Abstract
Background:
The regimen of raltegravir (RAL), ritonavir-boosted darunavir (DAR/r), and etravirine (ETR) for HIV treatment-experienced patients in a non-clinical trial setting in the rural/semi-urban United States had not been evaluated.
Objective:
A retrospective cohort analysis was performed of adult patients prescribed the regimen from 2008 to 2013 at a HIV clinic serving such a population.
Results:
In all, 51 patients met inclusion criteria including 15 with suppressed viral loads at regimen initiation. Of the 36 patients with detectable viral loads, 22 (61.1%) achieved a plasma HIV-1 RNA level < 50 copies/ml at 28 weeks and 17 maintained viral suppression at 56 weeks (50% of those surviving without death). Of 42 patients with long-term follow-up, mean of 216 ± 83 weeks following regimen initiation, 31 (73.8%) had viral suppression. Suppression was significantly more likely in those patients that maintained adherence.
Conclusions:
In a non-urban clinic population from the United States with considerable treatment experience, the combination of RAL, DAR/r, and ETR was well tolerated and resulted in viral suppression in those that maintained adherence. Future prospective studies may better define the role of such a regimen in the context of revised recommendations for first-line medications in the HIV treatment naïve.
Background
For heavily treatment-experienced patients for whom drug-resistance is compounded or medication-related adverse effects limit drug choice, so-called ‘salvage’ regimens are constructed in an attempt to treat with three active drugs. 1 One such combination regimen of raltegravir (RAL), etravirine (ETR), and ritonavir-boosted darunavir (DAR/r) was first demonstrated to confer viral suppression in 86% [confidence interval (CI): 80–93%] of 103 patients with multidrug-resistant HIV-1 from a clinical trial in France. 2 However, reports of RAL, ETR, and DAR/r for use in non-clinical trial settings are limited in populations from the United States and absent from non-urban sites. 3 Few studies have examined differential HIV outcomes between rural and urban settings of the United States, though recent outbreaks of rural HIV highlight the potential importance.4,5
Objectives
People living with HIV in rural areas have limited access to HIV care services based on geography and other factors, and yet the recommendation for and monitoring of a salvage regimen requires expert consultation. 6 The University of Virginia’s HIV clinic cares for more than 650 people living with HIV principally in rural and semi-rural Virginia. The catchment area includes approximately 24,000 square miles with 19 counties designated as underserved by the Department of Health and Human Services. 7 A recent local study found that the distance a patient with HIV lived from hospital was an independent predictor of lower CD4 count and hospital mortality from sepsis. 8 Such a clinical setting may therefore be important to study the efficacy and tolerability of drug regimens that may be dependent upon factors like adherence. Thus, we sought to describe the clinic’s experience with RAL, DAR/r, and ETR for salvage therapy to inform the use of this approach beyond the clinical trial setting and specifically in a clinic serving a majority non-urban population.
Methods
Study design and study population
A retrospective analysis was performed of HIV-1 infected adults who were prescribed the regimen of RAL (400 mg twice daily), DAR/r (600 mg/100 mg twice daily), and ETR (200 mg twice daily) for the first time at the University of Virginia’s HIV clinic from 2008 to 2013 to allow for longer term follow-up. Criteria for inclusion consisted of patients with confirmed HIV, diagnosed by ELISA, and confirmed with Western blot, who had detectable or undetectable plasma HIV-1 RNA levels (viral load) within 12 months prior to starting RAL, DAR/r, and ETR. Patients with prior use of all 3 drugs in combination were excluded. Patients were not on concomitant nucleoside reverse transcriptase inhibitor (NRTI) backbones while on the regimen.
Clinical data were gathered from the electronic medical record, including demographics (age, sex, date of HIV diagnosis, number of years from diagnosis to initiation of the regimen), plasma viral load and CD4 cell count at time points before and after regimen initiation, prior antiretroviral regimens, HIV resistance genotype profiles, and indications for the regimen prescription, and comorbidities existing prior to initiation or acquired during the initial 56 weeks of the regimen. Comorbidities were as follows: coronary artery disease defined by a history of acute coronary syndrome or presence of obstructive lesions on angiographic imaging (CAD); chronic kidney disease stage III or greater defined by a <glomerular filtration rate (GFR) 45–59 ml/minute [chronic kidney disease (CKD)]; diabetes mellitus type II defined by HgbA1c >6.5% (DMII); liver disease, including hepatitis B, hepatitis C, or cirrhosis; hyperbilirubinemia or hepatic dysfunction defined by a total bilirubin >3x upper limit of normal or aspartate transaminase/alanine transaminase (AST/ALT) >3 x upper limit of normal; mental health disorder defined by regular appointments with psychiatry, prescription of antidepressants, antipsychotics, or mood stabilizers; and substance abuse disorder defined by chart documentation. HIV resistance genotypes were assessed by laboratory records and transcribed medical notes, and interpreted using the Stanford University HIV drug resistance database. 9
Given the limited number of studies applying three novel agents simultaneously in treatment-experienced patients, antiretroviral efficacy standards used in studies conducted with treatment naïve patients were utilized. Thus, we considered ⩾60% of the population of patients having viral suppression at 56 weeks as effective.10,11 Viral suppression was defined as a viral load levels <50 copies/ml given the early generation assays employed at the beginning of the study period. Adherence was assessed by patient self-report, clinician documentation, and pharmacy records in the medical record. Given that this was not a prospective study designed to monitor adherence, we elected to define non-adherence more conservatively as missing more than three scheduled doses of any of the RAL, DAR/r, and ETR regimen in one month in line with earlier studies of adherence that found improved viral suppression and treatment outcomes with 95% adherence or better.12,13
The protocol was approved by the institutional review board for human subjects research at the University of Virginia and granted exemption from written consent.
Statistical analysis
The primary endpoint was the proportion of patients reaching viral suppression measured at week 28, at week 56, and to the last time point available. These time points were used to mimic as closely as clinically feasible common clinical trial endpoints of 24 and 48 weeks, but allowing for a reasonable visit scheduling window and for blood tests that may be have been ordered at a visit but not performed until days after. Time was quantified as starting at the initiation of the RAL, DAR/r, and ETR regimen. Categorical variables were summarized by frequency tables, and continuous variables were summarized by standard descriptive statistics (range, quartiles, means, standard deviations). Due to the small group sizes between those achieving or not achieving viral suppression, categorical variables were analyzed with an exact logistic regression model, and quantitative variables were analyzed with two-sample t-tests or non-parametric methods. The differences in viral suppression between each time point and prior to initiating the RAL, DAR/r, and ETR regimen were assessed by McNemar’s exact test. An exact multivariable logistic regression model was used to assess candidate risk factors simultaneously. SPSS Version 21.0; (SPSS, Chicago, IL) and SAS 9.4 (SAS Cary, NC) were used for analysis.
Results
In all, 56 patients were screened and 51 patients met study inclusion criteria. The mean age was 51 ± 11 years (see Table 1). The majority of patients were male (72.5%), and the mean time from HIV diagnosis to initiation of the RAL, DAR/r, and ETR regimen was 21 ± 7 years. Patients with prior exposure to an individual drug in the regimen (but not in combination) were as follows: RAL 15 (29.4%), DAR/r 20 (39.2%), and ETR 10 (19.6%). The primary documented indication for prescribing the RAL, DAR/r, and ETR regimen in 48 (94.1%) was drug resistance on the basis of treatment failure history and/or drug resistance testing. Additional contributing indications that were not mutually exclusive from the primary indication were hyperbilirubinemia or hepatitis associated with atazanavir in 5 (9.8%), and CKD necessitating a switch from tenofovir disoproxil fumarate in 2 (3.9%), and nephrolithiasis in 1 (2.0%) patient. Of the 35 patients with available genotyping for review prior to the initiation of the RAL, DAR/r, and ETR regimen, 16 (45.7%) had mutations in three or more classes of antiretrovirals predictive of phenotypic resistance (see Table 2).
Baseline characteristics of patients started on RAL, DAR/r, and ETR.

CAD, coronary artery disease – CAD (history of ACS, presence of obstructive lesions on angiographic imaging); CKD III, chronic kidney disease (GFR 45–56); DAR/r, darunavir; DMII, diabetes mellitus II; ETR, etravirine; HBV, Hepatitis B; HCV, Hepatitis C; SD: staULN, upper limit of normal, hyperbilirubinemia or liver failure – T Bili >3x ULN or AST/ALT>3 x ULN; RAL, raltegravir.
Overall sample of 51.
HIV genotype resistance profiles,

IQR, inter quartile range; SD, standard deviation.
Major protease inhibitor mutations as defined by the Stanford University HIV drug resistance database.
A single patient may have contributed multiple mutations.
Primary endpoint
Prior to initiation of all three drugs simultaneously, RAL, DAR/r, and ETR, 15 (29.4%) patients had a suppressed viral load over the previous year. Those patients that had a suppressed viral load over the previous year were excluded from analysis of treatment efficacy. Of the remaining 36 patients with detectable viral loads before initiation of RAL, DAR/r, and ETR, the median viral load was 4203 (285–7.5 x 104) copies/ml. At week 28, 22 patients (61.1%) had achieved viral suppression, and the median viral load had dropped to 48 (48–430) copies/ml (Figure 1). Two patients died between week 28 and 56 and were hence excluded from week 56 analyses. The first patient did not achieve viral suppression and died from complications of ESRD. The second patient also did not achieve viral suppression and had multiple episodes of non-adherence with death attributable to AIDS. At week 56, 17 (50%) patients had achieved viral suppression.
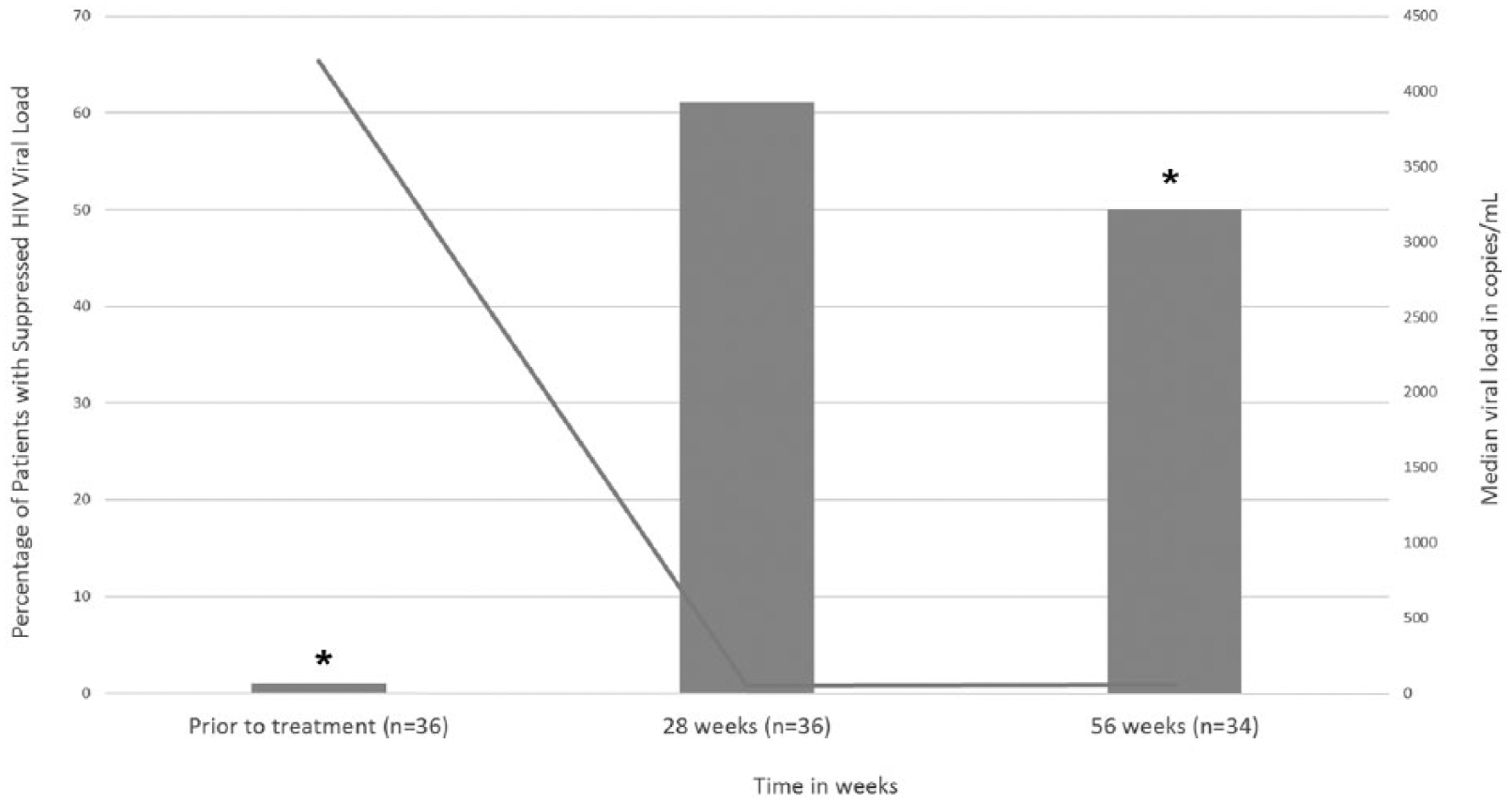
Proportion of patients with viral suppression and the median detectable viral load during the study period.
Predictors of viral suppression at 56 weeks
A descriptive analysis was also completed for all patients who met inclusion criteria for the study, including those 15 patients that had suppressed viral load at the time of RAL, DAR/r, and ETR initiation. One patient with suppressed viral load at the time of initiation died between week 28 and 56, hence 48 patients were analyzed (Table 3). Drug intolerances (pill burden, rash, nausea) occurred in 2 (11.1%) patients without viral suppression compared to 1 patient (3.3%) with viral suppression (
HIV parameters from initiation to week 56.

CAD, coronary artery disease-CAD (history of ACS, presence of obstructive lesions on angiographic imaging); CKD III, chronic kidney disease (GFR 45–56); HBV, Hepatitis B; HCV, Hepatitis C; ULN, upper limit of normal, hyperbilirubinemia or liver failure (T Bili >3x ULN or AST/ALT>3 x ULN).
Overall sample of 51 as noted there were 3 deaths occurred between week 28 and 56.
Statistically significant in univariate analysis (
Patient reported difficulties with pill burden but never discontinued therapy.
Long term follow-up
Patients were assessed up to their most recent available viral load measurement. The last time point available for patients was at a mean of 216 ± 83 weeks following RAL, DAR/r, and ETR initiation, and of those 42 patients who had complete documentation past 56 weeks, 31 (73.8%) achieved viral suppression, including 14 (93.3%) of those 15 patients that originally had undetectable viral loads at the time of initiation. In one patient with documented adherence but lack of viral suppression, new integrase inhibitor resistance (N155H mutation) was found after 120 weeks of RAL, DAR/r, and ETR. RAL was replaced with dolutegravir and the patient subsequently achieved viral suppression.
Discussion
This study is to our knowledge the first to assess the application of RAL, DAR/r, and ETR outside of the clinical trial setting in the United States. Our results demonstrate that in treatment-experienced patients infected with detectable drug-resistant HIV, viral suppression with RAL, DAR/r, and ETR occurred in 61.1% and 50.0% of cases at 28 weeks and 56 weeks, respectively. Suppression rates did not meet the primary endpoint of 60%, and were lower than the 86% of treatment experienced patients achieving viral suppression at 48% and 88% at 96 weeks in the ANRS 139 Trio trial.2,14
There were important differences between the ANRS 139 Trio trial design and the clinical practice for patients in this cohort. All patients in the ANRS 139 Trio trial had a detectable viral load at initiation, and the degree of resistance was greater, for example, a median number of 4 major PI mutations at enrollment. 2 Importantly, the majority of patients in the clinical trial were also treated with an optimized background regimen of NRTIs or enfuvirtide. It remains unknown if increased use of other NRTIs or enfuvirtide could have improved the rates of viral suppression, but for enfuvirtide in particular, this practice was avoided because of concern for drug intolerance. Also, patients in ANRS 139 Trio were excluded if they had previously been treated with RAL, DAR/r, or ETR. Instead, prior exposure to one study drug was common in our observed clinic practice and may have been related to the timing of when individual drugs were available for use and was explanative of why some patients had an undetectable viral load when the final combination of RAL, DAR/r, and ETR was initiated. These 15 patients with undetectable viral load when the final combination were therefore excluded from the primary efficacy analysis and could be more aptly compared to patients in regimen ‘switch’ trials of sustained viral load suppression.
The higher rates of viral suppression achieved in ANRS 139 Trio may also have been secondary to the structured monitoring and follow-up which consequently improved medication adherence. While one study by Imaz,
Indeed, in our population from rural United States, viral suppression was significantly associated with clinician documented adherence in the medical record. While we did not have the ability to systematically assess all barriers to adherence, rural populations face unique challenges to adherence and engagement in care which are especially important when patients have progressed to the complexity of a salvage regimen.16 –18 Different than other studies, substance abuse and/or mental health problems did not independently predict a lack of viral suppression. 19 Yet given our sample size, interactions between adherence and having a history of substance abuse and/or mental health problems may not have been adequately analyzed. Furthermore, the clinic had implemented a program to improve adherence with mobile technology in patients with substance abuse that overlapped in time with the study population and may have additionally confounded such analysis. 19 The data from the mobile technology study were not available in the clinical documentation, so analysis of adherence for the present study was prepared in a uniform manner for all patients from the available clinical documentation alone.
The study had other limitations. Chart documentation of self-reported adherence rates may have overestimated average adherence. Yet studies in the era of boosted protease inhibitors have suggested that adherence to as low as 80% of prescribed doses may be all that is necessary to achieve viral suppression. 20 Furthermore, our definition of adherence, missing no more than 3 doses per month, was intended to be conservative. Additionally, patients in this study had a mean time from HIV diagnosis to RAL, DAR/r, and ETR initiation of more than 20 years, and thus findings regarding adherence and tolerability may be most generalizable to other clinic populations with considerable treatment experience. 21
Overall, the RAL, DAR/r, and ETR combination was well tolerated, and no patient discontinued the regimen due to medication-related adverse events. Nevertheless, we observed one case of acquired resistance to RAL with the N155H mutation in a patient with documented adherence. The patient was naïve to RAL, DAR/r, and ETR at initiation of the regimen. While the 96-week follow-up of patients in the ANRS 139 Trio trial did not find acquired integrase mutations, other reports of deep sequencing have suggested that patients with virologic failure to RAL-based salvage regimens with N155H mutants or other signature mutations in the integrase catalytic center originate from pre-existing minority variants that escaped under suboptimal drug pressure. 22 Such cases highlight the ongoing need for sound clinical judgment and informed patient participation in decisions regarding regimen choice and the preservation of drugs within multiple classes that could be used in the case of salvage.
Conclusion
In a non-urban clinic population from the United States with considerable treatment experience, the combination of RAL, DAR/r, and ETR was well tolerated and resulted in viral suppression in those that maintained adherence. Future prospective studies may better define the role of such a regimen in the context of revised recommendations for first-line medications in the HIV treatment naïve.
Footnotes
Acknowledgements
The authors would like to thank University of Virginia Ryan White HIV clinic. A.M.E. contributed to conception and design, or acquisition of data, or analysis and interpretation of data; was involved in drafting the manuscript or revising it critically for important intellectual content; and gave final approval of the version to be published. Y.A. contributed to acquisition of data, or analysis and interpretation of data, and given final approval of the version to be published. J.S. contributed to acquisition of data, and gave final approval to the version to be published. B.W. was involved in drafting the manuscript or revising it critically for important intellectual content, and given final approval of the version to be published. R.D. was involved in drafting the manuscript or revising it critically for important intellectual content, and given final approval of the version to be published. X.W. contributed to analysis and interpretation of data, and gave final approval to the version to be published. S.K.H. contributed to conception and design, or acquisition of data, or analysis and interpretation of data, was involved in drafting the manuscript or revising it critically for important intellectual content, and gave final approval of the version to be published.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Andrew Ebers is on the 5T32AI007046 training grant.
Conflict of interest statement
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Presentation of research
Virginia American College of Physicians research abstract competition, 2015.
