3, p = 0.011), intake of sleeping pills in the previous 6 months (β = 6.6, 95% CI: 1.0 to 12.1, p = 0.020), a higher number of antiretroviral treatment lines before switching (β = 0.7, 95% CI: 0.2 to 1.2, p = 0.006) and duration of last line of antiretroviral therapy (β = −1.1, 95% CI: −1.6 to −0.5, p < 0.001).
Overall treatment evaluation
Mean score ± SD for OTE was 0.5 ± 1.2 both at M1 (n = 239) and M6 (n = 229).
Adherence to treatment
The mean adherence score on the 10-point visual analog scale was 9.1 ± 2.1 at D0, 9.5 ± 1.5 at M1, and 9.2 ± 2.0 at M6. The percentage of participants who did not miss their treatment dose during the previous 4 days was 87.4% at D0, 90.8% at M1, and 87.3% at M6. In addition, 93.7% of participants did not miss their treatment dose during the previous weekend at D0, 95% at M1, and 92.1% at M6.
Clinical and biological outcomes
The percentage of individuals with an undetectable HIV viral load (i.e., below 50 cp/mL) was 99% (258 out of 260) at D0, 100% (227 out of 227) at M1, and 99% (239 out of 241) at M6. Specifically, at D0, two individuals presented with viral loads of 93 and 2490 cp/mL, but by M1 and M6, both had undetectable levels. At M6, two individuals registered viral loads of 53 and 2278 copies/mL; the latter individual had briefly discontinued treatment at M6, which likely contributed to the elevated viral load level. Both had undetectable viral loads by 12 months.
Mean ± SD CD4 T-cell count (/mm3) was 732 ± 294 at D0 (n = 259) and 754 ± 300 at M6 (n = 220). Mean ± SD CD4 T-cell percentage was 36 ± 9 at D0 (n = 257), and 36 ± 10 at M6 (n = 221). Mean ± SD weight of participants was 75 ± 15 kg at both D0 (n = 257) and M6 (n = 221). Further clinical metrics are presented in Supplemental Data Table 1.
Discussion
The PROBI cohort study provides insight into the use of the dual oral therapy dolutegravir and lamivudine thanks to real-life data collection in a sample of 260 PLWH followed-up in HIV care centers in France. Findings set an excellent foundation for clinicians and researchers to understand the perceptions of PLWH receiving dolutegravir and lamivudine . The study population presents a diverse demographic and geographic distribution, including one-third of non-French native participants and 35% of women, thereby offering insights into understudied subpopulations. The study results show significant improvements in self-reported symptoms, acceptability of treatment, and treatment impact related to HRQL during the 6-month follow-up as well as high levels of self-reported treatment adherence. The occurrence of treatment discontinuations, which concerned 7.7% of the study population, underlines the need to monitor PLWH’s experience with therapy and perceived symptoms, especially among women, who appeared to have a higher risk of stopping treatment than men. PROQOL HIV scores values observed in the study population indicated HRQL levels higher than those observed in a representative sample of PLWH followed-up in French hospitals ten years ago 16 (82 vs 72 for physical HRQL and symptoms, 83 vs 73 for relationships with others (including social relationships, sexual relationships, and self-image), 83 vs 72 for treatment impact, and 69 vs 60 for mental and cognitive HRQL).
Similarly, in both studies, the lower scores in the Mental and Cognitive dimensions underscore the psychological and cognitive challenges faced by PLWH. The significant improvement throughout follow-up in the mean score for the treatment impact dimension of HRQL and positive scores obtained at M1 and M6 on the OTE scale, underscore a globally positive impact of the change for dolutegravir and lamivudine treatment on patients’ lives.
Other studies demonstrated improvements in PLWH's experience with treatments, assessed using different psychometric instruments. For instance, in the Salsa study, 17 virologically suppressed adult participants who switched to DTG/3TC reported greater enhancements in treatment satisfaction, as measured by the HIV Treatment Satisfaction Questionnaire (TSQ), compared with those who continued with a variety of three- or four-drug antiretroviral regimens. These improvements were noticeable as early as 4 weeks post-switch and remained stable throughout the first year of treatment. However, the magnitude of change remained limited, with the mean change from baseline in the HIV-TSQ score (ranging from 0 to 60) falling between 1.3 and 1.8 points. In the real-world URBAN study in Germany, 18 pre-treated PLWH receiving DTG/3TC and completing questionnaires at baseline and Year 2 reported a significant increase in the mean ± SD total HIV TSQ score, from 53.5 ± 8.4 to 56.7 ± 5.1, with a change of 3.2 ± 8.1 (p < 0.001).
In the qualitative phase of the PROBI study, 8 PLWH articulated the anticipated benefits of the dual therapy, which encompassed the possibility of initiating an antiretroviral regimen containing fewer drugs and the potential for preserving overall health with reduced long-term toxicity. They also reported trust in this new treatment due to its endorsement by their healthcare provider and saw the possibility they were offered to switch to dual therapy as indicative of overall good health status.
In our study, we did not observe significant improvements in the other dimensions of PROQOL HIV, such as the physical dimension. These findings align with that of the Gemini study, 6 where the general HRQL scores (measured with generic tools) were high at baseline; changes observed at Week 48 were minimal and did not significantly differ between treatment groups.
Neither of the two dimensions of the Toxicity and Treatment Preference Scale exhibited a significant mean score change from baseline to M6, despite a tendency to lower scores which could be related to scale items inquiring about PLWH’s perception of the long-term efficacy of treatment, a factor that may be challenging for some individuals to evaluate. During the qualitative phase of the PROBI study, 8 some PLWH, particularly individuals with a longer history of HIV, expressed concerns about the risk of a lower efficacy of the oral dual therapy.
We observed a significant increase in the mean score of the Acceptability of Treatment Scale at both M1 and M6. Higher acceptability often correlates with better adherence and consequently, improved health outcomes. 19 As expected, patients being previously on dolutegravir, abacavir, and lamivudine (DTG/ABC/3TC) had less potential to perceive a change after treatment switch.
Interestingly, the number of self-reported symptoms, including bothersome symptoms, significantly decreased from D0 to M6, suggesting reduced symptom distress with the dual oral therapy. PLWH's favorable rating on the OTE scale underscores a moderately positive perception of the impact of the switch to dolutegravir and lamivudine treatment on everyday life. This may be related to the fact that most PLWH in the study population were diagnosed with HIV for many years and probably settled in a daily routine of treatment.
The consistently high adherence scores recorded across the study duration reaffirm the acceptability of treatment and participants' commitment to their health regimen. This is promising, given that adherence is critical to the success of antiretroviral therapy. 20
Despite high adherence levels in the study population, 20 PLWH stopped treatment with dolutegravir and lamivudine, without resumption. The reasons provided for treatment discontinuation, including neuropsychiatric symptoms and digestive disturbances, emphasize the need for close monitoring and management of side effects. Despite high adherence scores, the subtle discrepancies in reported consistency, especially concerning missed doses over weekends, underscore potential challenges in sustained medication intake, possibly due to lifestyle factors, forgetfulness, or other unforeseen disruptions. There was not a treatment-specific pattern of tolerability and the reasons to stop were various and similar to the reasons observed in other studies.4–6 No new or unexpected problems of tolerability emerged in the present study. Almost all patients maintained an undetectable viral load throughout this real-life study, considering an adherence rate of approximately 90%. Of note, the two participants with HIV-RNA > 50 cp/mL at D0 had undetectable viral load at M6.
This study presents several limitations. First, it was not able to assess the information about the screened population and the reasons for refusal. Second, participation was restricted to a single country, albeit with a significant proportion of PLWH born abroad and the inclusion of one center in the Caribbean. The cultural, healthcare system, and socioeconomic context of France may limit the generalizability of the study findings to other global settings. In addition, while it would be beneficial to have data collected over a longer period, the study duration allows for an evaluation of the dolutegravir and lamivudine treatment effects on PROs in both short-term (M1) and medium-term (M6) scenarios. The limitation to include only French-speaking participants was mitigated by the fact that those with limited literacy could fill in the questionnaire with the help of a research assistant. Lastly, the implemented measures of HRQL treatment acceptability and adherence are based on self-reported scales, which are susceptible to biases such as recall bias and social desirability bias. 21
Conclusion
The PROBI study largely portrays a positive impact on PROs of the switch from standard multi-drug therapy to the dolutegravir and lamivudine (DTG/3TC) combined therapy. Beyond high virological suppression, this drug showed improvements in several dimensions of HRQL: treatment impact and mental and cognitive dimensions, acceptability of treatment, toxicity and treatment preference, and number of symptoms. Regarding the individualization of therapeutic strategies in real life, one should remain vigilant about the potential reasons for treatment discontinuation, especially among women. Dolutegravir and lamivudine dual oral therapy delivers a simplified treatment regimen designed for eligible patients, supporting a patient-centered approach to managing HIV care.
Supplemental Material
sj-docx-1-tai-10.1177_20499361251321728 – Supplemental material for Patient-reported outcomes among people living with HIV switching to dual therapy with dolutegravir/lamivudine: results from the PROBI study
Supplemental material, sj-docx-1-tai-10.1177_20499361251321728 for Patient-reported outcomes among people living with HIV switching to dual therapy with dolutegravir/lamivudine: results from the PROBI study by Martin Duracinsky, Tristan Alain, David Zucman, Olivia Rousset Torrente, Eva Brown Hajdukova, Laurent Hocqueloux, Clotilde Allavena, Fabienne Marcellin and Olivier Chassany in Therapeutic Advances in Infectious Disease
Supplemental Material
sj-docx-2-tai-10.1177_20499361251321728 – Supplemental material for Patient-reported outcomes among people living with HIV switching to dual therapy with dolutegravir/lamivudine: results from the PROBI study
Supplemental material, sj-docx-2-tai-10.1177_20499361251321728 for Patient-reported outcomes among people living with HIV switching to dual therapy with dolutegravir/lamivudine: results from the PROBI study by Martin Duracinsky, Tristan Alain, David Zucman, Olivia Rousset Torrente, Eva Brown Hajdukova, Laurent Hocqueloux, Clotilde Allavena, Fabienne Marcellin and Olivier Chassany in Therapeutic Advances in Infectious Disease
Footnotes
Acknowledgements
This paper was written on the collaborative platform Mypubli.online. Participating centers: European Hospital of Marseille, Infectious disease department: Barbara Gasse, Dr. Patrick Philibert, Dr. Christine Psomas; Foch Hospital, “Ville-Hôpital” Network: Camille Vassord-Dang, Dr. David Zucman, Dr. Erwan Fourn, Dr. Catherine Majerholc; University Hospital of Nantes, Infectious disease department: Dr. Antoine Asquier Khati, Dr. Eric Billaud, Morane Cavellec, Dr. Antoine Gregoire, Dr. Renate Mahot, Dr. Pascale Morineau, Ernesto Paredes Manyari, Dr. Véronique Reliquet, Albane Soria; Paris Hospitals—Necker-Sick Children Hospital, Adult infectious and tropical diseases: Dr. Claudine Duvivier, Dr. Cléa Melenotte, Dr. Perrine Parize, Elisabete Pires, Dr. Claire Rouzaud, Fatima Touam; Paris Hospitals—Hôtel-Dieu Hospital, Infectious disease department: Marie-Pierre Pietri, Pr Dominique Salmon-Ceron, Dr. Dorsaf Slama, Dr. Olivier Zak Dit Zbar; Paris Hospitals—Bicêtre Hospital, Infectious disease department: Françoise CHURAQUI; Saint-Denis Hospital Center—Delafontaine Hospital, Infectious and tropical diseases: Emilie Chenot, Dr. Benoît Frison, Dr. Marie-Aude Khuong, Randa Maamar, Awa Ndiaye, Dr. Marie Poupard; La Rochelle Hospital, Infectious disease department: Pascale Camps, Lucie Marchandeau, Dr. Mariam Roncato-Saberan; University Hospital of Poitiers, Infectious and tropical diseases: Dr. Gwenael Le Moal, Dr. Mélanie Catroux, David Plainchamp, Pr France Cazenave-Roblot; University Hospital of Rouen—Charles Nicolle Hospital, Infectious diseases department: Pr Kevin Alexandre, Dr. Yasmine Debab, Dr. Elise Fiaux, Dr. Anaïs Lesourd, Dr. Véro Rasoldier, David Theron, Guillemette Unal, Dr. David Vautrin; University Hospital of Reims, Infectious diseases department: Pr Firouzé Bani-Sadr, Isabelle Kmiec; Local Hospital Center of Vendée - La Roche-sur-Yon Hospital, Infectious disease department: Dr. Olivier Bollengier-Stragier, Hélène DURAND, Laetitia Laine, Dr. Sophie Leautez-Nainville, Dr. Dominique Merrien, Dr. Marine Morrier, Armelle Pégeot, Hélène Pelerin, Dr. Philippe Perré; University Hospital of Guadeloupe—Pointe-à-Pitre Hospital, Infectious and tropical diseases: Emelyne Duvallon, Dr. Isabelle Lamaury, Ketty Samar; University Hospital of Clermont-Ferrand, Infectious and tropical diseases: Emilie Goncalves, Dr. Christine Jacomet, Dr. Magali Vidal; Cornouaille Hospital Center—Quimper Hospital, HIV-HCV network: Jean-Charles Duthe, Dr. Nolwenn Hall, Dr. Pasacale Perfezou; Marseille Hospitals—Sainte-Marguerite Hospital, Clinical immunohematology department: Dr. Sylvie Bregigeon, Alena Ivanova, Hélène Laroche, Anne-Suzel Ritleng, Teresa Rojas, Dr. Olivia Zaegel-Faucher; Sud-Francilien Hospital, Infectious disease department: Nouara Agher, Dr. Amélie Chabrol, Dr. Thomas Gabas, Dr. Aurélia Henn, Dr. Olivia Son; Villeneuve-Saint-Georges Hospital, Infectious and tropical diseases department: Pauline Caraux-Paz, Laurent Richier, Dr. Sophie Dellion; Hospital of Toulon, Infectious diseases department: Dr. Gilles Hittinger, Véronique Lambry, Dr. Fanshen Lionetto; Local Hospital of Créteil, Internal medicine and Infectious diseases department: Dr. Isabelle Delacroix, Dr. Valérie Garrait; Paris Hospitals—Antoine Béclère Hospital, Internal medicine clinical immunology department: Pr Sophie Abgrall, Dr. Renato Fior, Sandrine Poirier; University Hospital of Caen Normandie, Infectious diseases Department: Dr. Arnaud de La Blanchardiere, Manuela Le Cam, Dr. Anne Martin, Dr. Jocelyn Michon, Dr. Vincent Noyon, Anne Ricci, Pr Renaud Verdon; Paris Hospitals—Jean-Verdier Hospital, Infectious and tropical diseases: Dr. Johann Cailhol, Flory Mfutila Kaykay. We also would like to thank the patients of the study.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
