Abstract
Background and aim:
The RADICAL trial has been funded by the National Institute for Health Research (NIHR) to evaluate the clinical and cost-effectiveness of radiofrequency denervation (RFD) for low back pain. Recommendations have been published which aim to standardise selection of patients and RFD technique. However, it is important to ensure these recommendations are acceptable to clinicians within the context of the trial. The aim of this work was to develop standardised criteria for the trial entry and RFD technique for implementation within the RADICAL trial.
Methods:
Fourteen pain clinicians completed a survey, which involved reviewing the current recommendations and indicating whether they disagreed with any of the recommendations and if so why. Responses were collated and presented at a half-day workshop with 14 attendees. During the workshop, the National Low Back and Radicular Pain Pathway (NLBRPP) guidelines for patient selection and an article by Eldabe and colleagues presenting recommendations on the RFD technique were reviewed. Attendees discussed whether each component of the recommendations should be mandatory, mandatory with alteration or clarification or optional within the RADICAL trial.
Results:
Attendees agreed during the workshop that 5 of the 10 criteria for patient selection described in the NLBRPP should be mandatory within the RADICAL trial. Three were agreed as mandatory criteria but required further clarification, one of which involved defining a positive response to a diagnostic medial branch block as ⩾60% pain relief. Two criteria had optional components. After reviewing the recommendations on the RFD technique from Eldabe and colleagues, seven components were agreed as mandatory, three were mandatory with alterations and three were optional.
Conclusion:
When evaluating complex interventions, such as RFD, it is important to ensure agreement and clarity on the clinical protocol, so that the intervention can be reproduced, if found to be effective.
Background
Low back pain (LBP) is the leading global cause of years lived with disability 1 and is associated with high personal, societal and economic burden. 2 Non-surgical interventions for LBP recommended by The National Institute for Health and Care Excellence (NICE) are self-management, exercise, psychological therapy, combined physical and psychological programmes and non-steroidal anti-inflammatory drugs. LBP can sometimes be localised to the facet joints and periarticular structures supplied by the medial branches of the primary dorsal rami. NICE guidelines recommend that patients with clinical features suggestive of pain with a facet joint component and who do not experience an improvement in moderate to severe pain symptoms with conservative management can be offered a diagnostic medial nerve (to the facet joint) branch block (MBB) with local anaesthetic. Patients who respond positively to the MBB can undergo radiofrequency denervation (RFD), a minimally invasive procedure which aims to reduce pain by interrupting the pain signal from the medial branch nerves in the spine to the brain by denaturing the target nerve. RFD is endorsed by NICE and implemented in clinical practice through inclusion in the National Low Back and Radicular Pain Pathway (NLBRPP) and British Pain Society Low Back and Radicular Pain Pathway.3,4
Based on Hospital Episode Statistics data, there were 13,046 RFDs of the lumbar facet joints performed in the NHS in 2017–2018. 5 However, there is uncertainty regarding the effectiveness of RFD due to a lack of high quality evidence. 6 A number of systematic reviews on the effectiveness of RFD have been published with conflicting findings and interpretation.7–11 A Cochrane review, published in 2015, concluded that there was no high-quality evidence that RFD provides pain relief for patients with chronic LBP. 6 The recent Myocardial Ischemia and Transfusion (MINT) trial from the Netherlands concluded that RFD combined with an exercise programme was not superior to an exercise programme alone. 12 The MINT trial was commissioned by the National Health Care Institute and aimed to reflect usual practice in RFD, which included variability in technique. However, this trial was criticised for a number of reasons, including wide variation in RFD operator protocols, resulting in a suboptimal technique and inconsistent delivery.13–17
The recent call to action on LBP in The Lancet recommends that research to evaluate treatments without supporting evidence should be commissioned to contribute to the development of evidence-based care pathways to enable people to receive appropriate, effective treatments. 18 NICE recently made a research recommendation that high-quality evidence on the clinical and cost-effectiveness of RFD compared to placebo RFD in the NHS is needed to inform clinical practice guidelines and commissioning of care. 3 This subsequently informed a commissioned call for research from the National Institute of Health Research Health Technology Assessment Programme in 2018 (HTA 18/49). The RADICAL trial was funded in response to the call and will provide definitive evidence on the clinical and cost-effectiveness of RFD to inform NHS service provision. The RADICAL trial is a 20-centre, double-blind, parallel group, superiority randomised controlled trial of RFD versus placebo, with an internal pilot phase, embedded qualitative research and cost-effectiveness analysis. Further information on the trial design can be found on the funder website (https://www.fundingawards.nihr.ac.uk/award/NIHR127457), and full details will be published separately in the study protocol.
Prior to the commencement of the trial, it was important to establish best practice in the RFD technique for use in the trial and ensure that this would be acceptable to participating clinicians. Recently, a consensus on the best practice technique for RFD, based on the Nath technique, 19 was reached among experts in the United Kingdom. 20 In addition, detailed guidance was published from the worldwide clinical community. 21 National guidance is available on the pathway to RFD and selection of patients through the NLBRPP. 4 However, guidance on the technique has not been tested among the wider clinical community, and the NLBRPP has not been fully implemented. The aim of this work was to develop agreement on standardised trial entry criteria and best practice RFD technique, guided by previous consensus documents, for implementation in the RADICAL trial.
Methods
Survey
The consensus articles on the RFD technique by Eldabe et al. 20 and Cohen et al. 21 and a copy of the NLBRPP were e-mailed to the 17 consultants who had expressed interest in participating in the RADICAL trial at 17 NHS Trusts. The clinicians were asked to review the guidance and complete a short online survey to determine their level of agreement with each piece of guidance. The survey asked responders to indicate if they disagreed with any of the recommendations in the three pre-circulated documents and, if they did, to state their concerns in a free-text box. Non-responders were sent a reminder. Responses were collated and summarised for presentation at the workshop.
Workshop
The 17 clinicians (or a representative on their behalf) and 2 patient representatives for the trial were invited to attend a half-day workshop in Southampton in November 2019. The meeting was facilitated by Dr Cathy Price, an experienced pain clinician and Clinical Lead for the RADICAL trial. The meeting was audio-recorded with participants’ verbal consent. The meeting began with a presentation giving an overview of the RADICAL trial design by the Chief Investigator of the trial (V.W.), followed by a presentation from C.P. summarising the collated survey responses. The attendees were then introduced to the structured process that would be used for agreeing on the standardisation of patient selection and RFD technique within the RADICAL trial. This process was based on an adaption of the published framework for standardising surgical procedures which describes each step of a surgical procedure as prohibited, mandatory or optional. 22 The ‘prohibited’ option was not used during te workshop because the focus was on published guidance. It was decided after the survey to only discuss one guidance document on the RFD technique during the workshop to allow time for in-depth discussion. The article by Eldabe et al. was chosen, as there was less disagreement with these recommendations in the survey. Therefore, the international guidance from Cohen et al. was included in the survey but not the workshop. Paper copies of two of the three documents previously circulated were distributed to attendees: the NLBRPP recommendations for the selection of patients for RFD 4 and the recommendations given by Eldabe et al. 20 on the RFD technique.
In small groups, the attendees discussed the two guidance documents and decided whether all the components of the two documents should be mandatory or optional. In a facilitated whole group discussion, attendees reviewed and discussed each component from the guidance documents until participants agreed on whether it should be mandatory or optional in the RADICAL trial. For some components, it was decided that alterations or additional clarification were required. The approach taken to reach agreement during the workshop was informal and flexible with facilitated group discussion to engage clinicians and encourage pragmatic decisions to be taken to allow attendees to reach agreement on trial processes.
During the discussion about the criteria for selecting patients for RFD, attendees also reviewed anonymised and aggregated data shared by the MINT trial team relating to the distribution of participants’ pain relief response to MBB. A review of these data informed the decision about the percentage pain relief that would define a positive response to an MBB for the purposes of the RADICAL trial. The cut-off for defining a positive response to MBB was chosen pragmatically, balancing the importance of the following: including patients likely to benefit from RFD; reflecting current NHS practice; and providing a large pool of patients to be invited to take part in the trial. Workshop minutes were written, and the agreed outcomes were summarised into tables.
Results
Survey
Fourteen clinicians responded to the survey (82%). Seven clinicians disagreed with one or more aspects of the United Kingdom consensus guidelines published by Eldabe et al. 20 Aspects of disagreement included the following: 0.5 mL of levo-bupivacaine 5 mg/mL as the preferred solution for MBB (n = 1); the need for intravenous (IV) access to be routinely established (n = 1); use of an 18G RF needle (n = 1); use of a curved needle (n = 2); no sensory testing (n = 1) and lesioning at 80°C for 120 seconds (n = 3). Other issues raised included the lack of a definition for a positive response to MBB (n = 1) and lack of clarity on the motor testing (n = 1).
The international guidance published by Cohen et al. 21 also raised disagreement, with seven clinicians disagreeing with aspects of the guidance. These included the following: requirement of conservative treatment before RFD (n = 1); definition of a positive response to MBB as ⩾ 50% pain relief (n = 2); MBB should be performed with a volume ⩽ 0.5 mL (n = 1); use of an 18G RF needle size (n = 1); inclusion of sensory testing (n = 1); continued use of anticoagulants in the peri-procedure period (n = 1); complications in patients with pacemakers or implantable cardioverter defibrillators (n = 1) and performing repeat RFD if pain relief lasts ⩾ 3 months (n = 3).
Four people disagreed with one or more components of the NLBRPP. Areas of disagreement related to the recommendations for MBB without steroids (n = 1); entry criteria for RFD of no sacroiliac joint pain (n = 1); use of physical examination for lumbar joint pain (n = 1); use of the EQ-5D, Oswestry Disability Index and pain visual analogue scale (VAS) to measure outcomes (n = 2) and performing repeat RFD if pain relief lasts 16 months or more (n = 1).
Workshop
The workshop had 14 attendees: the facilitator and Clinical Lead for the RADICAL trial (C.P.), RADICAL trial Chief Investigator (V.W.), nine consultant pain management specialists working in NHS centres across the United Kingdom, two patient representatives and one academic representative from a pain management centre.
Criteria for the selection of patients for the RADICAL trial
The group decision on whether each criterion for the selection of patients for RFD from the NLBRPP should be mandatory or optional within the RADICAL trial is provided in Table 1. It was agreed that of the 10 criteria, 5 criteria should be mandatory and 3 should be mandatory but with further clarification. The eligibility criterion of patients having to have had insufficient improvement despite previous conservative management was expanded on two points: (a) to clarify that patients should have tried a minimum of one conservative management option that failed to improve symptoms and (b) to acknowledge that combined physical and psychological programmes are not available to patients in all areas of the United Kingdom and are therefore an optional component with respect to the entry criterion. Workshop participants also agreed that a positive response to MBB with 1 mL or less of bupivacaine 0.5% at each level of the spine (no steroids) was mandatory but with further clarification that a positive response should be defined as ⩾60% pain relief at 3 hours after the procedure. In their routine clinical practice, six of the clinicians defined a positive response to MBB as ⩾50% pain relief and four of the clinicians used ⩾80%. There was a strong preference for the threshold for defining a positive response to be higher than 50% to ensure that patients most likely to benefit from RFD were selected for the RADICAL trial. Setting the threshold at ⩾80% was discussed; however, review of the MINT-aggregated data showed that this would greatly reduce the number of patients eligible for the trial. The criterion of ‘no sacroiliac joint pain’ was considered mandatory, although clarification was added regarding the assessment of sacroiliac joint pain using a provocation test. The group agreed that assessment could be based on a provocation test or clinical suspicion due to the limited reliability and validity of provocation tests. 23
National low back and radicular pain pathway recommendations on the selection of patients for radiofrequency denervation.
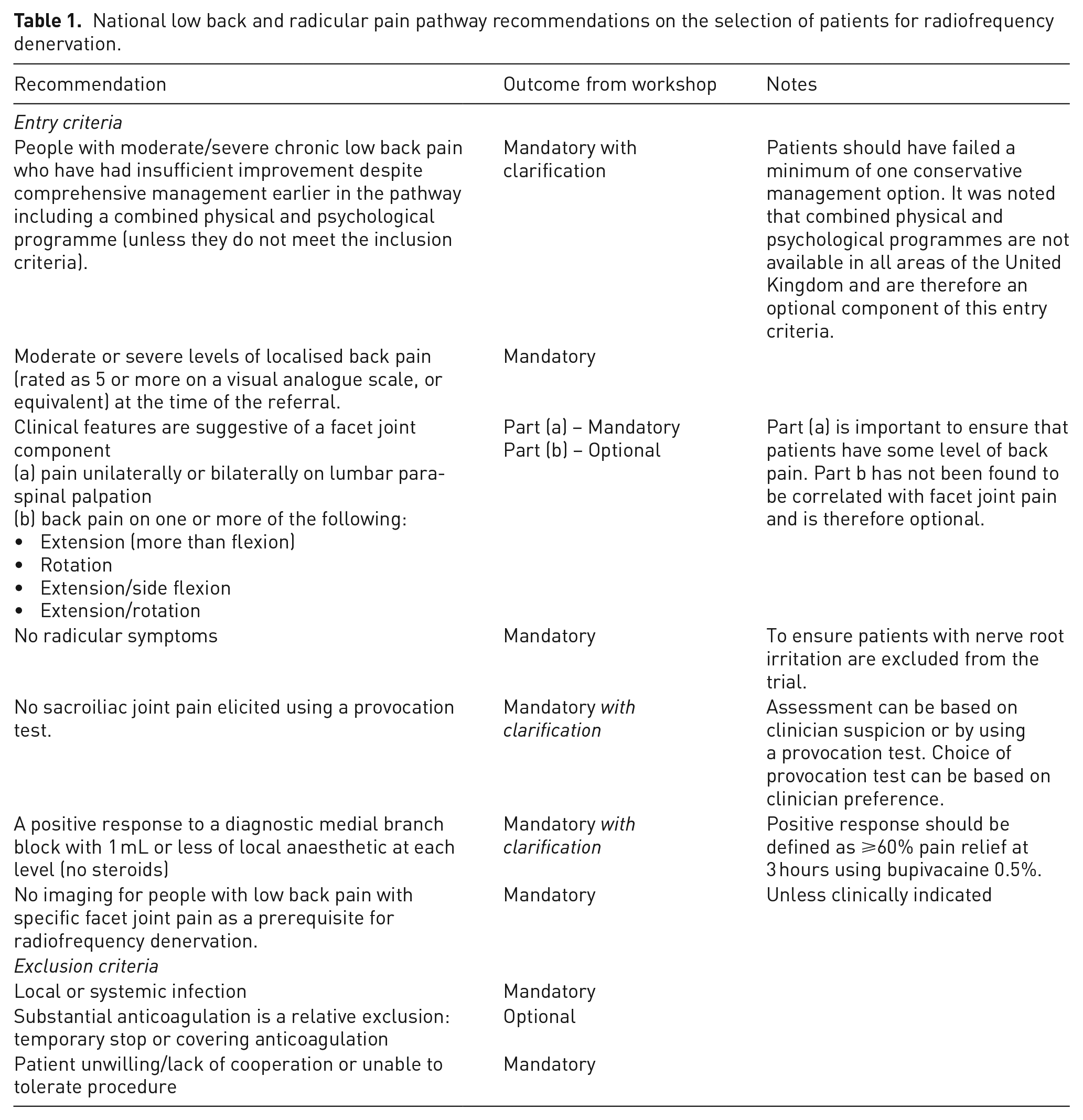
Two of the NLBRPP criteria for selecting patients were considered as optional or containing an optional component for the RADICAL trial. Experiencing ‘increased back pain on one or more of the following: extension (more than flexion), rotation, extension/side flexion or extension/rotation’ was considered optional because evidence suggests a lack of correlation between these measures and pain arising from the facet joints. 24 Substantial anticoagulation as an exclusion criterion was also considered optional.
Technique for the RFD procedure in the RADICAL trial
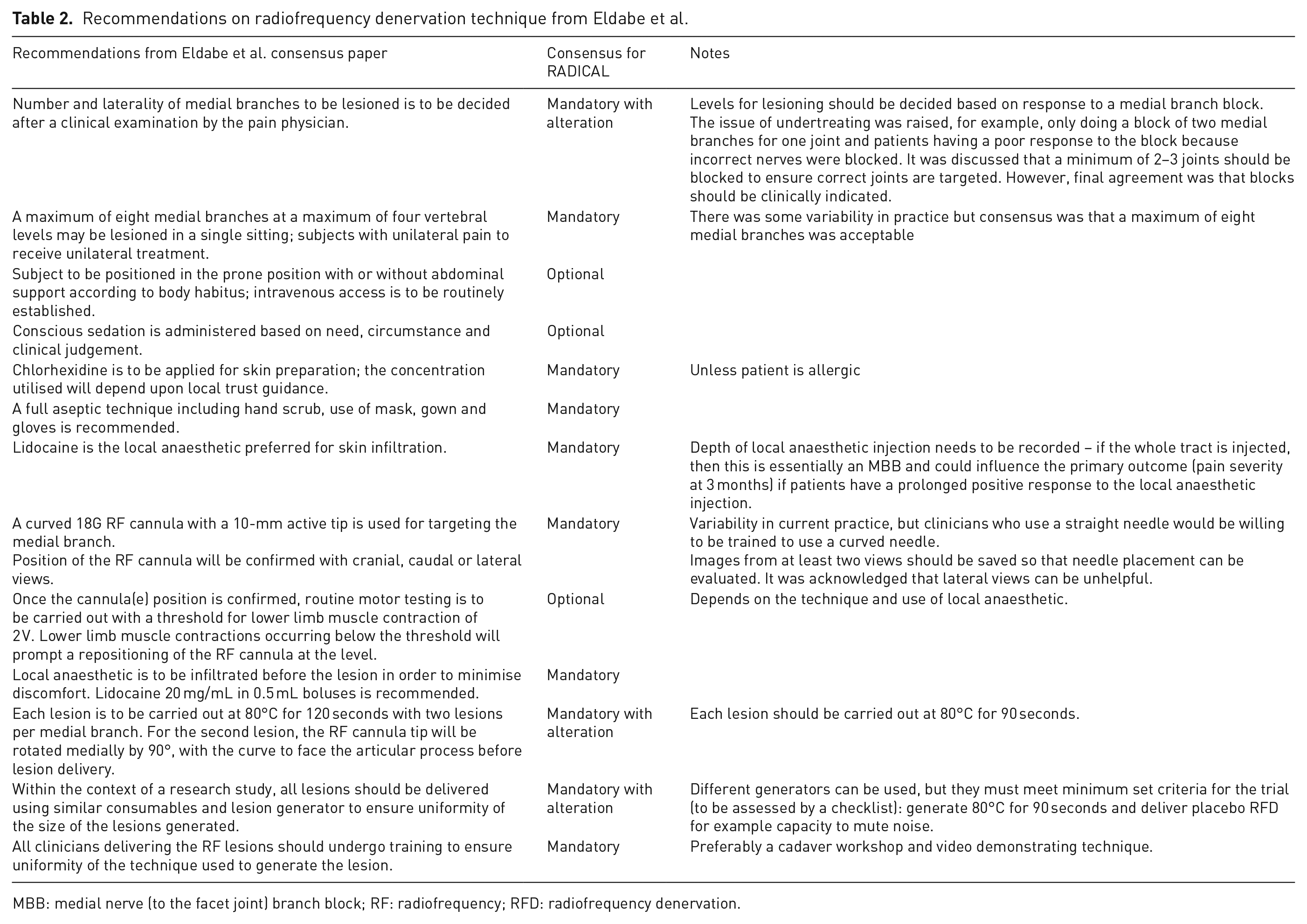
A summary of the group decision on whether each component of the RFD procedure recommended by Eldabe and colleagues (based on the Nath technique) should be mandatory or optional within the RADICAL trial is provided in Table 2. Seven components were agreed as mandatory, three were mandatory with alterations and three were optional. The first mandatory recommendation that required alteration was ‘number and laterality of medial branches to be lesioned is to be decided after a clinical examination by the pain physician’. It was decided that the levels of lesioning should be decided based on response to an MBB, rather than clinical examination by a pain physician. The other two components that were mandatory with alterations were related to the lesioning duration and the lesion generator. The recommendation that RFD should be delivered at maximum temperature (80°C) for 120 seconds was discussed in light of the considerable variability in current practice of the group with respect to the duration of RFD (varying from 60 to 120 seconds for each lesion) and inconsistent evidence from studies on optimal duration.25,26 A pragmatic decision was made to alter the recommendation, so that RFD must be carried out for 90 seconds, since all the participating pain clinicians were willing to implement this in the trial. The second altered recommendation was related to lesions being delivered using similar consumables and lesion generators to ensure uniformity of lesion size. Due to existing supplier contracts at hospital sites, it would not be possible for all sites to use the same generator in the RADICAL trial. Therefore, it was agreed that different generators could be used, but they must meet minimum set criteria for the trial. These will be assessed by a checklist to ensure that the generator can deliver RFD at 80°C for 90 seconds and deliver placebo RFD, for example, have the capacity to mute noise associated with RFD.
Recommendations on radiofrequency denervation technique from Eldabe et al.
MBB: medial nerve (to the facet joint) branch block; RF: radiofrequency; RFD: radiofrequency denervation.
Three components of the RFD technique recommendations were agreed as optional for the RADICAL trial: placing patients in the prone position with intravenous access; administration of conscious sedation (considered to depend on clinical need, judgement and local practice) and routine motor testing (considered to depend on the technique and the local anaesthetic used).
Discussion
There is considerable variation in the RFD technique within UK clinical practice, 20 and it is important that a pragmatic trial employs a consistent and defined approach to technique, based on best practice. Previous work by Eldabe et al. 20 defined best practice in the RFD technique using a rigorous process involving reviewing relevant literature and guidelines and conducting a survey and a one-day consensus meeting with 24 clinical experts. Our approach was to build on this work to assess the acceptability of these recommendations, alongside guidance from the NBLRPP on selecting patients for RFD, 3 to avoid constraining practice unnecessarily while ensuring that best practice is followed. To achieve this, we conducted a survey and workshop with clinicians and patient representatives. Through this process, we reached agreement on the entry criteria and clinical protocol for RFD for implementation in the RADICAL trial. Using a standardised and consistent approach in the trial, based on best practice, will overcome some of the criticisms of the MINT trial 17 and facilitate the uptake of findings into clinical practice. The principles of the RFD technique to be used in the RADICAL trial are the same as the Spine Intervention Society technique and therefore reflects best practice. 27
It is important to acknowledge the strengths and limitations of this work. There are recognised and structured methods that can be used for the process of gaining consensus, including the Delphi technique and the nominal group technique. 28 However, rather than aiming to gain formal consensus, this work focused on assessing the acceptability of the best practice technique defined by Eldabe and colleagues and selection criteria recommended by the NBLRPP to optimise implementation in a trial setting. A strength of the work is that this process was guided by the use of a framework for standardising surgical procedures which describes each step of a surgical procedure as prohibited, mandatory or optional. 22 Another strength of the work was that funding for the workshop was provided as part of the National Institute for Health Research (NIHR) grant funding the trial, and therefore the work was independent from industry. A potential limitation was the small number of clinicians who attended the workshops. Although the clinicians were drawn from a range of types of services to reflect services in the United Kingdom, from large teaching hospitals to smaller district general hospitals, and they were encouraged to discuss the workshop with colleagues in their department, it is acknowledged that they may not have been representative of the UK clinical opinion, which likely limited the generalisability of the results. However, they provided a good representation of hospitals to be included in the RADICAL trial.
In conclusion, when evaluating complex interventions, such as RFD, it is important to test for agreement on the clinical protocol so that the intervention can be reproduced in the context of a trial or implemented into usual practice if found to be effective. 29 We will use these outcomes to inform the development of a detailed protocol for the RADICAL trial.
Footnotes
Acknowledgements
The authors acknowledge the wider RADICAL trial team for their input into trial design, Dr Ester Maas and collaborator Professor Raymond Ostelo for sharing anonymised data from the MINT trial and Dr Khalid Alaib, Dr Dalvina Hanu-Cernat and Dr Mike Williams for attending the workshop.
Conflict of interest
The authors declared the following potential conflicts of interest with respect to the research, authorship and/or publication of this article: C.P., B.C.R., A.A., M.B., R.C., T.M., M.S., B.V., M.G., K.P. and V.W. have no conflicts of interest to declare. G.B. is on the advisory board, received Honorarium and Educational/Research Grant from Abbott, Boston Scientific, Nevro Corporation and Nalu Medical.
Contributorship
C.P., B.C.R. and V.W. designed the work. C.P., V.W., A.A., M.B., G.B., R.C., T.M., M.S., B.V., M.G., and K.P. were involved in data collection. W.V. drafted the manuscript, and all authors reviewed the final version of the manuscript.
Ethical approval
This work was not a research project and therefore did not fall under the remit of an ethics committee.
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This project was funded by the National Institute for Health Research (NIHR) (Health Technology Assessment programme (project reference NIHR127457)). The study was also supported by the NIHR Biomedical Research Centre at the University Hospitals Bristol NHS Foundation Trust and the University of Bristol. The views expressed are those of the authors and not necessarily those of the NIHR or the Department of Health and Social Care.
Guarantor
C.P. is the guarantor of the article.
Informed consent
The meeting was audio-recorded with participants’ verbal consent.
