Abstract
Cardiac catheterization for congenital heart disease (CHD) performed under fluoroscopic guidance still lacks definition and requires exposure to ionizing radiation and contrast agents, with most patients needing multiple procedures through their lifetime, leading to cumulative radiation risks. While fusion overlay techniques have been employed in the past to aid, these have been limited to a single plane, while interventions are traditionally performed under biplane fluoroscopy. We describe our initial experience performing cardiac catheterizations guided by an enhanced biplane GuideCCI system© (Siemens Healthcare, Germany) augmented by 3D magnetic resonance imaging and computed tomography modeling. Twenty-one children and young adults with CHD undergoing catheterization procedures between October 2019 and May 2021 were chosen based on their degree of complexity of cardiac anatomy. 3D stereolithography models were generated, overlayed, and displayed in real time, alongside angiographs in both planes on the screen during these procedures. We report successful implementation of this novel technology for performance of 26 interventions including stent placements, balloon dilations, vessel occlusion and percutaneous valve and transvenous pacemaker implantation all in patients with various complex cardiac anatomies. A statistically significant reduction in radiation and contrast use was noted for coarctation of the aorta stent angioplasty and transcatheter pulmonary valve replacement when compared with national benchmarks and local institutional metrics (with and without single plane overlay). No complications were encountered with the use of this technology. Use of a tracheal registration technique provided very good correlation in most cases. Operators preferred using biplane augmented catheterization over traditional fluoroscopy in patients with complex cardiac anatomy undergoing interventions.
Introduction
Traditionally catheter-based interventions in patients with congenital heart diseases (CHDs) have been performed using biplane X-ray fluoroscopic guidance. These X-ray based projectional images lack soft tissue definition and require exposure to ionizing radiation and potentially nephrotoxic contrast agents. The patients are frequently children or young adults in whom the cardiovascular anatomy is variable and complex. Children and young adults in general have shown to be more susceptible to the effects of ionizing radiation and have a longer life-expectancy over which this risk is expressed.1–3 There is well-demonstrated evidence of long-term chromosomal damage children with CHD exposed to ionizing radiation procedures, with the risk increasing in those needing repeated examinations through their lifetime due to a high cumulative dose.4,5 More complex cardiac interventional procedures are now being performed with advancements in available hardware, with a consequent higher radiation dose exposure. Thus, radiation dose reduction is paramount. A second important problem is procedural failure associated with more complex percutaneous interventions, in which case surgery is often required for these patients.
Augmentation of x-ray-guided interventional cardiac procedures using high-resolution three-dimensional magnetic resonance imaging (MRI) and computed tomography (CT) modeling has been developed and exploited commercially for this application.6,7 This potential has not been fully realized for CHD interventional procedures; the main obstacle to implementation being that MRI augmentation is available on single-plane systems, whereas CHD intervention is invariably performed using a biplane system due to the complexity and variability of the anatomy. Recently, an enhanced biplane system has been developed with advanced X-ray control, institution of an effective augmented virtual display and respiratory motion correction. The potential advantages for such a system would be a reduction in radiation usage, reduction in procedure time by efficient guidance, and the reduced use of nephrotoxic x-ray contrast agents. We performed a study to assess the feasibility and benefits of this new technology for children and young adults undergoing various types of CHD catheter-based interventions, and to quantitate the hypothesized reduction in radiation and contrast metrics.
Materials and methods
Patients with CHD between the ages of 0 and 50 years, undergoing vascular intervention using biplane X-ray fluoroscopy at the Children's Medical Center (Dallas, Texas, USA) were enrolled in this prospective study between October 2019 and February 2021. The study was approved by our Institutional Review Board (STU 092017-042), and all patients consented, with assent obtained (when appropriate) to participate in the study. Subjects included in the study were chosen to participate based on higher degree of complexity of their cardiac anatomy, and at the request of the interventional cardiologist. A sizable subset of patients undergoing a stent angioplasty of a coarctation of the aorta, or a transcatheter pulmonary valve placement (TPVR) were included due to the availability of comparative metrics for these procedures and the complexity and higher radiation burden associated with the procedure (TPVR). No specific anatomy, interventions, or patient demographics were excluded from this study, except those who could not receive contrast due to severe renal dysfunction (estimated glomerular filtration rate <30 mL/min/1.73 m2) or those who had ferromagnetic implants that would preclude them from undergoing an MRI. Patients who had not already undergone any form of advanced imaging prior to catheterization (either a cardiac MRI or a cardiac CT) underwent a contrast-enhanced cardiac MR Angiography (CEMRA) [Phillips Ingenia 1.5T, Philips Healthcare, Best, NL] just before the procedure. Children under the age of 9 years underwent this MRI under the same general anesthesia encounter. Segmentation of the relevant anatomic structures was performed by postprocessing from the DICOM (Digital Imaging and Communications in Medicine) data using Materialise Mimics Research 21.0 [Materialise, Belgium]), after which 3D stereolithography (STL) files were created using 3-Matic Research 13.0 [Materialise, Belgium]. The airway (trachea and bronchi) was also segmented as shown in Figure 1. These 3D STL models along with DICOM data were exported into the Siemens biplane fluoroscopy system [Guide CCI system©, Siemens Healthcare, Erlangen, Germany] (Figure 2).

The use of a quick cardiac magnetic resonance imaging (MRI) protocol to generate cross-sectional Digital Imaging and Communications in Medicine (DICOM) data, which was volume rendered for a quick view of complex anatomy and also segmented into 3D STL models of relevant cardiac structures.
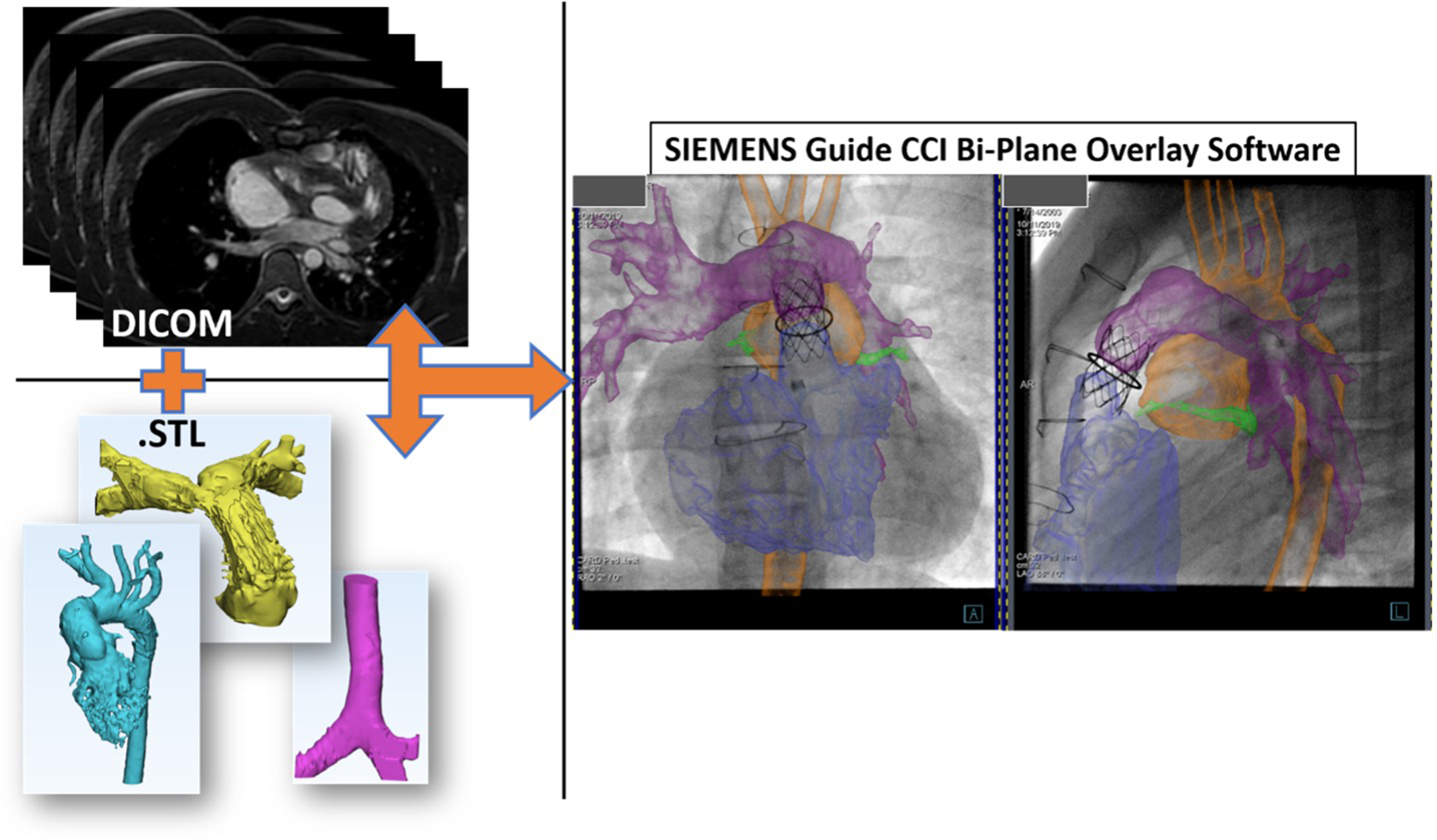
Stereolithography (STL) files exported along with Digital Imaging and Communications in Medicine (DICOM) datasets into the Siemens biplane overlay software which was displayed on an augmented reality display in both planes on the screen for the interventionalists alongside their routine fluoroscopic display.
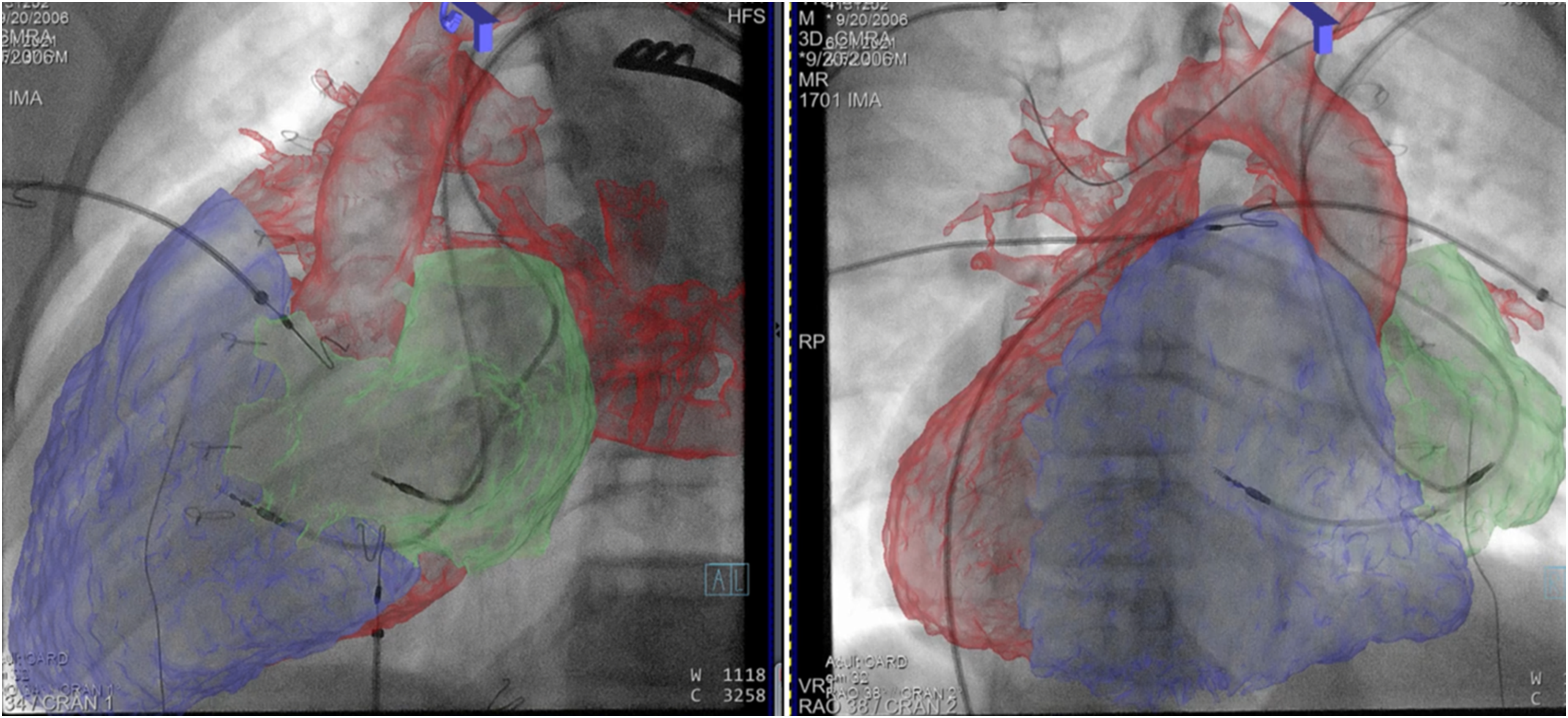
While the initial biplane scout fluoroscopic images were obtained to assess patient and camera position, the 3D model of the tracheobronchial tree was used as a reference landmark to align the digital data with the fluoroscopic data (“registration”) (Figure 3). In patients who underwent a cardiac MRI just prior to the procedure, radio-opaque markers were placed on the chest wall to serve as additional reference points for digital overlay. The overlay augmented fluoroscopic images were thus generated and displayed, in real-time, alongside angiographs in both planes on the screen during the procedure. The interventionalist had the option to view both the native fluoroscopic images and the augmented fluoroscopic models, side-by-side, on the same display. Respiratory motion compensation was enabled when useful. Patient demographics along with imaging, and catheterization and anaesthesia procedure data were collected and compared to local institutional and published national radiation benchmarks for the same age group and procedure. For patients who underwent a stent angioplasty for coarctation of the aorta, these data were also compared to published single plane overlay data for the same procedure.

Use of a 3D tracheabronchial model for registration of the digital data on the fluoroscopic data.
Statistical analysis was performed using SPSS 28.0.1 (IBM, USA) with an unpaired one-tailed t-test to compare radiation, contrast, and procedure metrics between national, single plane, and biplane overlay assisted catheterization data.
The Guide CCI system was developed at King's College, London, in conjunction with Siemens as an industry partner. The Monaco platform has an accurate and robust biplane overlay system developed by Siemens Healthcare, with an automatic device detection and tracking module to enable catheter detection and tracking, in conjunction with development of an advanced X-ray system and the development of a virtual guidance system. As this is a novel technology, minor technology upgrades were made to our current cardiac catheterization lab setup. This included adaptation of the current biplane fluoroscopic biplane system with an auxiliary workstation in the control room which would run the software (Guide CCI fusion system) and help a physician or a technologist setup the initial registration after importing the 3D STL models into the system, and the institution of an effective virtual display. The augmented display was mirrored to the preexisting large display screens that were already in use for the interventionalist.
Results
The Guide CCI biplane overlay system was successfully used in 21 patients undergoing cardiac catheterization. The median age of our study population was 8.8 years (ranging from 11 days to 19.5 years) and weight was 31.3 kg (weight range 3.5–97 kg), with a median body surface area of 1.1 m2 (range 0.2–2.1 m2). Further demographic and procedure data are outlined in Table 1. Ten patients had prior CT angiography, and six patients had prior cardiac MRI. Only five patients underwent a preprocedural CEMRA the same day as the procedure, with only one patient requiring preprocedural anaesthesia. Median time between cardiac imaging and the catheterization procedure was 29 days (IQR from 0 to 96 days). As listed in Table 2, 18 patients underwent a total of 26 biplane overlay assisted interventions. The spectrum of interventions included stent and balloon angioplasties, vessel and collateral coiling, and device occlusion, along with transcatheter pulmonary valve (including prestent) placement. Median and IQR values of catheterization data showed a total Dose Area Product (DAP) of 657 µGy.m2 (267,5447), with an absolute radiation dose of 70 Gy (11,194) in the anteroposterior plane (A plane), and 62 mGy (17,287) in the lateral plane (B plane). Median contrast dose usage was 3.8 mL/kg (2.4,4.8), and median total fluoroscopy time was 29 min for all procedures (11,42). Median procedure time was 137 min (96,174) and time from anaesthesia induction to anaesthesia stop was 205 min (150,274).
Patient demographics and catheterisation data for all procedures.

Overview of Guide CCI system cases to assist cardiac catheterisation diagnostics and interventions.

Table 3 specifically lists patients who underwent stent angioplasty of a coarctation in the aorta in whom a stepwise reduction in the total and body weight indexed DAP was noted (21 ÂμGy.m2/kg with biplane overlay) from the mean national benchmark (90 ÂμGy.m2/kg) and cases from our institution without overlay guidance (72 ÂμGy.m2/kg), and using single plane overlay guidance (49 ÂμGy.m2/kg). There was a statistical difference between our institutional DAP/kg when biplane overlay was used versus fluoroscopy alone (p = 0.021).Similar reduction was noted in the median total air kerma, 486 mGy without overlay to 344 mGy with single plane overlay and 102 mGy with biplane overlay (p = 0.013); and median fluoroscopy times of 26 min without overlay to 20 min while utilizing biplane overlay (p = 0.023). A statistically significant stepwise reduction in contrast usage was also noted from 3.8 mL/kg without overlay to 3.3 mL/kg with single plane overlay and 2.1 mL/kg with use of biplane overlay (p = 0.007). Procedure times were noted to be shorter (by a median 106 min) with single plane versus a biplane overlay guidance.
Results of radiation metrics when compared to the national C3PO-QI database benchmark8 for stent angioplasty of coarctation of the aorta (CoA) showed improvement in DAP indexed to body weight, when using augmented bi-plane overlay as an alternate to traditional fluoroscopy. (DAP=Dose Area Product).

Statistical significance of biplane overlay vs no overlay assessed using an unpaired t-test (p < 0.05). Values in bold are to highlight results of current study compared to other values, which show statistically significant reduction.
Among age stratified patients older than 15 years, who underwent a TPVR, as demonstrated in Table 4, statistically significant reduction in the median total and indexed DAP for body weight (85 ÂμGy.m2/kg with biplane overlay) when compared with the national (271 ÂμGy.m2/kg), and local statistics (105 ÂμGy.m2/kg) (p = 0.03 for institutional no overlay vs biplane overlay).A reduction in the total air kerma was noted from a national 271 mGy to an institutional 105 mGy without overlay, to 85 mGy with biplane overlay (p = 0.028). Fluoroscopy exposure time reduced from an institutional 47 to 39 min (17% reduction), and contrast usage reduced from 3.8 mL/kg to 2.9 mL/kg (24% reduction), although these were not statistically significant.
Results of radiation metrics when compared to the national C3PO-QI database benchmark8 for Trans-catheter Pulmonary Valve Replacement (TPVR) show significant improvement in overall results and in sub-population >15 years of age, when using augmented bi-plane overlay as an alternate to traditional fluoroscopy. (DAP=Dose Area Product).

Statistical significance of biplane overlay vs no overlay assessed using an unpaired t-test (p < 0.05). Values in bold are to highlight results of current study compared to other values, which show statistically significant reduction.
Initial registration using tracheal positioning sufficed in 13 cases whereas eight required minor adjustments after initial x-ray angiography.
Physician satisfaction was objectively rated with this technology deemed “of some use” in 80% of cases where interventions were required. The highest rated value was for preprocedural planning and catheter navigation. It was noted as being “essential” during performance of procedures listed below.
Example cases
Specific cases where use of this technology was thought to be essential to their success and seemingly novel are listed here.
Biplane overlay helped preprocedural planning of approach to the intervention in an infant with a congenitally corrected transposition of the great arteries, who previously had undergone a “double switch” including a Senning procedure, in whom there was a diagnosed obstruction of the pulmonary venous baffle at the level of the superior vena cava (SVC). The interventional procedure involved a balloon dilation of this narrowed baffle, via a balloon catheter through the SVC after perforating into the pulmonary venous baffle. In this patient, the technology was extremely useful while planning camera angles for angiography and interventional planning with colored markers placed on the area of narrowing, region where the perforation would be performed, and approach to this area via the catheter. Novel placement of a transvenous dual chamber pacemaker system in a patient with atrial situs inversus, L-looped ventricles, with discordant ventriculo-arterial connections and dextrocardia {A;L;I} anatomy with complete heart block was performed. The technology helped the electrophysiologist not only provide guidance while mapping the His bundle but ultimately provide an accurate anatomic implantation of leads into the atrial appendage and ventricle (Figure 4).
Transvenous atrial and ventricular leads placed under biplane overlay guidance as seen on right and left oblique views of the heart in a patient with {A;L;I} heterotaxy with advanced third-degree heart block.
Similarly, area of landing or landing zones were identified based on the overlayed advanced imaging models for patients receiving a transcatheter prestent or a valve in the pulmonary position, and other stent placements in other systemic or pulmonary arteries, and device occluders in arterial and venous collaterals (Figures 5 and 6).

The upper panel shows a percutaneous Sapien valve positioned across the RVOT/ pulmonary valve (cyan) using advanced biplane overlay guidance, and then inflated over a balloon (lower panel). The aortic root and coronary relationship (blue) is helpful to plan for and avoid compression.

Final postintervention image with biplane overlay after placement of a pulmonary conduit stent (blue arrow), a melody valve (gold arrow) and a stent in the coarcted segment of the aorta (green arrow), all during the same procedure. The right ventricular blood pool is blue, the pulmonary arteries and distal conduit are pink, and the aorta is orange colored, with coronary arteries in green.
Discussion
We were able to implement and demonstrate feasibility of use, of a novel biplane fusion overlay system in the cardiac catheterization lab to augment fluoroscopic interventions. The two subgroups of patients were chosen with the intent to compare two procedures with a relatively large difference in complexity and radiation usage. Stent angioplasty of the aorta is considered a lower complexity, low-radiation dose procedure, whereas a transcatheter pulmonary valve implantation is inherently a procedure of higher complexity, and currently associated with the highest radiation exposure metrics in the CHD population. Comparative national and institutional metrics were also available for these interventions. The results as noted suggest that patients undergoing both low and high complexity cardiac interventional catheterization procedures can benefit with use of this technology. Children and adults who receive transcatheter pulmonary valves are also among those who have a high number of repeat interventions through their lifetime and may be the patients in whom reduction of radiation and nephrotoxic contrast usage is paramount. The patients selected for this study who underwent TPVR, also incidentally happened to be more complex, with three of the seven patients also undergoing other concomitant interventions during this procedure such as aortopulmonary collateral coiling; and other systemic or pulmonary artery balloon or stent angioplasty. A large proportion of these patients also underwent a pulmonary outflow tract prestenting, which made these cases more complex. The true benefit to this technology in this group may actually be larger, and over their lifetime, amounts to a significant reduction in the cumulative radiation damage incurred. No comparison of metrics were made between those who underwent TPVR under biplane vs single-plane assisted catheterization due to the very small number of patients (one) in the latter subset. The feedback received from the interventionalists suggested that the highest benefit during all procedures, but especially the low complexity procedures, was preplanning camera angles as the models would rotate in both planes based on the movement of the camera. This would completely avoid the extra radiation used while repositioning and realigning receivers after rotation. While this is a benefit that is shared with single-plane overlay, a biplane overlay was felt to be more intuitive for the interventionalist.
There have been studies which have previously showed benefits of using augmented overlay imaging to guide cardiac catheterization in CHD. Ehret et al. from Germany showed used both 2D and 3D models for overlay on various diagnostic and interventional cardiac catheterization procedures and showed significant accuracy of overlay models along with lower levels of radiation dose, contrast usage, and shortened fluoroscopy times. 9 While they used it for a larger number of interventions, they did not stratify their results based on procedures, and hence by which procedure benefited the most among all the interventions. There is growing literature, using single-plane overlay and rapid registration techniques, which have consistently demonstrated feasibility, and have helped aid decisions regarding access and planned intervention, improve accuracy for complex procedures, and reduction in radiation metrics.10–15 Use of a tracheal 3D model as a landmark of reference for the digital dataset overlay with the fluoroscopic images, and for the purposes of respiratory motion compensation were used for this project as pioneered by Rhode et al. 16 and has been reportedly used with success in other studies. 17 While our initial registration using the trachea and bronchial models was adequate in most patients, the models had to be readjusted based on initial angiography in 40% of patients. Other studies have demonstrated higher success with use of tracheal registration as a fiducial marker. 9 Respiratory motion compensation was enabled for a few patients, but was not used consistently as it was not operator-friendly in the beta-edition of the fusion software that was installed for this study. Its utility hence, could not be analyzed.
These studies have however all used single plane overlay techniques, whereas our study tried to go one step further to delineate and compare the benefit to using a biplane system over a single-plane system. Our data seem to correlate well with these studies, and in spite of the smaller number of patients studied, we were able to show significant reduction in radiation metrics, as initially hypothesized. The other patients were chosen usually based on the request of the interventionalist, to be those with high anatomic complexity.
In the current era, most patients with complex CHD undergo some form of cross-sectional diagnostic imaging prior to a decision being made for percutaneous intervention. This DICOM data can be easily utilized for purposes of generating the 3D models with no new imaging required. Preprocedural imaging in patients who have not had recent imaging may also help minimize the need for unnecessary interventions and help with intervention planning. Among the patients who needed to undergo preprocedural imaging, we obtained detailed anatomy with a CEMRA sequence and a 3D-VISTA sequence with a scan time less than 15 min. As we have an iCMR suite (Interventional Hybrid Cath/ MRI) at our center, the time taken to transfer this patient into the catheterization lab while anaesthetized was minimized. Technologically, we found it relatively easy to adapt to our current biplane fluoroscopic setup, with minimal need for training of operator and physician. Interventionalist comfort with this new technology is also a factor to consider. Interventionalists reported more confidence in proceeding with interventions without having to obtain an angiographic image to use as a reference during said intervention. The ease of setup and the use of this system were also a major driver for our adoption of this biplane fusion overlay system.
The major limiting factor to this technology noted was the “rigidity” of the models. Vascular anatomy tends to distort or move especially in younger patients with the use of rigid or larger sized catheters and sheaths; which alters the location of the area of planned intervention. The advantage remains in having both the augmented visuals and traditional fluoroscopic displays side-by-side to give the interventionalist the freedom to choose based on anatomy, and their degree of comfort with use of this technology. While an honest attempt was made at choosing patients and procedures for which comparative radiation and other data metrics were available, we were able to obtain eight patients undergoing a coarctation stent angioplasty of the aorta and five patients undergoing a transcatheter placement of a pulmonary valve. The time invested in postprocessing cross sectional images into 3D datasets is longer, and there is a need for a physician or a technologist for this purpose. We chose STL models as they had better fidelity compared to volume-rendered models, but this is time-consuming. While we could not assess the specific amount of time taken to segment these models, they were all performed by the primary author who took lesser time as he became more familiar with use of the software. The time taken to segment CT and CEMRA images was also lesser due to the higher signal-to-noise ratio afforded. Training advanced imaging physicians and technologists, and future developments in segmentation software including artificial intelligence-based segmentation tools will help minimize this burden. To date, no studies report any patient-related harm or morbidity with use of these technologies. While our study suggests that same, this has to be studied in the long term with consistent use, to ensure the safety of this technology.
With the above positive results, as an institution, we are currently utilizing this technology more comfortably for the more complex and high radiation cases such as TPVR, extensive collateral coiling, and lymphatic duct procedures as an adjunct to the current iCMR and single-plane overlay, with continuing good patient, and physician feedback. We have also been utilizing the models and overlay images and videos generated for patient education and surgical planning. Larger centers that take care of children and adults with CHD are now moving to adopt variations of methods including iCMR and single-plane overlay to help further patient care. While the current iteration of this technology is valuable, it is time consuming, and we foresee that as the technology improves we would achieve higher accuracy, reliability, and increased automation with less need for human intervention. Future advances in technology would likely automate the segmentation process for these models, and use advanced computer modeling and artificial intelligence to create “pliable” models that would bend and conform to vessel walls based on catheter position. We also hope this study lays groundwork for adoption of biplane overlay augmented cardiac catheterization technology, which other institutions around the world would build upon.
Footnotes
Acknowledgements
The authors would like to acknowledge that this was funded by a grant from Siemens, but the authors had full control over data inclusion, analysis, and manuscript preparation.
Authors’ note
Tweet: Check out the latest on 3D bi-plane augmented overlay guidance for cardiac catheter interventions in congenital heart disease. #pedscards
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical standards
The authors assert that all procedures contributing to this work comply with the ethical standards of the Belmont Report and with the Helsinki Declaration of 1975, as revised in 2008, and has been approved by the Children's Medical Center and University of Texas Southwestern institutional review boards.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Siemens Medical Solutions USA.
