Abstract
Background:
Skin prick testing (SPT) is an important investigation in the evaluation of allergy to fungal pathogens. However, the background sensitivity to fungal allergens among healthy people in Uganda is unknown. Our aim was to assess the background prevalence of Aspergillus fumigatus SPT positivity in apparently healthy adults without known atopic disease in Uganda.
Methods:
For this pilot study, we recruited 50 healthy volunteers using convenience sampling, 56% of whom were health workers. We performed the SPT for A. fumigatus according to manufacturer’s instructions. A wheal diameter of ⩾3 mm was considered positive.
Results:
The prevalence of A. fumigatus skin positivity was 60% (30/50). Participants with a positive A. fumigatus SPT were significantly younger than those with a negative result [median age (years): 28 versus 35; p = 0.005].
Conclusion:
There is a high skin positivity against A. fumigatus among non-atopic healthy Ugandan adults. There is an urgent need to establish a normal wheal cut-off value for this population. SPT alone may be an unreliable test for the diagnosis of A. fumigatus associated allergic syndromes. More studies are needed to define the prevalence of A. fumigatus skin positivity among non-atopic healthy population in Africa.
Introduction
Fungal allergy in the context of asthma, 1 non-cystic fibrosis bronchiectasis, 2 tuberculosis, 3 chronic obstructive pulmonary disease (COPD), 4 or fungal rhinosinusitis is one of the most common health problems globally. 5 However, the pathogenic significance of fungi from the genera Alternaria, Cladosporium, Penicillium and Aspergillus is poorly described in Africa. 6 The burden of serious fungal diseases in Uganda is high, with an estimated 2.5 million cases per year, 7 yet the index of clinical suspicion remains low, 8 despite recent evidence showing that Aspergillus species are significant causes of morbidity in different at-risk populations in Uganda.3,6–10
The lungs are the primary site of infection by Aspergillus species, causing disorders including allergic, chronic, sub-acute and invasive pulmonary aspergillosis, 11 with Aspergillus fumigatus the most commonly implicated species. 12 Human allergic disorders associated with A. fumigatus include allergic asthma, severe asthma with fungal sensitisation (SAFS), allergic rhinosinusitis, allergic bronchopulmonary aspergillosis (ABPA), and hypersensitivity pneumonitis. 13 In addition, A. fumigatus is isolated frequently from the respiratory tract of patients with asthma who do not meet the criteria for ABPA or SAFS, and occasionally in the respiratory tract of healthy individuals. 14 Diagnosis of Aspergillus sensitization requires demonstration of evidence of allergic sensitization to Aspergillus either by skin prick testing (SPT) or Aspergillus-specific IgE immunoassays. 1
There is a paucity of data on the frequency of SPT reactivity against A. fumigatus in healthy adults without known atopic disease. We therefore aimed to describe the distribution of A. fumigatus SPT positivity among healthy adults without known atopic disease in Uganda, which will, in turn, generate hypotheses to encourage further research in this field of fungal allergy in Uganda.
Methods
This was a cross-sectional study evaluating the frequency of A. fumigatus skin prick positivity among healthy adults without known atopic disease in Uganda. It was carried out between March and October 2019. This study was nested within the African Severe Asthma Program (ASAP) clinical study [ClinicalTrials.gov identifier: NCT03065920] at the Makerere University Lung Institute. 15 ASAP is a clinical study with the primary objective being to identify and characterize severe asthma in Uganda, Kenya, and Ethiopia. Participants provided written informed consent to participate in this study. Ethics approval for this sub-study was obtained from the School of Biomedical Sciences Research and Ethics Committee (SBS 598), the Uganda National Council for Science and Technology (HS 2532), and the Uganda National Drug Authority (9464). All participants in the current study were healthy adults (⩾18 years), without known atopic disease or respiratory conditions, and not taking steroids or oral antihistamines for any reason in the last 7 days. Since there is limited literature on the prevalence of Aspergillus skin positivity among healthy population, to understand prevalence of Aspergillus sensitisation in Ugandan healthy population, we consented and tested healthy volunteers. We used healthy individuals from the general population, including medical workers, medical students, and support staff at Makerere University and Kiruddu National Referral Hospital. A convenience sampling method was employed for this pilot study.
A. fumigatus SPT [Immunospec (Pty) Ltd, Johannesburg, Gauteng, South Africa] was performed and the results interpreted according to international guidelines. 16 Normal saline served as a negative control while histamine was the positive control, with a mean wheal diameter of at least 3 mm being considered positive, after 15 min of allergen application. We did not perform testing for total serum or A. fumigatus-specific IgE.
Data were analyzed using STATA® version 16 (STATA, College Station, TX, USA). Primary data analysis aimed to describe the distribution of A. fumigatus skin prick positivity at a 95% confidence interval (CI).
Results
Between March and October 2019, we enrolled 50 eligible participants, of whom 28 (56%) were female; the median age for all participants was 30 years [interquartile range (IQR) = 27–35). A total of 28 (56%) of the participants were health workers (Table 1).
Baseline characteristics of the study population.

IQR, interquartile range.
The prevalence of A. fumigatus skin positivity was 60% (30/50) (95% CI = 45.6–72.8). There was a significant difference in age between participants who were positive for A. fumigatus skin test and those who were negative (p = 0.005) (Figure 1). There was no significant difference in A. fumigatus reactivity by gender (p = 0.907) or occupation (p = 0.612). None of the participants reacted to the negative control. Only one participant had a wheal diameter of 2 mm and the rest had zero against the negative control. Figure 2 is a histogram of wheal sizes for A. fumigatus skin prick test for this population.
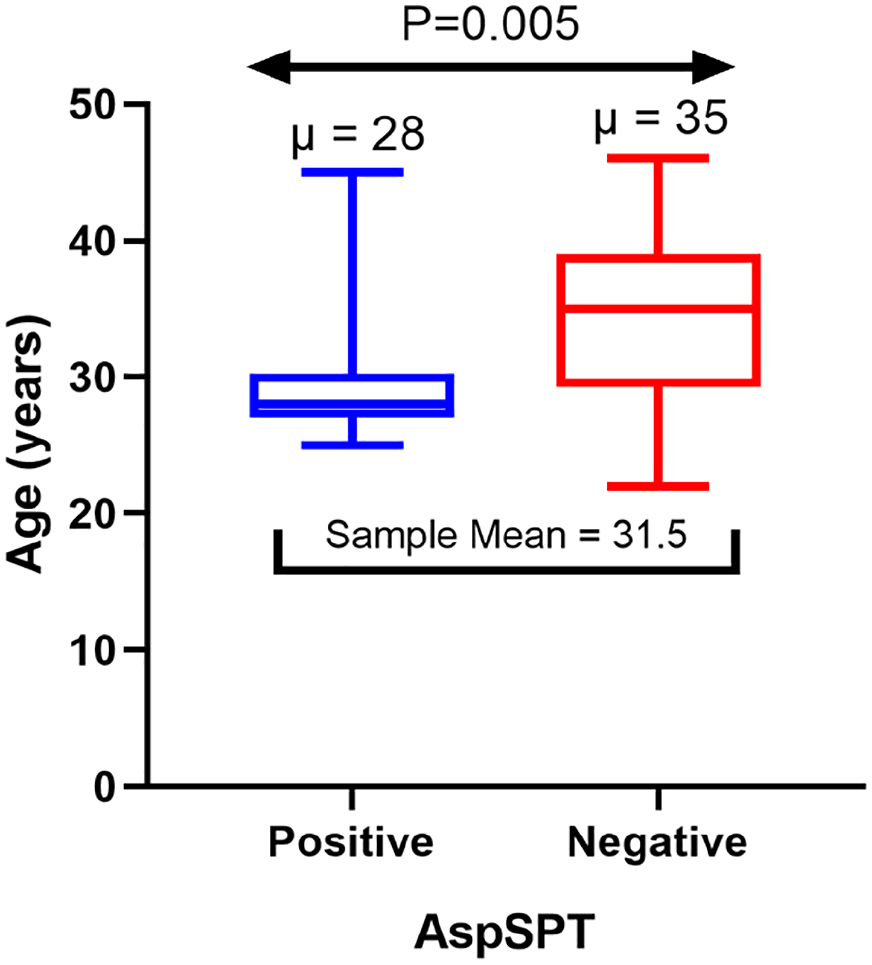
Distribution of age by Aspergillus fumigatus skin positivity.

Distribution of wheal size for Aspergillus fumigatus. Wheal size (µ) is measured in millimeters.
Discussion
In the present study, we report a high prevalence (60%) of A. fumigatus skin positivity among healthy adults without known atopic disease in Uganda. This could indicate the fact that apparently healthy people have an undiagnosed fungal atopy. Alternatively, this could also indicate that A. fumigatus is the commonest fungal allergen in Uganda, similar to what is reported in the developed countries. However, a study from India showed a 2% prevalence of A. fumigatus skin positivity among healthy controls. 17 In addition, our previous work among adult asthmatics in Uganda showed a lower prevalence (48%) of A. fumigatus skin reactivity than the 60% observed in this study despite the atopic nature of asthmatics. 10 We cannot rule out the possibility of cross-reactivity to A. fumigatus crude extracts, which often do not indicate genuine sensitization.13,18 Besides, products secreted after conidial germination into hyphae can be differentially recognized by protective T-cells in healthy non-atopic individuals. 19 Therefore, SPT should not be used alone to diagnose A. fumigatus sensitivity in Uganda since the background prevalence in healthy populations is apparently very high based on the results of this pilot study. However, we recommend that larger studies be carried out to confirm these data. The in vitro A. fumigatus-specific IgE may be more informative to reflect the actual burden. Those with skin positivity were significantly younger. However, the reason for this observation is unclear and not really clinically relevant.
Our study is limited by its very small sample size and sampling frame, which limits its generalization to the general population. The optimal cut-off wheal size to define A. fumigatus in our population is unknown. This could have led to an over-estimation of the prevalence of A. fumigatus SPT reactivity in this population. One uncertainty is whether the 3 mm cut-off is appropriate for Uganda; a 5 mm cut-off would reduce the prevalence of A. fumigatus sensitisation to 8%, which is more reasonable. Therefore, we propose to define a suitable cut-off wheal size in healthy adults. However, our estimate provides baseline information to encourage further research in the field of fungal allergy in Africa. More studies are needed to define the prevalence of A. fumigatus skin positivity among non-atopic healthy population in Africa. There is an urgent need to establish a normal wheal cut-off value for this population.
Conclusion
In conclusion, we found a high prevalence of A. fumigatus skin positivity in apparently healthy non-atopic individuals in Uganda. Those with skin positivity were significantly younger. Given a very high background A. fumigatus skin positivity rate in our setting, SPT alone may be an unreliable test for the diagnosis of A. fumigatus-associated allergic syndromes. We propose to define a suitable cut-off wheal size in healthy adults.
Supplemental Material
sj-xlsx-1-tai-10.1177_20499361211039040 – Supplemental material for Prevalence of Aspergillus fumigatus skin positivity in adults without an apparent/known atopic disease in Uganda
Supplemental material, sj-xlsx-1-tai-10.1177_20499361211039040 for Prevalence of Aspergillus fumigatus skin positivity in adults without an apparent/known atopic disease in Uganda by Richard Kwizera, Felix Bongomin, Ronald Olum, William Worodria, Freddie Bwanga, David B. Meya, Bruce J. Kirenga, Robin Gore, Stephen J. Fowler and David W. Denning in Therapeutic Advances in Infectious Disease
Footnotes
Acknowledgements
We would like to acknowledge all our participants. We thank the IDI research department for providing the PhD student with office space, internet access, printing services, research peer support, health insurance, and salary support during the period of study. David Denning and Stephen Fowler receive funding from the Manchester NIHR Biomedical Research Centre.
Author contributions
RK conceived the study. RK designed concept/protocol. RK, WW, DWD, and BJK participated in getting project funding. RK collected data. RK and RO reviewed, curated and analysed data. RK participated in initial manuscript drafting. RK, FB, RO, DBM, WW, FB, SJF, DWD, RG, and BJK participated in critical revisions for intellectual content. BJK participated in project administration. DBM, WW, FB, SJF, and RG participated in supervision.
Availability of data and materials
All data are available in the manuscript and supporting information.
Conflict of interest statement
The authors declare that there is no conflict of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported through the DELTAS Africa Initiative grant # DEL-15-011 to THRiVE-2. The DELTAS Africa Initiative is an independent funding scheme of the African Academy of Sciences (AAS)’s Alliance for Accelerating Excellence in Science in Africa (AESA) and supported by the New Partnership for Africa’s Development Planning and Coordinating Agency (NEPAD Agency) with funding from the Wellcome Trust grant # 107742/Z/15/Z and the UK government. ASAP [ClinicalTrials.gov identifier: NCT03065920] under the Makerere University Lung Institute provided SPT testing kits to the student. ASAP was funded by the GlaxoSmithKline (GSK) African Non-Communicable Disease (NCD) Open Lab (Project number: 8019) to Bruce Kirenga. Funders had no role in data collection, analysis, or decision to publish. Authors retained control of the final content of the publication.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
