Abstract
Cat scratch disease (CSD) is a zoonotic disease transmitted to humans, usually via scratches or bites. Bartonella henselae is the primary causative agent. It causes a mild, self-limiting disease. In immunocompromised patients, the course of the infection can be more serious because of the suppressed antibacterial response, causing a life-threatening disease. A 54-year-old male patient presented with ulcerative colitis. Five days after receiving the first dose of infliximab 400 mg intravenously and 0.5 mg/kg methylprednisolone, he presented with enlarged axillary lymph nodes and colliquation of the intraabdominal lymph node with intrahepatic colliquating areas caused by B. henselae after cat bites. Long-term treatment with multiple antibiotics and prednisolone resulted in clinical improvement and regression of the liver and intra-abdominal lymph nodes. After further treatment for ulcerative colitis, we assessed the possibility of reintroducing immunosuppressive therapy. Adalimumab was introduced after consulting an infectious disease specialist. At the follow-up visit, the patient was in remission of ulcerative colitis and without signs of reactivation of bartonellosis. Diseases such as CSD with a benign clinical appearance and prognosis can develop a severe and life-threatening course in immunocompromised patients. This requires a complex understanding of the immune processes in such patients, and the reintroduction of immunosuppressive therapy after successful treatment of CSD probably does not increase the risk of reactivation.
Introduction
Bartonella is a homotropic, facultative, intracellular, Gram-negative bacterium of the alpha-2 subgroup of Proteobacteria.1,2 One of its species, Bartonella henselae, is a causative agent of cat scratch disease (CSD). It is a zoonosis, with cats as its natural reservoir. CSD is usually transmitted to humans via cat scratch or bite. 3 B. henselae invades the phagocytes and macrophages and usually causes a mild, self-limiting disease. The typical clinical course includes a papule or pustule appearance at the site of injury, usually 10 days (3–50 days) after a cat scratch or bite. Regional lymphadenopathy, most commonly in the neck, axilla, or groin, develops within 6 or 7 weeks. These enlarged lymph nodes are mostly tender. They may suppurate but generally regress within 2–4 months without therapy. 4 If antibiotic treatment is required, most current antibiotics are clinically effective, with macrolide antibiotics as the recommended first-line agents.
However, in immunocompromised individuals, the course of the infection could be much more serious, owing to the suppressed antibacterial response, and even life-threatening disease. In addition to general symptoms such as fever and malaise, which are found in approximately one-third of patients, multiple organs can be affected, with development of hepatic peliosis, splenomegaly, pneumonia, pneumonitis, meningitis, encephalitis, osteomyelitis, mesenteric adenitis, and a variety of skin efflorescences (maculopapulous, scarlatiniform, morbilliform).5–8 Secondary diseases such as erythema nodosum, erythema multiforme, herpes zoster, and conjunctivitis (Parinaud oculoglandular syndrome) can also develop.6,7
The diagnosis of CSD is usually based on clinical findings. It requires the fulfillment of three of the following four criteria: (1) a history of animal contact (usually a cat) coupled with the presence of a primary skin or eye lesion, (2) positive results of a CSD skin test, (3) negative results of laboratory studies regarding other causes of lymphadenopathy, and (4) characteristic histopathology of a lymph node biopsy specimen. 4
We present a case of severe CSD in a 54-year-old patient with active ulcerative colitis. The disease has a severe course because of drug-induced immunosuppression. Moreover, specific treatment for CSD has been delayed because of difficulties in establishing a diagnosis.
Case report
A 54-year-old patient with a 14-year history of ulcerative pancolitis was treated with intravenous vedolizumab 300 mg every 2 months during the previous year. Three months prior to this presentation, disease exacerbation occurred, and 0.5 mg/kg methylprednisolone was orally administered. It was decided to stop vedolizumab therapy (due to loss of efficacy) and introduce infliximab 400 mg intravenously instead. Five days after receiving the first dose of infliximab, the patient presented with malaise, chills, and fever (up to 39°C). Physical examination revealed a tender, enlarged lymph node in the right axilla (Figure 1). No other physical abnormalities were observed.
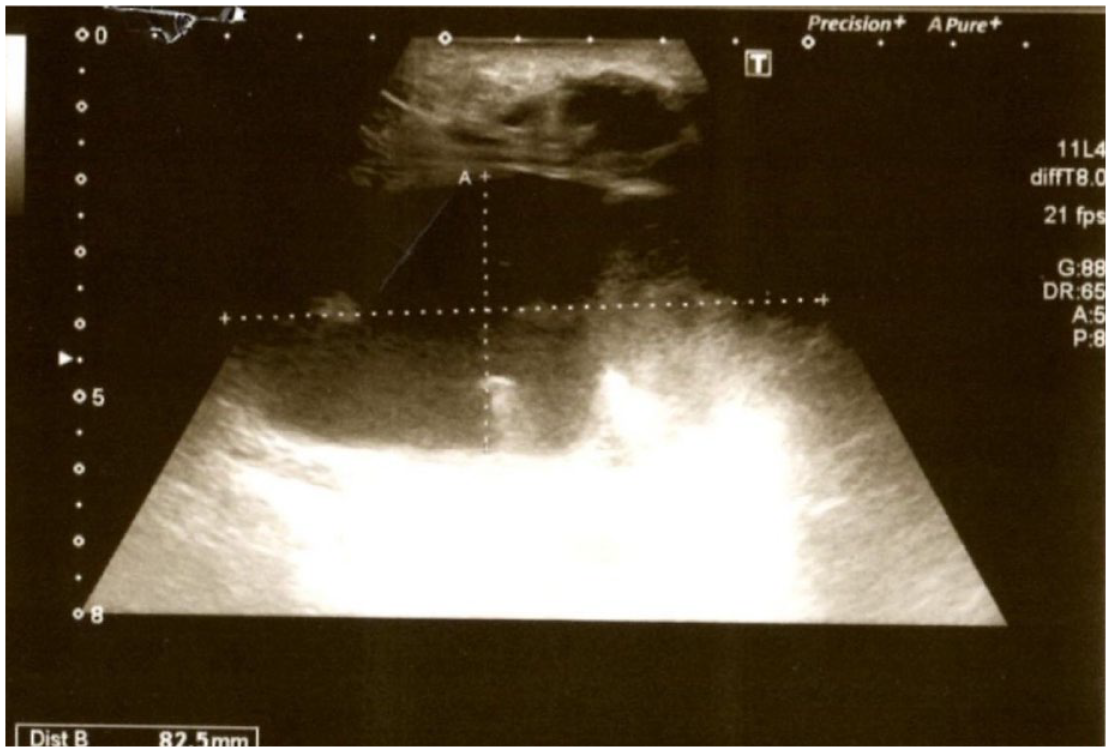
Ultrasound of right axilla: abscessed lymph node 3.5 × 8.2 cm.
History taking revealed that the patient had been bitten by a cat 4 weeks prior; therefore, CSD was suspected. The infliximab therapy had been stopped, and an incision of the axillary lymph node was made. The B. henselae serological IFA IgM test was positive (>1:40). During the next week, despite treatment with azithromycin 500 mg orally, the patient still had episodes of chills and temperature up to 39°C. Therefore, he was hospitalized. Surgical extirpation of the entire axillary lymph node was performed, and histopathological analysis confirmed CSD (Figure 2).

Light microscopy of lymph node (Olympus BX41; (a): magnification ×100 and (b): magnification ×400). The histologic features of cat scratches evolve over the course of the infection. Initially, small cortical abscesses appear beneath the subcapsular sinus, followed by mixtures of neutrophilic and granulomatous inflammation and non-necrotizing granulomas with multinucleated giant cells in the cortex and medulla. Eventually, the centers of some granulomas become irregularly contoured collections of neutrophils and necrotic debris surrounded by a rim of palisading macrophages. These so-called stellate microabscesses are considered the pathologic hallmark of CSD in the lymph nodes.
As the fever persisted, thoracic and abdominal multislice computed tomography (MSCT) scans were performed, and large colliquating intra-abdominal lymph node conglomerates as well as smaller intrahepatic colliquating areas were found (Figure 3).

Abdominal MSCT: (a) intraabdominal lymph node conglomerates and (b) intrahepatic colliquating areas.
Continuous therapy with 0.25 mg/kg methylprednisolone, rifampicin (2 × 300 mg), and sulfamethoxazole–trimethoprim (2 × 960 mg) orally was commenced, along with azithromycin (1 × 500 mg) 3 days weekly. Despite this therapy, the patient became subfebrile (<38°C), only after 3 weeks, and afebrile after 6 weeks. A control CT scan showed a regression of intraabdominal lesions. The patient was discharged with continuous therapy of prednisolone, azithromycin, rifampicin, and sulfamethoxazole–trimethoprim at the same dose as during regular checkups. He was on therapy for the next 3 months until the full regression of symptoms and normalization of laboratory findings. Due to the need for further treatment of ulcerative colitis, a multidisciplinary team (gastroenterologist and infectious disease specialist) assessed the possibility of reintroducing immunosuppressive therapy. Previously, the patient had received vedolizumab as the first line of therapy and had received no anti-TNF therapy except for one dose of infliximab. Ustekinumab was not an option because it was approved for patients who have failed other biological therapies. Antibodies against infliximab were found to be positive, and it was decided to introduce adalimumab 80 mg subcutaneously in combination with the continuing dose of 0.25 mg/kg methylprednisolone. Two months after induction, adalimumab and methylprednisolone were tapered and finally stopped. Subsequently, the patient was regularly controlled; he was in clinical and endoscopic remission of ulcerative colitis and without signs of bartonellosis reactivation.
Discussion
A standard diagnosis of CSD is usually established based on a typical history, exclusion of other causes, and typical histology and serology. However, the disease course is complex, particularly in immunocompromised patients. In our case, the patient was immunocompromised because of the biological therapy for ulcerative colitis. We observed a typical history of a right-sided skin lesion caused by a cat bite and right axillary lymph node enlargement. Positive Bartonella serology and lymph node histopathology are typical features of CSD. The lack of response to the initial antibiotic therapy was related to immunosuppression due to long-lasting corticosteroids and biological therapy for ulcerative colitis. The immunosuppression allowed for the development of a systemic form of the disease, which included colliquating masses in the intra-abdominal lymph nodes and liver parenchyma.
Most atypical cases of CSD in the literature regarding persistent fever following close contact with cats have been described in HIV and post-transplant patients, as well as in patients with chronic alcoholism. Immunosuppression leads to a weak, usually expected, regional response of the local lymphatic system and probably allows the development of a systemic form of disease.9–13 Santos et al. 11 report cases of fever and bacillary angiomatosis lesions, a vascular proliferation in the skin that presents as a purple nodule with central ulceration, as the most common manifestation of B. henselae infection in HIV/AIDS patients. The patient had complete regression of all lesions after a 6-month regimen of oral erythromycin. Persistent fever following close contact with cats is the most common presenting symptom among post-transplant patients infected with B. henselae. 14 Lienhardt et al. 14 report the case of a lung transplant recipient with skin furunculous hyperkeratotic nodule and multiple lesions in the bone marrow. Combination therapy with clarithromycin and ciprofloxacin failed so they added doxycycline and at the end rifampicin which is what finally led to control of the B. henselae infection. There were few cases of CSD reported in liver transplant patients. In that case, B. henselae presents as hepatic masses, hepatic granulomas, or disseminated disease.15–18 Thudi et al. 15 and Humarand et al. 16 report CSD as hepatic masses and hepatic granulomas. Response to therapy with doxycycline in one and azithromycin in the other was rapid within 7 days. On the other side, Bonatti et al. 17 report two liver transplant patients’ cases of disseminated CSD disease after undergoing therapy with azithromycin for 6 months.
Regarding CSD treatment, especially in patients with a generalized course, we must emphasize that there are no controlled trials or treatment guidelines on antibiotic therapy for CSD. Most authors maintain that antibiotic treatment does not affect the typical disease course. 4 By contrast, the removal of enlarged lymph nodes may rapidly abort the clinical symptoms of typical CSD, which could be explained by a sudden decrease in the number of pathogenic organisms contained in the removed lymph nodes. 4 Azithromycin and/or doxycycline are among the most commonly used antibiotic drugs for successful treatment of CSD in solid-organ recipients, but the duration of therapy is much less clear, varying greatly from 4 days to 6 months.17,19 In the case of our patient, who had no response to azithromycin monotherapy, long-term treatment with a combination of antibiotics caused gradual clinical improvement, and the patient became afebrile after 3 months, along with regression of the liver and intra-abdominal lymph node masses followed by MSCT.
This case raises an additional question regarding the causative agent of CSD. Because it is an intracellular bacterium, there is a possibility of disease reactivation after immunosuppressive therapy is reintroduced. After we reintroduced biological therapy, for example, a monoclonal antibody, in our patient, we observed no reactivation of the disease. Reviewing the literature, we found only two cases of CSD in rheumatoid arthritis patients treated with monoclonal antibodies tocilizumab. Two months after CSD treatment, tocilizumab was reintroduced, in both cases, no reactivation of the disease was observed.20,21
Conclusion
Diseases with a relatively benign clinical appearance and prognosis, such as CSD, can have a severe and life-threatening course in immunocompromised hosts. In such cases, virtually any organ system may be affected by the disease with unpredictable outcomes. It requires a complex understanding of the immune processes in such patients, as well as early diagnosis of the disease, to carry out an appropriate and efficient treatment. According to the present study, although scant data are available, the reintroduction of immunosuppressive therapy after successfully treating CSD does not increase the risk of reactivation.
