Abstract
Background:
Antifungal diagnostic capacity has been documented in various countries, there is a lack of comprehensive research on clinical mycology diagnostics and treatment in Hungary.
Methods:
We conducted an online survey encompassing questions that explored various aspects of the mycology diagnostic and antifungal therapy-related information. The survey aimed to gather details about institutional profiles, perceptions of invasive fungal infections (IFIs), and access to microscopy, culture, serology, antigen detection, molecular testing, and therapeutic drug monitoring.
Results:
As of May 2023, a total of 17 institutions responded to the questionnaire. Seven participants categorized the institutional incidence of IFI as ‘very low’, four as ‘low’, and six as ‘mild’. The majority of centers identified Candida spp. (94%) and Aspergillus spp. (82%) as the most prevalent fungal pathogens. Nearly half of the laboratories (47%) reported using matrix-assisted laser desorption/ionization-time of flight mass spectrometry for identification. All institutions had access to microscopy and culture-based diagnostic approaches. A significant number of centers had access to antigen detection (71%) and various molecular assays (59%). Regarding antifungal agents, all reporting sites used at least one triazole, with voriconazole (77%) being the most common mold-active azole. Furthermore, 71% of the centers applied at least one formulation of amphotericin B, and 65% to one echinocandin. However, only 18% of the centers used 5-flucytosine.
Conclusion:
Resource availability for diagnosing and treating IFI in Hungary varies across hospitals based on location. Surveys help identify gaps and limitations in this area. To address these challenges, interregional cooperation within Hungary could be a facilitating strategy.
Plain language summary
Background: While various countries have looked into their ability to diagnose fungal infections, there hasn’t been a comprehensive study on how Hungary deals with diagnosing and treating these infections.
Methods: We did an online survey with questions about how hospitals in Hungary handle fungal infections. We wanted to know about the hospitals’ characteristics, how they see these infections, and what tools they use for diagnosis and treatment.
Results: As of May 2023, we got responses from 17 hospitals. Some said they hardly ever see these infections, while others said they see them a bit more. Most hospitals found Candida and Aspergillus as the most common fungal culprits. Many used a tool called MALDI-TOF MS for identification. All of them had ways to look at samples under a microscope and grow them in a dish. Many hospitals had tests to look for certain things in the blood (71%), and they also used different genetic tests (59%). When it came to medicines, they all had at least one kind of medicine called a triazole, with voriconazole being the most common one. They also had amphotericin B and echinocandins. But only a few had a medicine called 5-flucytosine.
Conclusion: Hospitals in Hungary differ in how they handle fungal infections. Doing surveys like this can help find problems and limits. To fix these issues, hospitals in different parts of Hungary can work together.
Keywords
Introduction
Invasive fungal infections (IFIs) lead to death in up to 1.5 million people worldwide annually, displaying a remarkable public health problem along with lower respiratory tract infections, gastrointestinal tract-related diseases, and tuberculosis.1,2 The unacceptably high mortality rates (~30–50%) highlight the importance of prompt and accurate diagnosis of these infections.1,3 Despite the high mortality, about 80% of patients might be saved with appropriate diagnostic approaches and therapies. 1 Although, the number of Western European epidemiological studies and mycology specific laboratory diagnostic accessibility are steadily increasing, data derived from Central-Eastern European countries remain limited.4–7 Hungary is a Central European country with a population of 9.6 million inhabitants, and the gross domestic product is 24,380 EUR per person. 8 Hungary – similar to other Western European countries – has an aging society, which predisposes to an increase of the emergence of various chronic diseases associated with risk for IFI. 8 Hungary has a nationwide, universally accessible public healthcare system with a bed number of 6.8 per 1000 patients; however, there are several limitations, including underfunding and lack of human resources.
Approximately one-third of the Hungarian population has at least one chronic disease that might predispose to IFI, for example, diabetes (12%), coronary heart disease (12%), hematological disorders (6%), and asthma (5%), whereas the incidences are 2.8/100,000 and 683/100,000 for Mycobacterium tuberculosis infection and different cancers, respectively. 8 Chemotherapy-induced neutropenia in cancer patients may result in the emergence of IFI.9,10 Moreover, M. tuberculosis could be a major predisposing factor for IFI particularly in case of invasive aspergillosis.11,12 Based on Hungarian data reported by the European Center for Disease Prevention and Control (ECDC), Candida spp. are among the most frequently identified pathogens in intensive care unit-(ICU) acquired pneumonia (5.9%) and in ICU-acquired bloodstream infection (3.4%), whereas the urine Candida positivity rate in ICU is 13.3%. 13
In the past three decades, the number of fungal-based epidemiological studies derived from Hungary is very limited and they are primarily single center and/or population specific. In 2015, it was reported that more than 1.6 million (16.5%) Hungarian inhabitants suffer from fungal infection in each year, from which 33,000 (2.1%) cases are potentially life-threatening. 4 Based on crude estimation, approximately 370 cases of yeast and 52 of mold infections can be expected annually in the ICU population in Hungary. 4 In patients with acute leukemia and recipients of hematopoietic stem cell and solid organ transplants, the estimated incidence is more than 55 infections per year. 4
The study aims to thoroughly evaluate the current diagnostic and therapeutic approaches for managing IFI in Hungary, where data are currently lacking. This evaluation seeks to optimize IFI management and identify areas for improvement, focusing on enhancing diagnostic and therapeutic options within the Hungarian healthcare system to provide clearer guidance for healthcare professionals and ultimately improve patient outcomes.
Methods
In this study, conducted between March 2022 and May 2023, a multicentre questionnaire-based approach was employed. Data were collected through an online electronic case report form hosted at www.clinicalsurveys.net/uc/IFImanagementcapacity/, facilitated by TIVIAN GmbH in Cologne, Germany (Experience Management Software (EFS) Fall 2022). To ensure a wide pool of participants, 25 researchers from each of the 19 counties in Hungary were contacted, at least one per county. In counties with multiple tertiary care hospitals, more than one institution was reached. Mass e-mails were used as the main contact system, targeting both collaborators of the authors and members of key scientific organizations such as the International Society of Human and Animal Mycology (www.isham.org) and the European Confederation for Medical Mycology (www.ecmm.info). Validation of participants’ answers ensured the accuracy, coherence, and completeness of the data. Redundancy was also minimized by avoiding double data entry from the same institutions. In cases where provided data were unclear, participating institutions were contacted to ensure clarity. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology statement (Supplemental Appendix).
The survey included evaluating institution profiles, gauging perceptions regarding estimated IFI incidence and pathogenic fungi relevance at each institution, exploring microscopy techniques, culture, and fungal identification methods, analyzing serology approaches, antigen detection capabilities, molecular assays, and the availability of therapeutic drug monitoring (TDM). Participants were required to provide dichotomous responses indicating whether or not specific methods were accessible at their respective locations. For serology, antigen detection, molecular testing, and TDM, laboratories were additionally asked to specify if the respective services were available onsite or outsourced to external institutions. The survey, which is accessible in detail in Supplemental Table 1, was constructed by experienced international mycologists and used in previous experiences.5–7,14 Additionally, it underwent scrutiny by local mycologists to ensure that the data collection process was suitable for accurately depicting the unique characteristics of the Hungarian context, reducing any potential participation bias. To determine the estimated prevalence of IFI by the participants, a Likert scale, a five-point ordinal scale used to assess the extent to which survey participants concurred or disagreed with a statement, was employed. In this research, specifically regarding the significance of IFI incidence within their institution, participants are allowed to rate the incidence on a scale from 1 (extremely low incidence) to 5 (very high incidence) (Supplemental Table 1).
The collected data were summarized using frequencies and percentages. SPSS v27.0 (SPSS, IBM Corp., Chicago, IL, USA) was utilized for all statistical analyses. To offer a clear, meaningful, and uncomplicated data overview, we refrained from conducting any additional analyses.
Results
General overview
Responses from 17 of the 25 contacted (68.0% response rate) Hungarian hospitals were collected (Figure 1). The self-perceived incidence of IFI was reported as follows: seven (41.2%) of the participants indicated a very low incidence, four (23.5%) reported a low incidence, and six (35.3%) reported a moderate incidence. None of the participants reported a high or very high incidence of IFI. Regarding the most important pathogens reported to be associated with IFI, the respondents identified Candida spp. as the most prevalent pathogen (n = 16, 94.1%), followed by Aspergillus spp. (n = 14, 82.4%) (Table 1).

Map of participating institutions.
Characteristics and diagnostic and therapeutic capacities for fungal infections of participating institutions in Hungary.
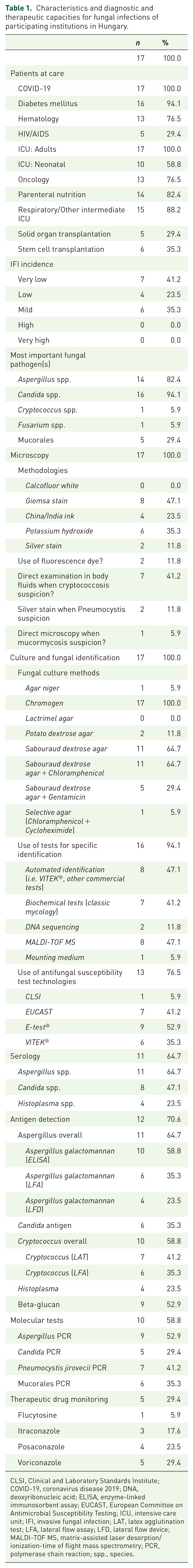
CLSI, Clinical and Laboratory Standards Institute; COVID-19, coronavirus disease 2019; DNA, deoxyribonucleic acid; ELISA, enzyme-linked immunosorbent assay; EUCAST, European Committee on Antimicrobial Susceptibility Testing; ICU, intensive care unit; IFI, invasive fungal infection; LAT, latex agglutination test; LFA, lateral flow assay; LFD, lateral flow device; MALDI-TOF MS, matrix-assisted laser desorption/ionization-time of flight mass spectrometry; PCR, polymerase chain reaction; spp., species.
Diagnostics tools
Microscopy was the most commonly employed technique and was reported by all respondents (n = 17, 100.0%). Among the microscopy methodologies, Giemsa stain was used by eight (47.1%) of the participants, followed by potassium hydroxide (n = 6, 35.3%), and China/India ink (n = 4, 23.5%). Only 11.8% of the respondents reported application of silver stain or fluorescent dye in microscopy. Fungal culture and identification could be performed in all laboratories (n = 17, 100%). The most commonly used culture method was chromogenic agar, utilized by all respondents (n = 17, 100%). Sabouraud dextrose agar, Sabouraud dextrose agar + Chloramphenicol (n = 11, 64.7% each), and Sabouraud dextrose agar + Gentamicin (n = 5, 29.4%) were also commonly employed. Regarding fungal identification, 16 (94.1%) of the respondents reported the execution of specific identification tests, with automated identification systems such as VITEK®(bioMérieux, Marcy-l’Étoile, France) or other commercial tests being the most commonly conducted (n = 8, 47.1%). Nearly half of the laboratories (n = 8, 47.1%) reported using matrix-assisted laser desorption ionization-time of flight mass spectrometry (MALDI-TOF MS) for identification purposes. Antifungal susceptibility testing technologies were implemented in 13 (76.5%) of the respondents. Among the available technologies, the most commonly undertaken methods included E-test® (bioMérieux, Marcy-l’Étoile, France) (n = 9, 52.9%), broth microdilution according to European Committee on Antimicrobial Susceptibility Testing (EUCAST) (n = 7, 41.2%), and VITEK® (n = 6, 35.3%) (Table 1).
Regarding newer diagnostic technologies, a total of 12 (70.6%) institutions were performing antibody detection, primarily focusing on Aspergillus spp. (n = 11, 64.7%), Cryptococcus spp. (n = 10, 58.8%), and beta-
Antifungal treatment and TDM
The routine administration of antifungal agents varied among the respondents as presented in Figure 2. Out of the participants, 12 (70.6%) reported administration of amphotericin B in its different formulations, with the liposomal formulation being the most common one (n = 9, 52.9%). Echinocandins were utilized by 11 (64.7%) of the respondents, with caspofungin consistently by all of them. Triazoles, on the other hand, were universally prescribed in all 17 participating institutions (100%), with voriconazole being the most prevalent among the mold-active agents (n = 13, 76.5%). However, the application of flucytosine (n = 3, 17.6%) and terbinafine (n = 9, 52.9%) was less common. In terms of TDM, five (29.4%) sites reported conducting such monitoring with voriconazole being the most frequent (n = 5, 29.4%) (Table 1 and Figure 2).

Applied antifungals by survey institutions in Hungary.
Discussion
Our survey provides data concerning the most important elements for mycological diagnostic capacity and treatment application in Hungary. Similar, regular, comprehensive surveys of the local and regional diagnostic and treatment opportunities have a pivotal role in revealing improved parts of mycological diagnostic services.4,6
Regarding epidemiological data, one of the most frequently observed IFI is candidemia.15–17 Approximately 50% of Candida bloodstream infection cases result in deep-seated infection due to hematogenous spread. 18 In 2015, it was reported that approximately 33% of candidemia cases occur in ICUs, which corresponds to a relatively high estimated incidence rate (11/100,000) followed by Thailand (13.3/100,000), Brazil (14.9/100,000), Qatar (15.4/100,000), or Pakistan (21/100,000).1,4 Concerning the available data of neighboring countries, the incidence rates were (5/100,000), (2.6/100,000), (7.3/100,000), (5/100,000), and (5.2/100,000) for Czech Republik, Austria, Serbia, Romania, and Slovenia, respectively.19–23 The last epidemiological data from a Hungarian university hospital reported that the incidence of Candida bloodstream infection increased between 2010 and 2013 from 1.7 to 3.5 episodes per 10,000 admissions then decreased to 2.66 per 10,000 admissions in 2014. 24 These incidence rate is similar to previous Hungarian data and approximately correspond to the Eastern European data.5,25,26 At the same time, a recently published comprehensive population-based meta-analysis revealed that the overall pooled incidence rate of candidemia is 3.88/100,000 in Europe. 27 According to Hungarian data, the estimated prevalence of IFI for patients with acute myeloid leukemia, recipients of hematopoietic stem cell and solid organ transplants are 12%, 6–8%, and 1–9%, respectively. 4 It is noteworthy that 29.4% of surveyed centers reported solid organ transplantation, whereas stem cell transplantation is performed in 35.3% of asked hospitals. Based on epidemiological data, the most common fungal pathogens from IFI are Candida spp. and Aspergillus spp., which coincides with our current survey. 17 Besides the above-described incidence of invasive Candida infection, the incidence of invasive aspergillosis and chronic pulmonary aspergillosis in Hungary was 3.2/100,000 and 6/100,000, respectively. 4
Regarding diagnostic capacity, microscopy and culture were the most commonly applied technique and were reported by all institutions. Chromogenic medium was available at all responded centers, which is a valuable diagnostic approach in resource-limited healthcare settings, supporting a prompt presumptive identification primarily in case of clinically relevant Candida species. 28 Different chromogenic solid media can detect mixed cultures of yeasts because various species frequently form colonies with different colors. These mixed populations may be indistinguishable on conventional medium such as Sabouraud dextrose agar with chloramphenicol. 29
Regarding fungal identification, the most reliable method in routine clinical laboratories is the Sanger sequencing of the internal transcribed spacer region; however, it is a time-consuming technique and requires qualified staff. 30 It is noteworthy, that 11.8% of the responder laboratories have direct access to DNA sequencing platforms. Nevertheless, the most widely available identification platform was MALDI-TOF MS. Out of 17 centers, 8 run mass spectrometry-based identification platforms.
In our study, the responder institutions reported antigen detection-based identification in 70.6%. Fungal antigens such as galactomannan, β-1-3-
Antifungal susceptibility testing is available at 13 of 17 laboratories. Surprisingly, only 41.2% of responders follow the EUCAST criteria. A higher ratio of laboratories with EUCAST directives may be needed to receive more reliable data about country-based resistance conditions because, since 2020, only data generated by laboratories using EUCAST breakpoints and methods are accepted by ECDC. 33 Regarding the availability of antifungal drugs, fluconazole was universally available to all 17 participants (100%), which coincides with the international availability patterns.5–7 Echinocandins are the first-line antifungal drugs in empirical therapy of fungal bloodstream infection; therefore, at least one type of echinocandins is available at 64.7% of reporting sites. 34 It is reassuring, that mold-active antifungal drugs – either voriconazole or posaconazole – are available at 76.5% of the centers. It is noteworthy that 5-flucytosine was available at only 17.6% of participant sites, which ratio is below average compared to other regional or country-specific surveys such as those in Latin America, Africa, or South-Eastern Europe (18%, 27%, and 29%, respectively).5,7,14,35 It may be alarming considering the high mortality of HIV-associated cryptococcal meningoencephalitis. 36 At the same time, the number of HIV-infected individuals is relatively low in Hungary (2.3/100,000 in 2022). 8 Different amphotericin B formulations are found in 70.6% of participant institutions in this survey. Interestingly, this ratio was higher in Latin America (72%) and Eastern European countries (83.9%) especially in the case of amphotericin B deoxycholate.5,37 Presumably, the increased cost of antifungals may result in the selection of cheaper options, particularly from liposomal amphotericin B to cheaper but more toxic deoxycholate, which is discouraged by guidelines.38–41
This study’s limitations stem from potential subjectivity introduced by relying on self-perceived incidence rates, possibly not accurately reflecting actual incidence. Moreover, the limited data collection period and absence of longitudinal data restrict insights into trends over time. The lack of direct comparisons with other countries and the subjective nature of the Likert scale for incidence rating also pose limitations. Nonetheless, the study offers valuable insights, emphasizing the importance of ongoing monitoring and improving diagnostic practices for IFI in Hungary.
In summary, the increasing number of difficult-to-treat and potentially life-threatening IFI in Hungary requires a comprehensive continuous monitoring of local diagnostic opportunities. To the best of our knowledge, this study was the first to document the strengths and limitations of diagnostic and treatment capacity of IFI. Overall, we conclude that general status of the fungal and therapeutic diagnostic capacity of Hungary is acceptable with regard to the population size; however, we found few considerable differences between responded university hospitals and smaller local city institutions. Further similar surveys with higher institute numbers have a pivotal role to reveal the diagnostic gaps and to allow closing them by defining those multidisciplinary actions, which improve the quality of IFI management in Hungary in the future.
Supplemental Material
sj-docx-1-tai-10.1177_20499361231219315 – Supplemental material for Unveiling the Hungarian landscape of laboratory and clinical management capacities for invasive fungal infections: navigating the frontlines against fungal menaces
Supplemental material, sj-docx-1-tai-10.1177_20499361231219315 for Unveiling the Hungarian landscape of laboratory and clinical management capacities for invasive fungal infections: navigating the frontlines against fungal menaces by Renátó Kovács, László Majoros, Jannik Stemler, Oliver Andreas Cornely and Jon Salmanton-García in Therapeutic Advances in Infectious Disease
Footnotes
Acknowledgements
The authors thank all participating institutions for their utmost contributions and support to the project during a pandemic situation and to all the individuals and associations that have disseminated the link to the survey. Additionally, we would like to greatly thank Attila Bátorfy for his support with the map preparation.
Declarations
Collaborators
Edina
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
