Abstract
Objectives:
Materials and Methods:
Routine clinical 30 or 40 mg/l doses of Flu were applied to
Results:
The results showed that this combination effectively promoted the degradation of the biofilm network, but unfortunately, also stimulated the growth of the yeasts population due to release of several glucose monomers during β-glucans hydrolysis.
Discussion:
AA lead to the hydrolysis of the β-glucans of the matrix, liberating glucose molecules which are used as carbon souce by the yeasts, thus suppressing the desired antifungal effect of the drug combination with Flu.
Conclusions:
Unlike to what happens in treatment of bacterial infection, AA should not be used together with Flu in the treating oral mucositis caused by
Introduction
Fungal infections are a major clinical health problem that affects more than 300 million people annually.
1
Mucositis (or oropharyngeal candidiasis) is a frequent infection among immunocompromised patients,33,34 which is characterized by the presence of creamy, white plaques on the tongue and buccal mucosa that generally leave a raw, painful, and ulcerated surface when scraped. Although not being a severe infection normally, it can be uncomfortable and detrimental to the patient’s nutritional status thanks to the diminished food and liquid intake. The latest 2016’s guidelines recommend the treatment general mucositis infections with oral fluconazole.
35
However, the number of effective antifungal drugs is limited, and resistance to frequently used agents is emerging.
24
Specifically, the resistance of
Ascorbic acid, also designated as vitamin C, has being used concomitantly in antibiotherapy for its capacity to boost the immune system and role as a helper in the treatment of several infections, for several years.
42
AA’s concentration in phagocytes and lymphocytes is very high compared with the level in plasma, indicating that AA may have functional roles in these immune system cells, for example, increasing the functioning of phagocytes, the proliferation of T-lymphocytes and the production of interferon, and decreasing the replication of viruses.
43
Furthermore, its metabolism is affected by various infections (e.g. common cold, pneumonia resultant from many microorganisms, scurvy or
So, the goal of this study was to check the
Materials and methods
Organisms and growth conditions
Flu and AA
Flu was kindly provided by Pfizer®. AA was purchased from Sigma-Aldrich (Roswell Park). Aliquots of 5000 mg/l were prepared using dimethyl-sulfoxide (DMSO), and the final concentrations used (30 and 40 mg/l for Flu and 200 and 300 mg/l for AA) were prepared with RPMI 1640. Controls were performed with DMSO in order to assure that the concentration used was not toxic (concentrations below 1% (v/v) DMSO).
Flu and/or AA effect on C. glabrata biofilm
Biofilm formation
Cell suspension was prepared and washed as described above and resuspended in RPMI 1640. Then, standardized cell suspensions (200 µl) were placed into selected wells of 96-well polystyrene microtiter plates (Orange Scientific, Braine-l’Alleud, Belgium). As a negative control, RPMI 1640 was used without cells and antifungal agent. As positive control, only cell suspensions were tested without antifungal agent. After 24 h, 100 µl of RPMI 1640 was removed and an equal volume of fresh RPMI 1640 plus the antifungal agent (2× concentrated) or the combination of flu and AA was used. The plates were incubated at 37°C for an additional 24 h period, a total of 48 h at 120 rpm. In addition, in order to assess the effect of glucose in the medium, the assay was performed in the same conditions explained above using RPMI supplemented with 2% of glucose.
Biofilm cultivable cells and biomass determination
The number of cultivable cells in the bioflim was determined by the enumeration of colony forming units (CFUs). For that, after the period of biofilm formation, all medium was aspired and the biofilms were washed once with 200 µl of PBS to remove non-adherent cells. After biofilms were scraped from the wells and the suspensions were vigorously vortexed for 2 min to disaggregate cells from the matrix. Serial decimal dilutions in PBS were plated on SDA and incubated for 24 h at 37°C. The results were presented as total of CFUs per unit area (Log10 CFUs/cm2). Total biofilm biomass was quantified by crystal violet (CV) staining. After the biofilm formation, the medium was aspirated and non-adherent cells removed by washing the biofilms with sterile ultra-pure water. Biofilms were then fixed with 200 µl methanol, which was removed after 15 min of contact. The microtiter plates were allowed to dry at room temperature, and 200 µl of CV (1% v/v) added to each well and incubated for 5 min. The wells were then gently washed twice with sterile, ultra-pure water and 200 µl of acetic acid (33% v/v) was added to release and dissolve the stain. The absorbance of the obtained solution was read in triplicate in a microtiter plate reader (Bio-Tek Synergy HT, Izasa, Lisbon, Portugal) at 570 nm. The results were presented as percentage of biomass. 48 All the assays were performed in triplicate and on three separate occasions.
Statistical analysis
Results were compared using one-way analysis of variance (ANOVA), Dunnett’s and Bonferroni’s multiple comparisons test, using GraphPad Prism 5 software. All tests were performed with a confidence level of 95%.
Results
The present study aimed to evaluate an alternative treatment for oral mucositis related to
Figure 1 shows the percentage of biomass of
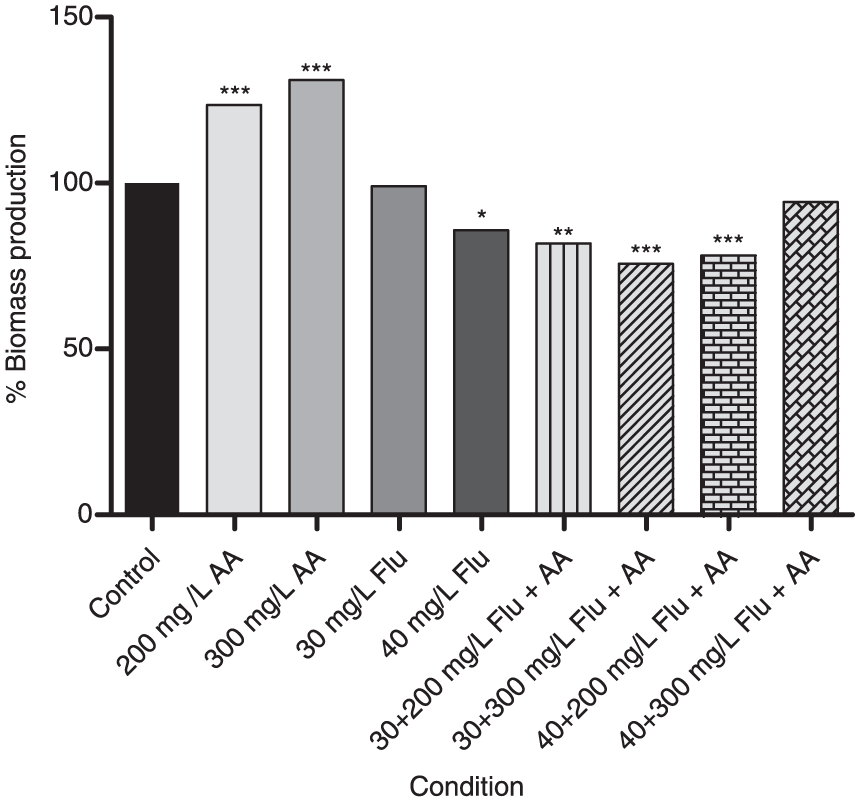
Percentage of biomass detected using CV staining with Flu and Flu + AA in biofilms of
CFUs count (Log10 CFUs/cm2) when using AA and Flu alone in biofilms of
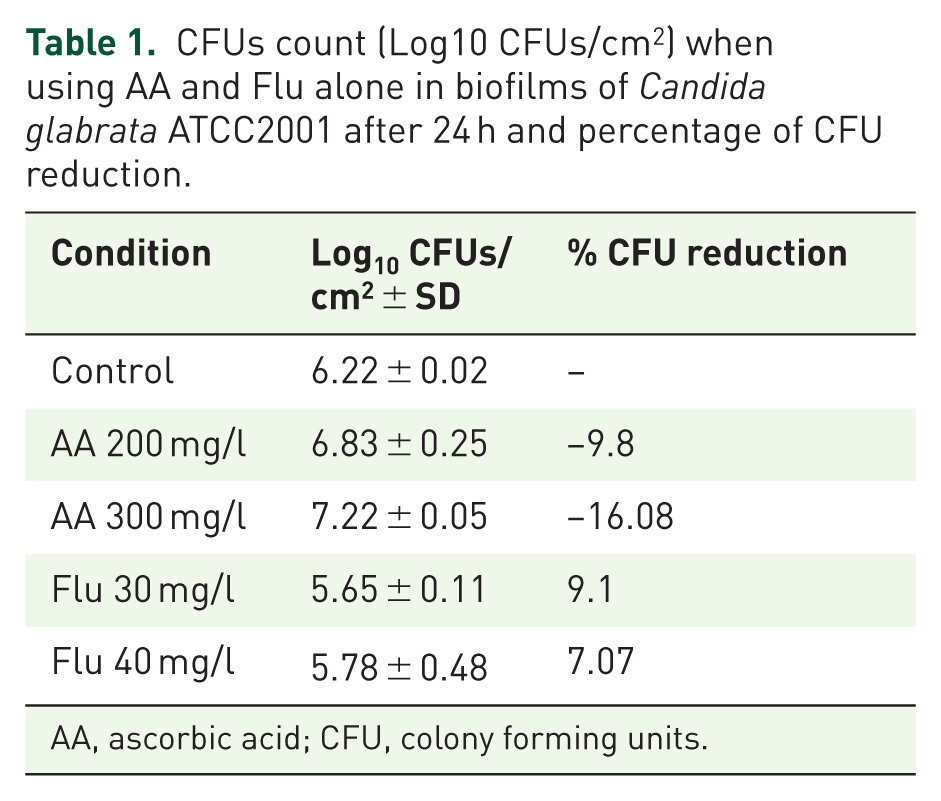
AA, ascorbic acid; CFU, colony forming units.
To verify if the possibility of whether the increased glucose concentration resulting from the β-glucans hydrolysis was contributing to the increase in the
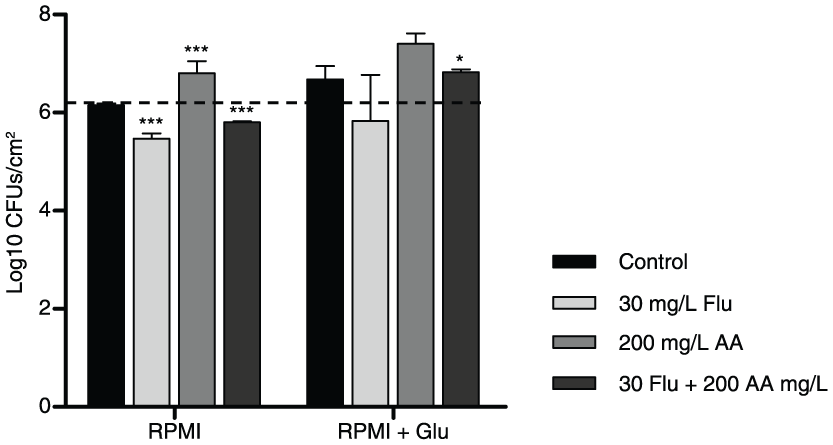
Cell production with Flu and Flu + AA in biofilms of
Lastly, concerning the drug combination (30 mg/l Flu + 200 mg/l AA), results show that the presence of glucose (Figure 2) in the medium was clearly harmful, since there was no variation in CFU count (thus, no cell death) comparing with the respective control. Additionally, when comparing this result with the control without glucose, there was an increase in cell population (Figure 2).
Discussion
Mucositis associated to erythematous ulcerations in the oral cavity causes pain, xerostomia, dysphagia, and lastly septicaemia. 49 It disturbs functions such as drinking, eating, speaking, dental and other mouth care practices, and it affects not only nutrition and quality but also can be life threatening.23,49
Despite the recognized resistance profile of
It was verified (Figure 1) that AA alone had no effect on the reduction of the biofilm biomass, compared with 30 mg/l of Flu, and it showed an increase in the growth of
The chemical events underlying this process were explained through the β-glucan degradation pathways.55–58 Thereby, there are, mainly, two paths: (1) the oxidative cleavage of β-glucan is initiated by the removal of a hydrogen atom from the anomeric carbon (C1) of the polysaccharide inducing the formation of an alkyl radical, and there are two possible courses for the cleavage of the glycosidic bond with the generation of a lactone in C1. The glycosidic bond may fragment due to delocalization of the unpaired electron, leading to the release of a β-glucan fragment with a lactone, and of a β-glucan fragment with an alkyl radical, which may react with O2 to form the corresponding peroxyl radical, which can further undergo transformations. Also, the glucan with an alkyl radical on the C1 can also suffer hydrolysis, which would point to the release of a non-radical β-glucan fragment (glucose monomers) and a β-glucan fragment containing a radical at C1; (2) the formation of peroxyl radical following the alkyl radical in C1. The carbon-centered radical in C1 would react rapidly with O2 to give a peroxyl radical, which can further combine with another peroxyl radical and fragment
In fact, a loss of viscosity in the biofilm was observed during the biofilm manipulation whenever AA was used. The biofilm was more flexible and easily breakable, which is related to the formation of hydroxyl radicals during β-glucan degradation.44,57,60,61 Actually, this glucose hydrolysis reaction is used as an alternative method to the DuBois et al. 62 for carbohydrate quantification.63,64
In the concomitant use of Flu and AA (30 mg/l Flu + 200 mg/l AA), the addition of glucose to the RPMI was very unfavorable. No cell death was achieved (there was no variation in the CFU count), compared with the respective control. In fact, there was even a small increase in cell population (Figure 2) because of the consumption of the free glucose derived from the hydrolysis of the β-glucans and/or the supplementation of the medium, which disrupted the Flu fungistatic activity.
To conclude, Flu is a drug that is significant in the treatment of mucositis. Although with certain side effects, it is generally well tolerated, it has low toxicity, and the usual therapeutic regimen is very appealing to the patient. Unfortunately, once more, the final data presented indicate that this drug might not work for infections derived from
AA leads to hydrolysis of β-glucan which forms glucose that is later used as carbon source by
Footnotes
Acknowledgements
The authors thank the Project ‘BioHealth – Biotechnology and Bioengineering approaches to improve Programa Operacional Regional do Norte’ (ON.2 – O Novo Norte), QREN, FEDER. The authors would also like to thank Pfizer(R) for the kind donation of Fluconazole.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Programa Operacional, Fatores de competitividade – COMPETE and by national funds through FCT – Fundação para a Ciência e a Tecnologia on the scope of the projects FCT PTDC/SAU-MIC/119069/2010, RECI/EBB-EBI/0179/2012 and PEst-OE/EQB/LA0023/2013 and Célia F. Rodrigues’ SFRH/BD/93078/2013 PhD grant.
Conflict of interest statement
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
