Abstract
Background
The prescription of opioids in emergency care has been associated with harm, including overdose and dependence. The aim of this trial was to assess restriction of access to oxycodone (ROXY), in combination with education and guideline modifications, versus education and guideline modifications alone (standard care) to reduce oxycodone administration in the Emergency Department (ED).
Methods
An unblinded, active control, randomised controlled trial was conducted in an adult tertiary ED. Participants were patients aged 18–75 years who had analgesics administered in the ED. The primary intervention was ROXY, through removal of all oxycodone immediate release tablets from the ED imprest, with availability of a small supply after senior clinician approval. The intervention did not restrict prescription of discharge medications. The primary outcome measure was oxycodone administration rates. Secondary outcomes were administration rates of other analgesic medications, time to initial analgesics and oxycodone prescription on discharge.
Results
There were 2258 patients eligible for analysis. Oxycodone was administered to 80 (6.1%) patients in the ROXY group and 221 (23.3%) patients in the standard care group (relative risk (RR) 0.26; 95% CI: 0.21 to 0.33;
Conclusions
Restricted access to oxycodone was superior to education and guideline modifications alone for reducing oxycodone use in the ED and reducing discharge prescriptions of oxycodone from the ED. The addition of simple restrictive interventions is recommended to enable rapid changes to clinician behaviour to reduce the potential harm associated with the prescribing of oxycodone in the ED.
Keywords
Introduction
The widespread misuse of prescription opioids is a ‘crisis’ that is being experienced in many countries, particularly in the United States of America (USA) and Canada. 1 There is currently evidence of harm associated with prescription opioids, including, but not limited to overdose and dependence. 2 Australia is rapidly following the same path and now ranks eighth internationally on the number of daily doses of prescription opioids per million population (at approximately 40% the level of USA). 3
Pharmaceutical opioids (primarily oxycodone) are a major contributor to deaths and poisoning-related hospitalisations in Australia which far exceeds that from illicit opioids. In 2016, it was estimated that 150 hospitalisations and 14 Emergency Department (ED) presentations per day involved opioid harm, and three people died per day from drug-induced deaths involving opioid use. 4 Opioids are often prescribed to patients who present to EDs.5,6 The prescription of oxycodone on discharge from the ED has been associated with long-term opioid use. 7
Education programs, prescribing guidelines and policies to reduce opioid use in the ED are being implemented. 8 However, the effects of such interventions have been modest, at best. For example, an educational program on safe prescriptions reduced discharge prescribing for opioids, but 16% of all patients discharged from the ED continued to receive a prescription of opioids. 9 Educational materials describing the background of the opioid crisis and emergency medicine's contribution to this crisis, combined with monthly distribution of provider-specific opioid prescribing rates were also successful in reducing opioid prescribing, but 14% of patients continued to receive opioid prescriptions. 10 Similarly, education sessions, staff information emails, posters within the ED and patient brochures were associated with significant, but small reductions in the rate of oxycodone prescriptions. 11 These results are consistent with other multimodal interventions that optimise oxycodone prescribing and have been associated with similarly modest reductions in rates of oxycodone prescription. 12 Current efforts demonstrate that despite admirable and significant improvements, even in the post-intervention arms of studies, a substantial proportion of ED patients continued to be prescribed oxycodone.
Consistent with interventions around the world, at our institution, after prescriber education and introduction of an analgesic ladder (Appendix 1), there was significantly reduced oxycodone prescription for back pain, but 51.4% of patients with pain continued to receive oxycodone in the ED. 13 In contrast to education programs, more extreme measures such as restriction to supply of a medication has had stronger effects. For example, the removal or restriction of pethidine availability was demonstrated to have profound reductions in use.14,15 Similar strategies using other medications from a formulary had immediate and persistent effects on use. 16 It is important that any policy to restrict availability or freedom to prescribe is based on sound evidence that harms of prescription outweigh benefits and the effectiveness of restrictive strategies. The collective evidence on oxycodone use suggests substantial harms that outweigh benefits in many patients.
The aim of this trial was to assess the effectiveness of a restrictive intervention to access oxycodone. The hypothesis for this trial was that restricted access to oxycodone in the ED, when combined with standard practice, will significantly reduce the proportion of patients prescribed and administered oxycodone compared to standard practice of safe-prescribing guidelines and educational programs.
Methods
This was an unblinded, active controlled, randomised controlled trial, undertaken at The Alfred Hospital in Melbourne, Victoria. The Alfred hospital is an adult tertiary referral centre, with an annual attendance of approximately 70,000 patients. The Emergency and Trauma Centre is the busiest adult level 1 Trauma Centre in Australia, with over 1200 major trauma presentations annually and 5000 trauma admissions. It has Victoria’s largest intensive care unit and provides multiple other state-wide services including burns, haemophilia, cystic fibrosis, heart and lung transplants, HIV and adult haematological malignancies.
A simple computer-generated randomisation technique was used, with allocation to intervention or standard care in the ED for all patients presenting in each 7-day period from 0800 hrs on a Monday to 0759 hrs the following Monday. At 0800 hrs on a Monday, the in-charge emergency physician and nurse, together with the clinical pharmacist, opened an envelope allocating the week to a restriction of oxycodone (ROXY) versus standard prescription week.
Eligible patients
All patients administered analgesia in the ED; patients aged 18–75 years were eligible for inclusion in this study. Eligible patients were identified retrospectively through medication dispensing logs recording administration of paracetamol (with or without codeine), oral non-steroidal anti-inflammatory drugs (ibuprofen, indomethacin diclofenac and naproxen), hydromorphone, celecoxib, ketamine, ketorolac, tramadol, tapentadol, morphine, fentanyl, pregabalin or oxycodone (with or without naloxone) in the ED. Data on initial level of pain was collected using an 11-point (0 to 10) Likert scale as reported by the patient. The patients’ primary presenting complaints were recorded and reported using the categorisation tool for presenting complaints in the Alfred registry for emergency care. 17
Intervention
The intervention was restriction of oxycodone (ROXY). In the week randomised to ROXY, the responsible clinical pharmacist removed all oxycodone 5 mg immediate release (IR) tablets from the main ED imprest. A small supply of oxycodone 5 mg IR was maintained in the Trauma Centre imprest, accessible only by the clinical pharmacist or selected senior nurses (nurse in-charge of clinical shift or critical care area). Access to oxycodone IR stock was available only after authorisation by the in-charge emergency physician. It was not possible for other nursing staff to access oxycodone. There were no restrictions on the prescription of discharge medications.
Comparator
The comparator group of patients were managed using standard clinical practice. When randomised to this week, nursing staff had access to and could administer oxycodone 5 mg IR when prescribed and no authorisation was required. All medications in The Alfred ED are stored in an Omnicell® automated medication dispensing machine and removal of oxycodone from the system requires dual confirmation of the prescription and checking of patient identification. Throughout the trial, standard processes of education regarding potential harm from opioid prescription and promotion of safe analgesic prescribing informed by the department’s analgesic ladder continued.
Outcome measures
The primary outcome was the proportion of patients administered oxycodone in the ED among those who were administered any analgesia. Secondary outcomes were the proportion of patients administered other analgesic medications and the proportion of patients discharged home with oxycodone and other analgesic medications. In addition, we assessed the time to first analgesic in the two groups.
Statistical analysis
A sample size of 3000 patients was planned to allow for patients presenting to the ED and not administered any analgesic medications. Among patients who were administered analgesia in the ED, the estimated proportion prescribed that oxycodone was 20%. Considering a clinically significant difference of 5% using power of 80% and 95% confidence intervals, the number of patients required in each arm was 906. After the last weeks were randomised, enrolment was continued till the end of that week and not stopped mid-week after the required sample size was achieved.
The primary outcome of oxycodone administration and secondary outcomes (administration of other analgesic medications and discharge prescriptions) were compared with unadjusted chi-square test for equal proportions, with results reported as frequency (percentage) per treatment group with a relative risk and risk difference, both accompanied by 95% confidence intervals (CIs). Time to first analgesic was assessed with Cox proportional hazards regression censored at 4 h, with results presented using Kaplan–Meier survival curves with corresponding log-rank test. All analyses were conducted with Stata v 15.1, College Station, Texas, USA. A two-sided
Ethics approval
The study was approved by The Alfred Human Research and Ethics Committee (Project no. 45/21). The requirement to seek informed consent from patients and clinicians was waived. The trial was registered with the Australian New Zealand Clinical Trials Registry (ACTRN12622001183774).
Results
During the study period, a total of 3949 patients were eligible for inclusion. After exclusion for age limits and pregnancy, 3014 patients were randomised to have restricted access to oxycodone or standard care. A further 756 patients did not have any analgesia administered in the ED, with equal distribution in the two arms, and were excluded. There were 1309 patients managed with restricted access to oxycodone and 949 patients with standard care (Figure 1). CONSORT flow chart.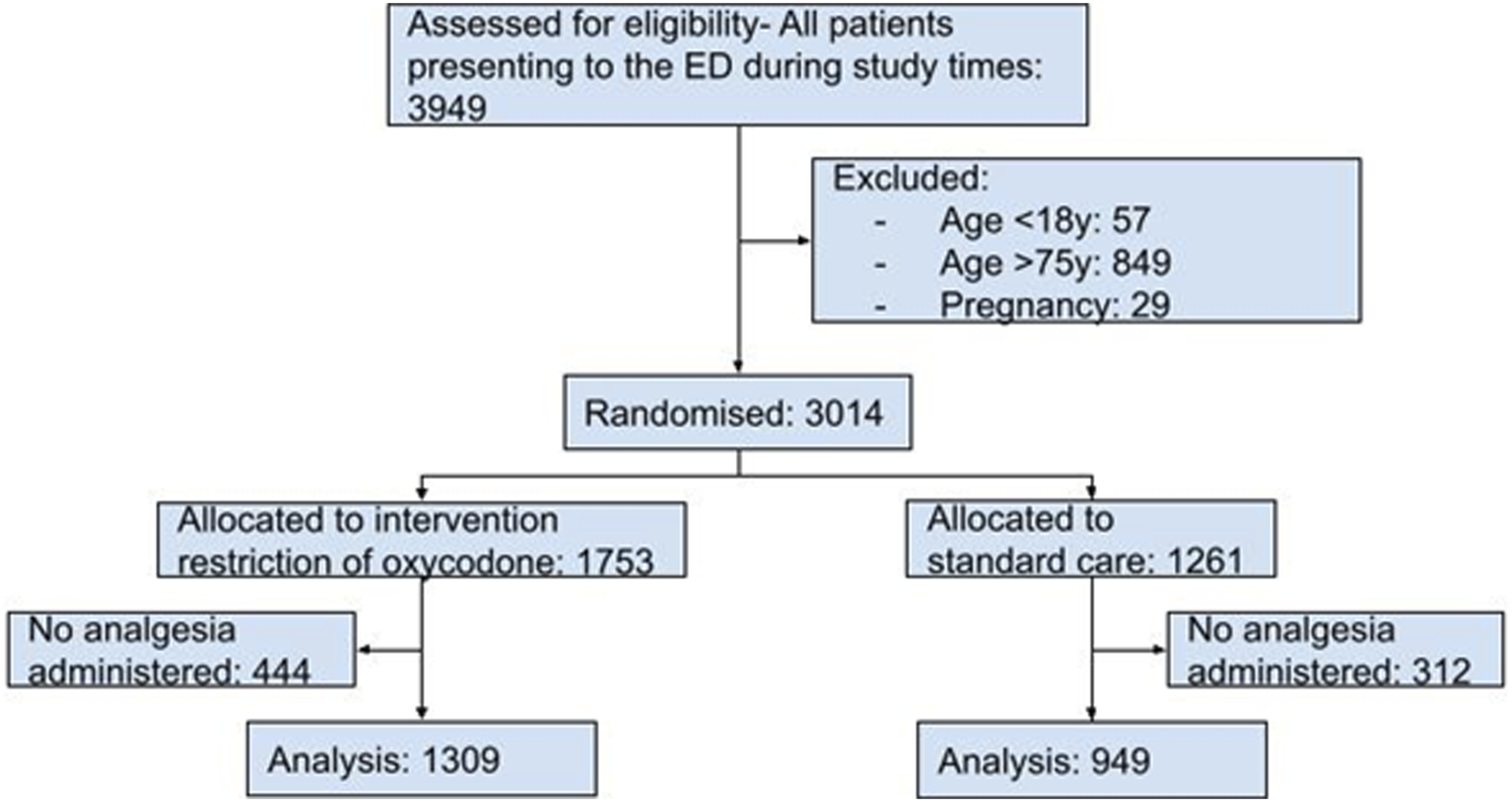
Demographic and baseline characteristics of included patients.
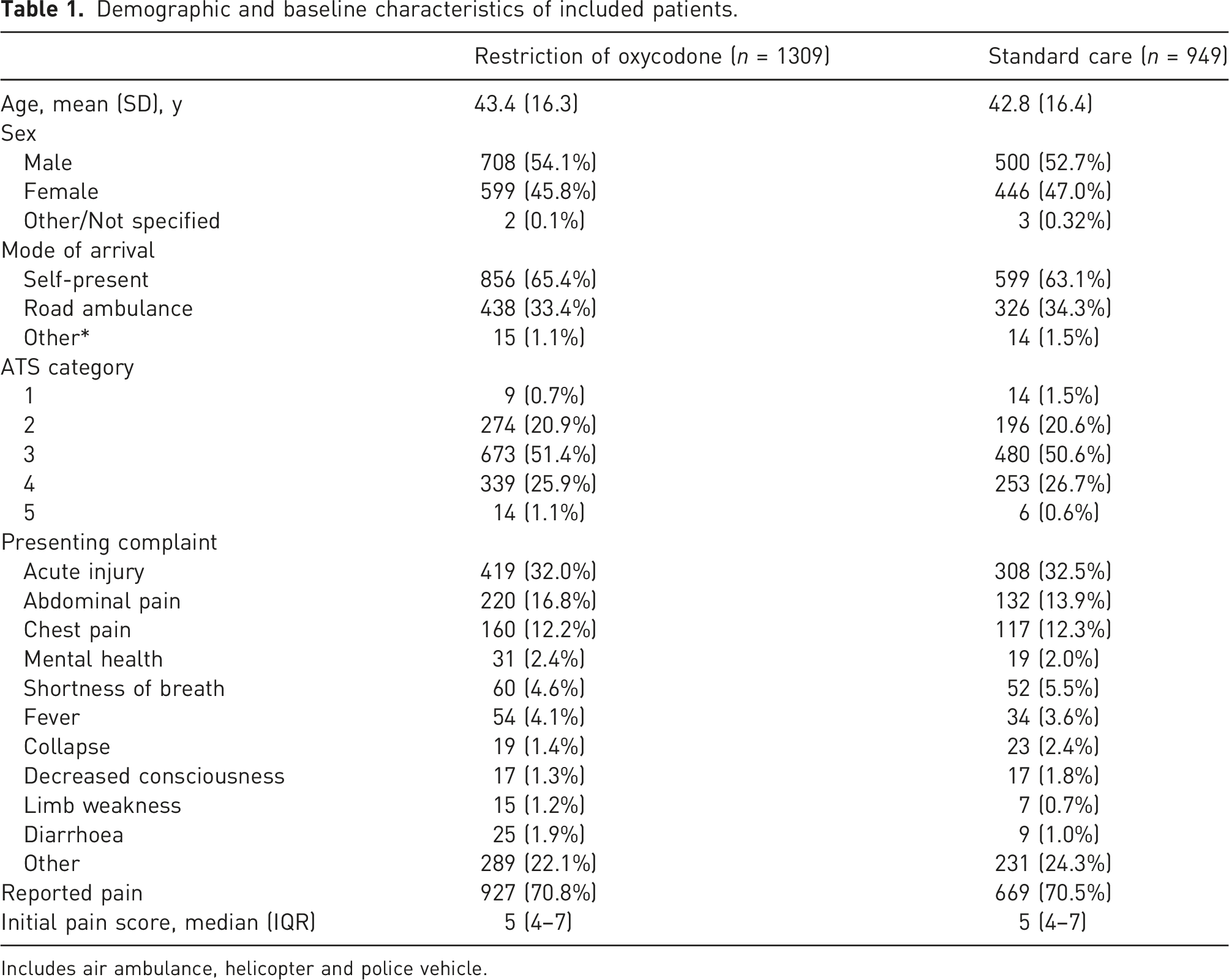
Includes air ambulance, helicopter and police vehicle.
Primary outcome
Primary and secondary outcomes.
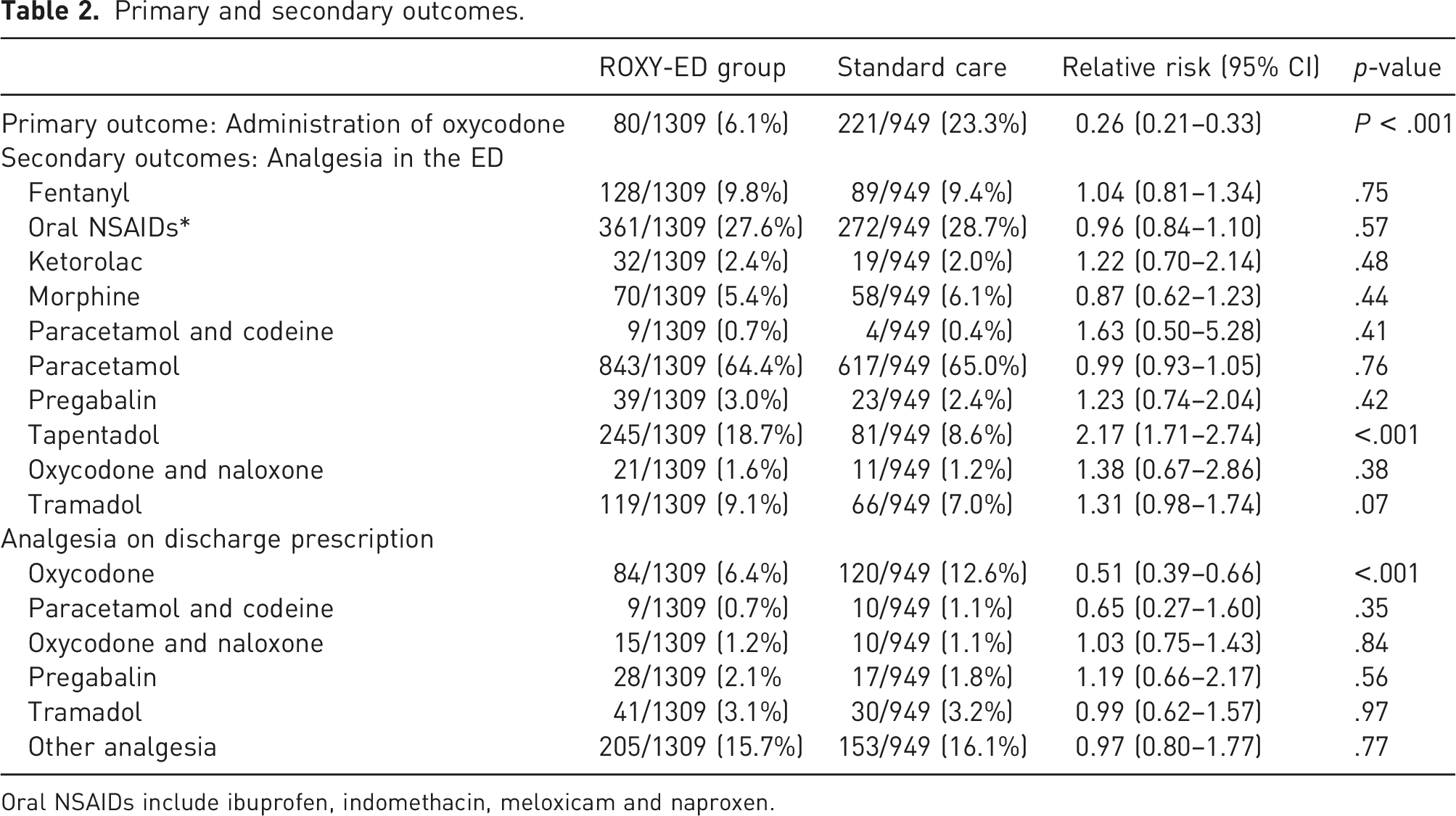
Oral NSAIDs include ibuprofen, indomethacin, meloxicam and naproxen.
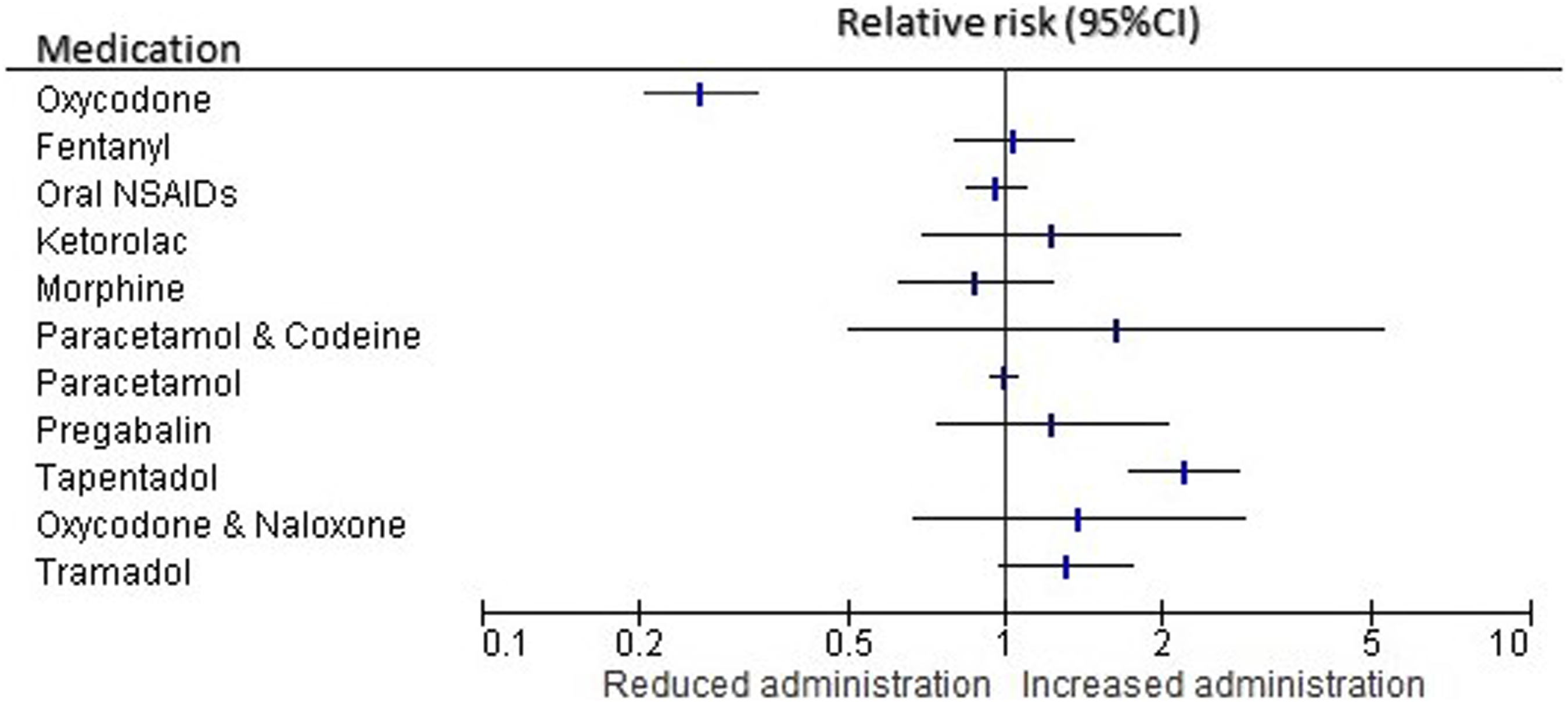
Differences in administration of analgesic medications in the ED.
Secondary outcomes
When prescribed, the mean dose of oxycodone in the ROXY group was 8.1 (SD 5.2) mg, compared to mean dose of 8.9 (SD 4.5) mg in the standard care group (difference in mean 0.79; 95% CI −0.61–2.19; Differences in discharge prescriptions of analgesic medications.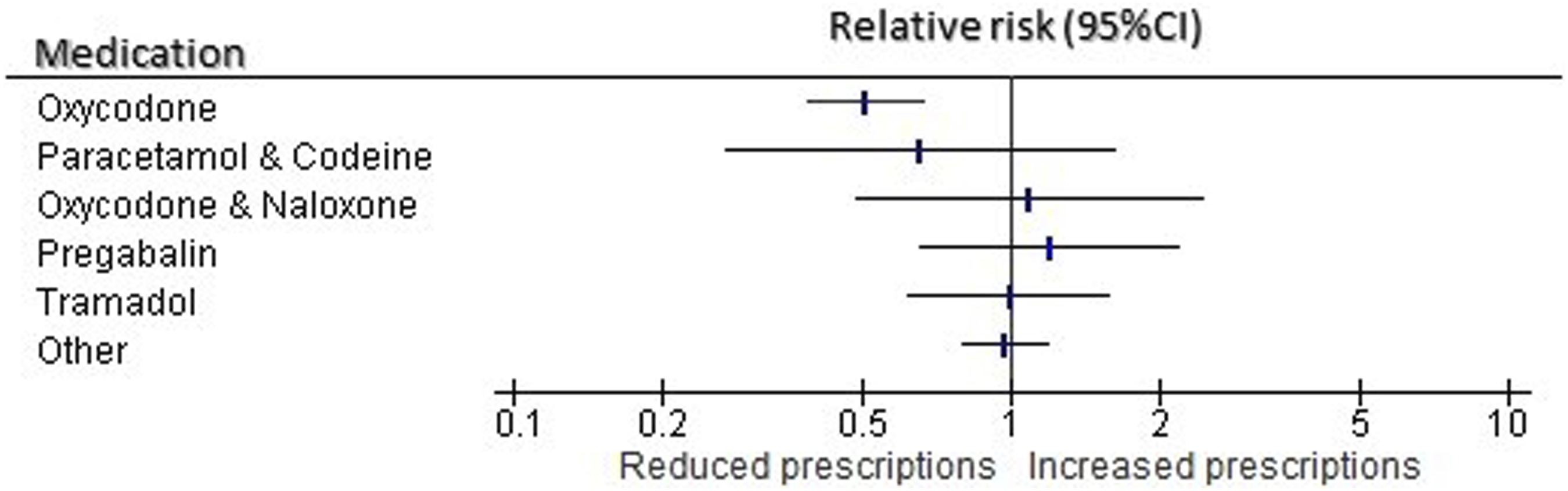
On discharge, significantly fewer patients were prescribed oxycodone (RR 0.51; 95% CI: 0.39–0.66). There were no significant differences in discharge prescription of other analgesic medications (Table 2 and Figure 3). There was no difference in time to first analgesic between the two groups (HR 0.94; 95% CI: 0.86–1.02); Figure 4. Time to first analgesic.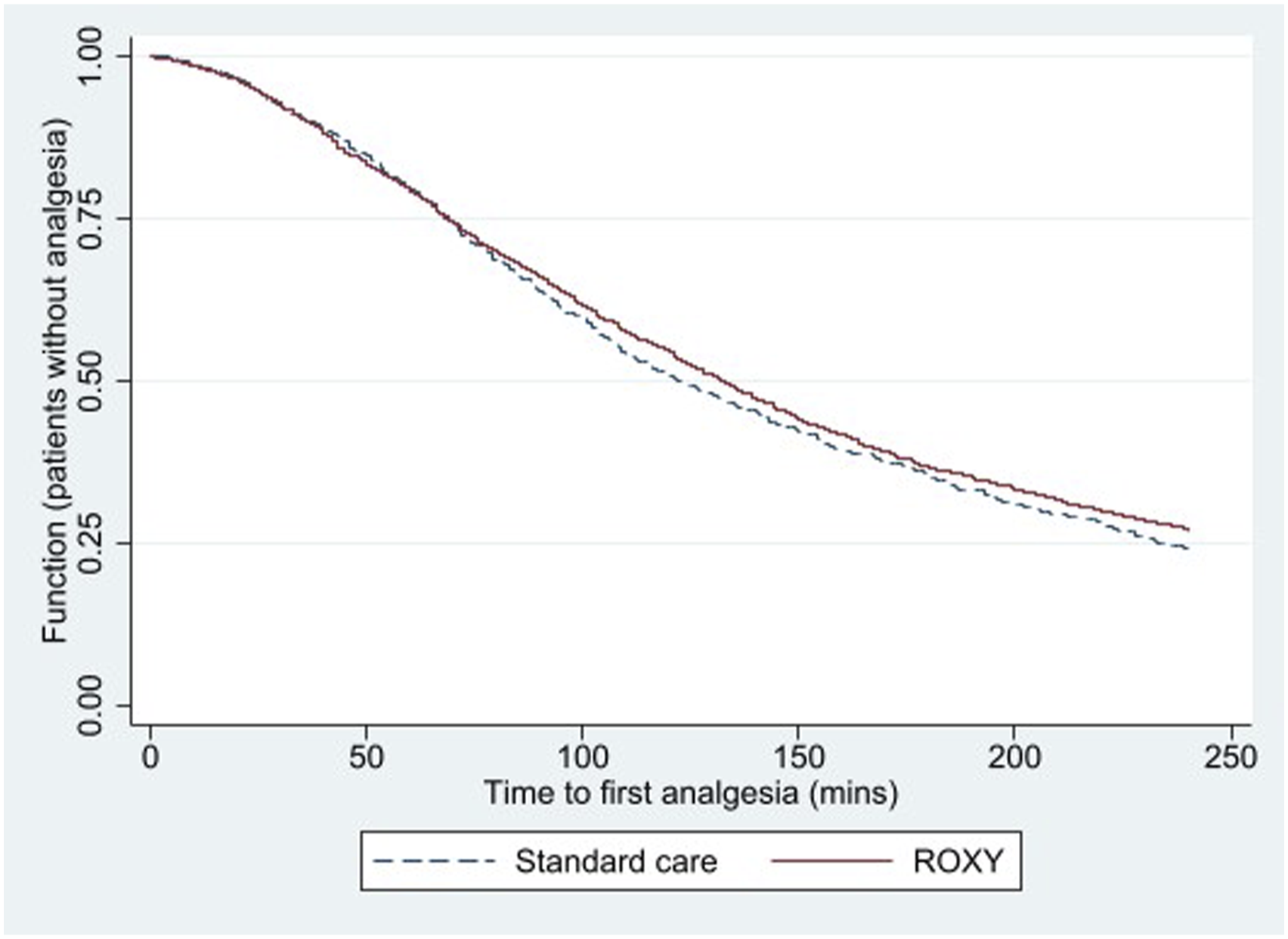
Discussion
Restriction of access, combined with education and promotion of guidelines, significantly reduced oxycodone administration in the ED, when compared to education and guidelines alone. In addition to oxycodone administration in the ED, the intervention was associated with significant reduction in discharge prescriptions of oxycodone. The dual benefit of this intervention is important. While reducing oxycodone administration in the ED may have avoided its use during the ED episode and inpatient care, reducing oxycodone prescribing on discharge may change the burden of opioid use in the general community and the potential for opioid-related harm. Encouragingly, restriction of access to oxycodone was not associated with any change in time to delivery of analgesia. The results support addition of restrictive interventions to educational and guideline reforms directed at reducing oxycodone use to ensure timely, responsible opioid use.
Combined with potential of harm from oxycodone use, the evidence for superior efficacy of oxycodone over other analgesic options is uncertain. The use of oxycodone is supported by equivalence to intravenous morphine after major orthopaedic, abdominal and cardiac surgery and for management of cancer-related pain.18–20 However, the results are inconsistent with others concluding that among patients who had undergone orthopaedic surgery, oxycodone did not provide superior pain relief compared with combination acetaminophen and codeine. 21 Similarly, among patients presenting to the ED with moderate pain after limb injury, non-opioid analgesia (paracetamol, ibuprofen and thiamine), in addition to codeine and oxycodone, demonstrated similar efficacy for acute (within 30 min) pain reduction. 22 Among patients with back pain, the addition of oxycodone to non-steroidal anti-inflammatory medications did not improve functional outcome among patients with acute, non-traumatic, non-radicular low back pain. 23 For post-operative dental pain, non-steroidal anti-inflammatory drugs were superior to oxycodone/paracetamol combinations. 24
Results of this study are consistent with others that demonstrated that oral oxycodone use can be replaced. Transition from parenteral to sublingual buprenorphine, compared to oxycodone, has been associated with significantly lower opioid use and pain scores. 25 Substituting oxycodone use with tapentadol has been supported by studies concluding equivalence of analgesia and a more favourable side-effect profile for tapentadol.26,27 It has been recently reported that tapentadol has replaced oxycodone and tramadol to become the most frequently prescribed oral opioid prescribed in an Australian local health district. 28 Similarly, equivalence of oral oxycodone and tramadol has also been concluded. 29 Previous studies had demonstrated wide variations in the practice of opioid prescription on discharge. Key determinants have been concluded to be patient age, time of arrival to the ED and diagnostic groups such as musculoskeletal complaints. 30 In addition, inpatient opioid use may influence discharge prescription. 31 This study therefore adds to the hypothesis that influencing prescription in the emergency department can lower discharge prescriptions.
As tapentadol is a relatively new analgesic, ongoing vigilance of evidence of improved safety over other opioids is indicated. A US-based study of the abuse potential of tapentadol found it to be higher than tramadol but lower than other strong opioids. 32 A subsequent study using US drug monitoring programs has demonstrated that the public health burden from tapentadol abuse is present but lower in comparison to other opioids. 33 We hypothesise that the risk of abuse is likely to be low when patients do not continue to receive doses on hospital discharge.
The combination of harms from oxycodone misuse, questionable superiority or at least equivalence to other analgesic options and results of this study demonstrating ability to achieve reduction in use through a relatively simple intervention provides a compelling case to restrict the availability of oxycodone in EDs. It is imperative, however, that patient comfort remains at the fore-front and effective analgesia is achieved by alternative therapies and/or interventions. Our observation that some patients with high pain scores were managed with non-opioid medications suggests that either non-opioids are effective for severe pain or that pain may settle over time. It is possible that clinician-driven analgesia by reliance on pain scores increases the potential for overuse of opioids. Detailed exploration of patient experience during the ED journey is therefore critical to rationalise analgesia. Ongoing patient education is also essential to prevent unwanted effects such as illicit trading of opioids. 34
This trial was limited in being unblinded. It is assumed that there was substantial contamination after initiation of the study with effect on the comparator arm. However, we expected that this contamination would have reduced the use of oxycodone in the comparator arm, thereby strengthening the conclusions of the trial. An effect in the opposite direction that the trial increased oxycodone use during standard care weeks was considered unlikely and not apparent. A further limitation of this trial is in the exclusion of paediatric and older patients from this study, who are potentially at higher risk of harm from oxycodone use. The setting of this study, being a major trauma centre, had a high proportion of patients with acute injury, and may have amplified the effect of restriction. Another limitation of this trial was that clinicians had the option to administer oxycodone despite the restrictions if they believed it was in the patients’ best interests. Thus, this trial does not provide evidence towards absolute restrictions to oxycodone access. Translation of this evidence should include patient-reported outcome measures, such as observation of pain scores or comfort levels at prescribed intervals, with a focus on optimal comfort for patients in the ED. Future work should also consider restrictive interventions as an additional strategy for incorporation in clinical guidelines such as the Australian Opioid Analgesic Stewardship in Acute Pain Clinical Care Standard.
Conclusions
The intervention to restrict access to oxycodone was superior to standard strategies of education and guideline modifications in reducing oxycodone use in the ED and on discharge prescriptions. The addition of simple restrictive interventions is recommended to enable rapid changes to clinician behaviour to reduce the potential harm associated with the prescribing of oxycodone.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The study was approved by The Alfred Hospital Human Research and Ethics Committee (Project no. 45/21).
Informed consent
The requirement to seek informed consent from patients and clinicians was waived.
Trial registration
The trial was registered with the Australian New Zealand Clinical Trials Registry (ACTRN12622001183774).
Guarantor
Biswadev Mitra
Contributorship
BM, DS and PC developed the hypotheses. BM, TA, HB and CR developed the methodology. TA, HB, BW and DG collected the data. BM and RH analysed the data. All authors were involved in drafting of the manuscript and revisions.
Appendix
