Abstract
Background and objectives
Previous studies suggest a link between high-sensitive cardiac troponin (hs-cTn) levels and coronary artery disease (CAD). However, the nature of this relationship is disputed. To address this, we conducted a study to gather and assess evidence on the association between hs-cTn and CAD prediction.
Data sources
Studies were systematically searched and collected from four databases and different types of gray literature to cover all available evidence. After the screening, the selected articles’ quality and risk of bias assessment were evaluated.
Synthesis method
Meta-analysis calculated std. mean difference on the extracted data. Furthermore, heterogeneity, sensitivity, subgroups, and publication bias analyses were assessed.
Results
Twenty-two studies were included in this systematic review, with a total of 844 cases and 2101 control people. The results of the meta-analysis on nine studies showed a significant and positive association between hs-cTn levels and CAD (pooled std. mean difference = 0.44; 95% confidence interval = 0.14–0.73; p < 0.003), with no publication bias (p = 0.9170). Among subgroups, std. mean differences were notably different only when the data were stratified by region or risk of bias; however, subgroup analysis could not determine the source of heterogeneity.
Conclusions
Available prospective studies indicate a strong association of hs-cTn with the risk of CAD and significant improvements in CAD prediction. Further investigations in both molecular and clinical fields with proper methodology and more detailed information are needed to discover more evidence and underlying mechanisms to clear the interactive aspects of hs-cTn level in CAD patients.
Systematic review registration number (PROSPERO)
CRD42021224527 https://www.crd.york.ac.uk/prospero/#recordDetails.
Summary
This systematic review aimed to investigate the association between high-sensitive cardiac troponin (hs-cTn) levels and coronary artery disease (CAD) prediction. A total of 22 studies were included, comprising 844 cases and 2101 control individuals. The meta-analysis revealed a significant and positive association between hs-cTn levels and CAD, indicating an increased risk of CAD in individuals with higher hs-cTn levels. Subgroup analysis suggested that the association varied by region and risk of bias. However, the source of heterogeneity could not be determined. The findings suggest that hs-cTn may serve as a valuable predictor of CAD and could contribute to improved CAD risk assessment. Nevertheless, further research is warranted to explore the underlying mechanisms and obtain more comprehensive evidence in this field. Future studies should employ rigorous methodologies and provide detailed information to enhance our understanding of the interactive aspects of hs-cTn levels in CAD patients.
Introduction
Coronary artery disease (CAD) is a major cause of death in the general population. 1 CAD may be present early in life, but its progression over time is very unpredictable. Age is the strongest factor associated with CAD. The prevalence of heart diseases is increasing due to factors such as obesity and worsening cardiovascular risk factors. 2 The progression of CAD can be gradual or sudden, leading to death. Identifying patients at risk is crucial for active secondary prevention. The prevalence of CAD increases with age, and certain risk factors, such as high blood pressure and smoking, contributes to its development. 3
Coronary atherosclerosis can also progress at varying speeds, from a gradual increase in duct stenosis to a sudden progression to complete duct obstruction, the second most common of which is often due to a disorder of non-stenotic plaque associated with rupture or erosion and subsequent thrombosis. Therefore, the progression of CAD may be gradual or sudden and catastrophic and lead to death.
4
Stable CAD, defined as obstructive or non-obstructive CAD with persistent symptoms, affects approximately 54 million patients worldwide and can range from a clinically stable condition to acute coronary syndrome (ACS). Therefore, early identification of stable patients at risk is essential for active secondary prevention of cardiovascular events.
5
The prevalence of symptomatic CAD increases steadily with age. NHANES (National Health and Nutrition Survey) reported a higher prevalence of coronary heart disease in men (30.6%) compared to women (21.7%) aged 80 years from 2011 to 2014. Similar findings have been reported from Framingham Heart Study and Cardiovascular Health Study. In the ARIC (Community Atherosclerosis Risk) study, the incidence of myocardial infarction (MI) was higher in patients aged 65 to 84 years in black than in whites.6,7 Studies have shown a high prevalence of obstructive CAD in patients 80 years of age, which often has advanced disease features (e.g. calcification (80% to 90%), multivessel disease (40%), and totality). High blood pressure, dyslipidemia, diabetes, kidney dysfunction, and a history of smoking are among the proven risk factors for CAD.
8
Cardiac troponin T, a highly sensitive and specific biomarker for myocardial damage, has been shown to be associated with the prognosis of CAD. Several reports have shown that an increase in cardiac troponin T can be seen in a proportion of patients after coronary artery bypass graft, ranging from less than 10% to approximately 50%. Elevated troponin T has also been reported to be relatively common in patients with stable CAD under coronary skin intervention and is a prognostic indicator independent of ischemic complications.
9
Cardiac troponin is a strong predictor of cardiovascular risk at the population level. It binds muscle by changing the concentration of intracellular calcium ions and producing muscle contraction
Several studies have investigated the relationship between CAD and serum troponin levels, but the results have been conflicting. Some studies suggest a significant relationship, while others deny its existence. The mechanism behind this relationship has not been determined. Therefore, this systematic review and meta-analysis aim to investigate the association between serum troponin levels and CAD incidence.
Materials and methods
Eligibility criteria
The design of the present study was based on the methodology of the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guideline [Moher D, Liberati A, Tetzlaff J, Altman DG, Group P. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS med. 2009; 6: e1000097. https://doi.org/10.1371/ journal.pmed.1000097 PMID: 19621072].
This review is registered in the international prospective register of systematic reviews (PROSPERO) with registration number CRD42021224527 (Available: https://www.crd.york.ac.uk/prospero/#recordDetails).
Definition
Inclusion/exclusion criteria
Inclusion criteria included: (a) case–control and suitable cohort, and brief-report studies that contain evidence about the relationship between serum cardiac troponin and CAD, (b) Human clinical study, (c) Having at least one parameter of the std. mean difference or can be calculated based on the information of serum cardiac troponin in both case and control groups.
The status of serum cardiac troponin should be detected by a blood test, high-sensitivity assays including Elecsys TNT hs STAT, Roche Diagnostics, TnI-Ultra assay, second-generation electrochemiluminescence immunoassay, and AccuTnI assay. Furthermore, the CAD should be confirmed by coronary angiography procedure and/or Exercise test. Articles without any of this information were excluded from further analysis.
Sources and search strategy
All relevant articles, published from 1990 to the end of the second month of 2022 were collected from the electronic database of PubMed, ProQuest, Scopus, and Web of Science. The ProQuest database was also reviewed for related dissertations, manually. Other related protocols were manually searched in PROSPERO. In addition, related articles will be obtained through various types of grays, such as conference and meeting summaries (such as the American Heart Association and the European Society of Cardiology). Experts in this field will also receive information via email. Several key journals (“The American Journal of Cardiology”, “European Heart Journal” and “Circulation”) will be manually reviewed for related articles. The reference list of previous systematic review studies 13 and preliminary articles will also be reviewed manually. No geographical restrictions will be considered. Articles will be included in the study and rating if they have full text. In the end, we performed a manual search in the references of the selected articles. The Medical subject heading (MeSH) database was used to find various terms of CAD and serum cardiac troponin. Two main keywords were “Troponin” and “coronary artery disease.” For better searching in the databases, we made syntaxes from a combination of free-text method, MeSH terms, the keywords, and Boolean operators (AND/OR/NOT). Moreover, calculating NNR (number need to read) helped us in evaluating the output of syntax and dedicating. The following syntax was applied in PubMed and adjusted for each search engine based on its search guidelines (“Troponin C”[TI] OR “Troponin I”[TIAB] OR “Troponin T”[TIAB] OR “Troponin Complex”[TIAB] OR “Troponins”[TIAB]) AND “Coronary Artery Disease”[TI] OR (“Artery Disease”[TIAB] AND coronary[TIAB]) OR (disease[TIAB] AND “Coronary Artery”[TIAB]) OR “Coronary Arteriosclerosis”[TI] OR (arteriosclerosis[TIAB] AND coronary[TIAB]) OR “Coronary Arterioscleroses”[TI] OR (arteriosclerosis[TIAB] AND coronary[TIAB]) OR “Coronary Atheroscleroses”[TI] OR (atherosclerosis[TIAB] AND coronary[TIAB]) OR “Coronary Atherosclerosis”[TI] OR (atherosclerosis[TIAB] AND coronary[TIAB]) AND 1990/01/01:2020/11/10[dp].
The search outputs were exported into the Endnote software (Version X7; Thompson Reuters Corporation, Toronto, ON, Canada) to remove any duplicate and screen articles. The first step of screening for determining the eligible primary articles was done based on the titles and abstracts. Two independent reviewers (M.D. and MA) evaluated the selected full-text articles and separately classified them into three relevant, irrelevant, and unsure groups, based on the eligibility criteria. Any discrepancy was supervised by a third reviewer (A.F.) and resolved via a consensus. If there was still a doubt in the selection of a study, the whole team made the final decision.
Quality and risk of bias assessment
The quality and risk of bias assessment of the selected articles were independently evaluated by two reviewers (M.Z. and M.D.), using the standard checklist of the Newcastle-Ottawa Scale (NOS) form [33]. The studies were classified into three poor, fair, and good categories based on getting a score between zero and eight in the selection, comparability, and outcome/exposure domains based on the standard guideline. Consensus and the opinion of the third reviewer (A.F) resolved any disagreements between the two reviewers.
Data extraction
Two authors (Y.Y. and A.A) independently performed data extraction from each paper based on a defined protocol. The data were divided into three categories and included (a) General information: the first author's name, year of publication, journal names, country, and region; (b) The risk of bias assessment; (c) Study setting: study design, study duration, sample size and total population recruited in each study, inclusion and exclusion criteria, age group and age range, sex, the definition of CAD, source of the data, diagnostic methods, std. mean difference and 95% confidence interval (CI), and the ethical approval. In cases of missing or additional necessary data, we further emailed each author, in charge of the related study.
Data synthesis and analysis
In the case of quantitative data, if possible, standardized mean difference and 95% confidence range from each study based on statistical software (Software for Statistics and Data Science) Stata-12 will be calculated. The composition method will be based on the methodological similarities of the studies between one of the two methods of FEM (Fixed Effect Model) and RM (Random Effect Model). A forest plot will be drawn as the combined effect size. The heterogeneity between the compound studies will be examined based on the standard chi (Cochran's Q test) and I2 scale. The I2 scale or more than 50% will be considered as severe heterogeneity. Where statistical consolidation is not possible, subgroup analysis or meta-regression method will be used for specified heterogeneity to determine its reasons and ultimately use sensitivity analysis. The Funnel chart, then Begg's and Egger's tests, are used for publishing error analysis. Trim and Fill are used if the publishing error is not ignored. One-out remove method is used to evaluate sensitivity analysis. Although the ultimate goal is to do a meta-analysis, it is only possible when the required data can be extracted from the systematic review.
Results
Search results
In total, the general searching step obtained 875 papers. After removing duplicates, 786 studies were screened, based on their titles and abstracts. Among them, 692 articles were excluded due to not meeting the inclusion criteria. Therefore, the full texts of the 73 remaining articles were completely evaluated. Finally, nine studies were included in the systematic review, and also meta-analysis (kappa (κ) = 0.84)1,5,7,14–20 and the others were excluded due to a reason mentioned in Figure 1. Ultimately, a total of five studies were classified as case–control, while nine studies were categorized as cohort.

Flow diagram of literature search, screening, and selection of studies for review and meta-analysis.
Characteristics of the included articles
The designs of all the included studies were case–control, with a total of 844 cases and 2101 control people. Among the studies Zethelius et al. 20 had the highest study population with 1203 participants (included 368 cases and 835 controls). Most studies were from Sweden and all studies had been performed only on adults. Table 1 shows the details of the included articles.
Main characteristics of the included studies in the meta-analysis on the high-sensitive cardiac troponin and coronary artery disease (CAD).

aFair.
bGood.
cPoor.
Overall, the quality of the included studies was good. The NOS assessment showed that only 44.4% of the studies had good quality.
Association between high-sensitivity troponin T level and CAD
By analyzing a combination of nine studies that addressed the association between the levels of serum hs-cTn and CAD, a positive association was found (std. mean difference = 0.44; 95% CI = 0.14–0.73; p < 0.001). The REM and Chi-squared method showed significant statistical heterogeneity between the studies I2 = 88%, p < 0.001, and Z = 2.92, p = 0.003, respectively (Figure 2).

The forest plot of the meta-analysis on the association of the hs-TnT and CAD. The random effects model was used to calculate pooled Std. Mean Difference with 95% CI. CI, Confidence interval; Std. Mean Difference, standardized mean difference; Hs-TnT, high-sensitivity cardiac troponin T; CAD, coronary artery disease.
Figure 3a shows a Drapery plot, presenting that the estimated effect is less than 1 in the majority of studies, similarly as shown in the Funnel plot. As the horizontal dashed lines show the CIs for common alpha levels (0.1, 0.05, 0.01, 0.001), at any level of prediction interval (90, 95, 99, 99.9%), the main triangle line (the red-bolded line in the online version) did not intersect the null effect line (Incidence Rate Ratio = 1). According to the results of the Funnel plot and Begg's tests, there was no significant publication bias (p = 0.9170, Figure 3b). The Baujat plot

(a) The drapery plot shows p-value functions for the included studies as well as pooled estimates and the two-sided confidence intervals for all possible alpha levels (confidence interval function). Each number represents a study. The red-bolded line (in the online version; bold line in the printed version) indicates the range of std. mean difference in each alpha level. (b) The Funnel chart shows no significant publication bias, as all studies are in a symmetric scheme. (c) The Baujat plot shows the effect of each study on the heterogeneity and overall influence of the results. As can be seen in the graph, the study of Walter et al. contributes more than the other studies to the overall heterogeneity. Each circle represents a study. (d) The Egger & Begg's test & plot assesses the significant correlation between the ranks of the effect estimates and the ranks of their variances.
(Figure 3c) showed that the study of Walter et al. contributes more than the other studies on the overall heterogeneity and influences the pooled effect size.
Subgroup and sensitivity analyses
The heterogeneity between studies was evaluated by multiple subgroup analyses in the four subgroups. Based on the Region of the studies, there were few studies in Europe, which made the results unreliable for statistical analysis.1,14,15,17,19,20 North America has a higher std. mean difference (std. mean difference = 1.16, 95% CI = 0.56–1.76) than Eastern Asia (std. mean difference = 0.4, 95% CI = 0.15–0.65).
However, further evaluation is needed due to the low rate of studies, especially in North America. Other subgroups could not determine the source of heterogeneity (Table 2).
Different subgroup analyses by stratifying the data.
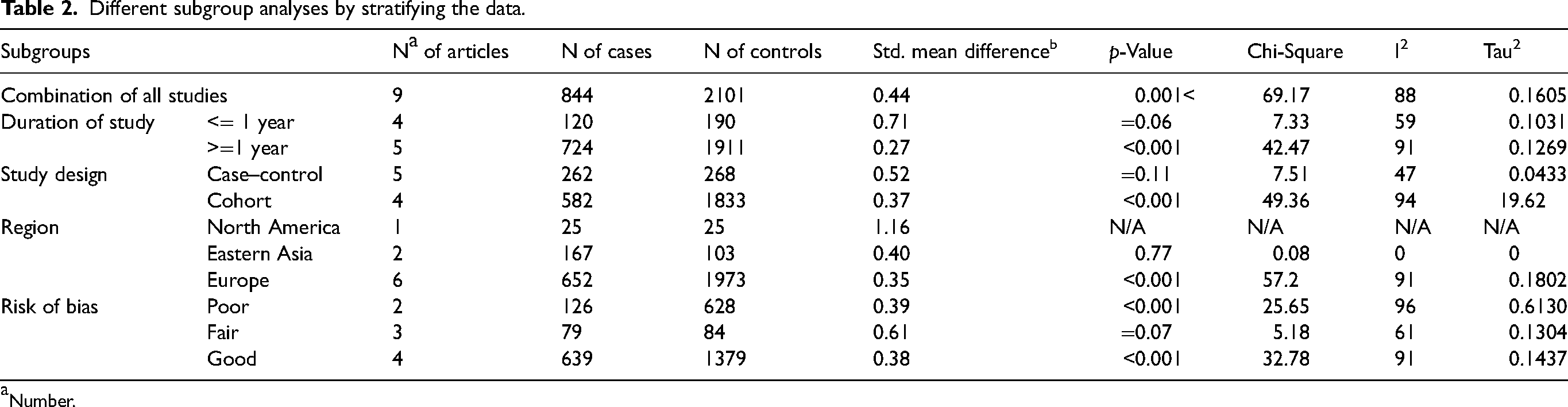
Number.
Discussion
Most information about the relationship between hs-cTn and CAD is inferred from case–control studies and more prospective cohort and /or systematic review are needed to provide the strongest evidence about this association. In this regard, the present study is the first systematic review and meta-analysis that aimed to clarify any possible association between hs-cTn and CAD. The meta-analysis showed a positive relationship between hs-cTn and CAD, and confirmed the findings of some previous studies, showing hs-cTn was significantly more prevalent in apparently healthy people than CAD patients.
In 2015, Inga Magdalena Huziuk et al. concluded that patients with multivessel CAD had remarkably higher levels of high-sensitivity troponin T (hs-TnT) (0.01 vs. 0.007, P = 0.0021) and fasting glucose (6.0 vs. 5.45, p = 0.0112). Established on the drawn receiver operating characteristic curves, the cut-off points were determined for hs-TnT ≥ 0.0085 ng/ml and fasting plasma glucose ≥ 5.85 mmol/l. Their multivariate analysis demonstrated that only hs-TnT in concentration higher than the cut-off point enhanced the risk of multivessel CAD (OR 4.286, 95% CI: 1.79–10.263, p = 0.001). In men with stable CAD, preserved systolic left ventricular function, and non-high cardiovascular risk determined from the initial concentration of hsCRP, elevated hs-TnT was independently associated with the risk of multivessel CAD. 21
A report by Philip D. Adamson et al. determined that high-sensitivity cardiac troponin I concentration is an independent predictor of obstructive CAD in patients with suspected stable angina and the use of this test may improve the selection of patients for further investigation and treatment. Higher cardiac troponin concentrations were associated with obstructive CAD with a fivefold increase across quintiles (9 to 48%, p < 0.001) independent of known cardiovascular risk factors (odds ratio [OR] 1.35 [95% CI 1.25–1.46] per doubling of troponin). The discrimination and calibration of the CADC model for identifying obstructive CAD were improved by Cardiac troponin concentrations (c-statistic 0.788 to 0.800, p = 0.004; χ2 16.8 [p = 0.032] to 14.3 [p = 0.074]). 22
The study by Askin et al. highlights that high levels of hs-cTnT in the blood are associated with increased cardiac events and mortality rates, independent of the underlying disease. However, the specific clinical conditions that cause hs-cTnT secretion in patients without ACS, as well as the appropriate diagnosis and treatment strategies for these individuals, remain largely unknown.
Also, it emphasizes that while hs-cTnT is commonly associated with the diagnosis of MI due to increased blood levels, it is not solely released as a result of ischemic myocardial cell necrosis. Rather, hs-cTnT can also be released in various non-ischemic acute and chronic heart diseases, such as myopericarditis, toxic injury, or severe cardiac overload.
According to the positive association of troponin and CAD in our study, Askin et al. suggest that understanding the various causes and prognostic significance of non-ACS-related hs-cTnT release is crucial. They also propose that elevated levels of hs-cTnT should not only be focused on cardiac events but also considered in relation to other acute events. It is possible that utilizing cardiovascular biomarkers such as hs-cTnT may aid in the early prevention of non-cardiac diseases. 23
Zhang-wei Chen et al. conducted a study with stable CAD patients admitted to their hospital for elective coronary intervention. They were divided into an elevated troponin T group and a normal troponin T group after the procedure by troponin T. Clinical factors, laboratory factors, and angiographic factors (such as sex, age, cholesterol, gender score, etc.) were analyzed. In the following, a total of 209 patients with an average age of 64.0 ± 9.9 years were included in the study: 70 patients with increased troponin T (.030.03 ng/ml) after the implantation of a drug-eluting stent and 139 patients with troponin normal T (<0.03 ng/mL). After univariate analysis, they found that age, blood pressure, total cholesterol, low-density lipoprotein cholesterol, gender score, the number of narrowed vessels, and the total number of implanted stents were associated with increased troponin T levels. According to the results of multivariate analysis, they also found that age, total cholesterol, the number of narrowed vessels, and the number of implanted stents is independent factors of increased risk of troponin T postoperatively. 9
In line with the results of a recent study by Askin et al., we have observed a positive correlation between elevated levels of high-sensitive troponin and the occurrence of CAD. The aforementioned study aimed to assess the significance of serum high-sensitivity cardiac troponin T (hs-cTnT) as a predictive marker for prehypertension, a condition known to be a precursor of hypertension and a substantial public health concern. The researchers prospectively enrolled a cohort of 100 subjects, consisting of 50 consecutive prehypertensive patients with blood pressures ranging from 120/80 to 139/89 mmHg, and 50 normotensive patients with blood pressures below 120/80 mmHg.
Their findings revealed a statistically significant elevation in hs-cTnT levels within the prehypertensive group compared to the normotensive group (p < 0.001). Moreover, a positive correlation was observed between hs-cTnT levels and prehypertension (r = 0.625, p < 0.001). The study further identified hs-cTnT as an independent predictor of prehypertension, with an odds ratio of 1.043 (95% CI 1.019–1.067, p < 0.001). Notably, an hs-cTnT threshold of 0.55 ng/L demonstrated predictive value for prehypertension, yielding a sensitivity of 86% and specificity of 60% (area under the curve = 0.861; 95% CI, 0.787–0.935; p < 0.001).
Drawing parallels with our own study on the positive association between high hs-cTnT levels and CAD occurrence, these findings support the notion that hs-cTnT may serve as a complementary diagnostic biomarker for identifying individuals at risk for both prehypertension and CAD. Further research is warranted to elucidate the underlying mechanisms linking hs-cTnT elevation to the pathophysiology of these conditions, thereby enabling the development of targeted preventive and therapeutic strategies. 24
The current findings showing a significant positive association between elevated hs-cTnT levels and prediction of CAD are consistent with the results of a recent cohort study examining hs-cTnT levels in non-dipper hypertension patients. 25 In their longitudinal analysis of 100 hypertensive adults, Askin et al. found patients with non-dipper hypertension had significantly higher hs-cTnT levels compared to dippers (p < 0.001). Furthermore, they reported hs-cTnT was an independent predictor of non-dipper status and was correlated with changes in left ventricular geometry over the 6-month follow-up. Although that study was limited to hypertensive patients and had a smaller sample size compared to our meta-analysis, the results provide further evidence that higher circulating hs-cTnT levels are linked to greater cardiovascular risk. The cohort findings also suggest hs-cTnT may have clinical utility specifically for identification of abnormal diurnal blood pressure patterns in hypertensive populations. Taken together, studies varied in methods and patient populations provide consistent evidence supporting the prognostic value of hs-cTnT for predicting adverse cardiovascular outcomes. 25
Samman Tahhan et al. conducted a study on 3087 patients (63.12 years old, 64% male) undergoing cardiac catheterization and without evidence of acute MI, the severity of CAD with the number of main coronary arteries with major stenosis 50% and calculated gender score. CAD progression was evaluated in a subset of 717 patients who underwent angiography more than two times and 3 months prior to enrollment. Patients were followed up regarding death due to accidents and cardiovascular events. Of the total population, 11% had a normal angiogram, 23% had non-occlusive CAD, 20% had 1-vessel CAD, 20% had 2-vessel CAD, and 26% had 3-vessel CAD. Furthermore, cardiac troponin I levels were independently associated with the severity of CAD as measured by the gender score (log 2 ß = 0.31; 95% CI, 0.18–0.44; p < 0.001) and with the progression of CAD (log 2 ß = 0.36; 95% CI, 0.14–0.58; p = 0.001). Cardiac troponin I level was also a significant predictor of accidental death, cardiovascular death, MI, and hospitalization, independent of the abovementioned variables and the severity of CAD. As a result, a higher level of cardiac troponin I was associated with baseline coronary atherosclerosis, faster progression of CAD, and a higher risk of mortality from all acute cardiovascular events. 4
The present study had some limitations. First, just a few numbers of the selected articles had a good quality method and many of them did not consider many confounding factors in the design of the study that could affect the results. Second, based on the results, the heterogeneity of outcomes was high, and the subgroup analysis could not recognize the source of it completely.
Third, most of the studies were conducted in Europe and Eastern Asia. There was no article from Africa. Moreover, only one articles reported the situation in North America. Studying other ethnicities can be a suitable target for further investigations.
Conclusion
The meta-analysis of nine clinical studies including 844 CAD patients showed that the high levels of serum hs-cTn are associated with an increased risk of CAD. However, it is important to acknowledge that there are certain limitations that need to be addressed in future research. One major obstacle in fully understanding the role of hs-cTn in CAD is the lack of accurate details in the clinical studies included in our analysis. Therefore, it is recommended to conduct cohort studies and further primary studies with robust methodology to provide more comprehensive and reliable evidence. Despite these limitations, preliminary data from our analysis indicate that hs-cTn assessment could potentially serve as a promising biomarker for measuring CAD risk as part of primary prevention. The findings suggest that incorporating hs-cTn assessment into routine clinical practice may enhance our ability to identify individuals at a higher risk of developing CAD.
Footnotes
Acknowledgement
Special thanks to all the fellow authors who contributed to this article.
Consent for publication
Not applicable.
Contributorship
Alireza Fatemi, Mahdi Zahedi, Maryam Daei, and Yasmin Yazdooei contributed in conceptualization. Alireza Fatemi, Mahdi Zahedi, Maryam Daei, Mohammad Mostafa Ansari, Yasmin Yazdooei, Kamineh Dehghan, Mohammad reza Hssanpour, and Arash Azizinejad involved in data curation. Ahmad Sohrabi involved in formal analysis. Alireza Fatemi, Mohsen Habibi, Arash Azizinejad, Marzieh Behrouzifar, and Hanieh Babapour involved in investigation. Ahmad Sohrabi, Alireza Fatemi, Mahdi Zahedi, and Mohammad Mostafa Ansari involved in methodology. Alireza Fatemi and Mahdi Zahedi involved in project administration. Ahmad Sohrabi contributed in software. Alireza Fatemi and Maryam Daei involved in supervision. Mahdi Zahedi and Alireza Fatemi involved in validation. Ahmad Sohrabi involved in visualization. Alireza Fatemi, Yasmin Yazdooei, Faegheh Aghdasi, Kamineh Dehghan and Mohammad reza Hssanpour wrote the original draft. Alireza Fatemi, Yasmin Yazdooei, Mahdi Zahedi, and Mohammad Mostafa Ansari reviewed and edited.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval and consent to participate
The study and all experimental protocols were approved by the Professional Ethics Committee of the Golestan University of Medical Sciences with the ethical code of ir.goums.rec.1396.74. The informed consent was also obtained from all patients for the implementation of this project.
All methods were performed in accordance with the relevant guidelines and regulations.
Funding
The Golestan University of Medical Sciences financially supported this work.
Guarantor
Alireza Fatemi accepts full responsibility for the study, including the conduct of the research, having access to the data, and controlling the decision to publish.
Supporting information
N/A.
