Abstract
Background:
Studies of nasopharyngeal secretions serve as reliable surrogate to evaluate the involvement of viruses in acute otitis media (AOM) and upper/lower respiratory tract infections (URIs/LRIs). We explored nasopharyngeal viral studies from children with uncomplicated AOM and examined their cost-effectiveness in relation to their age.
Methods:
We identified children aged 0–6 years admitted to our pediatrics department in a university-affiliated, secondary hospital with uncomplicated AOM and concurrent URI/LRI between 2012 and 2017, during October–April, when viral studies are performed. Studies were performed either using antigen detection tests, for respiratory syncytial virus (RSV) and influenza A/B (2012–2016) and for a variety of other common respiratory viruses, utilizing multiplex polymerase chain reaction assays (2017).
Results:
A total of 249 children were included (median age: 15 months). In 88 (35%) children, viral studies were positive, most of them in children ⩽24 months (78, 89%). RSV was positive in 52 (59%) children, followed by influenza A and B, in 11 (13%) and 5 (6%) children, respectively. First year switch to a molecular assay, 4.5-fold more expensive, resulted in a statistically significant higher yield: 69% positive results in ⩽24 months, and 66% in those aged ⩽12 months (p < 0.05). In those ⩽24 months, US$23 and US$95 were spent for one positive test in the antigen detection years and the polymerase chain reaction year, respectively, whereas in those >24 months, US$83 and US$878 were invested for one positive test in the same year, respectively.
Conclusion:
In cost-effectiveness terms, the greatest benefit of nasopharyngeal studies was highest in children ⩽24 months.
Keywords
Introduction
Acute otitis media (AOM) is a prevalent disease in children. By 1 year of age, 23% of the children residing in Galveston, TX, USA, experienced at least one episode of AOM; by 3 years of age, 60% had at least one episode of AOM, and 24% had at least three episodes. 1 Most episodes will be unnecessarily treated with antibiotics, thus, being certainly an important cause for antibiotic resistance. In the last few decades, the role of viral infections in the development of AOM during upper/lower respiratory tract infection (URI/LRI) has been established. AOM complicates around 35% of URIs in young children. 2 Viruses and bacteria cointeract in the inflammatory process in the nasopharynx and the middle ear.3–5 It was also shown that viruses alone can cause AOM: viruses were the only pathogens detected in 5–39% of middle ear fluid (MEF) samples obtained from AOM cases.6–8
Age, time during URI course, virus type, and seasonality are important factors in AOM development. Infants are considered to be more prone to develop AOM. In infants <12 months of age, AOM was diagnosed in about 20% of those with URI and in about 8% in those with LRI. 9 The cumulative incidence of URI, LRI, and AOM in the first year of life is 92%, 18% and 46%, respectively. 9 AOM is usually diagnosed in the first few days after URI symptoms have begun.10,11 However, almost half of children with URI would experience AOM within 28 days following the infection. 9 Of the viruses that can cause AOM, respiratory syncytial virus (RSV), influenza (INF), adenoviruses (AdV), and human metapneumovirus (hMPV) are considered to be ototropic.3,12 There is a seasonal correlation between URI and AOM development: AOM associated with RSV and hMPV infections peaks during winter time, when their activity is high. 12
Specimens obtained by nasopharyngeal (NP) swabbing/washing are often used for viral detection testing owing to the ease of specimen collection. In a real-life setting, NP cultures are not obtained in uncomplicated, nonhospitalized AOM children, and myringotomy and MEF testing for viruses or bacteria are not required for AOM diagnosis and treatment purposes. Previously, NP specimens were usually tested just for antigens for RSV and for INF A/B. In recent years, many laboratories switched to more sensitive and specific polymerase chain reaction (PCR)-based assays that also tested for other viruses.
The value of NP viral testing in a real-life, clinical setting is high. The practice of sending viral studies in hospitalized children is likely to confirm the presence of a viral disease, monitor RSV burden (especially in those younger than 3 years), search for INF cases (and give treatment) or to consider discontinuation of antibiotics and early discharge, if viral disease is evident and bacterial disease is ruled out. To date, there is no clear age cut-off from which children NP samples should be obtained and there are no clear guidelines when to perform NP testing or to whom in the setting of AOM.
In this article, we study the results of NP tests obtained from children with uncomplicated AOM and URI/LRI who were hospitalized during the cold months in six consecutive calendar years. A secondary objective was to examine the cost-effectiveness of such NP tests in relation to children’s age, to identify which age group would benefit the most.
Patients and methods
The study was approved by the local institutional review board. Owing to the nature of the study, written consent was not necessary.
Study aims
The aims of this study were as follows.
To study the positivity of NP tests obtained from children with uncomplicated AOM hospitalized during six consecutive URI/LRI seasons.
To study the cost-effectiveness of NP tests in relation to children’s age, to identify which age group would benefit the most.
Study design and population
We performed a retrospective chart review of children aged 0–6 years who were admitted to our pediatrics department in a university-affiliated, secondary medical care center between 2012 and 2017. Our study population was composed of children who required hospitalization owing to URI/LRI and concurrently presented with uncomplicated AOM and had NP testing results obtained on their admission. The reason for their hospitalization was supportive treatment (i.e. steroid therapy, inhalations, intravenous antibiotics) and vital-sign monitoring (i.e. saturation). Eligible children were identified from our electronic database, by using International Code of Disease-9 discharge codes: 381.0X, acute nonsuppurative/mucoid OM; 381.4, nonsuppurative OM, not specified as acute or chronic; 382.4, unspecified suppurative OM; and 382.9, unspecified OM.
In our institution, viral NP tests are performed during the cold months in Israel (October–April), but not during the summer months, and not automatically in all children. They are performed according to the physician’s discretion. We excluded children with complicated AOM (defined as AOM episodes associated with treatment failure, spontaneous otorrhea, or with complication, i.e. mastoiditis), forms of OM other than AOM (i.e. OM with effusion or chronic suppurative OM), charts with incomplete data, and children with faulty tests, that is, reported with technical errors.
The study years were chosen to blunt the effect of the preceding years characterized by substantial changes in the pneumococcal conjugated vaccine (PCV) policy, after PCV13 replaced the former PCV7 in 2010 in the local national immunization program. The study years were also not considered to be pandemic or exceptional for respiratory virus-associated morbidity in our country.13,14
We collected data concerning age (rounded to months), gender, NP viral studies results, and duration of admission. Owing to the disparity between the peak age of RSV associated illness in the first year of life and the peak age for AOM, at 2 years of age, we categorized the study children into two age groups to study our primary outcome: that is, which age group would benefit most from NP viral testing, measured by the rate of positive NP results. The two patient groups were: (1) <2 years (0–24 months) and 2–6 years (25–72 months) (AOM peak age), and (2) ⩽1 year (0–12 months, RSV peak age) and 1–6 years (13–72 months).
Viral studies
The decision to send NP samples was made by the attending pediatrician during the emergency department visit, shortly before admission, and when AOM was diagnosed when the child was symptomatic, using Regular FLOQ swabs (Copan, Italy).
Between 2012 and 2016, the panel kit included antigen assays for RSV, INF A, and INF B (BinaxNOW RSV and Influenza A & B cards, Alere, Abbott, Chicago, IL, USA), which are rapid qualitative detection immunochromatographic assays (for RSV, sensitivity 89% and specificity 100%; for INF A, sensitivity 70–89% and specificity 90–99%; for INF B, sensitivity 50–69% and specificity 94–100%). During these years, the testing for each virus was conducted upon the pediatrician’s order, and not automatically for all. The direct cost was US$9.45 for RSV testing and US$14.55 for INF A/B testing. Children in these years could have contributed more than one test result, for example, RSV and INF A, according to the order to the microbiology laboratory.
In 2017, we used a PCR-based assay. This qualitative in vitro test for the multiple detection of human respiratory viruses included INF A subtypes H1, H3, H1pdm09, INF B, and RSV (Allpex Respiratory Panel 1, Seegene, Seoul, Korea; detection limit = 100 copies/reaction), together with AdV, hMPV, enterovirus (EV) and parainfluenza 1–4 (PIV1–4) (Allpex Respiratory Panel 2, Seegene, Seoul, Korea; detection limit = 100–1000 copies/reaction). The direct cost of these two kits was US$43.15 per child. There was no change in the policy regarding the pediatricians’ judgement to send NP specimens for viral testing following the switch, but they were informed about the higher cost.
Statistical analysis
All positive results for respiratory viruses from either test were combined per virus.
Continuous variables were evaluated for normal distribution using histogram and Q–Q plot. Normal distributed continuous variables were described as mean and standard deviation (SD), whereas nonnormally distributed continuous variables were described using median and interquartile range (IQR). Age was evaluated for normal distribution, using histogram, and reported as median and IQR. Categorical variables were reported as frequency and percentage. χ2 test and Fischer’s exact test were used to compare categorical variables. All statistical tests were two sided, and p < 0.05 was considered as statistically significant. SPSS software was used for all statistical analysis (IBM SPSS STATISTICS for WINDOWS version 24.0, IBM Corp., Armonk, NY, USA, 2016).
Results
Demographics
A total of 249 children, with a median age of 15 months (IQR 7–22) were included. Demographic and clinical data are shown in Table 1. Of them, 155 (62%) were boys, 202 (81%) were <24 months old, and 92 (37%) were ⩽12 months old. The mean age of the >24 months old group (n = 47) was 34 months (range 24–72). Before comparing the two methods tested in two different groups, we verified that there were no statistically significant differences for age and gender distribution. Of note, there were fewer children in 2016, the last antigen detection year, when compared with previous years.
Clinical and demographic data.

INF, influenza; IQR, interquartile range; PCR, polymerase chain reaction; RSV, respiratory syncytial virus.
Antigen detection years (2012–2016)
In 188 children, a total of 280 tests were conducted: 126 for RSV (direct cost US$1190) and 154 for INF A/B (direct cost US$2240). Of them, 147 (7
PCR year (2017)
A total of 61 children (25% of the study children) were tested with both kits, because each kit tested for certain viruses, as described previously. Of them, 55 (90%) were aged <24 months and 35 (57%) were aged ⩽12 months. A total of 38 (69%) tests were positive in children aged <24 months,
RSV detection test results, per age.

RSV, respiratory syncytial virus
Numbers shown represent the total positive RSV cases per year from all the number of cases tested for RSV (2012–2016).
In 2017, all nasopharyngeal samples were tested for RSV and other respiratory viruses.
Positive tests
Overall, 88 (35%) children had positive viral studies. For RSV, tests were positive in 52 (59% of tested episodes for RSV) children, followed by INF A, in 11 (13%) and INF B, in 5 (6%) children. Positive NP viral results were mostly common in children aged <24 months (78, 89%), and were significantly more common in children aged ⩽12 months (43, 49%) (p = 0.025 and p = 0.004, respectively).
Table 3 shows the distribution of RSV results. Positive results were common in children ⩽12 months (31, 36%), which were statistically significantly higher than in older children aged 13–72 months (p = 0.038). Preadmission antibiotics were given in 66 (27%) children, and antibiotics during admission were given to all children. There was no statistically significant difference in the positive RSV results between those who were treated with antibiotics before admission (43%) and those who were not (29%) (p = 0.318). RSV positive tests distributed almost equally between boys and girls (Figure 1). The switch to a multiplex PCR assay resulted in almost a twofold increase in the RSV detection rate (Figures 1 and 2). Among those aged <24 months, 69% were RSV positive, and 66% in those aged ⩽12 months (Figure 3).
Proportion of other viruses detected, per age (2017).

In 2017, all nasopharyngeal samples were tested for RSV and other respiratory viruses.
AdV, adenovirus; EV, enterovirus; INF, influenza, PIV, parainfluenza; RSV, respiratory syncytial virus.
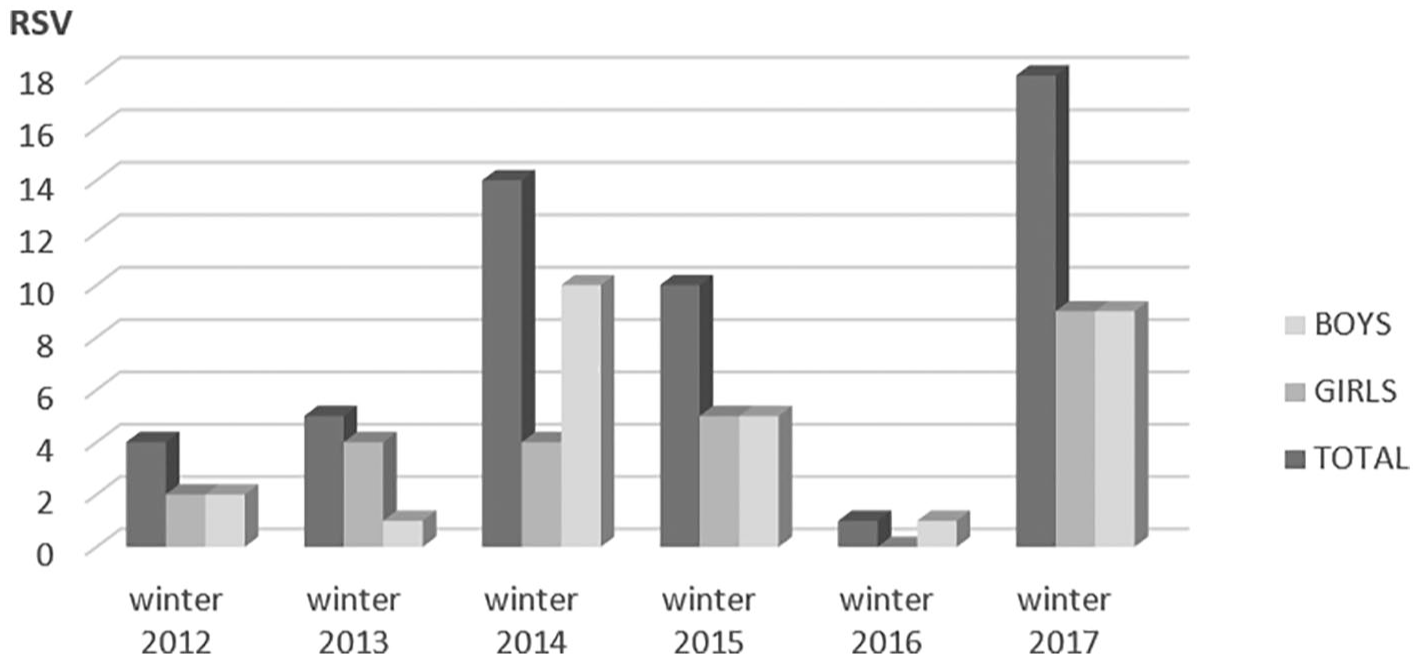
Respiratory syncytial virus positive cases, by gender and study year.

Positive nasopharyngeal tests, by age (⩽12 months versus ⩾13 months) and study year.

Number of positive RSV cases, per age (<12 months vs 13–72 months), per study year.
Cost-effectiveness
Overall, for each one positive test, one needed to invest US$73 in the antigen detection years, and US$124 in the PCR year. When children were grouped according to their age, there was a significant difference in the costs. In those ⩽24 months, one needed to invest US$23 and US$95 for each positive test in the antigen detection years and the PCR year, respectively, whereas in those aged >24 months, one needed to invest US$83 and US$878 for each positive test in the antigen detection years and the PCR year, respectively.
Discussion
We showed an overall low rate of positive NP samples when using antigen detection tests (2012–2016) and moderate–high rate of positive results when using a multiplex PCR assay (2017) in young children hospitalized with concurrent URI/LRI and AOM. During 2012–2016, the yield of viral testing in those aged ⩽24 months was low (27%) and even remarkably lower in those aged ⩽12 months (10%), despite their tendency to be more prone to respiratory viral infections. First year switch to a molecular assay, 4.5-fold more expensive, resulted in a statistically significant higher yield of this testing: 69% positive test results in <24 months and 71% in those aged ⩽12 months. From cost-effectiveness aspects, NP tests should be performed only in children ⩽24 months but not in older ones.
As expected, RSV was the most common virus detected in our NP viral tests. Studies that report on the viral etiologies of AOM episodes, such as the present one, emphasize again the role of RSV as the most detected virus in AOM cases from NP samples, and refine the need for an effective RSV vaccine, in light of decades of unsuccessful attempts. 15 Heikkinen et al. showed the high prevalence of RSV-associated AOM in young Finnish children at the outpatient setting in the early 2000s, especially in children <3 years of age. 16 They reported an average annual RSV illness incidence rates per 1000 children of 275 in children <3 years of age, compared with 117 in those 3–6 years of age, and 46 in those aged 7–13 years. AOM was the most frequent complication of RSV infection, occurring in 148 children in the study population (50%) and in 87 children <3 years of age (58%).
The higher overall positive viral results documented during 2017, when compared with 2012–2016, was due to the detection of viruses other than RSV/INF A/INF B that were not included the former antigen detection assay. In our opinion, there is a great importance in detecting viruses with ototropic abilities, 3 such as AdV, which was positive in 17 (31%) children. However, again, such detections did not substantially change the management of these children during their hospitalization in our experience.
The transition to a multiplex PCR testing showed that RSV was still the most common virus detected and was always positive if there was a coinfection. Ruohola et al. examined viruses in the nasopharynx of children with and without AOM. 5 Respiratory viruses were detected in 87% of the AOM group, and RSV contributed to the development of AOM, even when no bacteria were detected. In fact, they found that RSV increases by sixfold the risk of AOM.
Strengths of our study include: (1) generalizability to the entire Israeli pediatric population, as our child population is similar to the national child population with regards to many socioeconomic and health parameters (Israeli Central Bureau of Statistics, www.cbs.gov.il), (2) a relatively large sample size, and (3) entire study in the post-PCV13 years with no further changes in the immunization policy. Limitations include: (1) lack of bacterial data from MEF or NP studies, yet our focus was viruses; (2) during the antigen detection years, not all tests were conducted for RSV, INF A and B, as they were not always routinely ordered; (3) most of our results are from the antigen detection years, but this is true also for other reports;17,18 (4) our study population comprised hospitalized children, whose AOM is probably more severe than children who had not been hospitalized, yet NP sampling and testing for viruses was not done in the latter population; (5) INF vaccination data was incomplete, because of inconsistencies in the reviewed charts and the parents’ recall bias (INF vaccination is voluntary and up to the parents’ consent); this weakness is explained by the retrospective nature of this study; (6) in 2017, we had the highest frequency of included children (61 children, 25%) when compared with the previous years and with younger median age; possible explanations for the over-presentation of children tested in the PCR year is the technical ease of sending these samples and the need to test the new technology, but it could have also caused a selection bias; and (7) possible technical issues while obtaining NP swabs, as they depend on disease severity, anatomic differences between age groups, and other factors.
In an era when more sophisticated and more expensive laboratory assays replace old tests, as shown here in this study with PCR replacing antigen detection assay, it is obligatory to wisely consider their use. We showed that the money spent on NP tests in children >24 months with uncomplicated AOM and URI/LRI is almost always valueless and therefore obtaining NP test from these children should be discouraged.
Conclusion
Our recommendation, based on our results and limited to hospitalized children with concurrent URI and uncomplicated AOM, is to perform NP testing only in those aged ⩽24 months and not in older children.
