Abstract
Introduction
Mechanical chronic low back pain is often associated with impaired neuromuscular control of the lumbar multifidus muscles, the most important stabilizers of the lumbar spine. Restorative neurostimulation is a modality for the treatment for this specific subset of patients aimed to facilitate restoration of neuromuscular control by bilateral stimulation of the L2 medial branches. Evidence from both prospective and randomised clinical trials to date has demonstrated substantial improvements in clinical outcomes such as pain, disability and health-related quality of life.
Methods
This study is an open label prospective follow-up for the treatment of chronic mechanical low back pain of nociceptive origin with restorative neurostimulation. Patients completed assessments for pain, disability and health-related quality of life. Outcomes were collected at 45, 90 and 180 days, and 1, 2 and 3 years after the activation visit.
Results
Forty-two patients were implanted with the device and 33 (79%) were available at the 3-year appointment. Patients in this cohort presented with severe chronic low back pain (NRS = 7.0 ± 0.2) and severe disability (ODI 46.6 ± 12.0). The health-related quality of life was also severely impacted at baseline (EQ-5D 0.426 ± 0.061). Changes in pain, disability and quality of life at three-year follow-up demonstrated a statistically significant improvement between baseline and 1, 2 and 3 years. After 3 years of therapy, average NRS scores had reduced to 2.7± 0.3 and mean ODI score to 26.0 ± 3.1 while EQ-5D-5L index improved to 0.707 ± 0.036.
Conclusions
The ongoing follow-up of this post market cohort continues to demonstrate that restorative neurostimulation provides a statistically significant, clinically meaningful and durable response across pain, disability and quality-of life scores for patients suffering chronic mechanical low back pain that has been refractory to conventional management.
Trial Registration
ClinicalTrials.gov Identifier: NCT01985230.
Introduction
The global burden of low back pain is among the leading causes of years lived with disability in both high- and low-income countries alike. 1 The natural history of low back pain suggests that the majority of cases are self-limiting, though there is a significant portion of the population that go on to develop chronic pain low back pain (CLBP). 2 The source of pain can either be nociceptive, or neuropathic, sometimes mixed. Neuropathic pain has a relatively clear treatment pathway of managing inflamed neural structures, alleviating neural compression through surgery, and in certain circumstances, palliative pain management using spinal cord stimulation. 3 The therapeutic pathway for mechanical/nociceptive CLPB chronic nociceptive low back pain patients is less well defined, mainly because the efficacy and durability of traditional interventions is poor making it difficult for treating physicians and therapists to align on a single continuum of care. These patients will often start with physical therapy, the success of which is highly dependent on the skill of the practitioner and motivation of the patient. If this fails, patients will progress through a regimen of targeted injections, radio-frequency neurolysis and medical management using opioids (although not recommended, NICE NG59) 4 and sometimes a well-intentioned, but poorly indicated spinal fusion. 5
The pathophysiology of CLBP is complex and often a result of multiple overlapping mechanisms. Recently the International Society for the Study of the Lumbar Spine degenerative spinal phenotypes group suggested multiple pathways 6 including modifications to motor control, reflex inhibition and inflammatory mechanisms. Motor control dysfunction is of particular interest as it broadly describes a variety of changes in paraspinal muscle function. This includes changes in proprioception, 7 altered timing of activation, 8 disorganisation of the representation in the motor cortex 9 and modifications to the execution of motor planning strategies. 10 The combination of both peripheral and central aspects of motor control dysfunction has made it difficult to effectively target using conventional therapeutic approaches such as physical and exercise therapies. 11
The past decade has seen the development of restorative neurostimulation as a modality for the treatment for this specific subset of chronic nociceptive low back pain patients with motor control dysfunction resulting in neuromuscular instability.12–14 This therapy consists of two leads surgically implanted over the medial branch of the L2 dorsal ramus connected to an implantable pulse generator, located over the buttock. The nerves are stimulated with a waveform that induces smooth tetanic contractions of the multifidus muscle. Through a combination of peripheral and central mechanisms these contractions restore innate spinal motor control, reducing tissue nociceptor activation and alleviating the painful syndrome.
Clinical evidence in the form of both prospective and randomised clinical trials to date has demonstrated substantial improvements in clinical outcomes such as pain, disability and health-related quality of life.12–14 In this paper, we demonstrate the generalizability, durability and safety of these clinical effects by reporting the 3-year outcomes from the United Kingdom post market ReActiv8-A PMCF registry. This is a follow-up to the previous publication of the 2 year outcomes from this cohort. 15
Methods
Study design
Patients were consented to participate in an open label 5-year prospective follow-up for the treatment of chronic mechanical low back pain of nociceptive origin with restorative neurostimulation using the ReActiv8 (Mainstay Medical, Dublin, Ireland) device between September 2017 and September 2018. Here we present an analysis conducted after all subjects had reached 3 years of therapy or exited the study. Data was collected at five sites across the UK (ClinicalTrials.gov Identifier: NCT01985230). Outcomes were collected at baseline 45, 90 and 180 days, and 1, 2 and 3 years after the activation visit, with annual visits planned to continue to 5 years post-activation. Patients were screened at baseline for Depression, Anxiety and Stress using the DASS scale and completed assessments for pain (numerical rating scale – NRS), disability (Oswestry Disability Index – ODI) and health-related quality of life (EuroQol-5 Dimension 5 Level – EQ-5D-5 L) at baseline and each follow-up.
Ethics approval
The study protocol was reviewed and approved by a central ethics committee (NHS Health Research Authority North East—York Research Ethics Committee IRA’s project ID number 149412) as is required in the UK, and the protocol was followed in accordance with the Helsinki Declaration of 1964 and its later amendments. All subjects provided written informed consent to participate in the study. The study was funded by Mainstay Medical.
Patient selection
No formal inclusion or exclusion criteria were enforced outside the instructions for use and indications of the CE Mark. The aim was to reflect the “real world” clinical practice rather than the strictly controlled requirements for a randomised controlled study as needed by the Food and Drug Administration (FDA) by allowing the ultimate decision for inclusion or exclusion to be made by the treating clinician. By this definition, patients were eligible if they were adults with a history of predominantly mechanical CLBP for a minimum of 90 days that was refractory to physiotherapy and medication.
In practice mechanical/nociceptive low back pain presents as load dependent pain. In the initial diagnosis, there are a number of signs clinicians should look for, such as: • pain during flexion or return to neutral. • patients reporting a feeling of lumbar weakness or instability. • sudden pain or spasm during simple movements. • difficulty performing low load activities in sustained flexion, for example, brushing teeth or loading a dishwasher. • Pain during a prolonged period in a single position, for example, the patient canst sit or stand for long periods.
Additionally, prior to treatment with this device, patients should have tried and failed NICE approved conservative care such as pharmacological, physical therapies. Failed therapies included those that had only a temporary response or limited by side effects. Although not mandated in this study physicians are trained and encouraged to do physical testing for multifidus dysfunction, including the prone instability test (PIT),16,17 the multifidus lift test (MLF) and the aberrant movement patterns assessment. Observation of fatty infiltration of the multifidus by MRI imaging is also a concordant clinical sign. The history, continuity and severity of the symptoms should be significant enough for the clinician to conclude spontaneous resolution is unlikely and the severity sufficient to warrant interventional treatment.
Standard practice in the UK also implements assessment by a multidisciplinary team (MDT) that may include physical therapists, psychologists, neuromodulation nurse and pain management specialists to exclude serious co-morbidities and to ensure patient’s expectations and knowledge is aligned with the team.
Taken holistically, these patients are diagnosed with chronic neuromuscular instability of the lumbar spine that results in nociceptive low back pain.
Device description
A schematic of the device and its anatomical placement is shown in Figure 1. The system consists of two stimulating leads with distal fixation tines, an implantable pulse generator (IPG) and an external activator device. The proximal ends of the leads are connected to the IPG. The distal ends of the leads each contain four stimulating electrodes and two fixation tines to secure the leads that secure the leads in the intertransversarii muscle. The IPG can be programmed to deliver stimulation between any pair of electrodes on each lead. Device and Anatomical Placement.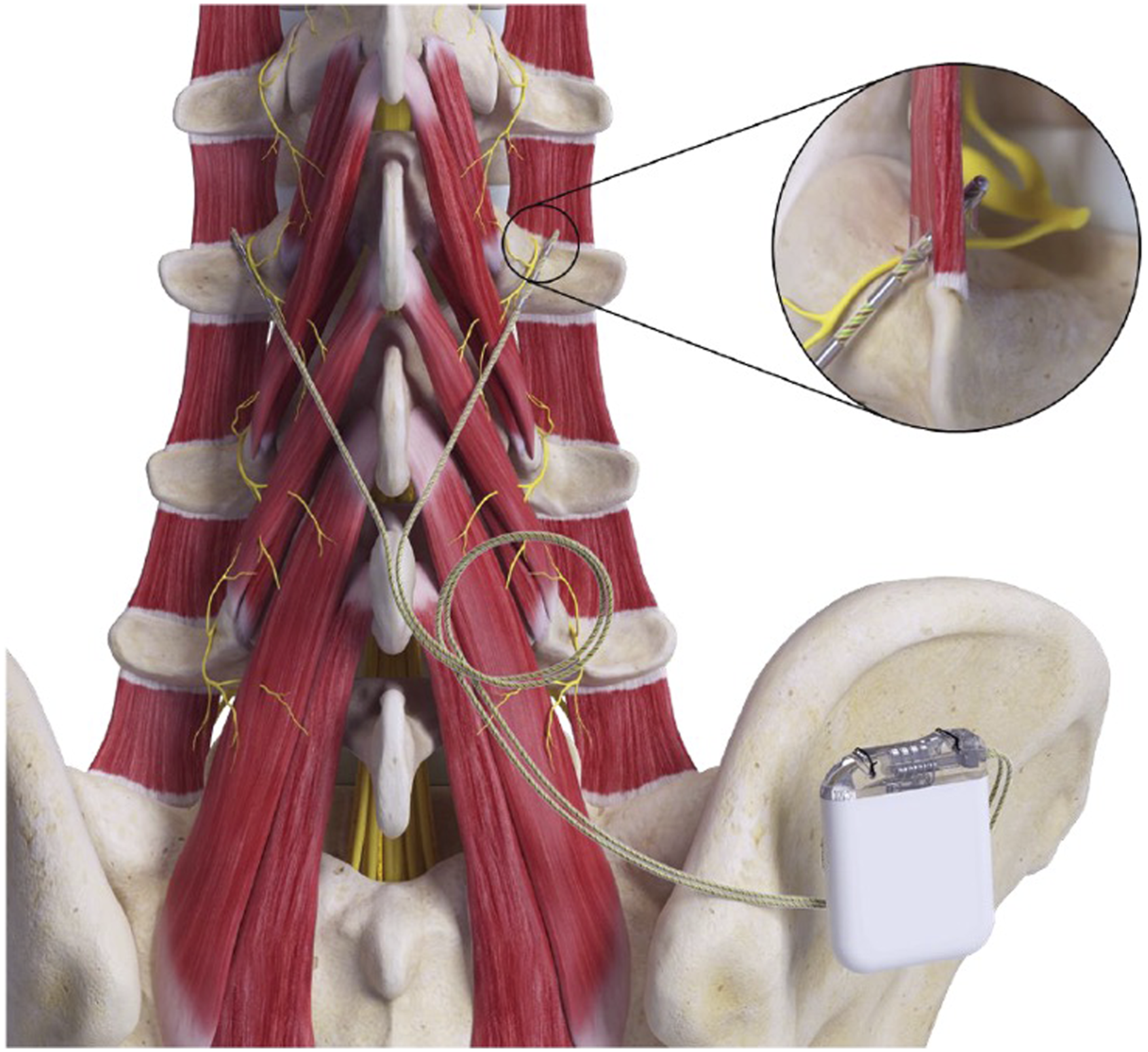
The implantation technique has been described elsewhere, 12 but briefly the distal electrodes are positioned adjacent to the medial branch of the L2 dorsal ramus as it passes over the junction of the L3 superior articular process and the transverse process, through a midline incision such that the leads pass through a minimum of fascial planes from medial to lateral. Once muscle contractions are elicited with on table testing, the leads are connected to an implantable pulse generator within a subcutaneous pocket in the flank or upper buttock.
Approximately 2 weeks after implantation the devices are activated and programmed with a waveform designed to elicit smooth strong contractions of the multifidus muscle. Patients are instructed to deliver the therapy by initiating up to two daily 30-min stimulation sessions while resting in either a prone or lateral position. The device was limited to provide a maximum of 60 min of therapy daily. Utilisation compliance was collected through interrogation of the IPG.
Data analysis
Patient reported outcomes for patients at 3-year follow-up were compared to baseline using repeated measures ANOVA with Bonferroni correction for multiple comparisons conducted in R version 3.6.1 and R Studio version 1.2.5019.
Missing patients included in imputation model.
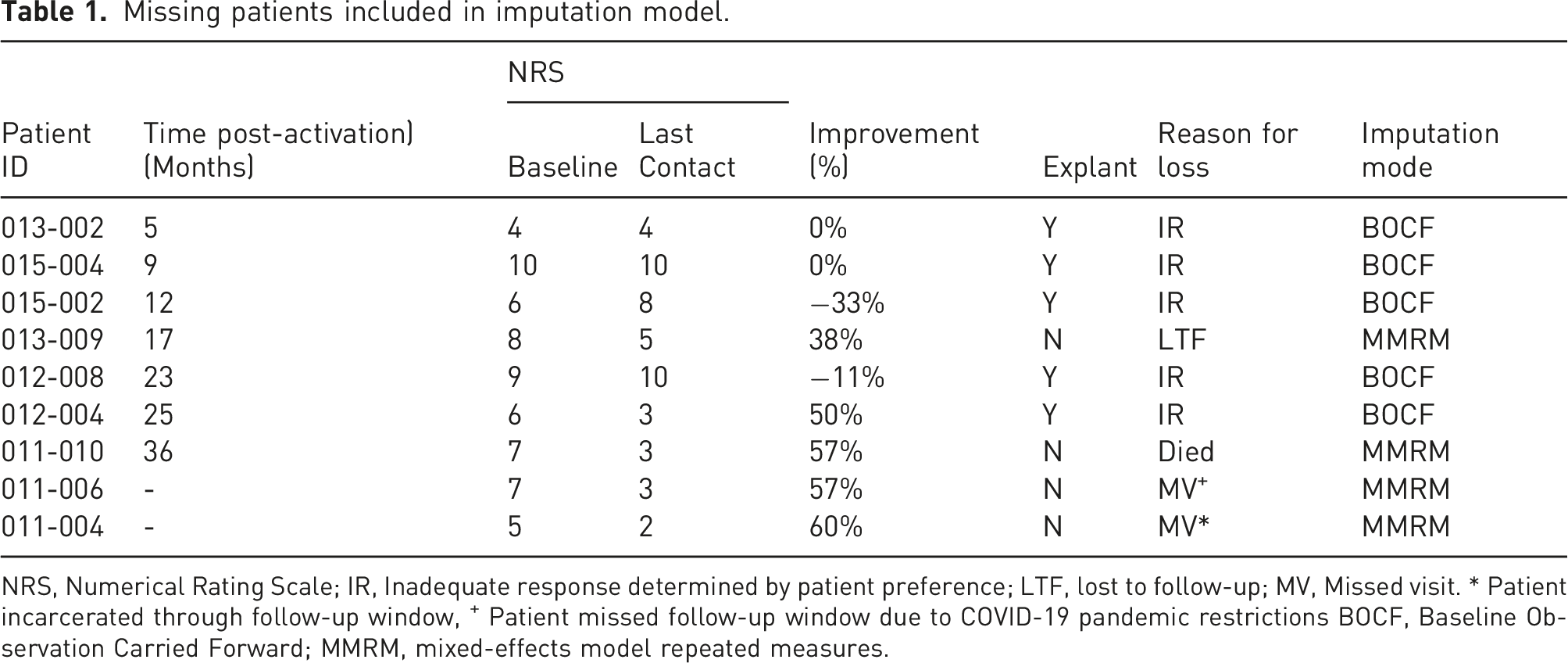
NRS, Numerical Rating Scale; IR, Inadequate response determined by patient preference; LTF, lost to follow-up; MV, Missed visit. * Patient incarcerated through follow-up window, + Patient missed follow-up window due to COVID-19 pandemic restrictions BOCF, Baseline Observation Carried Forward; MMRM, mixed-effects model repeated measures.
Baseline observation carried forward (BOCF), was used for participants withdrawn for reported lack of efficacy at any time. For those withdrawn or missing data for other reasons (death, unavailable or otherwise lost to follow-up) the mixed-effects model repeated measures (MMRM) approach was used to provide implicit imputations of missing data for continuous outcomes. 18 To evaluate mean changes from baseline, 95% confidence intervals and adjusted paired t-tests derived from MMRM contrasts were used. Two-sided p-values less than 0.05 were considered statistically significant.
Results
Patient demographics, disposition and follow-up
Forty-two patients were enrolled in the study and implanted with the device. The average age was 47.2 ± 11.0 years and 60% of patients were male. Mean duration of pain was 13.7 ± 10.2 years from the index event/onset. The patient disposition through to the 3-year follow-up is document in Figure 2 and the results of 33/42 patients are reported at this time point as the completer cohort. As previously reported 3/42 patients failed to complete year 1 and 2/42 patients year 2.
15
In this report four patients failed to complete year 3. Of these four patients one patient died from unrelated causes, one patient elected to have the device explanted, another was incarcerated at the time of follow-up and finally one patient was unable to attend due to COVID-19 restrictions. Patient disposition to three years post activation.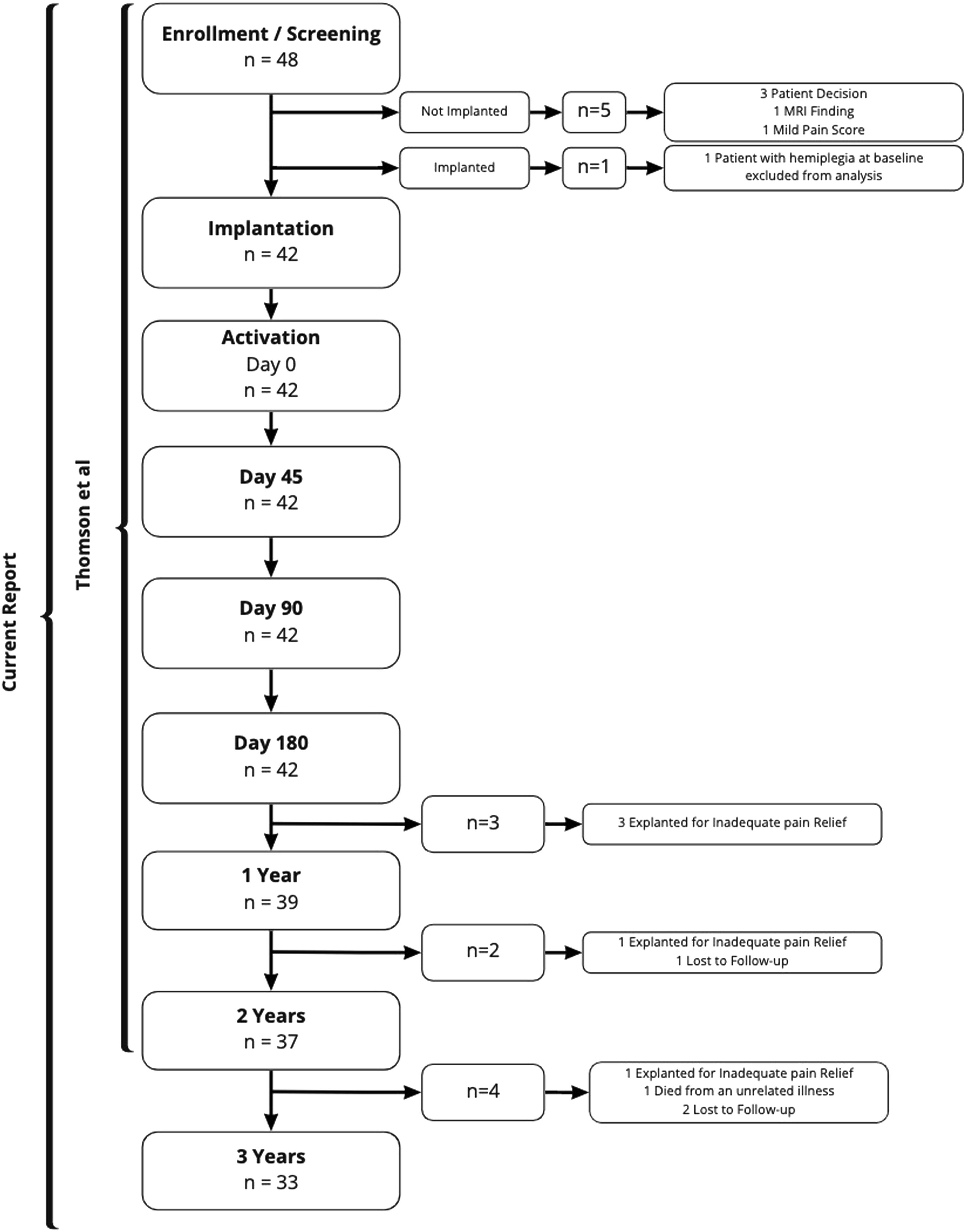
Patient demographics.
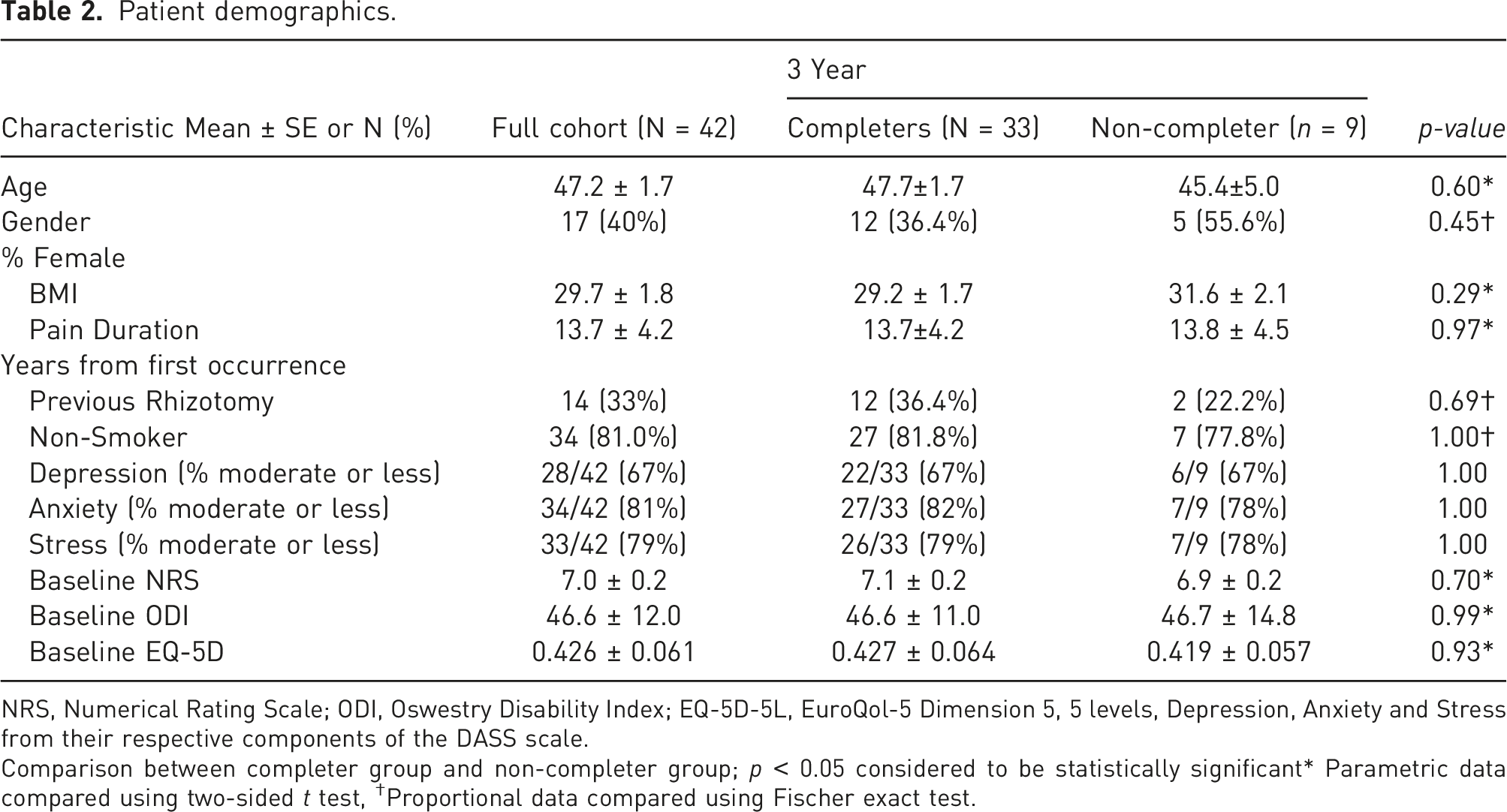
NRS, Numerical Rating Scale; ODI, Oswestry Disability Index; EQ-5D-5L, EuroQol-5 Dimension 5, 5 levels, Depression, Anxiety and Stress from their respective components of the DASS scale.
Comparison between completer group and non-completer group; p < 0.05 considered to be statistically significant* Parametric data compared using two-sided t test, †Proportional data compared using Fischer exact test.
Clinical outcomes
Patients in this cohort presented with severe chronic low back pain (NRS = 7.0 ± 0.2) and severe disability (ODI 46.6 ± 12.0). The health-related quality of life was also severely impacted at baseline (EQ-5D 0.426 ± 0.061).
Changes in pain, disability and quality of life at 3-year follow-up (Figures 3(a)–(c)) demonstrated a statistically significant improvement between baseline and 1, 2 and 3 years. After 3 years of therapy, average NRS scores had reduced to 2.7 ± 0.3 and mean ODI score to 26.0 ± 3.1 while EQ-5D-5L index improved to 0.707 ± 0.036. Imputation for missing data increased these values marginally but improvements remained both substantial and statistically significant. Longitudinal analysis showed that the improvements between 1 and 3 years were statistically significant, which is consistent with the restorative mechanism of action. Mean ratings over time for (a) low back pain NRS, (b) Oswestry disability index and (c). EQ-5D-5L index. Error bars represent the standard error of the mean.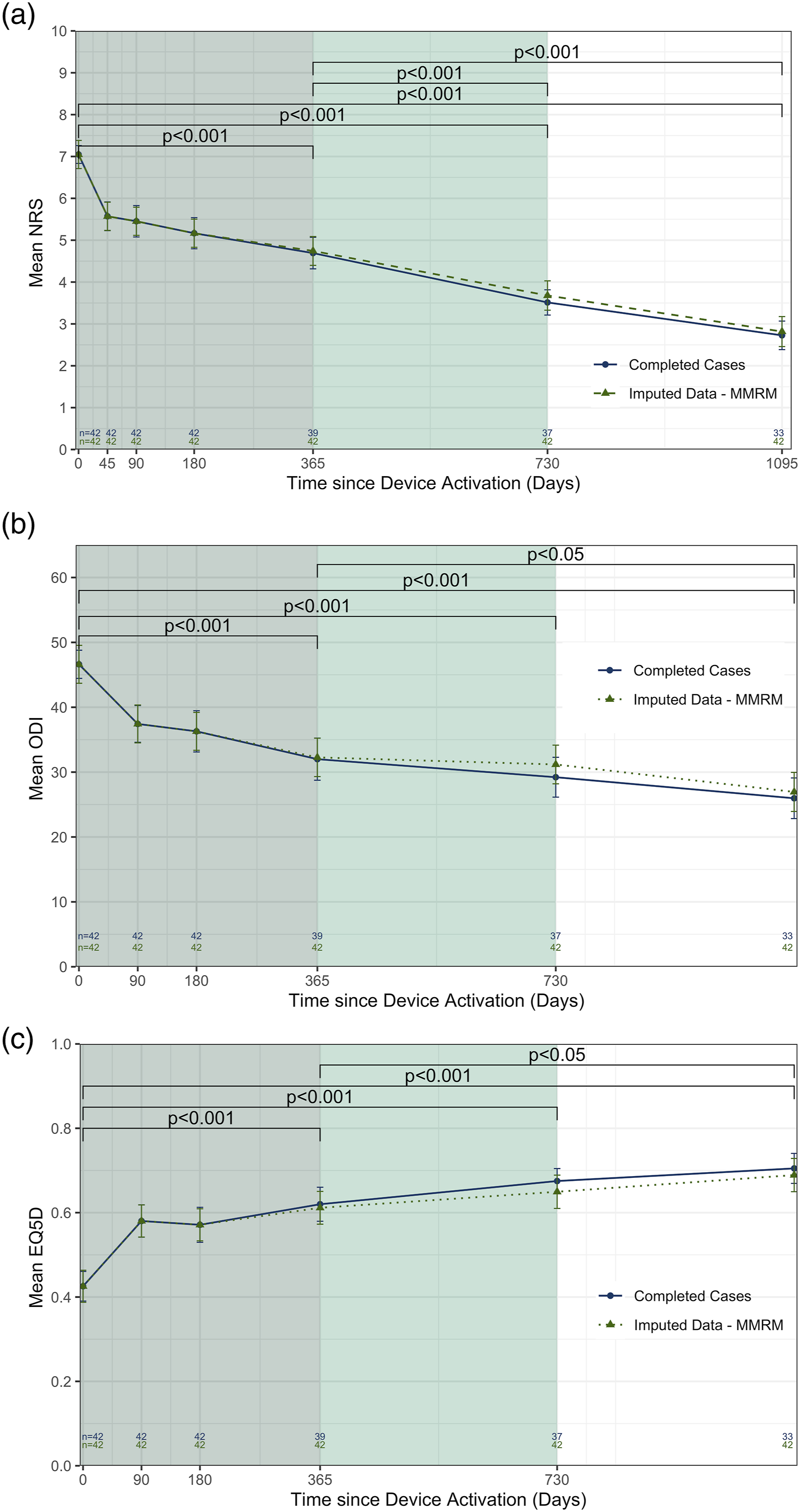
The proportion of patients deriving moderate (>30%) and substantial (>50%) improvements in pain (Figure 4(a)) and 15- or 20-point ODI improvements (Figure 4(b)) also shows accrual of benefit over time. By 3 years 70% of patients had reached the remitter threshold reporting mild to negligible pain (NRS ≤3) and 55% improved by more than 15 point on ODI. Response rates at common clinical importance thresholds for (a) NRS (reduction ≥30% and 50%, and absolute NRS ≤ 3 cm) and (b) ODI (reduction of ≥15 points and ≥20 points).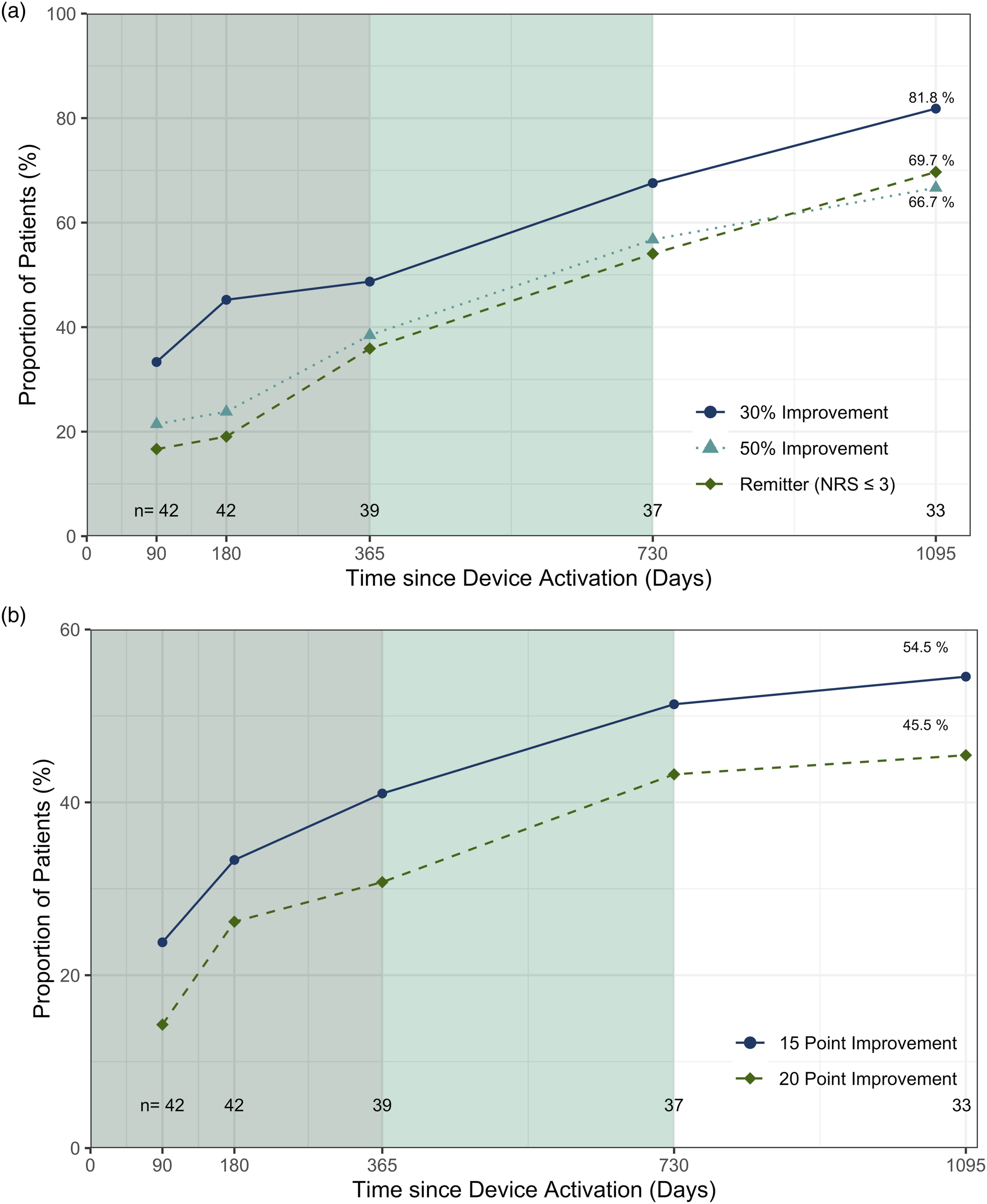
When the PROMS are combined, 75% of patients had a substantial or profound improvement in NRS (>50% improvement) and/or ODI (>20 point improvement). By 3 years patients reported a mean EQ-5D index was approaching an age matched UK population normal value of 0.861.
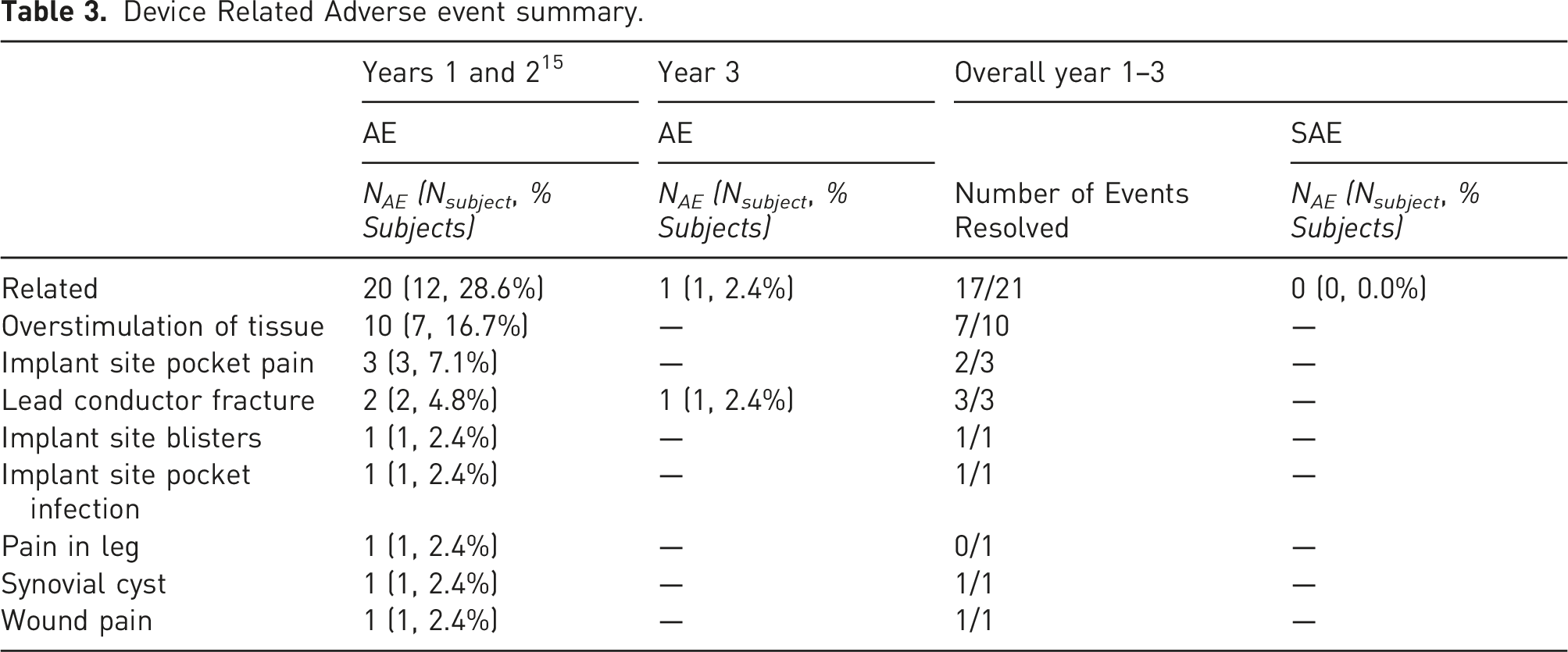
Safety outcomes
Discussion
The ReActiv8-A PMCF Study aimed to gather data on the performance and residual risks of ReActiv8 in the standard clinical practice post-approval/CE Mark. There were no specific inclusion or exclusion criteria beyond the regulated instructions for use. As such, this study demonstrates results that may be generalisable to clinical practice.
To contextualise the relevance of this therapy it is important to first consider the typical patient participating in this study, with an average age of 47 years and an average 14-year history of low back pain. Over this time, patients have tried and failed a variety of treatments, including physiotherapy, anaesthetic injections, radio-frequency neurolysis, opioids and psychological counselling. In many patients, these treatments are helpful, but it is well documented that there is a significant proportion where the effect is negligible or there is recurrence. When they fail the usual course is either to continue with ineffective therapies with questionable long-term safety when repeated or maintained or they receive a well-intentioned but poorly indicated fusion. The high levels of pain (NRS = 7), severe spine related disability (ODI = 46.6) and poor health-related quality of life (EQ-5D = 0.426) are a testament to the severity and chronicity of this patient cohort.
Patients consistently improved across the three major outcome domains over the 3-year period of follow-up, with final NRS of 2.7 ± 0.3, ODI of 26.0 ± 3.1 and EQ (5D) of 0.707 ± 0.036 and values obtained by missing data imputation within 5% of these (Figures 2(a)–(c)). These changes correspond to an average pain reduction of 61% of NRS, with 70% being classified as remitters and an average ODI reduction of 20 points or 44% of baseline. Additionally, the average 3-year EQ-5D improved from a severely disabled state to one approaching a population normal for UK adults.
Alternative therapies
Despite innumerable high quality clinical trials, the efficacy of physical therapy for CLBP is difficult to understand due to the heterogeneity of both the intervention and the patient population. An extensive Cochrane review for ‘Exercise therapy for chronic low back pain’ 19 concluded receiving exercise therapy resulted in small improvements in pain and disability at 3 months compared to no treatment, usual care or placebo but confidence was limited by the variety of ways exercise interventions are applied by clinicians and adopted by patients. Longer term follow-up (>12 Months) suggested that outcomes were no better when compared to other conservative strategies (−5.22 [−11.27, 0.84]) (Mean Difference [95% CI]). Regardless, the low complication rates, costs of intervention and almost ubiquitous access to at least some level of physical therapy it remains a frontline treatment for mechanical low back pain.
Radio-frequency ablation of the medial branch is another commonly applied management strategy in this patient group. A recent large clinical cohort 20 of 500 consecutive patients tracked longitudinal response rates in well selected RFA patients. They showed non-response rates 28% (135/482) in the first 30 days, 38% (195/490) at 90 days, 56% (269/484) at 6 months and 76% (382/500) by 1 year, suggesting that any clinical benefit wanes rapidly. This is consistent with the Cochrane Review of radiofrequency denervation for chronic low back pain 21 that concludes from a sample of 23 RCTs (n = 1309), 13 of which (56%) had low Risk of bias, there was no high quality evidence that RFA provides short-term pain relief for CLBP patients and that the lack of evidence of short-term effectiveness undermines the clinical plausibility of intermediate-term or long-term effectiveness.
The current evidence suggests that alternative therapeutic options for this population tend to provide either limited improvement or outcomes that are not durable.
Study strengths and limitations
At the 3-year timepoint, nine patients had left the study or missed follow-up appointments for various reasons. The completer analysis of the remaining 33 patients was consistent with conservative statistical imputation, providing confidence in the results and conclusions presented. An important consideration of this study was the more generalisable inclusion and exclusion criteria than those more closely aligned with the device instructions for use. This resulted in a more heterogenous cohort than may traditionally be enrolled in a clinical trial. This may also be a strength showing that this therapy is applicable to a broad patient group and the consistency with the 2- and 3-year data from the ReActiv8-B RCT gives confidence that real-world patient selection can achieve good clinical outcomes.
Conclusion
The ongoing follow-up of this post market cohort continues to demonstrate that restorative neurostimulation provides a statistically significant, clinically meaningful and durable response across pain, disability and quality-of-life scores for patients suffering chronic mechanical low back pain that has been refractory to conventional management. The safety of the procedure is favourable compared with other implantable neurostimulation devices such as spinal cord stimulators. Under conventional management strategies these patients accrue significant healthcare costs over the long course of their symptoms. Therapeutic options that provide durable relief by restoring function rather than palliating or masking pain perception should be considered when the right patient can be identified.
Footnotes
Acknowledgements
Editorial assistance in the preparation of this article was provided by Ben Goss, PhD (Mainstay Medical). Statistical and data management support was provided by Teresa Yurik, MS (NAMSA) and trial management by Diane Burnside, BS (Mainstay Medical) and Julian Mathew, MSc. (Mainstay Medical) The authors and sponsor would like to thank the participants in this study.
Author contributions
ST and SE were responsible for drafting the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript. SJT wrote the first and revised submissions. SE critically appraised and approved the submissions. All authors contributed patients to the trial and approved the submitted article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article: ST: Consulting agreement: Boston scientific, Galvani Bioelectronics, Mainstay Medical, Saluda Medical. Institutional research grants: Boston scientific, Mainstay Medical, Saluda Medical. *GV: Consulting Agreement: Abbot, Nevro Corp, Boston Scientific and Nalu Medical. *MS: Consulting Agreement: Nevro. *SL-J: Consulting Agreement: Boston Scientific, Medtronic and Nevro Corp. Institutional research grants from Boston Scientific, Medtronic, Nevro Corp, Abbot and Saluda Medical. *SE: Consulting Agreements Mainstay Medical and Medtronic. Institutional research grants Medtronic. Other from Abbott. AW and RC report no COI.
Ethical approval
The study protocol was reviewed and approved by a central ethics committee (NHS Health Research Authority North East—York Research Ethics Committee IRA’s project ID number 149412).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Mainstay Medical.
Informed consent
Written informed consent was obtained from the patients for their anonymised information to be published in this article.