Abstract
Introduction
Regular review of patients prescribed opioids for persistent non-cancer pain (PCNP) is recommended but not routinely undertaken. The PROMPPT (
Methods
Interviews (n = 15) and an online discussion forum (n = 31) with patients prescribed opioids for PCNP and interviews with pharmacists (n = 13), explored acceptability of a proposed PROMPPT review. A prototype PROMPPT review was then tested and refined through 3 iterative cycles of in-practice testing (IPT) (n = 3 practices, n = 3 practice pharmacists, n = 13 patients). Drawing on the Theoretical Framework of Acceptability (TFA), a framework was generated (including a priori TFA constructs) allowing for deductive and inductive thematic analysis to identify aspects of prospective and experienced acceptability.
Results
Patients felt uncertain about practice pharmacists delivering the proposed PROMPPT review leading to development of content for the invitation letter for IPT (introducing the pharmacist and outlining the aim of the review). After IPT, patients felt that pharmacists were suited to the role as they were knowledgeable and qualified. Pharmacists felt that the proposed reviews would be challenging. Although challenges were experienced during delivery of PROMPPT reviews, pharmacists found that they became easier to deliver with time, practise and experience. Recommendations for optimisations after IPT included development of the training to include examples of challenging consultations.
Conclusions
Uptake of new healthcare interventions is influenced by perceptions of acceptability. Exploring prospective and experienced acceptability at multiple time points during early intervention development, led to mini-optimisations of the prototype PROMPPT review ahead of a non-randomised feasibility study.
Introduction
An estimated 43% of UK adults experience persistent non-cancer pain (PNCP), 1 many of whom are prescribed opioids. 2 However, evidence for the long-term benefits of opioids is lacking and their use is associated with adverse side-effects and the risk of serious harm, including addiction. 3 Therefore, regular review is recommended for people prescribed opioids for PNCP to assess treatment effectiveness and, where appropriate, support opioid tapering.4–6 However, implementation of best practice guidance is low7,8 and routine UK General Practitioner (GP) appointments offer limited opportunity for comprehensive opioid reviews. A move to multidisciplinary working is underway in UK primary care, with more pharmacists working in GP practices.9,10 Such practice pharmacists received additional training in patient care and conduct consultations with patients in general practice. Given their skills and knowledge around polypharmacy and complex medicines regimens, practice pharmacists (hereafter pharmacists) seem ideally placed to take a proactive role in reviewing patients prescribed opioids for PCNP, but there is currently no evidence about how they should do this.
The PROMPPT ( Four MRC phases applied to development and evaluation of the PROMPPT intervention.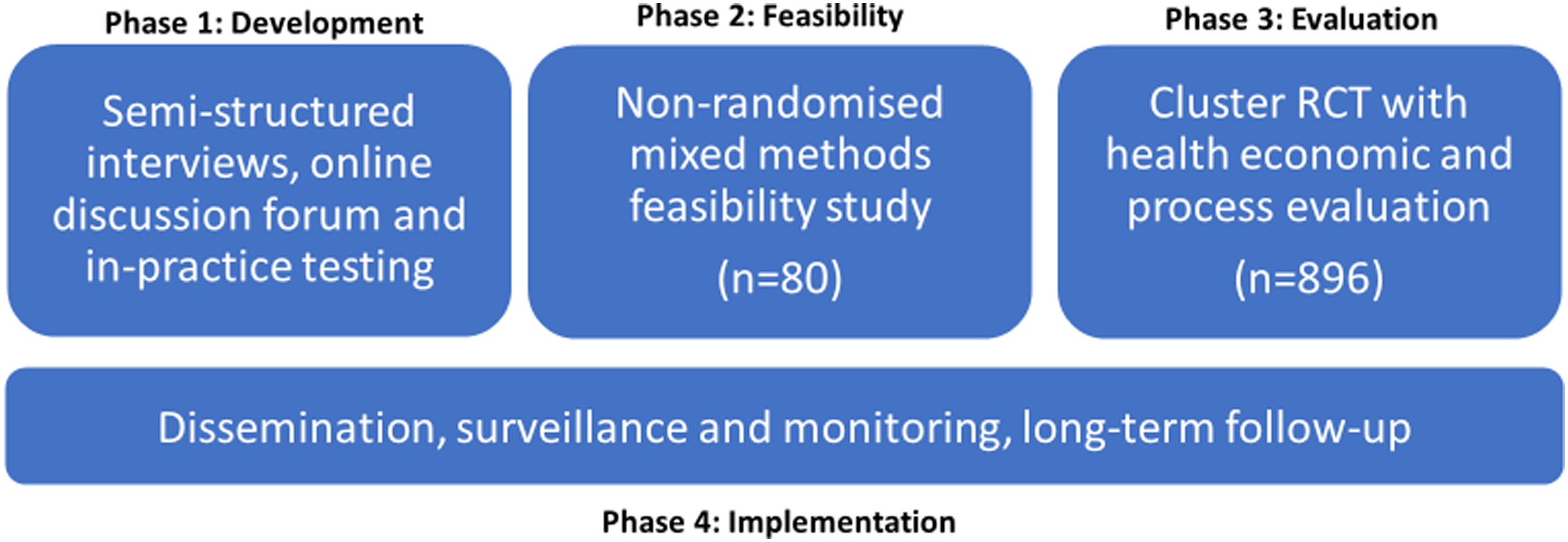
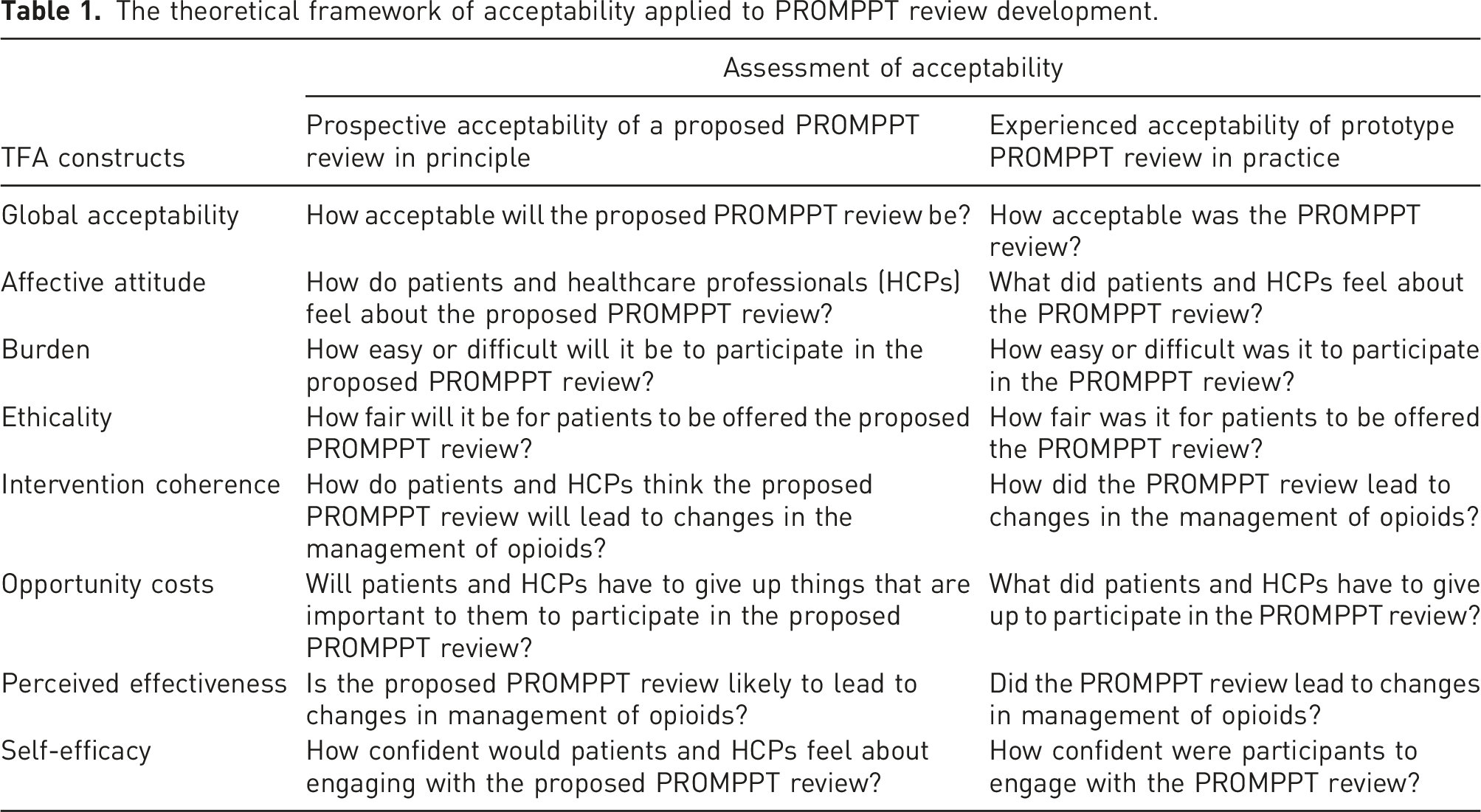
The current study sits in phase one of the MRC framework and focuses on one aspect of intervention development, developing an intervention that is acceptable to those who will use it. Perceptions of acceptability influence intervention uptake by patients and implementation by health care practitioners. However, until recently the construct of acceptability has been poorly defined and understood.12,13 We took a theory-informed approach drawing on the Theoretical Framework of Acceptability (TFA), to explore acceptability of a proposed PROMPPT review (prospective acceptability) and, through in-practice testing, explored acceptability of a prototype PROMPPT review (experienced acceptability). Recommendations for optimising the intervention were made at key timepoints (see Figure 2). Table 1 outlines how we aligned TFA constructs to PROMPPT. Evaluation of acceptability of PROMPPT. The theoretical framework of acceptability applied to PROMPPT review development.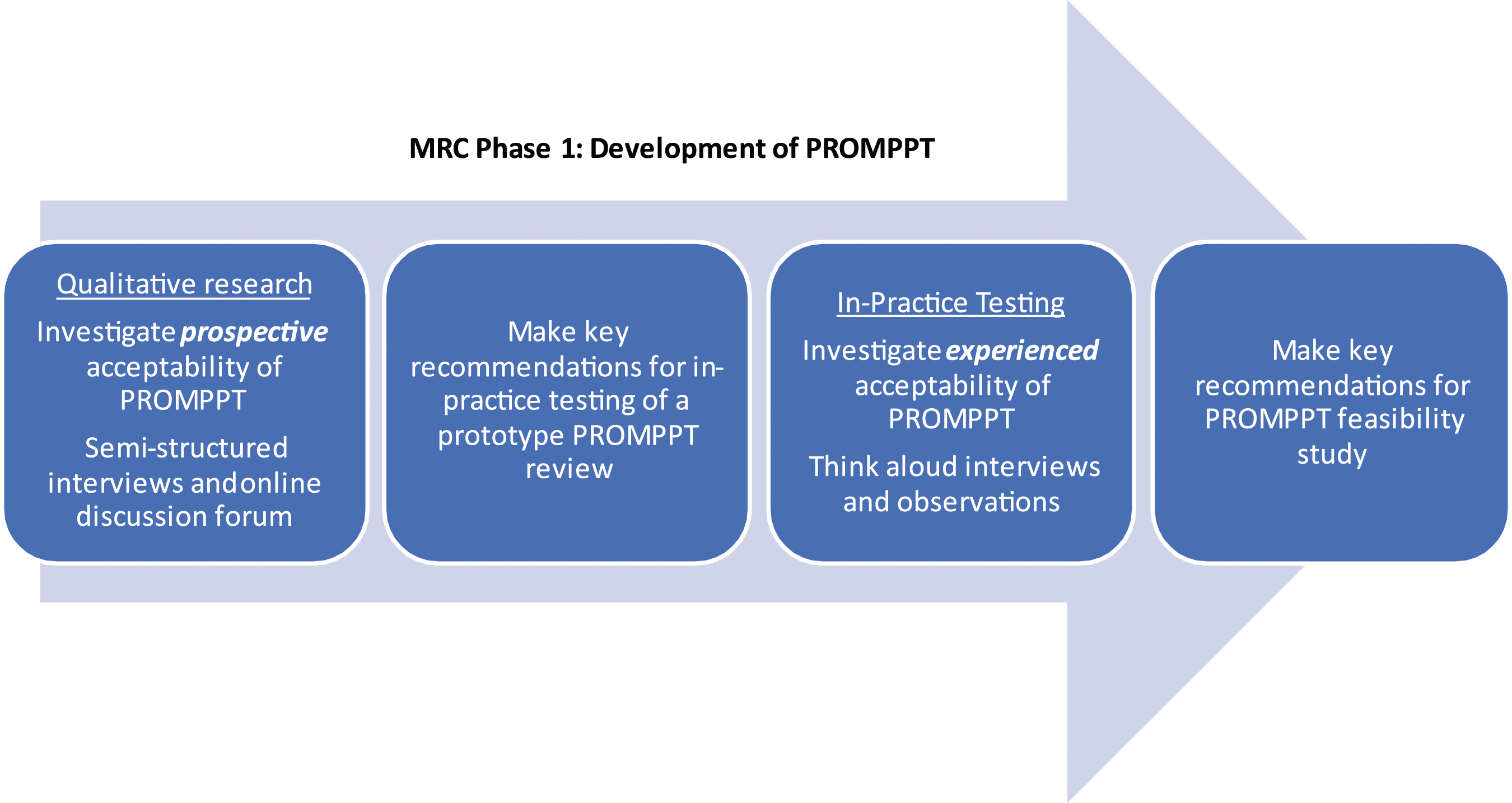
Methods
This study used semi-structured interviews, an online discussion forum and in-practice testing (IPT) with think-aloud interviews, in a UK Primary Care setting (July 2019–February 2020).
Recruitment and conduct
Prospective acceptability
Interviews (August 2019–October 2019)
Adult patients (>18 years) prescribed opioids for ≥6 months for PNCP were recruited, by postal invite, from two West Midlands GP practices without a pharmacist. Semi-structured interviews were conducted face-to-face or by telephone, according to participant preference. A topic guide informed by a patient advisory group included; experiences of long-term pain, using regular medication, experiences of discussing pain management with healthcare professionals, knowledge of pharmacists and thoughts about a proposed review with a pharmacist (supplementary material 1). We also included questions related to theoretical constructs of the TFA.
Pharmacists with experience of consultations with patients in UK General Practice were recruited opportunistically from professional networks. Semi-structured interviews were conducted via telephone or face-to-face using a topic guide, informed by the TFA. Questions included experiences of consulting with patients prescribed opioids for PNCP, thoughts about pharmacists delivering a proposed PROMPPT review and possible components of the PROMPPT review (supplementary material 2).
Interviews were conducted by NC, an experienced qualitative researcher, audio-recorded and recruitment stopped when the interviewer (NC) deemed data saturation had been reached whereby nothing new was being heard from the interviewee responses. 14
Online discussion forum (October–December 2019)
People with experience of using opioids for PNCP were recruited using posters displayed in GP practices, community pain services and community pharmacies across the West and East Midlands and Wessex in the UK, and online by regular posts and paid advertisements using social media. For ease of reading, the term ‘patient(s)’ will be used to represent all the patient and public participants with experience of using opioids for PNCP who took part in this study.
Advertisements directed potential participants to the PROMPPT discussion forum with links to a participant information sheet, electronic consent form and registration form. Upon registration, participants were assigned an anonymous username. To ensure acceptability and accessibility of the discussion forum, software and interface were user tested by members of Keele’s Patient and Public Involvement and Engagement (PPIE) group.
One of 10 topics was released weekly over 11 weeks (supplementary material 3). Facilitation prompts were posted approximately daily to aide discussions.
Findings were discussed with the intervention development team and the PROMPPT stakeholder group and key intervention components were agreed for the prototype intervention ready for in-practice testing.
Experienced acceptability
Pharmacists from three general practices in the West Midlands were recruited to IPT (November 2019 -February 2020), with adult patients (>18 years) prescribed opioids for PCNP, recruited from electronic practice records. Pharmacists attended an afternoon of face-to-face prototype training at Keele University, including practising PROMPPT reviews with simulated patients.
IPT comprised three iterative cycles of delivery, data collection, reflection, and revision of the PROMPPT review. Patients were asked to think-aloud during the review by saying out loud any thoughts or feelings as they came up. Reviews were audio-recorded and observed by two qualitative researchers (NC, CW, TH, SW). Immediately following each review, the patient and pharmacist were interviewed separately by one researcher, using a TFA-informed topic guide (supplementary material 4) and probes related to observing the review. Interviews were audio-recorded. Researchers categorised observations into six categories; visual cues, verbal cues, think-aloud, resources, potential changes required and other things to note.
Interview and observation data from each cycle of IPT were combined with observations highlighting aspects that worked well as well as identifying areas needing revision. Mini-optimisations were then made that were evaluated during the next cycle.
Data analysis
We used the TFA to understand the prospective and experienced acceptability of the PROMPPT review, including discussions about tapering opioids, among patients and pharmacists. To ensure rigour, a phased approach to analysis was adopted. Interview transcripts, including in-practice interviews, were professionally transcribed verbatim, anonymised and checked for accuracy against interview recordings. Online discussion forum posts were downloaded into Microsoft Excel® and forum user IDs replaced with deidentifying codes to further protect participant anonymity.
Transcripts were read and re-read for data familiarisation. A coding manual based on the TFA domains was created and discussed and an initial thematic framework developed using a priori theoretical constructs. Three transcripts were coded independently by a multi-disciplinary team including qualitative researchers, pain specialists and pharmacist (NC, CJ, CW, SH, SW) using NVivo v12 software to aid data management. The team met to discuss data and understanding of theoretical constructs before a second coding phase, followed by another coding discussion meeting. Two coders (NC and CW) then coded all remaining data (including discussion forum data) into the framework and ‘key aspects’ were generated within each TFA domain.
Ethics
Ethical approval was granted by the East of England – Cambridge East Research Ethics Committee (ref:19/EE/0151). Informed written consent was obtained from all participants.
Results
Participant characteristics
Prospective acceptability
Patient interview characteristics.
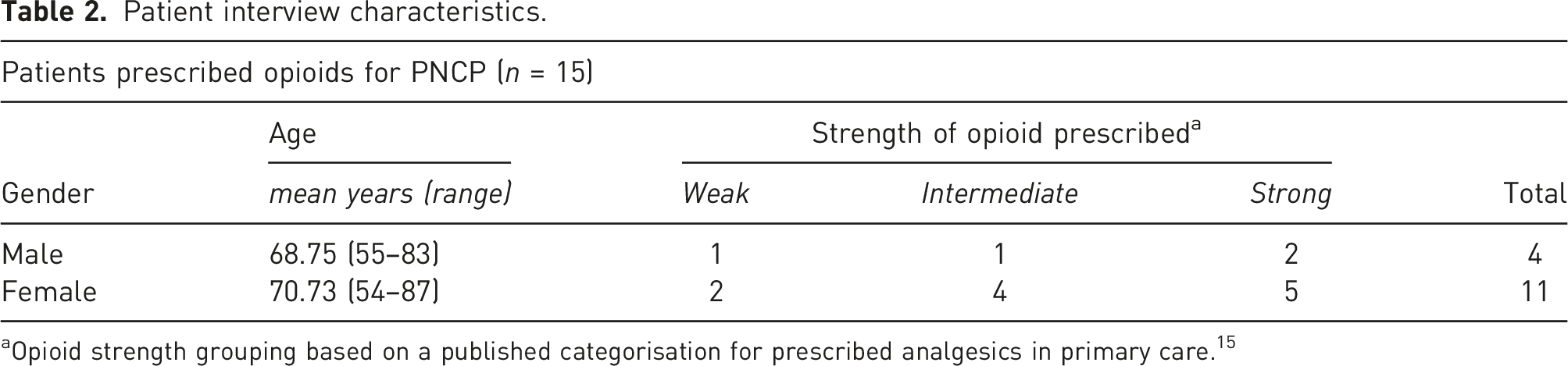
aOpioid strength grouping based on a published categorisation for prescribed analgesics in primary care. 15
The online discussion forum recruited 69 participants, posting 160 comments. As online discussion participants joined and participated anonymously, full participant demographics are unavailable.
Experienced acceptability
In-practice testing (IPT) participant characteristics.
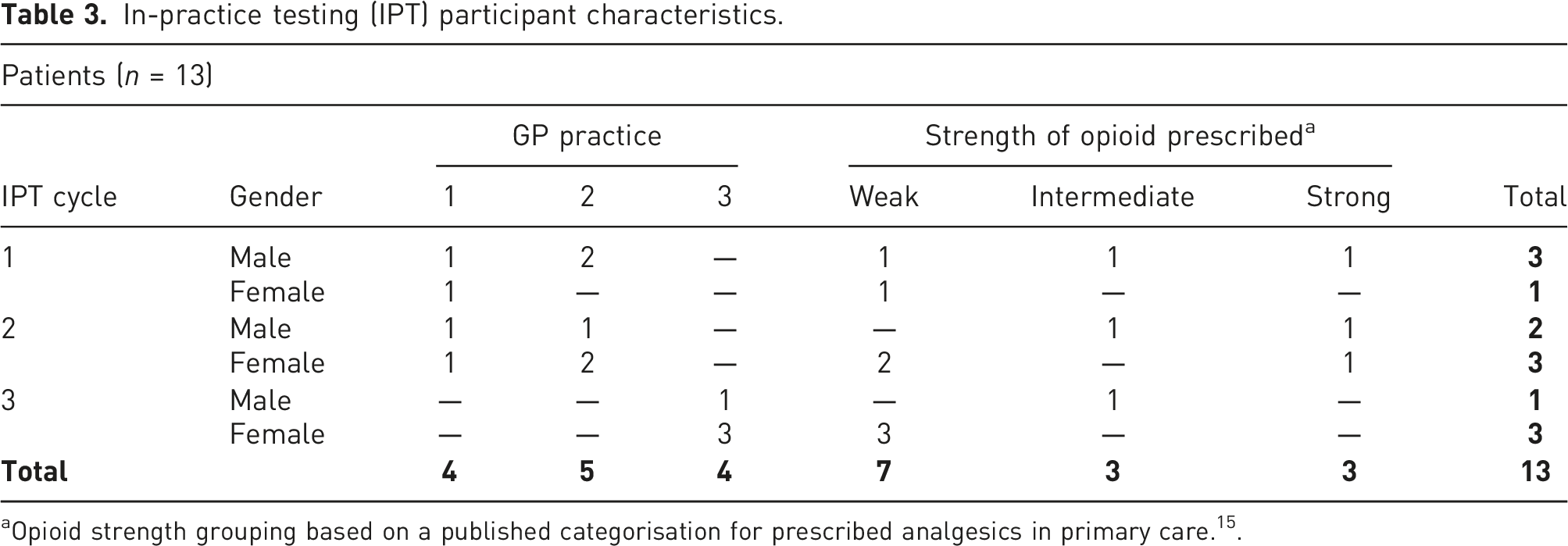
aOpioid strength grouping based on a published categorisation for prescribed analgesics in primary care. 15 .
Acceptability of PROMPPT: Patient perspectives
Summary of patients’ perceptions of acceptability of the PROMPPT review (*denotes aspect identified across all opioid strengths).
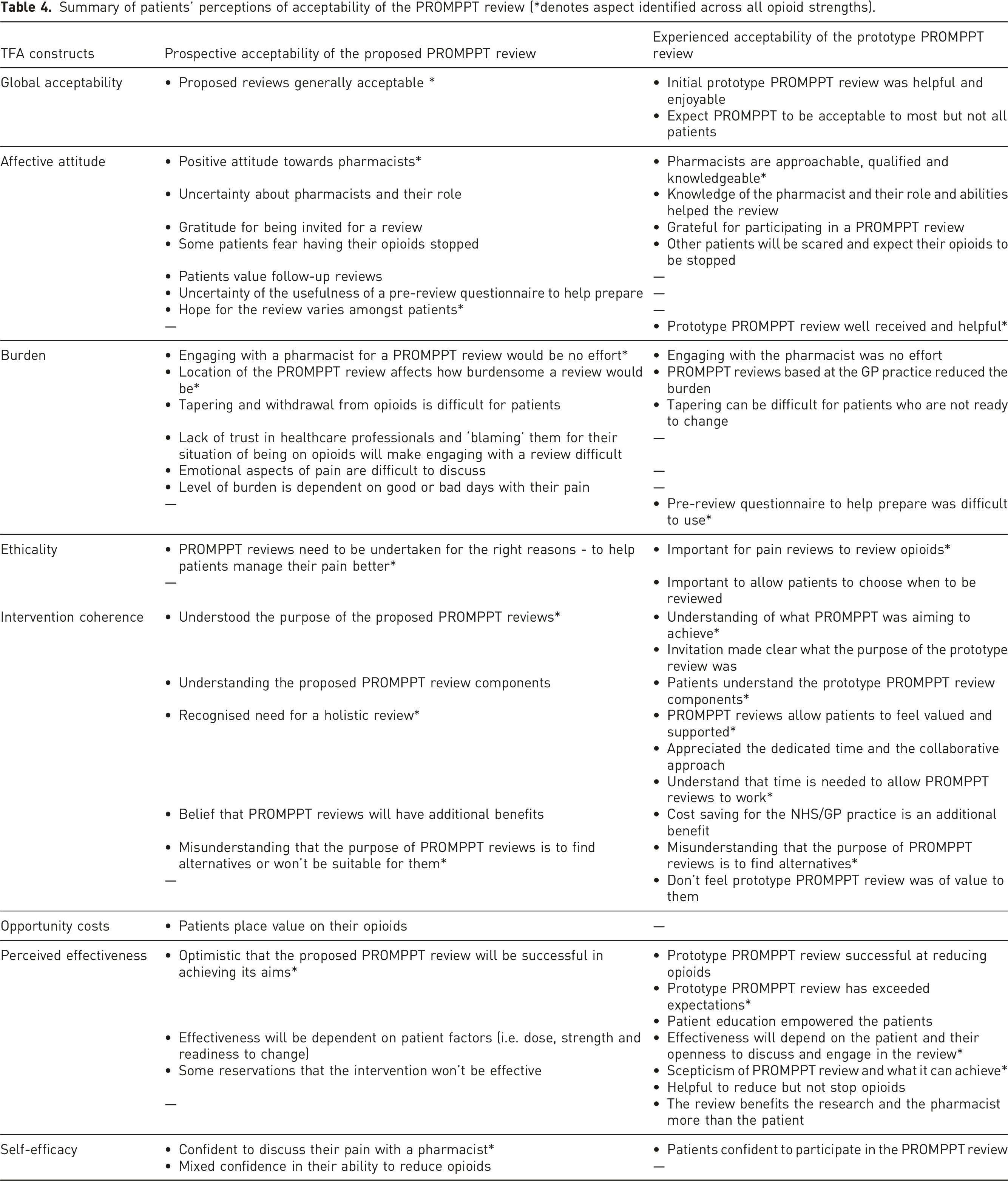
Affective attitude
When discussing their feelings towards the proposed PROMPPT review, patients were generally positive towards a pharmacist delivering the review. Some patients were uncertain of the role of a practice pharmacist, as they had not heard of them or consulted one previously. After experiencing a PROMPPT review, most patients felt that pharmacists were knowledgeable and qualified to deliver a review. Patients with prior knowledge of practice pharmacists and their role seemed more inclined to engage with the pharmacist during the review. ‘I hadn’t heard of a clinical pharmacist until now…if I got to see a regular pharmacist and had plenty of time to discuss my pain and ways to deal with it, I think I’d be happy to see one.’ (Forum participant_67)
Burden
Some patients discussed their lack of trust in healthcare professionals resulting from poor pain management experiences, often blaming them for being dependent on opioids that provide no relief. ‘I’m pretty damn miffed that I’m in an avoidable position here. This is literally prescribed harm.’ (Forum participant_19)
Although this lack of trust and discontent could have affected patients’ willingness to engage with a pharmacist during the review, they found pharmacists were approachable and voiced little effort required to engage with them during IPT.
Ethicality
Overall, it was important to patients that PROMPPT reviews are undertaken for the right reasons, to help patients manage their pain better and not for cost savings. ‘It would also help people to have the purpose and aims of the appointment laid out and what to expect from the discussion - is it just to try to get me off opioids or a genuine desire to get my pain under control to help me - or to help the practice to cut costs, or to make their statistics look good’ (Forum participant_61)
Once patients had experienced the prototype review during IPT, they felt that other patients like themselves should expect to have their opioids reviewed and that, although it would be important to have set reviews, it would also be important to allow patients to choose when they want or need reviews.
Intervention coherence
There were mixed perceptions of the purpose of the proposed PROMPPT review with some patients believing it would be to review prescribed opioids, reduce them where appropriate and support them to live better with pain, whilst others expected to be given an alternative pain medicine to replace opioids. After experiencing the prototype review, some patients still felt that it was not suitable for them as they were only taking a weak opioid or low dose that did not warrant any change. ‘If I was on a lot, I’d want to reduce it but I don’t take, I’m very careful what I do take.’ (IPT patient_5: female/weak opioid)
Once they had experienced PROMPPT, the majority of patients reported that the review left them feeling valued and supported and appreciated the dedicated time and collaborative approach to their pain management.
Opportunity costs
Some patients expressed concern about being invited and attending a review for fear of having their opioids stopped. They spoke of valuing opioids to help them get by day-to-day and feared stopping would impact on their priorities. This concern was not reported by any IPT patients once they had attended a PROMPPT review.
Perceived effectiveness
When discussing the potential of the proposed PROMPPT reviews, patients were generally optimistic that it would be successful, specifically in tapering down opioids, improving their quality of life and ability to manage their pain. However, some felt doubtful that a pharmacist would be able to help when a GP had not helped in the past. Some patients said the prototype review exceeded expectations, but some remained doubtful in how successful it would be in reducing their opioids completely, with a slight reduction seeming more realistic. ‘I mean I would love to, to get rid of them all. Er, I can't see that happening er, in the future, being off them all but it might get reduced which would be good.’ (IPT patient_29: female/weak opioid)
Self-efficacy
Although most patients said they felt confident that they would be able to discuss their pain with a pharmacist, confidence in their ability to reduce opioids was mixed. Patients with tapering experience felt confident about the prospect of engaging in the opioid reduction process and generally reported feeling very confident participating in the prototype review during IPT, allowing them to engage and be open with the practice pharmacist during the review. However, those with no experience expressed uncertainty and nervousness about trying.
Perspectives of prospective and experienced acceptability seemed largely similar regardless of opioid strength used. However, when thinking about the proposed PROMPPT review there were differences in the domains of burden and self-efficacy. Those on strong opioids felt the review would be more burdensome if they were having a bad day and were less confident/more concerned about their ability to reduce. Following IPT those on weak opioids perceived that patients taking higher doses would be scared that their opioids would be stopped (affective attitude). They felt that tapering could be difficult for patients who were resistant to changing opioids (burden) and some felt it may be more realistic to reduce rather than stop opioids (perceived effectiveness).
Acceptability of PROMPPT: Pharmacists’ perspectives
Summary of pharmacists’ perceptions of Acceptability of PROMPPT.
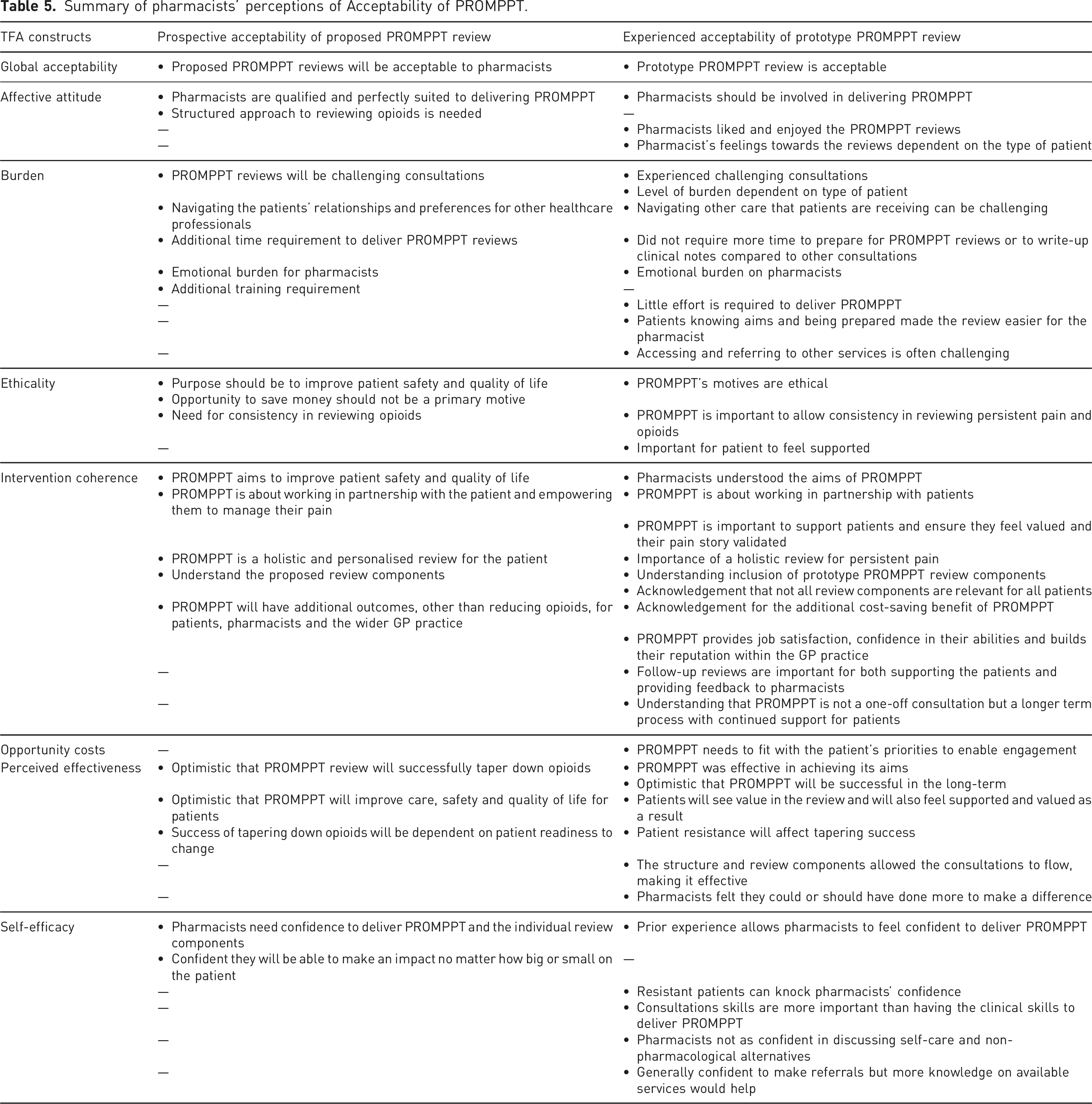
Affective attitude
When considering the proposed new pain review, pharmacists expressed a need for a structured approach to reviewing opioids making them more acceptable to deliver. ‘I think it would be very acceptable because if we’ve got like a structured approach then when we are reviewing these patients, we feel like we’ve done justice to them as well and we’ve done the appropriate review and gone through everything we need to for the safety of prescribing and management and safety for the patients as well.’ (Interview pharmacist_21)
After taking part in IPT, pharmacists expressed how they liked and enjoyed delivering the prototype PROMPPT reviews and confirmed pharmacists’ prospective thoughts that they should be involved in delivering PROMPPT as they are appropriately qualified and experienced.
Burden
Reflecting on their previous experiences of consulting with patients prescribed opioids for PCNP, pharmacists had an expectation that they would have to deal with resistance from some patients to make a change. There was a perception that these patients are often complex, with multiple health and social concerns which make consultations more challenging. Some IPT pharmacists confirmed this expectation when describing the challenging consultations experienced during IPT, with patients often having a different agenda and not wanting to discuss making changes. ‘I think her expectation at that time initially was maybe thinking about more medication or different types of medication, rather than perhaps my agenda which was more of a like trying to reduce the medication. So it was a bit of sort of internal conflict right at the start.’ (IPT pharmacist_3)
Despite these predicted challenges, pharmacists felt that the prototype PROMPPT reviews required no extra effort compared to regular consultations and found they became easier to deliver with practise and experience.
Ethicality
Pharmacists agreed that dedicated review for opioid-treated patients with persistent pain was important, with most saying these patients should be reviewed regularly and consistently to improve patient safety and quality of life. After delivering prototype PROMPPT reviews, all pharmacists said it provided patients the opportunity to be fully informed about their health and access to support.
Intervention coherence
Pharmacists felt the proposed PROMPPT review needed to be a holistic review, aligned with individual patient’s needs, allowing time to discuss pain stories and strategies for living well with pain. They spoke about working in partnership with patients to empower them to manage their pain and their opioids. Reflecting on the prototype reviews after IPT, pharmacists recognised that PROMPPT is not just a one-off consultation but a longer term process and highlighted the importance of follow-up reviews to provide continued support for patients and feedback to pharmacists. ‘the useful bit was getting her back and seeing how it was working, rather than the unknown. You send them off and you don’t have a clue. You hope your plan comes to fruition, but it could’ve just gone completely pear-shaped (IPT pharmacist_2)
Opportunity costs
One of the IPT pharmacists acknowledged the need for the PROMPPT review to fit with the patients’ priorities, to ensure best possible engagement from patients and the best chance of making changes.
Perceived effectiveness
Pharmacists were optimistic that the proposed review would improve patient care. Most expressed confidence about opioid tapering but highlighted that success would depend on the patient’s readiness to change. ‘I think it will be very effective and I think it would definitely help, just purely because of the time that we have and that dedication that we can have’ (Interview pharmacist_21)
After delivering the prototype reviews, pharmacists felt that PROMPPT was effective in tapering opioids and improving patients’ ability to manage their pain and hoped it would improve their quality of life in the long-term.
Self-efficacy
Overall, pharmacists felt they would be confident to deliver the proposed PROMPPT reviews including discussing medicines, having honest conversations and communicating with patients. Creating management plans with patients was a skill some said they felt less confident with and highlighted this to be a training need. Prior experience in doing medication reviews, prescribing and discussing medicines helped pharmacists to feel confident to deliver the prototype review but consulting with patients they perceived as ‘difficult’ or ‘resistant’, negatively affected the pharmacist’s confidence. ‘Started off quite confident and then as soon as I hit the brick wall I felt a little bit like, ‘Oh no, where do I go now?’ So my confidence sort of just took a bit of a knock halfway through.’ (IPT pharmacist_2)
Discussion
This work is timely, given NHS England’s medicines optimisation aim to reduce inappropriate prescribing of potentially dependence-forming medicines. Structured medication reviews (SMRs) are a key component of this medicines optimisation strategy 16 and it is expected that clinical pharmacists working in primary care will lead on and undertake SMRs. 17 The prototype PROMPPT review is consistent with recently published NHS England guidance on optimising care for adults prescribed medicines associated with dependence 18 and includes key recommended components of SMRs, namely: shared decision-making and a personalised approach to exploring the balance of safety and effectiveness of current treatment. 16
We used a novel approach by applying the TFA early in the intervention development pathway, to explore acceptability of the proposed PROMPPT review (prospective acceptability) and the acceptability of a prototype PROMPPT review (experienced acceptability), within MRC phase 1 evaluation. 11
Generally, the PROMPPT intervention was deemed acceptable by patients and pharmacists at both the prospective ‘in-principle’ phase and experienced prototype testing phase. Patients were grateful to be reviewed and for someone to listen and help them. Pharmacists felt that they were suited to deliver PROMPPT as they have the knowledge of pain medicines and the skills to consult with patients prescribed opioids for PNCP. Patients and pharmacists acknowledged that PROMPPT might not be acceptable to all patients, for example those who are fearful of having their opioids stopped or not being ready to make any changes to their pain management.
Key findings and associated recommendations for PROMPPT from prospective and experienced acceptability.
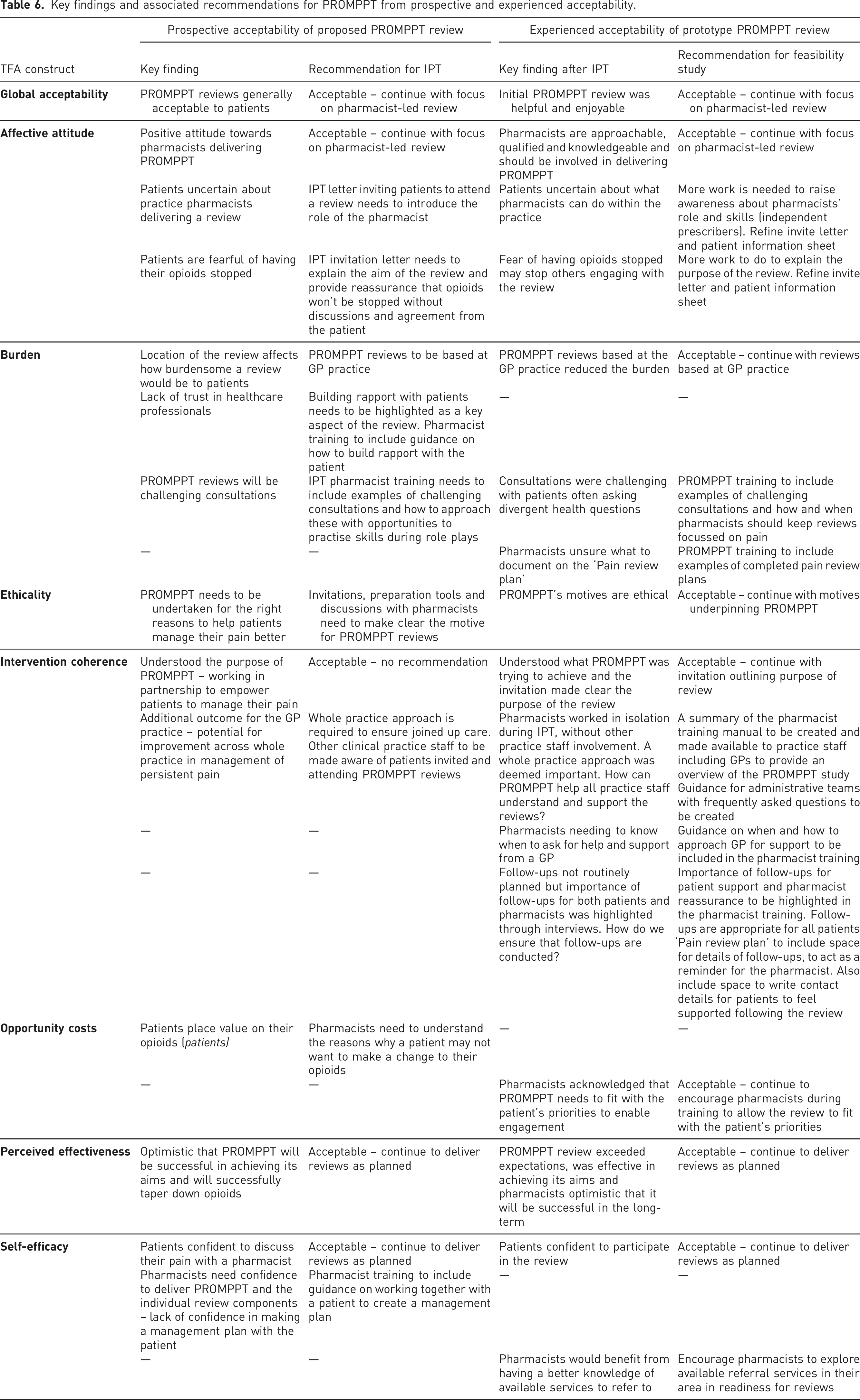
Reflections on using the TFA
The TFA has been used at singular timepoints (prospective,19,20 concurrent 21 and retrospective22,23) and across timepoints24,25 in intervention development and evaluation. We applied the TFA during the first phase of PROMPPT intervention development, including both prospective and experienced acceptability, allowing key concerns (about implementation or uptake) to be identified and addressed. Exploring prospective and experienced acceptability, highlighted how different constructs apply at different times. For example, patients expressed their concerns that PROMPPT reviews would need to be undertaken for the right reasons to help patients manage their pain and not be for NHS cost-saving when discussing ethicality. Following IPT, patients understood that the purpose of PROMPPT was to support them to live well with pain and that cost saving for the NHS and the GP practice was an additional outcome, demonstrating a shift from ethicality to intervention coherence.
Using the TFA to identify and address key concerns according to the different constructs, supports the assertion from Sekhon and colleagues that acceptability is not a fixed construct with binary (acceptable/not acceptable) outcomes. This is shared by Deja et al., 25 who acknowledged the merit in adopting the TFA to identify and address key issues that threatened the acceptability of their trial. 25 The TFA was developed as a tool to help understand what makes health interventions acceptable and what needs to be addressed to improve its acceptability12,13 rather than defining criteria to assess whether something is acceptable or not according to set cut-offs. We used the construct of ‘Global acceptability’ as an overall marker of whether the intervention was acceptable or not and used other constructs to identify areas that required no change and recommendations for improvement. As no ‘red flags’ were identified in the key findings for each construct, we were reassured that the intervention was generally acceptable but could be improved by actioning the recommendations. We also observed a significant overlap in the constructs ‘Global acceptability’ and ‘Affective attitude’ when participants were discussing their feelings towards the PROMPPT intervention. This supported our decision to use ‘Global Acceptability’ as a marker to judge overall acceptability rather than to use it to identify aspects of acceptability as a separate construct. To support future research using the TFA, some clarification is required with regards to assessing acceptability and whether when an intervention or aspects of, can be classified as ‘acceptable’ according to the constructs.
Strengths and limitations
We have adopted a multi-component approach using complementary qualitative research methods to support the early development phases of PROMPPT. Recruiting participants through a range of routes allowed us to capture perspectives from a wider range of people with experience of taking opioids for PNCP, including both those currently using opioids and those who had experience of tapering and stopping their opioids. Interviewing patients and pharmacists means that we have investigated potential problems for those either receiving or delivering the intervention. The study was further strengthened with the inclusion of prototype testing during IPT, allowing for potential barriers to implementation and issues of acceptability to be addressed to increase probability of intervention success.
During IPT, patients were asked to ‘think-aloud’ during the PROMPPT review by saying any thoughts or feelings that came up concurrently. However, none of them did this, and so we asked retrospectively about the review in the interviews afterwards. Concurrent think-aloud methods have been used previously in questionnaire design26–28 and intervention development studies. 29 However, in the context of a primary care consultation, ‘think-alouds’ did not work so well as it disrupts the flow of the review and the dialogue between the patient and the pharmacist.
Issues highlighted during each stage of IPT were addressed where possible, but a short timeframe between each cycle limited how much could be addressed. Any recommendations not implemented during cycles (e.g. summary of pharmacist training for other practice staff including GPs and guidance for administrative teams) were addressed following the final cycle of IPT and helped to inform the intervention tested in a single-armed feasibility study to assess the acceptability and credibility of the pain management review.
Conclusion
This paper highlights how assessing acceptability at multiple time points during early intervention development allows for refinement and development to optimise implementation in relation to acceptability. 12 Using ‘Global acceptability’ as a marker, we were reassured of the overall acceptability of the PROMPPT intervention. Recommendations identified according to the TFA constructs allowed an acceptable intervention to be developed that was ready to be tested in a formal feasibility study. The TFA was used during feasibility testing and is now currently being used for evaluation of the intervention in a main cluster-randomised controlled trial as recommended.12,13
Supplemental Material
Supplemental Material - Acceptability of a proposed practice pharmacist-led review for opioid-treated patients with persistent pain: A qualitative study to inform intervention development
Acceptability of a proposed practice pharmacist-led review for opioid-treated patients with persistent pain: A qualitative study to inform intervention development by Nicola Cornwall, Charlotte Woodcock, Julie Ashworth, Sarah A Harrisson, Lisa Dikomitis, Simon White, Toby Helliwell, Eleanor Hodgson, Roger Knaggs, Tamar Pincus, Miriam Santer, Christian D Mallen, Clare Jinks: on behalf of the PROMPPT team in British Journal of Pain.
Supplemental Material
Supplemental Material - Acceptability of a proposed practice pharmacist-led review for opioid-treated patients with persistent pain: A qualitative study to inform intervention development
Acceptability of a proposed practice pharmacist-led review for opioid-treated patients with persistent pain: A qualitative study to inform intervention development by Nicola Cornwall, Charlotte Woodcock, Julie Ashworth, Sarah A Harrisson, Lisa Dikomitis, Simon White, Toby Helliwell, Eleanor Hodgson, Roger Knaggs, Tamar Pincus, Miriam Santer, Christian D Mallen, Clare Jinks: on behalf of the PROMPPT team in British Journal of Pain.
Supplemental Material
Supplemental Material - Acceptability of a proposed practice pharmacist-led review for opioid-treated patients with persistent pain: A qualitative study to inform intervention development
Acceptability of a proposed practice pharmacist-led review for opioid-treated patients with persistent pain: A qualitative study to inform intervention development by Nicola Cornwall, Charlotte Woodcock, Julie Ashworth, Sarah A Harrisson, Lisa Dikomitis, Simon White, Toby Helliwell, Eleanor Hodgson, Roger Knaggs, Tamar Pincus, Miriam Santer, Christian D Mallen, Clare Jinks: on behalf of the PROMPPT team in British Journal of Pain.
Supplemental Material
Supplemental Material - Acceptability of a proposed practice pharmacist-led review for opioid-treated patients with persistent pain: A qualitative study to inform intervention development
Acceptability of a proposed practice pharmacist-led review for opioid-treated patients with persistent pain: A qualitative study to inform intervention development by Nicola Cornwall, Charlotte Woodcock, Julie Ashworth, Sarah A Harrisson, Lisa Dikomitis, Simon White, Toby Helliwell, Eleanor Hodgson, Roger Knaggs, Tamar Pincus, Miriam Santer, Christian D Mallen, Clare Jinks: on behalf of the PROMPPT team in British Journal of Pain.
Supplemental Material
Supplemental Material - Acceptability of a proposed practice pharmacist-led review for opioid-treated patients with persistent pain: A qualitative study to inform intervention development
Acceptability of a proposed practice pharmacist-led review for opioid-treated patients with persistent pain: A qualitative study to inform intervention development by Nicola Cornwall, Charlotte Woodcock, Julie Ashworth, Sarah A Harrisson, Lisa Dikomitis, Simon White, Toby Helliwell, Eleanor Hodgson, Roger Knaggs, Tamar Pincus, Miriam Santer, Christian D Mallen, Clare Jinks: on behalf of the PROMPPT team in British Journal of Pain.
Supplemental Material
Supplemental Material - Acceptability of a proposed practice pharmacist-led review for opioid-treated patients with persistent pain: A qualitative study to inform intervention development
Acceptability of a proposed practice pharmacist-led review for opioid-treated patients with persistent pain: A qualitative study to inform intervention development by Nicola Cornwall, Charlotte Woodcock, Julie Ashworth, Sarah A Harrisson, Lisa Dikomitis, Simon White, Toby Helliwell, Eleanor Hodgson, Roger Knaggs, Tamar Pincus, Miriam Santer, Christian D Mallen, Clare Jinks: on behalf of the PROMPPT team in British Journal of Pain.
Supplemental Material
Supplemental Material - Acceptability of a proposed practice pharmacist-led review for opioid-treated patients with persistent pain: A qualitative study to inform intervention development
Acceptability of a proposed practice pharmacist-led review for opioid-treated patients with persistent pain: A qualitative study to inform intervention development by Nicola Cornwall, Charlotte Woodcock, Julie Ashworth, Sarah A Harrisson, Lisa Dikomitis, Simon White, Toby Helliwell, Eleanor Hodgson, Roger Knaggs, Tamar Pincus, Miriam Santer, Christian D Mallen, Clare Jinks: on behalf of the PROMPPT team in British Journal of Pain.
Supplemental Material
Supplemental Material - Acceptability of a proposed practice pharmacist-led review for opioid-treated patients with persistent pain: A qualitative study to inform intervention development
Acceptability of a proposed practice pharmacist-led review for opioid-treated patients with persistent pain: A qualitative study to inform intervention development by Nicola Cornwall, Charlotte Woodcock, Julie Ashworth, Sarah A Harrisson, Lisa Dikomitis, Simon White, Toby Helliwell, Eleanor Hodgson, Roger Knaggs, Tamar Pincus, Miriam Santer, Christian D Mallen, Clare Jinks: on behalf of the PROMPPT team in British Journal of Pain.
Footnotes
Acknowledgements
We would like to thank all the participants for sharing their views and experiences in interviews or online via the discussion forum. We would also like to thank all participants who gave us their time to take part in the in-practice testing. Our appreciation is also extended to members of the PROMPPT research user group, and members of the public, who helped develop topic guides. We would like to thank Dr Mandeep Sekhon who helped us to understand TFA constructs and provided an example TFA topic guide for us to use. We would also like to thank Keele University’s Clinical Trials Unit in supporting study management and the NIHR West Midlands Clinical Research Network in supporting recruitment processes.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the NIHR Programme Grant for Applied Research under Grant number RP-PG-0617-20005. CM, CJ and CW are part funded by the National Institute for Health Research (NIHR) Applied Research Collaboration (ARC) West Midlands. CM is also funded by the National Institute for Health Research (NIHR) School for Primary Care Research and the School of Medicine have received funding from BMS to support a non-pharmacological Atrial Fibrillation Screening Trial.
Ethical statement
Contributorship
CJ, JA, LD, SW, CM conceived the study idea. CJ, NC, CW, JA, SA, LD, SW, TH, CM all contributed to research design and protocol writing. NC, CW, TH and SW collected data. NC, CW, CJ, SH, SW and EH analysed the data. NC wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Trial registration
Research Registry: researchregistry4796
Guarantor
CJ
Data Availability Statement
Supplementary materials for this article are available online including topic guides and illustrative quotes.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
