Abstract
Background:
Bacterial infections frequently occur in haematological patients, especially during prolonged neutropenia after intensive chemotherapy, often leading to bloodstream infections and pneumonia.
Objective:
Routine antimicrobial prophylaxis (AMP) for high-risk haematology patients is still debated while prevalence of multi-drug resistant (MDR) Gram-negative bacteria (GNB) is rising globally. We aimed to assess the current practice of AMP in this population.
Design:
Cross-sectional observational survey study.
Methods:
Haematologists and infectious diseases physicians Europewide were invited to an online survey including questions on routine screening for GNB, incidence of MDR-GNB colonization, antimicrobial prophylaxis practices, rates of bloodstream infections (BSI), ICU admission and mortality differentiated by infections due to GNB versus MDR-GNB.
Results:
120 haematology centres from 28 countries participated. Screening for MDR-GNB is performed in 86.7% of centres, mostly via rectal swabs (58.3%). In 39.2% of routine AMP is used, mostly with fluoroquinolones. Estimates of GNB-BSI yielded higher rates in patients not receiving anti-GNB prophylaxis than in those who do for E. coli (10% vs 7%) Klebsiella spp. (10% vs 5%), and Pseudomonas spp. (5% vs 4%). Rates for MDR-GNB infection were estimated lower in centres that administer AMP for MDR E. coli (5% vs 3%) Klebsiella spp. (5% vs 3%), and Pseudomonas spp. (2% vs 1%). In an exploratory analysis, Southern and Eastern European countries expected higher rates of MDR-GNB infections with lower ICU admission and mortality rates which may be subject to estimation bias.
Conclusion:
Screening for MDR-GNB is frequently performed. AMP against GNB infections is still often implemented. Estimated BSI rates are rather low, while the rate of MDR-GNB infections rises. Tailored prophylaxis including antimicrobial stewardship becomes more important.
Keywords
Introduction
Bacterial infections occur frequently in patients with acute myeloid leukaemia (AML), high-risk myelodysplastic syndrome (MDS) as well as in acute lymphoblastic leukaemia (ALL) and other patients with haematological malignancies. They occur mostly during long-lasting (i.e., ⩾7 days) periods of neutropenia (i.e., absolute neutrophil count < 500/µl) after intensive chemotherapy, mostly as bloodstream infection (BSI) and pneumonia. 1 After chemotherapy, translocation of gram-negative bacteria (GNB) from the gut to the bloodstream and dissemination to other organs may occur through an impaired mucosal barrier, often leading to severe infections with sepsis and a subsequent high mortality rate. 2 Routine antimicrobial prophylaxis is recommended with moderate strength by some guidelines despite lacking evidence regarding a survival benefit from randomized clinical trials (RCT).3–6 A rising incidence of multidrug-resistant (MDR) GNB has been observed over past decades, particularly in Southern Europe posing a significant challenge for selection of antibacterial therapy or prophylaxis. 7 This fuelled a debate regarding efficacy of prophylaxis.4,5 While MDR-GNB colonization rates and associated infection and mortality risk have been well described for the intensive care unit (ICU), transplant and abdominal surgery setting,8–16 few studies in the haematological population exist. 17
More recent guidelines restrict prophylaxis to certain settings and clinicians discourage the use of routine fluoroquinolone prophylaxis with a variety of arguments, including lack of efficacy, quality of data included in guideline recommendations, increasing antimicrobial resistance, and adverse effects, among others.6,18
Owing to the heterogeneity of recommendations, it is currently unclear what is routinely performed in haematology centres. We conducted a web-based survey to better understand the current practice of antimicrobial prophylaxis. This survey aimed (1) to assess the current practice of prophylaxis in this high-risk patient population and (2) to assess practice of screening for MDR-GNB and (3) to include estimated rates of infections due to frequently detected GNB.
Methods
A web-based cross-sectional observational survey was designed and made accessible via www.clinicalsurveys.net – a health services research platform facilitated by EFS Summer 2021, TIVIAN GmbH in Cologne, Germany. Data collection specifically targeted professionals specializing in clinical microbiology, haematology, infectious diseases, internal medicine, and oncology, as these are the main medical specialities managing patients with GNB infections and prescribing AMP. Targeted professionals were reached out to via scientific societies (see Supplemental Table 1) and professional networks as well as direct contacts. This approach aimed to ensure a comprehensive, reliable data collection while accounting for differences in service quality and quantity across the selected hospitals.
Participants were encouraged to respond on behalf of their clinic/hospital and distribute the survey among their personal network. Participants were obligatorily asked to provide their country and the institution on behalf of which they responded to the survey, whereas other personal data, affiliated scientific organizations, and their specialty were voluntary responses. Participants were asked to provide estimated numbers of patients with underlying disease of ALL, AML and high-grade MDS who receive induction or re-induction chemotherapy and have long-term neutropenia (i.e., ANC < 500/µl for at least 7 days) at their haematology centre. For this patient group we asked whether screening for colonization with GNB prior to chemotherapy is in place and if so, which proportion of patients have resistant GNB colonization. Furthermore, we evaluated whether antimicrobial prophylaxis is routinely administered and which antibiotic was used for this purpose. Estimated rates of development of BSI due to GNB (resistant or not) in those patients were to be provided as well as causative species and proportion of MDR-GNB-BSI in patients who had received or who had not received antimicrobial prophylaxis, if this data was available.
The collected data were summarized using frequencies and percentages. Descriptive statistical analysis was done with SPSS v27.0 (SPSS, IBM Corp., Chicago, IL, USA).
Results
Between May and September 2023, participants from 28 European countries representing 120 institutions responded to the survey (Figure 1). After clearance of double-answers and incomplete answers not eligible for analysis, 120 respondents were included in the descriptive analysis.

Distribution of participating institutions.
A total of 104 participating centres (86.7%) routinely screen for colonization with MDR-GNB before chemotherapy. Most frequently, this is performed via rectal swab, followed by anal swab, and stool culture (Table 1 and Figure 2(a) and (b)).
Survey results: overall and separately for Italy, Spain and other European countries.

Categorical variables are summarized with n and percentage (%); continuous variables are summarized with median, (interquartile range) and [absolute range].
Other European countries include Albania, Armenia, Belgium, Bulgaria, Croatia, Czech Republic, Denmark, Faroe Islands, France, Germany, Greece, Hungary, Ireland, Israel, Lithuania, Moldova, Netherlands, Norway, Poland, Portugal, Russia, Serbia, Slovakia, Slovenia, Switzerland, and the United Kingdom.
Full details in Supplemental Table 2.
AMP, antimicrobial prophylaxis; GNB, Gram-negative bacteraemia; ICU, Intensive Care Unit; MDR, multi-drug resistant; spp., species.
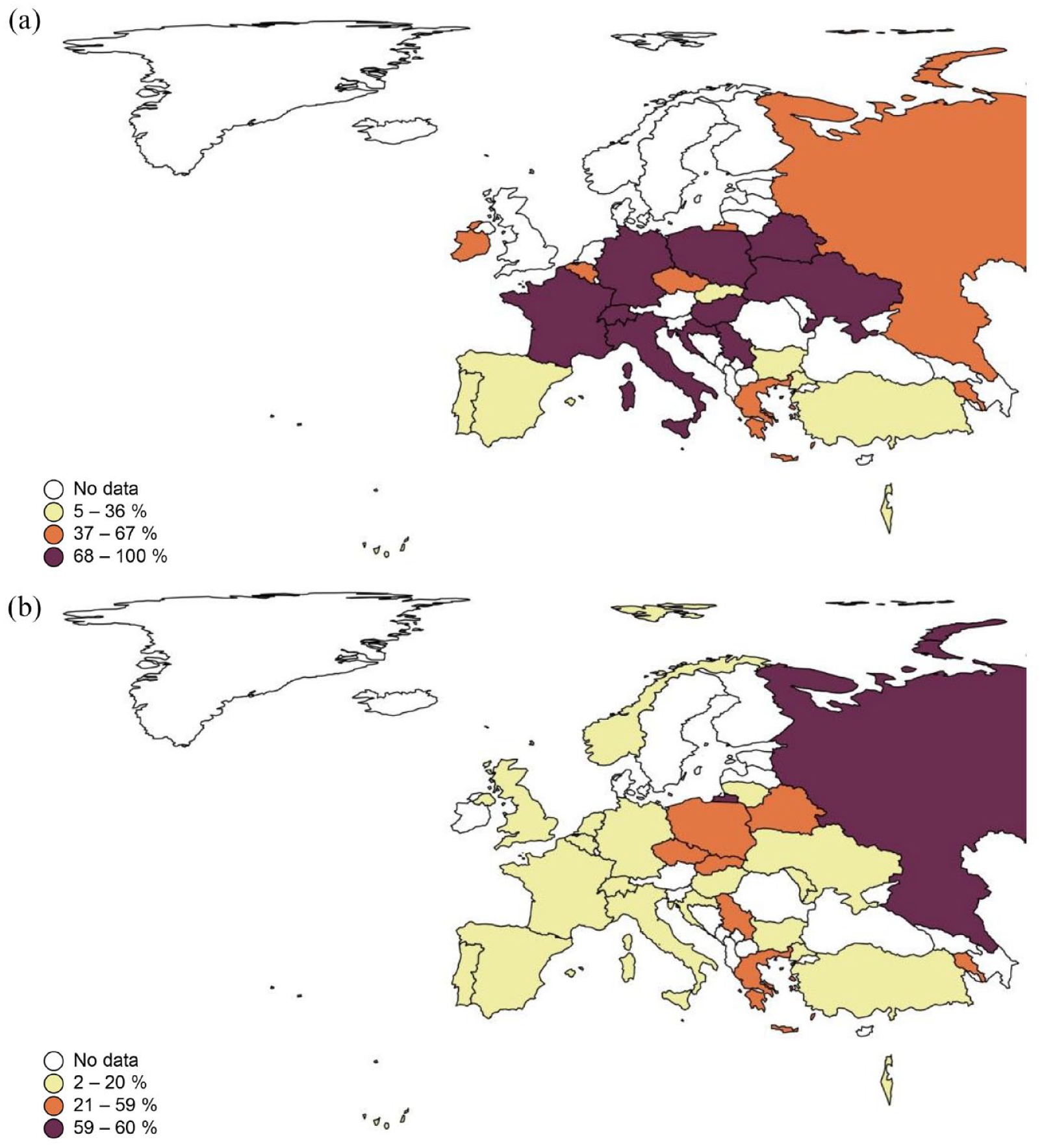
Distribution of routine testing practices for Gram-negative bacterial colonization: overall and multidrug-resistant strain considerations: (a) percentage of patients tested for colonization with Gram-negative bacteria and (b) percentage of tested patients colonized with a multi-drug resistant Gram-negative bacteria.
Only 39.2% of participating centres administer antimicrobial prophylaxis against GNB, mostly using levofloxacin or ciprofloxacin in patients without known resistant GNB colonization. However, even in patients with resistant GNB colonization, fluoroquinolones are routinely administered in 32.5% of participating centres, but antibiotics with a broader spectrum of action are used as well for prophylaxis, such as amikacin, carbapenems or colistin (Table 1).
Estimates of BSI development due to GNB yielded slightly higher rates of bacteraemia in centres not administering AMP than in those who do for E. coli (10% vs 7%) Klebsiella spp. (10% vs 5%), and Pseudomonas spp. (5% vs 4%). Rates for MDR-GNB BSI were also lower in centres that administer prophylaxis for MDR E. coli (5% vs 3%), MDR Klebsiella spp. (5% vs 3%), and MDR Pseudomonas spp. (2% vs 1%) (Table 1). Median mortality was estimated to range around 5% (IQR 2–15) but was excessively higher in patients with MDR-GNB colonization with 52% (IQR 30–66.7) (Figure 3(a) and (b)).

Comparison of overall mortality and mortality from multidrug-resistant Gram-negative bacteria after bacteraemiaemia development: (a) overall mortality after development of bacteraemia and (b) mortality due to MDR Gram-negative bacteria (from overall) after development of bacteraemia.
With high participant rates from Italy and Spain, countries where the prevalence of MDR-GNB isolates is increased, we split results for the two countries compared to the overall participants. Rectal swab for screening for GNB is significantly more often used in the two countries than in the rest of Europe (80% vs 58.3%, p = 0.010). No differences compared to other centres in Europe were detected regarding the indication for administration of routine antimicrobial prophylaxis, as well as the selection of antibiotics for that purpose, mostly fluoroquinolones (Table 1). In patients without MDR-GNB colonization, prophylaxis is administered in 32.5% (40% in Italy, 20% in Spain, 34.1% in other European countries, p = 1.000). In patients with MDR-GNB colonization prophylaxis is administered in 32.5% (33.3% in Italy, 35.0% in Spain, 31.8% in other European countries p = 1.000)
Resistance rates for GNB were estimated to be as frequent as in other European countries, in around one-third of the patients within this high-risk population (Table 1). With lower participant numbers from South-Eastern Europe where the prevalence of MDR-GNB isolates exceeds 50%, we did not perform a separate analysis for this region.
The estimated development of bacteraemia with any of the three given GNBs: E. coli, Klebsiella spp. and Pseudomonas spp., did not differ between Italy, Spain, and other European countries (Table 1). In patients not receiving Gram-negative prophylaxis, overall, 69.2% were estimated to develop bacteraemia, whereas this was 66.7%, 80% and 67.1% for Italy, Spain and other European countries, respectively.
However, ICU admission rates are lower in Spain and Italy compared to other European countries, with an overall median of 15% of those patients (IQR 5–30) versus 5% (IQR 2–5) in Italy, 15% (IQR 5–20) in Spain, and 17.5% (IQR 5–35) in other European countries (p = 0.005).
Discussion
Our survey demonstrated that prophylactic antibiotics are used in 39% of participating centres treating patients with AML, ALL and high-risk MDS.
In RCTs, levofloxacin reduced the incidence of fever, infection, and hospitalization compared to placebo, but did not impact survival.19,20 In patients with haematological malignancy, no causality has been established between colonization by MDR-GNB and mortality rate. Older data hint towards higher prevalence of MDR-GNB, namely ESBL-producing Enterobacterales (ESBL-E), than in our survey in the haematological population, with colonization rates up to 23% while only 1% of these patients developed BSI with ESBL-E, in 80% with the same strain that was identified previously as colonizer. 17 Antibiotic administration for prophylaxis may significantly impact the intestinal microbiota of patients exposed. There is evidence that intestinal microbiota diversity impacts the development and severity of infections. 21
Estimated BSI rates by E. coli, Klebsiella spp. and Pseudomonas spp. were comparably high in this survey in up to 10% of patients. Current studies suggest those three species remain the dominant causative pathogens of GNB-BSI in patients with leukaemia. 22
Selection of resistant GNB strains may occur in these high-risk patients under extensive empiric antimicrobial exposure as well as in other patient populations under immunosuppression, those with a travel or migration history and those with other antimicrobial exposure, for example, in agriculture. These infections mostly comprise AmpC- or extended-spectrum β-lactamase producing Enterobacterales, carbapenem-resistant Enterobacterales, Pseudomonas aeruginosa with difficult-to-treat resistance patterns, carbapenem-resistant Acinetobacter baumannii, and Stenotrophomonas maltophilia. 23 Surprisingly, in this survey study, rates for MDR-GNB infection were lower in centres that do administer prophylaxis, despite probable missing antimicrobial coverage. On the one hand, this is a finding which could be interpreted as an artefact as BSI rates were only provided as estimates. On the other hand, this may indicate that centres with high resistance rates are less likely to employ universal antibacterial prophylaxis assuming that this measure may be ineffective for a large proportion of patients.
The shift in prevalence of MDR-GNB may lead to more severe infections which cannot be prevented by routine antimicrobial prophylaxis, especially in geographic regions with high rates of antimicrobial resistance, such as – in this survey – Southern and Eastern Europe. 24 This does not preclude prophylaxis in general but calls for a more targeted approach to optimize antimicrobial exposure and efficacy in high-risk haematology patients. 25 Furthermore, prophylactic antimicrobial administration is critical regarding the risk of drug-related adverse events, which is of concern, especially for fluoroquinolones, as well as from a general antimicrobial stewardship viewpoint. 26 Those concerns are part of an ongoing debate around antimicrobial prophylaxis in high-risk patients. They are critically appraised by scientific societies and have been associated with abandoning of routine antimicrobial prophylaxis in many haematology centres. 27 Interestingly, our survey showed that in both patients with and without MDR-GNB colonization, AMP is administered in 32.5% while reasons and rationales for or against AMP in a specific subset of patients (e.g., those with MDR-GNB colonization) were not assessed, and should be approached in future studies.
Screening for MDR-GNB colonization appears frequent practice, as 87% of the participating centres in this survey do so prior to initiation of chemotherapy. The risk of subsequent infection in patients colonized with MDR bacteria is substantial in carriers of carbapenem-resistant GNB, whereas it is lower for patients with vancomycin-resistant enterococci colonization. 28 However, the downstream management impact of the screening results in case of MDR-GNB detection is heterogeneous and guideline recommendations are incoherent.29,30 These include, among others, contact precautions (CP), less contact with health-care providers, delay in diagnostic and therapeutic procedures (e.g., radiological imaging and surgery), and potential psychiatric sequelae of isolation.31,32 The transmission of MDR bacteria needs to be prevented under most circumstances, however, it is still debated if CP is effective to achieve this purpose while other measures, such as conventional but strictly performed hand hygiene seem to be more efficacious.33–35
This study has several limitations. The nature of survey studies does not allow for a balanced selection of participants according to their geographic distribution, kind of hospitals they work at etc., and is dependent on their responses only allowing estimates limits the accuracy of the results. Thus, real rates of MDR-GNB colonization and bacteraemia development may differ. It is also important to note, that anchoring bias may be a limitation on the estimation of bacteraemia development, ICU admission and mortality by the participants. Furthermore, detailed aspects of MDR (e.g., local pathogen distribution and resistance mechanisms as well as the used microbiologic methodology) were not captured to discourage participation due to time constraints. Lastly, due to the exploratory scope of the survey, no sample size was calculated.
Conclusion
In summary, screening for MDR-GNB in high-risk haematology patients is a common practice, and a subsequent infection prevention strategy for high-risk patients – including antimicrobial prophylaxis – is implemented at most participating centres. The heterogenous results of this survey highlight a need for tailored infection prevention measures as well as further research to optimize infection management.
Collaborators
Lukas van den Ven, Thomas Louie, Khrystyna Zapotochna, Mar Laya, Divya Vijayakumar, Xiaomeng Feng, Fernando Riera, Juan Carlos Ramos-Ramos, Raghunadharao Digumarti, Tafese Beyene Tufa, Christopher Inchley, André Pinto, Nathalie Layios, Francesca Farina, Silvia Corcione, Qing Cao, Gianpaolo Nadali, Marina Popova, Methee Chayakulkeeree, Galina Solopova, Corrado Zuanelli-Brambilla, Marie-Pierre Ledoux, Natalia Ana Mendoza-Palomar, Nicola Giesen, Alfredo Jover-Sáenz, Martin Čerňan, José María Valle-T-Figueras, Lubos Drgona, Sulieman Abu Jarir, Zekaver Odabasi, Andreas Groll, Paul Georghiou, Milan Navratil, Farhan Rasheed, Gustavo Adolfo Méndez, Elio Castagnola, Zaira Palacios, Stefan Zimmerli, Sanjay Bhattacharya, Olha Dashchakovska, Ilhomidin Niyazov, Zsófia Müller, Marija Elez, Miki Nagao, Inaam Bizid, Thomas Lehrnbecher, Hussain Alizadeh, Martha Aviles, Mariana Guaraná, Paloma Merino-Amador, Sabina Cviljevic, Enrico Schalk, Christopher H. Heath, Sofya Khostelidi, Ilana Reinhold, Andreas Glenthøj, Stef Meers, Lior Nesher, Olga Kozlova, Holger Rohde, Angelo Pan, Alaa Elhaddad, Danguole Vaznaisiene, Edmond Puca, Roman Fishchuk, Philipp Kohler, Peter Hermans, Bastian Dirk, Jan Jubřica, Diana Dimova-Vasileva, Mehmet Sait Yıldırım, Mehmet Doymaz, Secil Deniz, Seda Yılmaz Semerci, Rodrigo Alonso-Navarro, Jorge Labrador, Emanuele Pontali, Sutthichai Sae-Tia, Chia-ying Liu, Jeongsoo Park, Hee Jae Huh, Jaehyeon Lee, Nousheen Iqbal, Musa Garbati, Juan Pablo Ramirez-Hinojosa, Tebuka Toatu, Baiduissenova Aliya Uteshovna, Rekha Sharma, Ritin Mohindra, Jorge Reyes, Mohammad Naeem, Przemyslaw Zdziarski, Lenka Součková, Vladimir Ivanov, Athanasios Tragiannidis, Khalid Shoumariyeh, Anahit Zakharyan, Óscar Pérez-Olaso, Stefan Borgmann, Tinsae Alemayehu, Josip Batinić, Giovanni Cenderello, Shahin Gaini, Luis Ostrosky-Zeichner, Liping Zhu, Oxana Munteanu, Samuel McConkey, Alexander Schauwvlieghe, Rafat Alkadi, Sherif Mossad, Igor Stoma, Basil Xavier, Carolina Garcia-Vidal, Miguel Salavert-Lleti, Chelsea Gorsline, Marijana Virijevic, Jorge Alberto Cortés, Daniele Avenoso, Tomas Garcia-Lozano, Jan Novák, Olga Vladimirovna Gerasimovich, Pratibha Kale, Branislava Milenkovic, Raquel Rodrigues, Ángela Cano-Yuste, David Allsup, Daniel Aguilar-Zapata, Sergei Liapustin, Packia Nancy Romald, Marcio Nucci, Yasemin Cag, Aleksandar Savic, Gloria Pérez-Caballero, David Aguilera-Alonso, Caterina Buquicchio, Wing-yan Au, Balint Gergely Szabo, Marisa Miceli, Salvador López-Cárdenas, Esma Eryilmaz-Eren, Raeseok Lee, Jose A Vazquez, Emmanuel Roilides, Natalia Minaeva, Abraham Samuel, Fabio Forghieri, Maja Subelj, Giovanni Breda, Agnieszka Wierzbowska, Andrea Visentin, Fatma Ebeid, Lorenzo Brunetti, Natasha Ali, Javier Cornago-Navascués, Paloma Geijo-Martinez, Anjum Khan, Jiri Sramek, Krzysztof Mądry, Gökhan Metan, Gin Gin Gan, Moshe Mittelman, Malgorzata Mikulska, Yavuz M. Bilgin, Francesco Marchesi, Chiara Cattaneo, Billie Caceda, Monika Biernat, Andreas Voß, Indira Erimbetova, Ata Nevzat Yalcin, Ariful Basher.
Supplemental Material
sj-docx-1-tai-10.1177_20499361241271863 – Supplemental material for Current practice of screening and antimicrobial prophylaxis to prevent Gram-negative bacterial infection in high-risk haematology patients: results from a pan-European survey
Supplemental material, sj-docx-1-tai-10.1177_20499361241271863 for Current practice of screening and antimicrobial prophylaxis to prevent Gram-negative bacterial infection in high-risk haematology patients: results from a pan-European survey by Jannik Stemler, Eleni Gavriilaki, Oksana Hlukhareva, Nina Khanna, Dionysios Neofytos, Murat Akova, Livio Pagano, José-Miguel Cisneros, Oliver A. Cornely and Jon Salmanton-García in Therapeutic Advances in Infectious Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
