Abstract
Objective
Persons with chronic pain suffer from additional symptoms and deficits that are directly or indirectly related to their condition. Attentional deficits are a common problem in these individuals. The same is true for sleep disturbances. It is well-known that night sleep affects attention the next day. Therefore, sleep disturbances might be responsible for the association between chronic pain and deficits in attention.
Methods
We studied the gaze behaviour (absolute and relative fixation times, i.e., attentional biases) in patients with chronic musculoskeletal pain (N = 20) and pain-free individuals (N = 28). For that purpose, we used an eye-tracker to monitor gaze behaviour during the presentation of affective stimuli, that is, pictures of faces displaying joy, anger, pain, and neutral expressions. In addition, we assessed subjective/wellbeing parameters, as well as objective sleep parameters with a portable polysomnography (PSG) device during two nights at home, with the aim of testing the mediating role of sleep for the relationship between chronic pain and alterations of attention.
Results
Patients with chronic pain exhibited overall shorter fixation times (on average ∼160 ms shorter fixation times). With respect to the attentional biases, all participants looked longer at the affective faces compared to the neutral ones, independent of group or type of affective stimulus. Several subjective sleep/wellbeing parameters, together with two PSG parameters (sleep efficiency and duration of awakenings), were significantly worse in patients with chronic pain. Despite this, only subjective sleep quality and sleep efficiency mediated the association of chronic pain and attentional processes.
Conclusions
Our findings confirm previous studies showing attentional deficits and sleep disturbances in patients with chronic pain. However, our results only partially support the hypothesis that the relationship between chronic pain and altered attentional processes is due to disturbed sleep. Other mechanisms might be involved, highlighting the need for further studies.
Introduction
Chronic pain is one of the leading causes of long-term disability affecting several aspects of health and wellbeing. 1 Along with the progressive ageing of population, the prevalence of chronic pain conditions has been increasing in the last decades, 2 stimulating research on consequences of chronic pain as well as mechanisms involved in its development and maintenance.
Attentional deficits are among the various comorbid symptoms reported by persons with chronic pain. 3 Experimental studies have shown that persons with chronic pain perform worse and have longer reaction times in tasks assessing selective, sustained, and divided attention.4–8 Attentional biases (preference for one stimulus over the other when they are presented simultaneously) have also been investigated in relation to chronic pain. Studies based on reaction time paradigms (e.g. Stroop task and visual dot-probe task) have often reported a bias towards pain-related stimuli in persons with chronic pain.9,10 Such attentional biases have been interpreted as the result of a hypervigilant state towards pain-related information, which, in turn, has been proposed to play a central role in the development and maintenance of chronic pain.11,12
More recent studies have employed eye-tracking paradigms, as a more direct and continuous measure of attentional processes and gaze behaviour, compared to analyzing accuracy and reaction times in attentional tasks. Findings from eye-tracking studies are mixed, but with a tendency towards ubiquitous effects of preferentially gazing at pain-related words and pictures, which is not dependent on the pain status, as summarized in a recent meta-analysis. 13 Many studies could not detect any significant difference between groups of persons with and without chronic pain either when presenting pain-related stimuli paired with neutral ones or paired with other emotional stimuli.14–18 Still, some other studies showed that persons with chronic pain have a stronger attentional bias towards pain stimuli, either at earlier or later stages of attentional processes, compared to pain-free individuals.19–24 Possible explanations for these mixed results could be, among others, the heterogeneity of stimuli (e.g. words vs images) and of paradigms used (dot-probe together with eye-tracking or only eye-tracking, i.e., free gazing behaviour), and the heterogeneity of individual characteristics (e.g. severity and duration of pain symptoms) of persons with chronic pain included in the studies. 25
To clarify these incongruencies in the literature, the mechanisms behind the co-occurrence of chronic pain and attentional deficits and/or biases should be explored. There is good reason to believe that pain directly disturbs attention because of the disruptive nature of pain.26,27 Thus, attentional differences between patients may be due to individual differences in the type and temporal dynamics of chronic pain. Alternatively, indirect effects on attention transferred via co-occurring disturbances in chronic pain such as those of mood or sleep, which both are affected by chronic pain and known to disturb attention, may play a role. These two factors (and likely others) are equally plausible in mediating the effect of chronic pain on attention. Due to our expertise in sleep research, we studied the role of disturbed sleep as mediator. Sleep disturbances are often reported as a comorbid condition in persons with chronic pain.28,29 Longitudinal studies have shown that the relationship between pain and sleep is likely to be bidirectional, although the influence of pain on sleep appears to be more established.30,31 For instance, pain symptoms have been found to be predictive of the new emergence of sleep disturbances. 32 While sleep plays a central role for health, it is also important for cognitive performance, and several laboratory studies have shown that sleep restriction protocols have detrimental effects on, among others, attentional processes.33,34 Typically, reaction times become longer and performance decreases in participants performing cognitive/attentional tasks after undergoing a sleep deprivation protocol. 35 Not only laboratory, but also clinical studies have underscored the importance of sleep for cognitive performance and in particular for attentional processes. According to a recent systematic review, persons with a sleep disorder perform significantly worse on attentional tasks compared to healthy controls. 36 In summary, research has shown that chronic pain can negatively influence sleep, and that sleep can influence attentional processes. Thus, it is possible that sleep acts as a mediator of the relationship between chronic pain and attentional deficits, as already suggested in previous studies.37,38 Regarding attentional biases towards pain-related stimuli, it remains unclear, whether sleep might play a role. A recent review from 2022 has identified only two studies that have investigated the relationship between sleep and attentional biases in persons with chronic pain. 39 However, attentional biases were self-reported and not objectively assessed with methods such as eye-tracking, thus highlighting the need for more studies.
The aim of this study was therefore to explore the relationship between chronic pain and attention, proposing sleep (assessed both subjectively and objectively) as a possible mediating variable of this relationship. Specifically, we formulated the following research question: Can differences in attentional processes between persons with and without chronic pain be in part explained by differences in their sleep?
Methods
The data collection for this study was part of a series of studies aiming at comparing persons with and without chronic pain in several domains, such as pain, sleep, and attention (e.g. Ref. 40). Here we report the results about attention mediated by sleep. The experimental protocol was approved by the ethics committee of the University of Bamberg, Germany. The study was conducted in accordance with the Declaration of Helsinki. The experimental protocol was not pre-registered (see also the limitation paragraph in the Discussion section).
Participants
Patients with chronic pain (N = 20) were recruited from a specialized chronic pain outpatient unit (Sozialstiftung Bamberg, Bamberg, Germany). To be included in the study, a diagnosis of primary musculoskeletal pain (e.g. neck pain, upper back pain, low back pain, or fibromyalgia) was required. Tension-type headache was allowed as a secondary diagnosis. Patients were excluded if they had another predominant pain disorder, surgical interventions during the last year, or if they suffered from mental or severe affective disorders. Ongoing therapies with analgesic medications were allowed during the study. Pain-free individuals (N = 29) were recruited via local newspapers or advertisement at the University of Bamberg as control group. They were pre-screened via phone call and excluded from the study if they had any diagnosed physical or mental illness, suffered from acute or chronic pain, and took regularly pain medications or any other type of medications. Pain-free participants were also asked to not take any pain medication the day before and the day of each study session. All participants were asked to refrain from drinking alcohol the day before and the day of each study session. All participants received financial compensation for their participation in the study and signed an informed consent.
Study protocol
Participants visited the laboratory a total of four times. The first test session was held at 6 p.m. prior the first polysomnographic (PSG) recording night; participants were expected the following morning at 8 a.m. in the laboratory for the second test session; the same procedure was repeated before and after a second non-consecutive recording night. The PSG recording was performed by use of a portable device at home. The interval between nights varied between 1 and 13 days. During the test sessions in the laboratory, participants completed several psychophysical pain tests (not reported here) and attentional tasks, the latter of which were an eye-tracking paradigm and a dot-probe task, both with affective facial stimuli (affective attentional task). In addition, participants filled out two questionnaires to assess subjective sleep quality and wellbeing. Since the eye-tracking procedure covered a wider attentional time span (2000 ms) compared to the dot-probe task (500 ms) and produced continuous measures of attention, we had at this time already decided to use only eye-tracking procedures in our next studies (starting with Priebe et al., 2021). 18 Moreover, the dot-probe paradigm has been criticized due to poor consistency and reliability, which additionally motivated us to not further analyze the dot-probe data. 41
Measures
Assessment of attentional processing of affective pictures
We used an eye-tracking paradigm (see also Priebe et al., 2015 for more details on the eye-tracking paradigm) 42 to study a) the ability to keep the attentional focus on emotional stimuli and to prevent distraction and b) the attentional bias towards affective stimuli with personal relevance (symbolic representations of pain). Participants sat in front of a computer-screen with their head placed on a chin rest 70 cm away from the screen. They were instructed to look at the pictures that were about to be displayed on the screen. Thirty-two monochrome photographs of affective facial expressions (anger, joy, pain, neutral) extracted from the Montreal Pain and Affective Face Clips 43 were used as stimulus material. Pictures (size: 7.8 × 6.1 cm) were presented in the middle of a 19-inch screen (monitor resolution: 1280 × 1024 pixels) on a black background. Each affective stimulus (anger, joy, pain) was simultaneously presented with a neutral picture (the distance between the two pictures was 4.8 cm). In addition, neutral–neutral picture pairs were also presented as control trials. There were 24 pictures per each affective stimulus. Eye movements were recorded with an Interactive-Mind System, comprising of a Desktop-PC, a 19-in. LED screen, and the monocular eye-tracking-system Eyegaze EdgeTM (LC Technologies, Inc., VA, USA). For stimulus presentation and registration of ocular movements, the system was driven by the software NYAN 2XT (version 2.3.3, Interactive Minds GmbH, Dresden, Germany).
After calibration, the affective attentional task started. First, a white fixation cross appeared in the middle of the screen for 500 ms. Next, two pictures (either affective–neutral or neutral–neutral) were presented simultaneously, left and right to the central fixation point, for 2000 ms. The presentation of the 2 pictures was followed by a black screen (2000 ms) and then the next fixation cross appeared. Participants were presented with a total of 64 paired pictures (i.e. 64 trials). The order of affective categories and side of appearance of the affective pictures at the screen (affective picture left and neutral right or affective picture right and neutral left) were randomized at the beginning of the experiment and then applied in the same order for all participants. Thus, the number of times that the affective pictures were presented right or left was balanced. Moreover, the number of affective pictures for joy, anger, and pain was the same. Together with the calibration time, the task lasted about 6 minutes. Fixations were defined as a period of at least 100 ms where participants’ gaze did not deviate more than 0.7° from the centre of the actual fixation consistent with previous eye-tracking studies. 44 Thus, fixations <100 ms were excluded from further analyses. Regarding very long fixations, no threshold was set. Only fixations on the target area (pictures) were recorded from the software and later analyzed (e.g. fixations on the screen, but outside the target area, were discarded – task instructions not followed). Two variables were extracted for further statistical analyses: absolute and relative fixation times. The absolute fixation times correspond to the accumulated time (in ms) that the participants spent looking at both paired pictures during a total of 2000 ms presentation time (i.e. total viewing time calculated as the sum of all fixation durations of both pictures). For example, an absolute fixation time of 1500 ms would mean that the participants kept their gaze on the designated target area (part of the screen where the two pictures were presented) for a total of 1500 ms. The relative fixation times were calculated as differences between the fixation times of affective and neutral pictures, with positive values indicating longer fixation times for affective pictures, that is, a preference (bias) for these over neutral pictures.
Assessment of objective sleep parameters
Objective sleep parameters were assessed using the PSG portable device SOMNOwatchTM plus EEG6 (SOMNOmedics, Randersacker, Germany). At the end of each of the two evening sessions, participants were prepared for the sleep recordings in the laboratory and then sent home to sleep in a familiar environment. They were instructed to follow their regular sleeping habits. Four EEG-channels (C3, C4, O1, O2), bilateral electrooculogram (EOG; left and right), and two channels of electromyogram (EMG; M. submentalis) were recorded using gold disc electrodes (Grass Technologies, West Warwick, USA). Positioning of EEG-electrodes was based on the international 10-20-system. All electrodes were referenced towards Cz. Before attaching electrodes, skin was cleaned with cleansing gel (Nuprep, Weaver and Company, Aurora, USA) to reduce contact resistance. Electrodes were attached using electrode cream (EC2 Electrode Cream, Grass Technologies, West Warwick, USA) and fixed using a piece of mull and a plaster. PSG recordings were analyzed according to the standard PSG protocol. 45 First, the DOMINO light software (SOMNOmedics, Randersacker, Germany) automatically scored sleep and wake stages in epochs of 30s, then a visual inspection was performed to check whether the automatic analysis performed correctly according to the Rechtschaffen and Kales criteria. In case of discrepancy, the automatic analysis was overruled, and stages were rescored. The following sleep parameters were extracted from the PSG recordings: Time in bed (TIB), total sleep time (TST; defined as the time between ‘lights off’ and ‘lights on’ without sleep onset latency and time spent awake), sleep efficiency (SE; total sleep time/time spent in bed after sleep onset * 100%), sleep onset latency (SL; time from ‘lights off’ to the first appearance of non-REM stage 2), total number and total duration of awakenings as well as durations of rapid eye movement sleep (REM sleep), non-REM stages 1 and 2, and slow wave sleep (SWS; non-REM stages 3 and 4).
Assessment of subjective sleep and wellbeing parameters
Subjective sleep quality was assessed with the Pittsburgh Sleep Quality Index (PSQI 46 ) after the first morning session. The overall index was calculated (scores ≤5 indicate good sleep). Four additional items 47 were used to assess subjective sleep and wellbeing after each PSG recording night. On a 5-point scale (ranging from ‘very restful’ to ‘not restful at all’) participants evaluated the restfulness of their night of sleep; on 6-point scales participants reported their mood (ranging from ‘depressed’ to ‘untroubled’), freshness (ranging from ‘run down’ to ‘refreshed’), and tension (ranging from ‘tense’ to ‘relaxed’).
Statistical analyses
The sample size was estimated using G*Power based on previous studies run by the same research group with similar patients, which found moderate to strong effects when comparing persons with and without chronic pain in several domains (test family: t test; effect size d = 0.8; α = 0.05, power = 0.80, total sample size = 52).18,40 Statistical analyses were performed using R software (R version 4.4.0 48 ). Figures have been prepared in R software using the package ‘ggplot2’. 49 We first performed linear mixed models to compare attentional processes and sleep characteristics between persons with and without chronic pain. We then performed a multilevel mediation analysis to investigate the role of sleep as potential mediator of the relationship between chronic pain and attentional processes.
Linear mixed models
Linear mixed models (R packages ‘lme4’ 50 and ‘lmerTest’ 51 ) with random intercepts (ID) and slopes (time of day: morning/evening and experimental session: day 1/day 2) to model the repeated nature of the data were used to analyze the eye-tracking variables. We first compared the absolute fixation times between the two groups (‘group’ was added to the model as fixed factor) to investigate whether attentional processes – independent of the type of affective stimulus – differ between persons with and without chronic pain (model 1). We then compared the relative fixation times between the two groups (‘group’ was added to the model as fixed factor) to investigate whether attentional biases (here preference for the affective stimuli over neutral ones) differ between persons with and without chronic pain and whether there is any difference also depending on the type of affective stimulus (‘affect’ was also added to the model as fixed factor with three levels: joy, anger, and pain) (model 2). Linear mixed models with ID as random factor were also used to compare the subjective/wellbeing sleep parameters and the PSG-derived sleep parameters (two nights) between persons with and without chronic pain (‘group’ was entered in the model as fixed factor) (PSG-derived sleep par: models 3–13; subjective/wellbeing sleep parameters: models 14–18). The Bonferroni–Hochberg correction for multiple testing was applied using an online tool, 52 separately for models 3–13 in which 11 PSG-derived sleep parameters were compared between groups and for models 14–18 in which 5 subjective sleep and wellbeing parameters were tested between groups. Partial eta squared (η2p) is reported as measure of effect size for main effects in all models. Age and sex (see the Sample Characteristics section) were not significantly associated with any of the dependent variables (all p-values >0.05) and therefore were not included in any of the models as covariates (also in the mediation models described below).
Mediation models
To test whether sleep characteristics mediate the association between group (persons with and without chronic pain) and fixation times, we performed a multilevel mediation analysis with group as independent variable, fixation times as dependent variable (fixation times were averaged across affective stimuli and were clustered within individuals with ID as random factor), and sleep characteristics (only the parameters that emerged as significantly different between the two groups) as mediating variables using the R package ‘bmlm’.53,54 The Markov Chain Monte Carlo (MCMC) method with 10000 sampling iterations to estimate the posterior distribution of the model parameters was used. Direct and indirect effects with 95% credible intervals (CI) not including zero were considered significant. Given that sleep quality (PSQI) had been measured just one time, we performed a simple mediation analysis for this variable using the PROCESS macro for SPSS. 55
Results
Sample characteristics
The control group consisted of 14 females and 15 males, while the chronic pain group consisted of 14 females and 6 males, which was to be expected given the higher prevalence of chronic pain in women. 56 The two groups did not significantly differ in terms of age (persons with chronic pain: M = 47.25 years, SD = 9.02, range = 25–62 years; healthy controls: M = 44.69, SD = 9.01, range = 29–59 years; t47 = −0.977, p = .333). Patients with chronic pain had the following diagnoses: neck pain (2), low back pain (8), upper and low back pain (1), neck and upper back pain (3), neck and low back pain (2), neck, upper, and low back pain (1), and fibromyalgia (3). The duration of the chronic pain varied between participants: 6 months (1), between 6 months and 1 year (3), between 1 and 2 years (4), between 2 and 5 years (5), and longer than 5 years (7). Two patients were not taking any medication, and the rest were taking the following medications: analgesics (e.g. nonsteroidal anti-inflammatories, nonopioid analgesics, opioid analgesics, and muscle relaxant) (17) and antidepressant (2). One of the participants in the control group had markedly longer fixation times, especially for the affective face displaying joy (more than 3 SD from the average of the control group), and we thus decided to exclude this participant from all further analyses (i.e. sample size for the control group N = 28).
Comparison of absolute and relative fixation times between groups
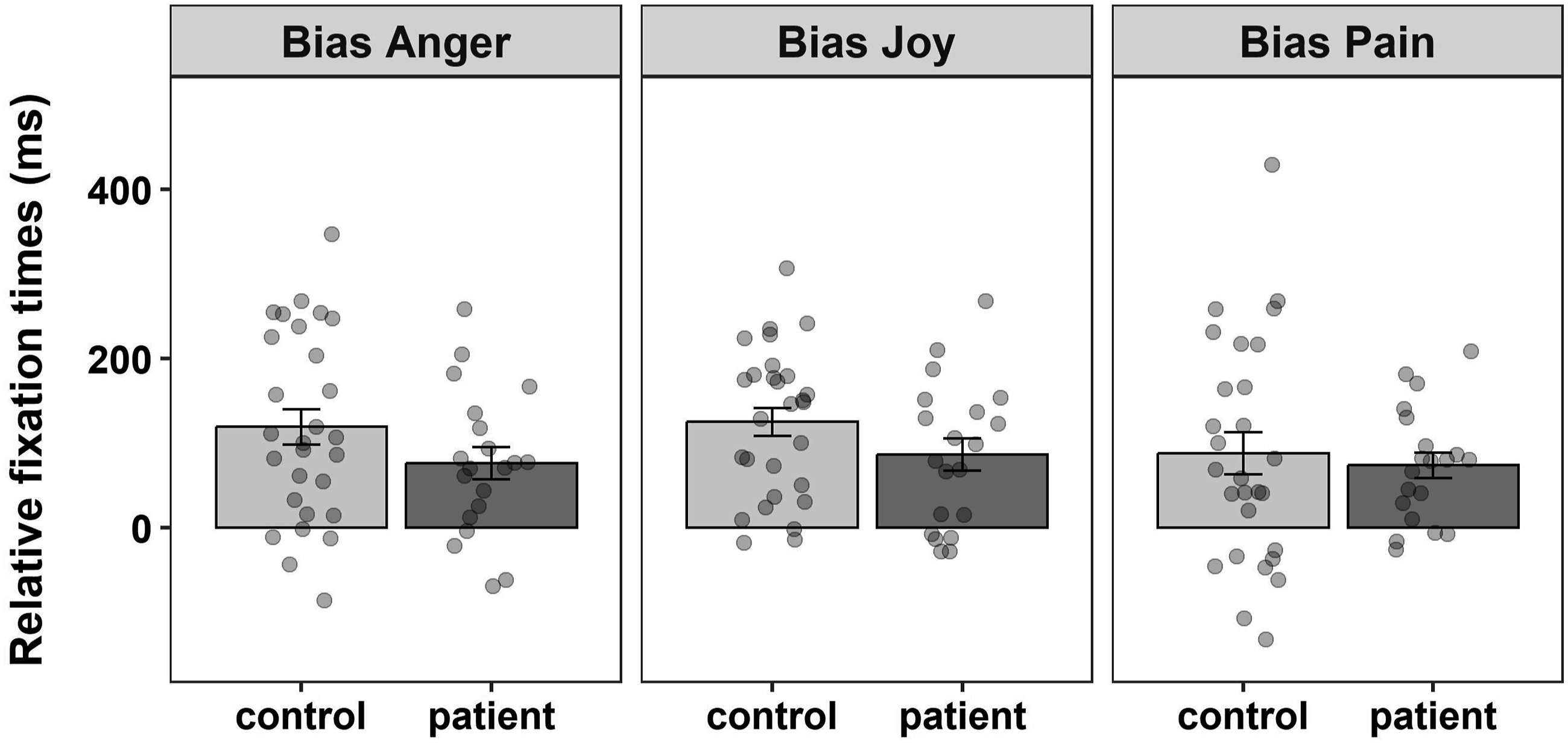
Patients with chronic pain showed overall shorter fixation times (on average ∼160 ms; F1,46 = 5.822, p = .020, η2p = .11; Figure 1). Attentional biases were calculated by subtracting the fixation times of the neutral stimuli from the fixation times of the affective stimuli. Positive values indicate a bias (preference) towards the affective rather than the neutral stimuli. Persons with and without chronic pain both fixated longer the affective stimuli, that is, showed a bias towards affective stimuli, which did not significantly differ between groups (F1,46 = 2.361, p = .131, η2p = .05). The attentional bias did not differ across affective stimuli (F2,428 = 1.588, p = .206, η2p < .01; Figure 2). The interaction effect between group and affective stimulus was also not significant (F2,428 = 0.592, p = .554, η2p < .01). Absolute fixation times by group. Bar plots represent the average absolute fixation times (in ms ± standard error of the mean), that is, how long the participants looked at either one of the paired pictures (affective and neutral) during the 2000 ms presentation time, separately for the control and the chronic pain group (control group: 1198.2 ± SD 237.7; chronic pain group: 1037.3 ± SD 322.6). Data points represent the average absolute fixation times across trials for each participant. *p < .05. Relative fixation times by group and affective stimuli. Bar plots represent the relative fixation times (in ms ± standard error of the mean) for the three affective stimuli, separately for the control and the chronic pain group (control group: anger (119.2 ± SD 111.8), joy (125.2 ± SD 87.8), pain (88.0 ± SD 132.8); chronic pain group: anger (76.5 ± SD 85.4), joy (86.3 ± SD 85.3), pain (73.8 ± SD 67.4)). Positive relative fixation times indicate a bias for affective pictures (i.e. pictures displaying affective facial expressions are fixated longer compared to neutral ones).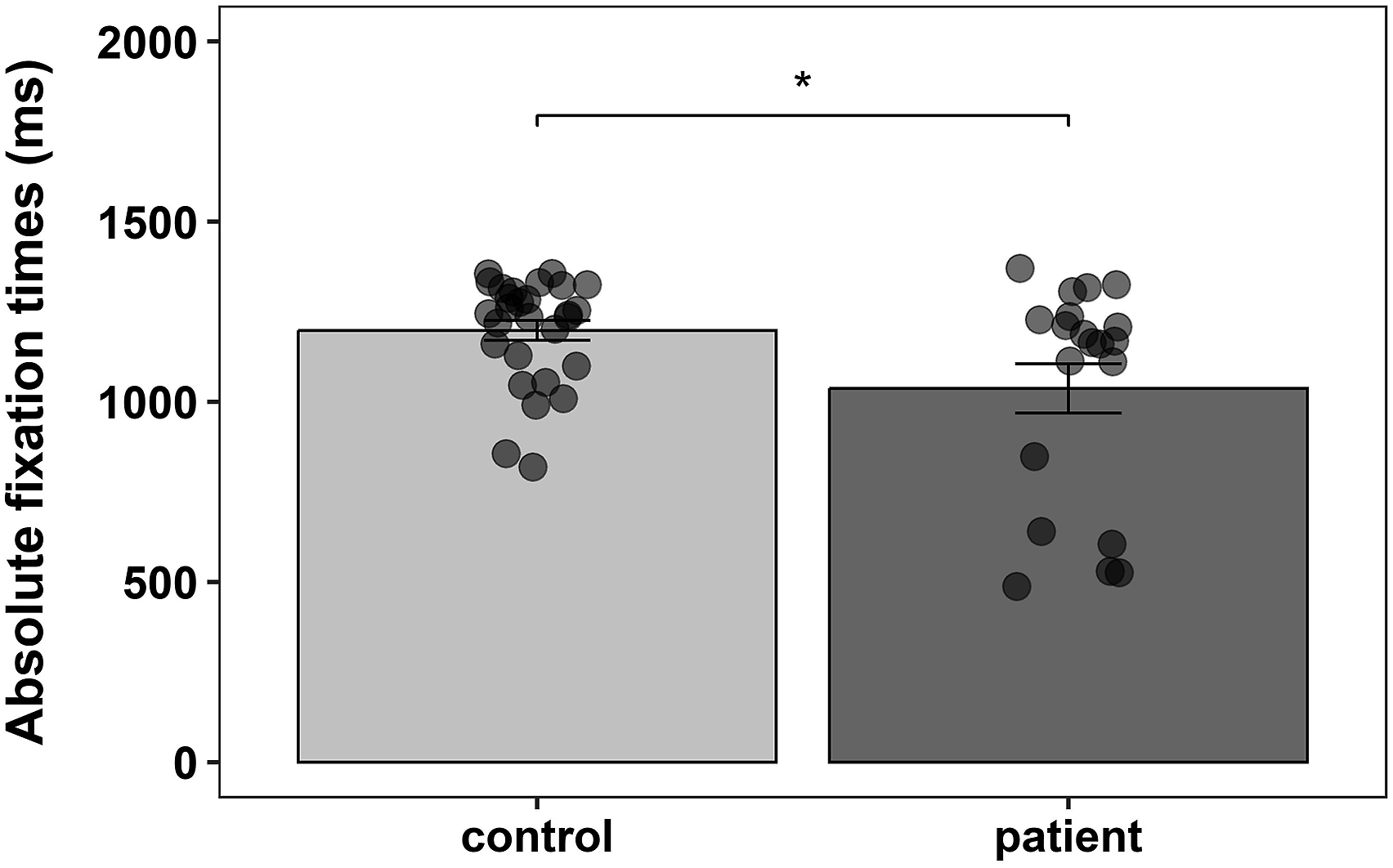
Comparison of PSG and subjective sleep/wellbeing parameters between groups
Means and SD for objective and subjective sleep/wellbeing parameters in the control and chronic patient group.
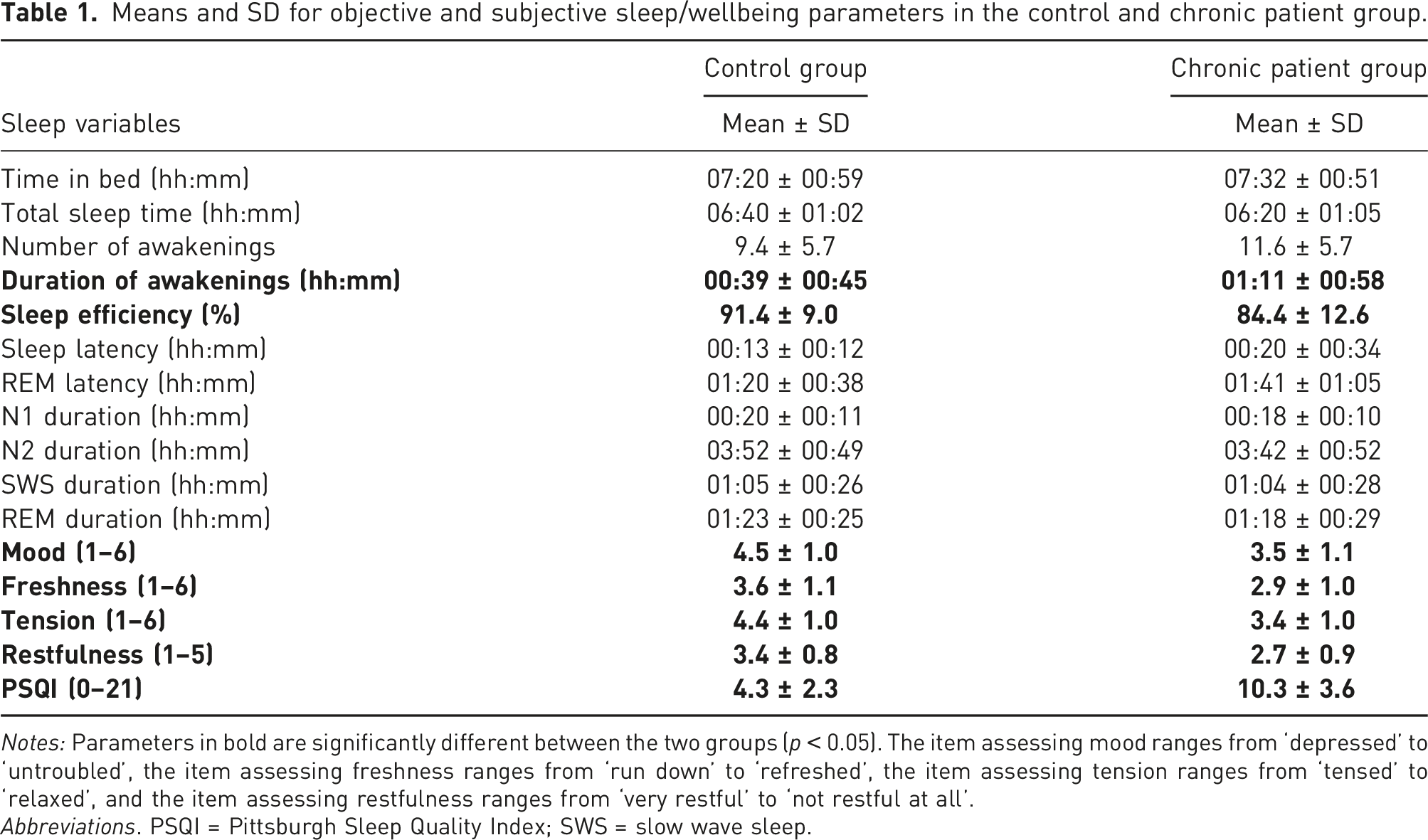
Notes: Parameters in bold are significantly different between the two groups (p < 0.05). The item assessing mood ranges from ‘depressed’ to ‘untroubled’, the item assessing freshness ranges from ‘run down’ to ‘refreshed’, the item assessing tension ranges from ‘tensed’ to ‘relaxed’, and the item assessing restfulness ranges from ‘very restful’ to ‘not restful at all’.
Abbreviations. PSQI = Pittsburgh Sleep Quality Index; SWS = slow wave sleep.
The subjective variables, on the contrary, were all significantly different between groups, with patients with chronic pain reporting to feel more depressed (F1,46.205 = 14.053, p < .001, η2p = .23), more tensed (F1,46.039 = 14.602, p < .001, η2p = .24), less refreshed (F1,46.094 = 9.003, p = .004, η2p = .16), and less rested (F1,45.199 = 12.011, p = .001, η2p = .21) after waking up. Sleep quality (assessed with the PSQI) was also lower in the chronic pain group (F1,46 = 48.767, p < .001, η2p = .51).
Mediation analysis of the relationship between group and fixation times via objective and subjective sleep/wellbeing parameters
After establishing that persons with and without chronic pain differed in several objective and subjective sleep/wellbeing parameters, we explored whether these parameters were mediators of the relationship between group and fixation times. We thus performed seven multilevel mediation analyses with group as independent variable, sleep efficiency, duration of awakenings, mood, freshness, tension, restfulness, and sleep quality (PSQI) as mediators and absolute fixation times as dependent variable. We did not perform a mediation analysis with relative fixation times (i.e. attentional bias) as dependent variable because we did not find a significant effect of group on attentional bias (F1,46 = 2.361, p = .131, η2p = .05). In the first model with sleep efficiency as mediator, the total estimated effect of group on absolute fixation times was −164.6 (95% CI [−325.8, −3.5]), indicating that fixation times in patients with chronic pain were on average ∼160 ms shorter (as previously described). When accounting for the effect of sleep efficiency on absolute fixation times, the group direct effect was reduced to −68.3 (95% CI [−229.8, 91.9]) and became non-significant. The indirect effect (mediated by sleep efficiency) was significant (−96.2; 95% CI [−190.1, −23.5]; Figure 3(a)). Subjective sleep quality emerged also as a significant mediator of the relationship between group and fixation times (−143.4; 95% CI [−322.1, −19.2]; Figure 3(b)). In all the other models, the total estimated effect of group on absolute fixation times was reduced when accounting for the mediating variables, but the mediating effects were all non-significant (duration of awakenings: −31.6 (95% CI [−132.9, 61.1]); mood: −17.7 (95% CI [−70.1, 27.4]); freshness: −19.9 (95% CI [−68.4, 15.0]; tension: −0.6 (95% CI [−64.2, 61.9]; restfulness: −32.7 (95% CI [−90.6, 7.3])). Path diagram for multilevel (sleep efficiency) and simple (sleep quality) mediation analysis. A) Path diagram of the effect of group (control group as reference) on absolute fixation times mediated by sleep efficiency estimated from PSG. Point estimates (posterior means) of the parameters and associated 95% CI are reported. B) Path diagram of the effect of group (control group as reference) on absolute fixation times mediated by subjective sleep quality assessed with the PSQI. Coefficients with bootstrapped 95% CI are reported. Effects with 95% CI that do not contain zero are statistically significant. Notes: a = effect of X on M; b = effect of M on Y; c’ = indirect effect of X on Y mediated by sleep variables.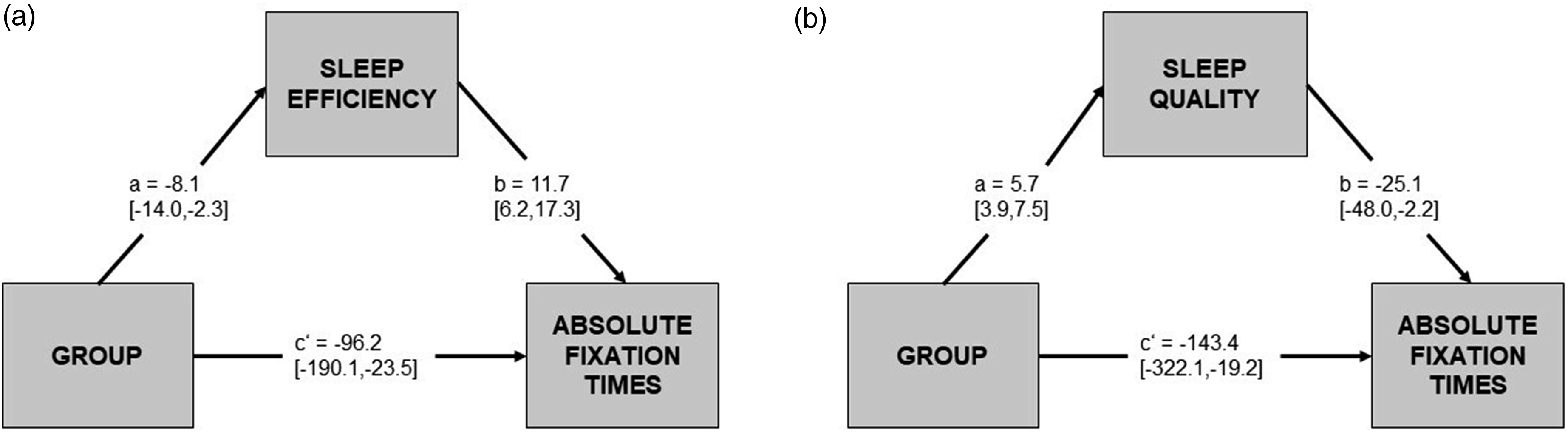
Discussion
Persons suffering from chronic pain are often impaired and debilitated in their everyday life, not only because of their chronic pain condition but also because of co-occurring additional deficits, symptoms, and/or psychological and medical comorbidities. 57 Alterations of attentional processes are common in patients with chronic pain and have been previously studied.13,25,58,59 In the present eye-tracking study, we found that attentional processes (i.e. gaze behaviour) of patients suffering from chronic pain differed from those of pain-free individuals. The patients had overall absolute shorter fixation times (on average 160 ms shorter in an interval of 2000 ms). The group effect was medium to large (η2p = .11). Attentional deficits in individuals with chronic pain have been already reported both in reaction time paradigms as well as eye-tracking studies.4–8 Our results therefore provide additional evidence that attentional processes are altered in persons with chronic pain. It is important to note that we used picture of faces as stimulus material, and thus, our findings are specific to attentional processes of this type of stimuli and cannot be generalized to other stimuli (e.g. neutral abstract stimuli).
The possible mechanisms to explain attentional deficits and/or alterations in chronic pain have not been extensively explored yet. Pain could act as direct distractor during attentional tasks. 26 However, certain patients with chronic pain suffer episodically from pain attacks like in migraine and show attentional deficits even outside the attacks. 60 Furthermore, attentional deficits have shown to persist also under analgesia, at least in animal models, 61 suggesting that other factors might play a role. In our study, we wanted to explore the role of sleep as mediating variable of the relationship between chronic pain and attentional deficits. We chose to investigate sleep because of the ample evidence regarding the detrimental effects of sleep restriction and deprivation on cognitive performance33,35 and because sleep disturbances are a common comorbid symptom in chronic pain. 28 In our sample of patients with chronic pain and pain-free participants, subjective and objective sleep/wellbeing parameters were assessed.
We found significant group differences for some objective and all subjective sleep variables assessed. Patients with chronic pain had lower sleep efficiency and spent more time awake after sleep onset. They also reported worse sleep quality, worse mood, feeling less refreshed, less rested, and more tensed. This finding is in line with many previous studies, reporting sleep disturbances as well as subjective complaints in chronic pain.28,62,63 A meta-analysis by Mathias et al. (2018) 63 showed that sleep efficiency and duration of nocturnal awakenings were among the PSG variables producing the strongest effect sizes for the difference between persons with and without chronic pain. In our mediation models, sleep efficiency (but not duration of awakenings) and subjective sleep quality significantly mediated the relationship between group and fixation times. In all the other models, the total estimated effect of group on fixation times was reduced, when accounting for the sleep-mediating variables, but the sleep-mediating effect was not significant. Since only a subset of sleep variables were significantly different between groups and since not all of them were significant mediators, it is likely that persons with and without chronic pain might differ in other additional characteristics that could further explain the relationship between chronic pain and attentional deficits. For instance, depressed mood might also play an important role, given that it is a common comorbid symptom in chronic pain,64–66 it is associated with sleep disorders, 67 and it can affect cognitive functioning. 68 We did not find a significant mediating effect of mood, but we assessed mood with just one item. Thus, future studies could explore the relationship between chronic pain, mood, and attentional processes using a more detailed assessment of mood.
Another possibility for the non-ubiquitous sleep-mediating effects is that the differences in sleep characteristics between patients with chronic pain and pain-free participants were not large enough to elicit observable effects. Our patients had a sleep efficiency of 84% (compared to 91% in our control participants), which indicates only slight sleep problems. It could be that sleep needs to be manipulated or drastically disturbed (e.g. sleep deprivation protocols) for meaningful associations with the pain system to appear. 69 Without a sizeable linkage between sleep and pain, sleep cannot qualify as mediator for explaining the relationship of chronic pain and other variables such as – in our case – attention. Future studies could screen persons with chronic pain based on their sleep quality and only recruit participants with clinically relevant sleep problems. If our mediation hypothesis is correct, we would expect first a stronger group effect in terms of attentional deficits and second a more ubiquitous sleep mediation effect (confirmed across various sleep parameters). Another possibility for future studies could be to recruit only persons with chronic pain and to then compare attentional processes between persons with and without sleep problems.
While patients with chronic pain showed a clear general difficulty in keeping gaze at the pictures presented on the screen, indicating a deficit of basal selective attention, significant group differences in terms of attentional biases could not be detected in the present dataset. All participants exhibited a clear preference for those pictures displaying affective faces over the neutral ones. Descriptively, the biases for joy and anger appeared stronger than the bias for pain in the control, but not in the chronic pain group. The patients with chronic pain spent less time looking at any affective state, which could indicate a possible emotion-avoidance regulatory mechanism, similar to what has been already observed in patients with depression.70,71 However, given that we did not find any significant effect (group nor affective stimulus) on the attentional biases, these are only speculations. We also did not find a significant stronger bias towards pain-related stimuli in patients with chronic pain. Although some studies found the opposite,19–24 our finding is in line with several eye-tracking studies that found similar results.14–18 A recent meta-analysis and a review corroborated that patients suffering from chronic pain and pain-free individuals do not differ in their attentional biases towards pain-related stimuli.13,25 One reason might be that, from an evolutionary point of view, pain is a very relevant stimulus. As a result, persons with and without chronic pain both tend to look longer at pain-related stimuli compared to neutral ones. 13 Furthermore, the overall attentional deficits of our patients with chronic pain could have hindered the appearance of a stronger bias towards pain-related stimuli. It may well be that stronger attentional biases might appear, if the patients watched the faces equally long as the pain-free participants.
Limitations
The small sample size might be seen as a limitation. We were in fact only able to detect medium to large effect sizes, highlighting the need for more studies with larger sample sizes. We can offer the following justification: Each participant was studied four times in two morning and two evening sessions (fixed schedule) while in-between two mobile sleep recordings took place at night. Thus, the personal and technical costs as well as the participants’ effort were high. Another limitation concerns the diagnostic heterogeneity of the sample of patients with chronic pain. Although all patients could be classified as suffering from musculoskeletal pain, there was great variation in terms of type of pain and pain duration among the chronic pain patients (as described in the first paragraph of the Results section). Given the small sample size, we decided however to not statistically control for these two variables (the restricted number of degrees of freedom did not allow to reliably estimate the effects of type of pain and pain duration on attention in the model). Another limitation is that our study was not pre-registered, which reduces the transparency of our planned statistical analyses (e.g. possibility of post-hoc changes in the statistical analyses performed) and does not allow to clearly distinguish between exploratory and confirmatory analyses. Finally, our mediation model assumes a causal link between chronic pain, sleep, and attentional deficits. However, given the cross-sectional nature of our design, we can only assume and not prove causality. Future studies with a longitudinal or experimental design are warranted to test the proposed causal link between chronic pain, sleep, and attentional deficits.
Conclusions
Altogether, our data confirm the presence of a perturbation of the attentional system in chronic pain. In the present study, patients with chronic pain clearly showed a reduced ability to keep the attentional focus on the pictures presented on the screen. The attentional bias towards affective stimuli was equally present in both groups and was not modulated by the type of affective stimulus. Typical sleep problems (reduced sleep efficiency, prolonged duration of awakenings as well as subjective complaints) could be found in the patients with chronic pain to a moderate degree. These sleep problems partially explained the attentional deficit of the patients. However, given the partial role of sleep in explaining this phenomenon, future studies are warranted to clarify the relationship between chronic pain and attentional deficits, and to identify possible further mediators involved.
Footnotes
Author contributions
SL conceived the study and obtained ethical approval. SL and MK supervised the project. CS, AK, and PL were involved in patient recruitment, data collection, and data curation. GZ analyzed the data and prepared figures and tables. GZ wrote the first draft of the manuscript. SL, MK, and GZ reviewed and edited the manuscript. All authors reviewed and approved the final version of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was financially supported by Dr Robert-Pfleger-Stiftung Bamberg.
Ethical statement
Data Availability Statement
The data is available from the corresponding author upon request.
