Abstract
Sepsis is a common, complex condition that requires early recognition and aggressive management to improve outcomes. There has been significant improvement in the management of sepsis and septic shock in the last decade; however, it continues to be a leading cause of mortality, morbidity and burden on healthcare services globally. Several guidelines with evidence-based recommendations for the management of children with septic shock and associated organ dysfunction have been produced with the objective of helping clinicians in various settings to provide standardised high-quality care. This article aims to increase awareness among all clinicians, including those working in emergency departments, general paediatric wards and primary care physicians, about the management of sepsis in children.
Introduction
Sepsis is a common, yet hugely complex syndrome caused by the body’s dysregulated response to infection. 1 Sepsis continues to be a major burden on health care services 2 in both developed and developing countries, despite implementation of good hygiene practices and robust immunisation programmes. If not treated in a timely manner, sepsis can rapidly progress to septic shock associated with organ dysfunction and death. Early recognition and diagnosis of sepsis, together with appropriate management consisting of prompt use of empiric antimicrobials, aggressive fluid resuscitation and early escalation of care has been shown to reduce mortality.
Evolving definitions of sepsis
The International Pediatric Sepsis Conference in 2005 published a definition and criteria for diagnosing sepsis, severe sepsis and septic shock in children. 3 This was based on the then existing understanding and definitions for adult sepsis with modifications based on age and differences in physiology in children (see Table 1 for definitions).
Definitions of systemic inflammatory response syndrome (SIRS), infection, sepsis, severe sepsis and septic shock in children.

SD, standard deviation; SIRS, systemic inflammatory response syndrome.
Published by the International Pediatric Sepsis Conference in 2005.
In 2016, the ‘adult’ definitions of sepsis and septic shock were revised, and new criteria were published (sepsis 3). ‘Sepsis’ was defined as life-threatening organ dysfunction caused by a dysregulated host response to infection; and ‘septic shock’ was defined as the subset of sepsis with circulatory and cellular/metabolic dysfunction associated with a higher risk of mortality 4 (see Table 2 for ‘adult’ definitions of sepsis).
Definitions for sepsis and septic shock in adults.

ICU, intensive care unit; MAP, mean arterial pressure; qSOFA, quick SOFA; SOFA, Sequential (Sepsis-related) Organ Failure Assessment.
Published by the Third International Consensus Definitions for Sepsis and Septic Shock.
In 2020, the Surviving Sepsis Campaign published Guidelines for the Management of Septic Shock and Sepsis-Associated Organ Dysfunction in Children. 1 There is debate regarding whether the ‘adult’ definition of sepsis is applicable to children 5 and formal revisions to the 2005 paediatric sepsis definition are still pending. 6
For the purposes of this article, septic shock in children is defined as severe infection leading to cardiovascular dysfunction (including hypotension, need for treatment with a vasoactive medication, or impaired perfusion) and ‘sepsis-associated organ dysfunction’ in children is defined as severe infection leading to cardiovascular and/or non-cardiovascular organ dysfunction.
Because several methods to identify acute organ dysfunction in children are currently available, we preferred not to require a specific definition or scheme for this purpose.
Aetiology and causative organisms
Evolution of sepsis is not merely dependent on the causative organism but includes several factors such as age of the patient, likely source of infection, underlying host factors, or susceptibility such as immunocompromised state, in-dwelling devices or chronic co-morbidities and time to or delay in recognition.
The common causative organisms for sepsis in children vary based on age, underlying condition as well as geographical location. While the most frequently identified pathogens are bacteria and viruses; fungi and parasites can also cause sepsis. It is important to remember that less typical bacteria may not be covered by the usual broad-spectrum antibiotics and would require further thought to identify and treat.
Group B streptococci (GBS) and Escherichia coli (E. coli) and Listeria monocytogenes are commonly found to be responsible for neonatal sepsis. 7 Increased vigilance and the use of peri-partum antibiotics has reduced the incidence of GBS sepsis. Herpes simplex virus (HSV) can cause fulminant sepsis in neonates, leading to rapid deterioration and death. Enterovirus and Parecho viruses may also cause a similar sepsis syndrome in neonates and young infants. Bordatella pertussis can cause severe illness and should be suspected in infants presenting with episodes of apnoea or respiratory failure.
H. influenzae type b (Hib), Neisseria meningitidis and Streptococcus pneumoniae are common causes for bacterial illnesses in children under 5 years of age. Improved availability of vaccines has significantly reduced mortality due to sepsis caused by these organisms. 7 Toxic shock syndrome caused by toxin-producing strains of Staphylococcus aureus and Streptococcus pyogenes (Group A streptococci) continue to remain important additional causes of sepsis in children. Children with frequent hospital admissions may be prone to infections with methicillin-resistant Staphylococcus aureus (MRSA) and vancomycin-resistant enterococci. Immunocompromised children and those with chronic co-morbidities are at increased risk for sepsis caused by gram-negative organisms, particularly Pseudomonas aeruginosa, as well as alpha-haemolytic streptococci and fungi.1,7 Coagulase-negative Staphylococcus should be suspected in children with in-dwelling vascular catheters.1,7 Children with splenic dysfunction or congenital or acquired asplenia are prone to pneumococcal diseases.
Viral-induced sepsis is not uncommon and while respiratory syncytial virus (RSV) and influenza virus are well known causes for hospitalisation of infants, human metapneumovirus, rhinovirus and adenovirus are being increasingly identified.
Covid-19-related illness in children may present with a hyper-inflammatory syndrome, often affecting the myocardium and presenting with shock, mimicking septic shock and should be evaluated in children as part of the differential diagnosis of sepsis. 8
It is important to be aware that sepsis can also be caused by malaria, dengue fever, rickettsial and other endemic or epidemic infections, so inquiry for travel or other epidemiological clues should be sought on evaluation. 7
Identifying the child with sepsis
Not all children with sepsis will present with fever, and many may have non-specific symptoms. Thus, clinicians should have a high index of suspicion when reviewing a child who presents with non-specific features, as this may be the early presentation of sepsis. A large number of health care systems now use Paediatric Early Warning Scores (PEWS) in both the emergency department as well as for patients admitted to the ward, which may help to improve early identification of the deteriorating child.9,10
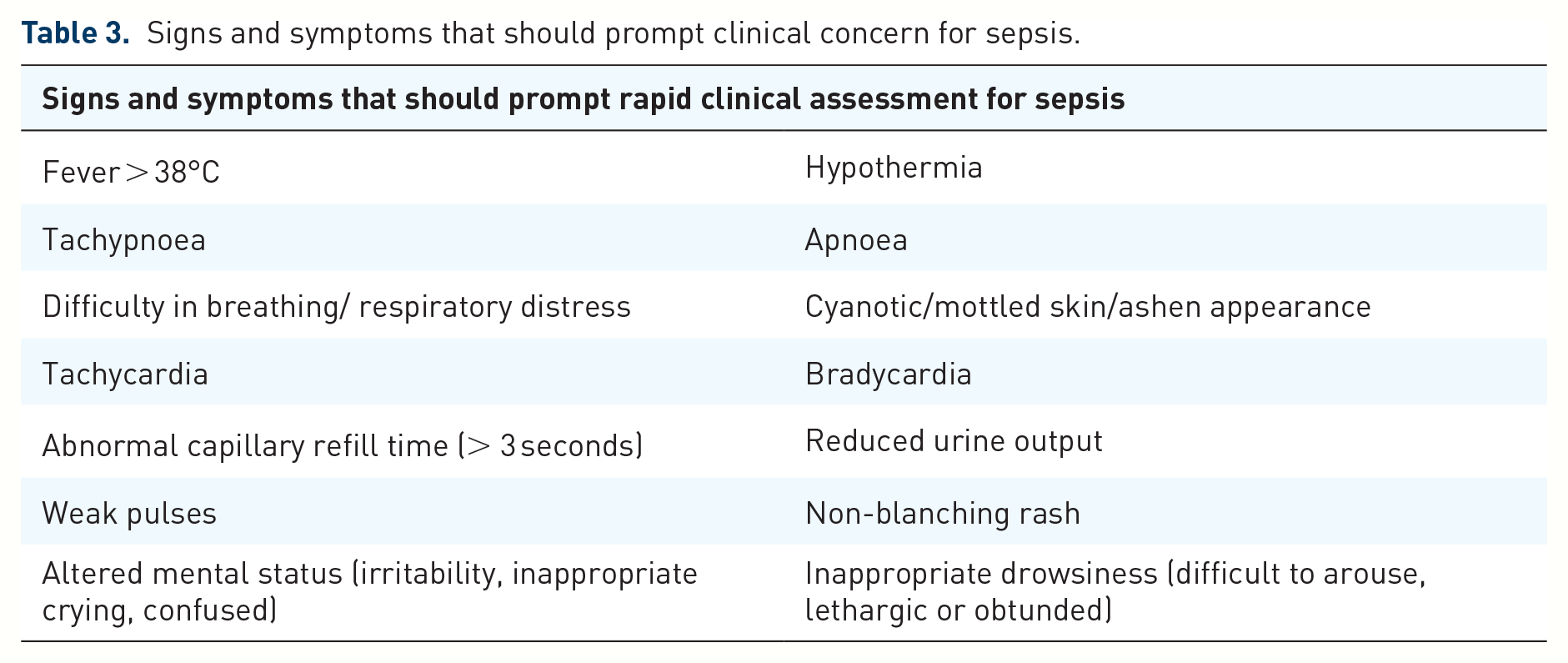
Clinicians should take a thorough history to ascertain whether the patient has increased risk factors for sepsis, followed by a detailed physical examination. Signs and symptoms that should raise suspicion of sepsis are listed below in Table 3. 11 All emergency departments should have a screening tool and sepsis bundle to help early identification of the septic child with timely management and appropriate escalation. Multiple quality improvement projects have demonstrated that the activation of a sepsis bundle is associated with improved outcome in patients with sepsis.1,12
Signs and symptoms that should prompt clinical concern for sepsis.
Diagnostic tests to identify sepsis are limited to non-specific biomarkers indicative of an ongoing inflammatory response such as White Blood Cell count and differential, C-reactive protein (CRP) and procalcitonin (PCT). However, none of these tests are sensitive or specific for diagnosing sepsis and evidence to suggest they accurately predict sepsis is lacking. 13 The cost to perform PCT test is significantly higher when compared with CRP or white cell count. In addition, there are situations in which a markedly elevated procalcitonin is not predictive, for example, in the setting of moderate to severe acute kidney injury. Based on studies performed in adults, PCT is a good prognostic marker that can be used for monitoring therapeutic response and can help guide safe antibiotic de-escalation/rationalisation. 14
Newer molecular tests (e.g. polymerase chain reaction, 16S ribosomal nucleic acid (RNA)) to identify both bacterial and viral infections can be useful if standard microbiological techniques fail to identify the infecting pathogen, but these are expensive and are not widely available.
Blood lactate measurement provides a rapid and indirect estimation of cardiac output, available as a point of care test to assist in the evaluation of the child with signs of shock. In adults and children, a blood lactate measurement of > 2 mmol/L is an indication of poor perfusion and/or cellular/metabolic dysfunction, associated with septic shock. 15 This may aid in decisions regarding adequacy of shock resuscitation. 16
While tissue hypoperfusion is probably the most common cause for elevated lactate levels, persistently elevated lactate levels or those not responding to fluid therapy in the absence of signs of hypoperfusion should prompt the clinician to consider alternate causes of lactic acidosis such as liver dysfunction, state of nutritional deficiency (thiamine deficiency), ingested drugs/toxins affecting cellular metabolism or malignancies.
Pathophysiology
The pathophysiology and mechanism of sepsis is complex. The host’s immune system is activated by bacterial cell wall components (e.g. endotoxin, peptidoglycan, lipoteichoic acid) or bacterial exotoxins (e.g. Staphylococcal enterotoxin B, toxic shock syndrome toxin-1, Pseudomonas exotoxin A, M protein of haemolytic group A streptococci) or components of fungi, viruses or parasites.
Activation of host inflammatory cells including neutrophils, macrophages, lymphocytes and endothelial cells leads to the production of proinflammatory cytokines such as tumour necrosis factor alpha (TNFa) and interleukin-1 (IL-1). 17 This amplifies host cell activation and production of downstream mediators such as prostaglandins, leukotrienes, platelet-activating factor and phospholipase A2. When these proinflammatory mediators are released in an uncontrolled and dysregulated manner, dysfunction of the endothelium, capillary leakage, prothrombotic states and myocardial dysfunction occurs. These changes ultimately resolve in organ dysfunction. 18
Hypotension in sepsis is thought to be a consequence of vasodilation due to impaired endothelial function, upregulation of nitric oxide secretion and redistribution of intravascular fluid. Myocardial dysfunction results from reduced venous return, decreased nitric oxide release, cytokine dysregulation and release of myocardial depressant factors. There is impaired secretion of antidiuretic hormone, which may also contribute to the vasodilation.
Hypoperfusion secondary to vasodilation results in acute tubular necrosis, which is one of the presumed mechanisms leading to acute kidney injury in sepsis. 19 Other factors contributing to multiple organ dysfunction and failure includes disseminated microvascular coagulation.
Antimicrobial therapy
Evidence suggests that early and prompt administration of antibiotics is crucial to optimise outcomes in patients with sepsis and septic shock. 20 We would recommend that broad spectrum antibiotics should be given within 1 hour of presentation. 1 Empiric broad-spectrum antimicrobials should be selected, depending on known epidemiology and local antimicrobial resistance patterns, factoring in the likely source of infection, presence of any indwelling devices or catheters, immunocompromised state if present, recent hospital admissions and known colonisation with specific pathogens. 11 Clinicians should be guided by their local antimicrobial policy but can also refer to the recommendations from NICE guidelines and the Surviving Sepsis Campaign Guidelines, which have been summarised in Table 4.
Antimicrobial therapy guidance based on recommendations from NICE and SSC.

IVIG, intravenous immunoglobulin; MRSA, methicillin-resistant Staphylococcus aureus; NICE, National Institute for Health and Care Excellence.
All efforts should be made to collect relevant microbiological samples of blood, urine, sputum and CSF prior to starting antibiotics. However, diagnostic tests should not delay antibiotic administration. The lumbar puncture should be deferred due to haemodynamic instability, concerns of raised intracranial pressure, abnormal coagulation, or infection at the lumbar puncture site.
As soon as clinically feasible, interventions to achieve source control should be implemented. 1 This includes removal of infected indwelling devices, abscess drainage, debridement of necrotizing soft tissue and drainage of a septic joint or empyema.
Antimicrobial stewardship should be employed, which includes de-escalation of antibiotics when appropriate to reduce toxicity as well as avoidance of prolonged use. Appropriate antimicrobial stewardship involves cessation of antimicrobials if an alternative non-infectious aetiology for the clinical presentation is determined and switching to a narrower spectrum antimicrobial as soon as possible based on microbiological data. 21 This decision should be made based on clinical improvement, site of infection and whether source control has been achieved, and identification of infecting pathogen with accompanying antimicrobial sensitivities. Close consultation with infectious disease specialists in complex cases is advised.
Vascular access and fluid therapy
Hypovolaemia in a child with sepsis is a consequence of capillary leak, vasodilation and fluid loss, which can be corrected with appropriate fluid resuscitation. Vascular access must be obtained immediately when sepsis or septic shock is suspected. Ideally, the child should have two wide bore peripheral venous cannulae inserted. If the child is in septic shock and venous access has not been established within 5 minutes, interosseous access should be obtained. A blood gas should be obtained for lactate, glucose and acid base status. Other investigations including full blood count, clotting screen, renal and liver function tests should be obtained. In children who have no evidence of cardiovascular compromise demonstrated by tachycardia, abnormal perfusion or hypotension, fluid bolus therapy should not be given; they should be commenced on maintenance fluid therapy instead. 22 Children who are in shock should receive fluid boluses of 10–20 mL/kg with strict monitoring of heart rate, capillary refill time, blood pressure, urine output and lactate levels. To assess response and need for further fluid administration after each bolus, it is important to repeat clinical examination for evidence of fluid overload by evaluating for new or worsening hepatomegaly, new or increasing oxygen requirement, basal crepitations, or radiographic evidence of pulmonary oedema. Fluid boluses up to 60 mL/kg can be given within the first hour in settings with access to advanced intensive care.22,23 In lower resource settings that do not have provision for advanced intensive care, fluid blouses in the first hour should not exceed 40 mL/kg.22,23 The child should be re-assessed regularly following each fluid bolus to evaluate response and to check for signs of fluid overload. Early consultation and consideration of escalation to the Paediatric Intensive Care Unit is recommended.
While there is limited data in the paediatric population, 24 randomised control trials (RCTs) in adults have shown that the use of crystalloid fluids containing high concentrations of chloride for resuscitation is associated with hyperchloremic acidosis, acute kidney injury, coagulopathy and mortality when compared with balanced or buffered crystalloid solutions such as Ringer’s lactate or PlasmaLyte©. 25 Routine use of colloid solutions such as Human Albumin Solution or Gelofusin for resuscitation is not recommended as they exhibit no advantage over crystalloids, are more expensive, less easily available and carry an increased risk of infection or coagulopathy.1,22
Vasoactive agents
In children who have received 40–60 mL/kg of fluid resuscitation within an hour and who remain in shock, commencement of vasoactive medications is strongly recommended. 1 These children may continue to receive fluid boluses if appropriate alongside vasoactive agents to try to reverse shock, but if shock persists, these children will require intubation and ventilation. Some children may be managed with non-invasive positive pressure ventilation, but this depends on the clinical situation, patient response and resources.
Previously dopamine was commonly used as the first line vasoactive drug in the management of children with septic shock. Based on randomised control trials which have concluded that there are increased adverse effects with the use of dopamine in comparison to epinephrine and norepinephrine, current practice has moved towards the use of epinephrine and norepinephrine as first line vasoactive agents, both of which have vasopressor as well as inotropic effects.1,26,27
If there is a delay or difficulty in obtaining central access, vasoactive agents can be administered peripherally.1,28 Epinephrine is the only vasoactive agent approved to run peripherally, though many practitioners will administer norepinephrine peripherally if necessary due to lack of central venous access and excessive tachycardia precludes the use of epinephrine. The published data on the safety of administering vasoactive medications peripherally in children is limited and clinicians should attempt to obtain central venous access as soon as feasible.
As there is no good evidence comparing the use of epinephrine and norepinephrine in children with fluid refractory shock, the choice of agent depends on the treating clinician’s preference, local policy and an assessment of physiology. Epinephrine is often used to manage shock associated with a low cardiac output state while norepinephrine is often used to manage shock where vasodilatation and decreased systemic vascular resistance is present. The previously used classification of shock into ‘warm shock’ and ‘cold shock’ is now outdated as there was poor correlation between clinical assessment, cardiac index and systemic vascular resistance when measured using advanced monitoring. 1
Vasoactive agents should be titrated to a mean arterial pressure (MAP) between the 5th and 50th percentile for the age, adequate urine output and adequate peripheral perfusion.
Vasopressin-receptor agonists (vasopressin or terlipressin) may be used in catecholamine-resistant shock, and inodilators (milrinone) can be considered if the child remains in shock with evidence of low cardiac output. These therapies are typically initiated in the intensive care unit setting where advanced haemodynamic monitoring is available.
Corticosteroids
Children who are at high risk for adrenal insufficiency, such as those with chronic or recent treatment with steroids or those with congenital or acquired adrenal insufficiency, should receive stress dose hydrocortisone early. Low dose hydrocortisone can also be used in children who remain in refractory shock after use of fluid and vasoactive agents; however, there is not good evidence of their benefit.29,30
Respiratory support
All children with sepsis should be given additional oxygen by facemask early, unless they are able to maintain their transcutaneous oxygen saturation > 95% while breathing in room air. Children who need additional respiratory support but who do not have a clear indication for tracheal intubation and mechanical ventilation can be trialled on non-invasive respiratory support. This may reduce the work of breathing and improve oxygenation, thereby avoiding the need for intubation. 31 Definitive indications for mechanical ventilation include acute or incipient respiratory failure, not responsive to non-invasive respiratory support, persistent shock despite acute fluid resuscitation of 40–60 mL/kg in 1 hour, persistent hypoxemia, or inability to safely maintain the airway.
Rapid sequence induction of anaesthesia followed by tracheal intubation by the most experienced clinician is advised. Administration of general anaesthetic drugs and muscle relaxants along with the transition to positive pressure ventilation can reduce venous return and precipitate cardiac arrest. Induction agents such as propofol or benzodiazepines that may cause cardiac depression or vasodilatation should be avoided. Using reduced dosing of induction agents in the child with shock is also recommended. 1
Mechanical ventilation should be lung protective with minimal tolerated tidal volumes and peak pressures, high positive end expiratory pressure, together with efforts to minimise the amount of inspired oxygen while ensuring adequate oxygenation.32–34 Based on the possibility of pulmonary oedema due to capillary leak, and the development of sepsis-induced paediatric acute respiratory distress syndrome (PARDS), a higher positive end expiratory pressure (PEEP) may be required for recruitment of alveoli and to prevent collapse and improve oxygenation.32–34
Nitric oxide therapy is not routinely used in the management of children with sepsis induced PARDS. However, children who have pulmonary hypertension or severe right ventricular dysfunction confirmed by echocardiography should be commenced on nitric oxide if refractory hypoxaemia remains a challenge despite optimisation of oxygenation strategies. 35
Monitoring the child with septic shock
All children with sepsis should have basic monitoring which includes continuous monitoring of peripheral oxygen saturation and electrocardiography (ECG) and non-invasive blood pressure measurement.
In children who are in shock and receiving resuscitation fluid therapy and vasoactive agents, an arterial line for continuous invasive blood pressure measurement should ideally be inserted. High blood lactate levels may be an indication of incomplete resuscitation or should prompt the clinician to think of an alternative diagnosis.
Previously central venous pressure (CVP) measurements were used to guide the on-going resuscitation of patients with septic shock. This practice is no longer recommended as CVP monitoring has poor predictive value for assessing fluid responsiveness and is affected by several physiological factors. 1 However, placement of a central venous line may aid rapid fluid administration and the use of high-dose vasoactive agents.
There are a variety of advanced tools to monitor ongoing resuscitation in children with septic shock. Several devices exist which are used for the non-invasive measurement of cardiac output and peripheral vascular resistance. These newer tools may not only give us more accurate information about fluid responsiveness and volume status but can also provide values for cardiac output and tissue perfusion. The detailed description of these methods is beyond the scope of this review.
Electrolytes and metabolites
Electrolyte disturbances are commonly found in children with sepsis and septic shock, specifically hypoglycaemia, hypocalcaemia, hypokalaemia and hypomagnesaemia. Plasma electrolytes should be monitored regularly and optimised when necessary. If present, hypoglycaemia should be corrected by rapid infusion of IV dextrose. The routine use of insulin to maintain glucose within a tight normal range is not recommended.1,36 Adjunctive insulin treatment should only be considered if hyperglycaemia is associated with clinical compromise despite control of glucose administration. 36 Calcium plays an important role in myocardial contractility; hypocalcaemia should be actively corrected to maintain ionised blood calcium levels above 1 mmol/L. Hypomagnesemia may exacerbate cardiac arrythmia but should be treated cautiously as magnesium sulphate can worsen hypotension.
Blood and blood product transfusion
Blood haemoglobin level determines the oxygen carrying capacity of blood, thus affecting tissue oxygen delivery. Children who remain in shock despite fluid resuscitation and initiation of vasoactive agents may benefit from packed red blood cell transfusion if haemoglobin is < 7 g/dL. 37 Disseminated intravascular coagulopathy is common in children with septic shock and may require transfusion with platelets, fresh frozen plasma and/or cryoprecipitate to manage significant bleeding. There is no evidence to recommend prophylactic transfusion of red blood cells, platelets or plasma in children with septic shock, even with coagulopathy. Children with very low platelet counts (<20,000/mm3) are at a significantly higher risk for bleeding and should be given platelet transfusion. 1
Refractory septic shock
The European Society of Paediatric and Neonatal Intensive Care has defined refractory septic shock in children as the association of high blood lactate with high vaso/inotrope doses associated with myocardial dysfunction. 38 Possible reversible causes should be evaluated and treated if found. The presence of refractory shock is associated with a significantly higher mortality, and these patients should be considered for adjunctive therapies or extra-corporeal support.
Renal replacement therapy
Fluid overload is associated with increased morbidity and possibly mortality in critically ill children. 39 There is no evidence that routine use of renal replacement therapy (RRT) is associated with improved outcomes. 40 Common indications for initiation of RRT in children with septic shock include fluid overload unresponsive to fluid restriction and diuretic therapy, acute kidney injury and persistent lactic acidosis. The use of RRT requires both clinician and nursing expertise, insertion of large vascular catheters for haemofiltration or dialysis in patients with coagulopathy and is not available in all centres. There is no guidance on the optimal time to initiate RRT and it should be commenced at the discretion of the treating team after the benefits have been weighed against possible risks.
Therapeutic plasma exchange/plasmapheresis
While the role of therapeutic plasma exchange in the management of children with septic shock is not clearly established, small trials have shown some benefit for patients with thrombocytopaenia-associated multi organ failure (TAMOF). 41 TAMOF is an inflammatory phenotype of sepsis-induced multiple organ dysfunction in children. It is identified by new-onset thrombocytopenia and evolving multiple organ dysfunction. If suspected, management should be discussed with a paediatric haematologist.
Immunoglobulin therpay
A meta-analysis has demonstrated the benefits of intravenous immunoglobulin (IVIG) in the management of children with Toxic Shock Syndrome and it is used as an adjunct to antibiotics and source control. 42 However, there is no evidence that supports routine use of IVIG in the management of children with septic shock apart from those with Toxic Shock Syndrome. 43
Extracorporeal membrane oxygenation (ECMO)
Extracorporeal Membrane Oxygenation (ECMO) has been suggested as a treatment for children with refractory septic shock, where anecdotal reports of its use have demonstrated up to 50% survival. 44
Conclusion
Sepsis and septic shock continue to be associated with significant morbidity and mortality in children worldwide. All children with septic shock should receive early empiric antimicrobial therapy and aggressive fluid therapy. All emergency departments should have guidelines and protocols in place for the management of sepsis. Sepsis bundles, protocols and guidelines ensure standardised care and have been shown to improve outcome.
Footnotes
Author contributions
N.P drafted the original manuscript. S.N supervised and revised the manuscript.
Conflict of interest statement
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
