Abstract
Some studies consider the different physical properties of the stent graft when compared with the blood vessel on the basis of vascular lesions that may require further intervention. We present a case in which a patient developed an intramural hematoma at the distal landing of previous thoracic endovascular aortic repair (TEVAR) that required the relining with a flared prosthesis. During follow-up, we observed the appearance of more caudal hematoma. We decided to observe this lesion with close radiological controls. In order to prevent serious complication after the induction of TEVAR, accurate planning of the procedure is very important to study the impact of the prosthesis implanted in the cardiovascular system. In particular, oversize, radial forces and length of coverage have been taken into account. The adherence to follow-up is very important to precociously detect the lesions to avoid the onset of complication.
Key message
Several studies present the impact of TEVAR on the mechanical feature and histological composition of the aortic wall. These changes could promote the progression of lesions that require new treatment procedures.
Case report
A 77-year old woman suffering from hypertension with history of isteroannessectomy for uterine fibroma was admitted to the cardiological unit following an episode of syncope due to severe transitory bradycardia. During hospitalization, an aortic angio-TC scan revealed the presence of a 6 cm aortic aneurysm in the post-isthmic tract extended to the descending aorta and an infrarenal aortic ectasia (29 mm) (Figure 1). The arrhythmia was treated with the implantation of Pace Maker (PM), and the previous chronic therapy was integrated with the introduction of beta-blocker and a calcium antagonist.
Preoperative CT scan.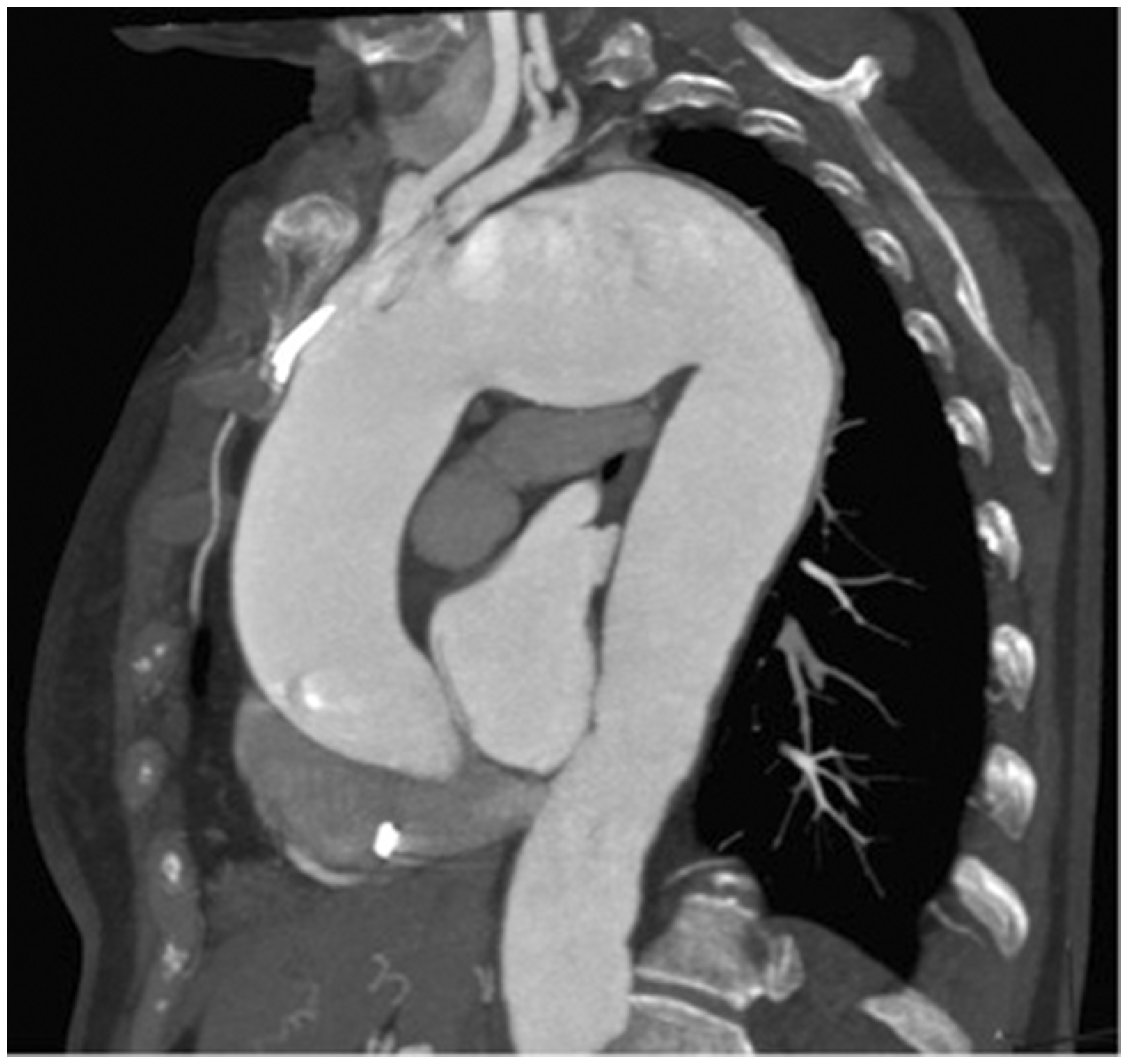
We performed, after two months, a surgical intervention for the endovascular exclusion of the thoracic aortic aneurysm (landing zone 1) after the debranching of both the left common carotid artery (LCCA) and the left subclavian artery (LSA) and their revascularization from a bypass coming from right common carotid artery (RCCA).
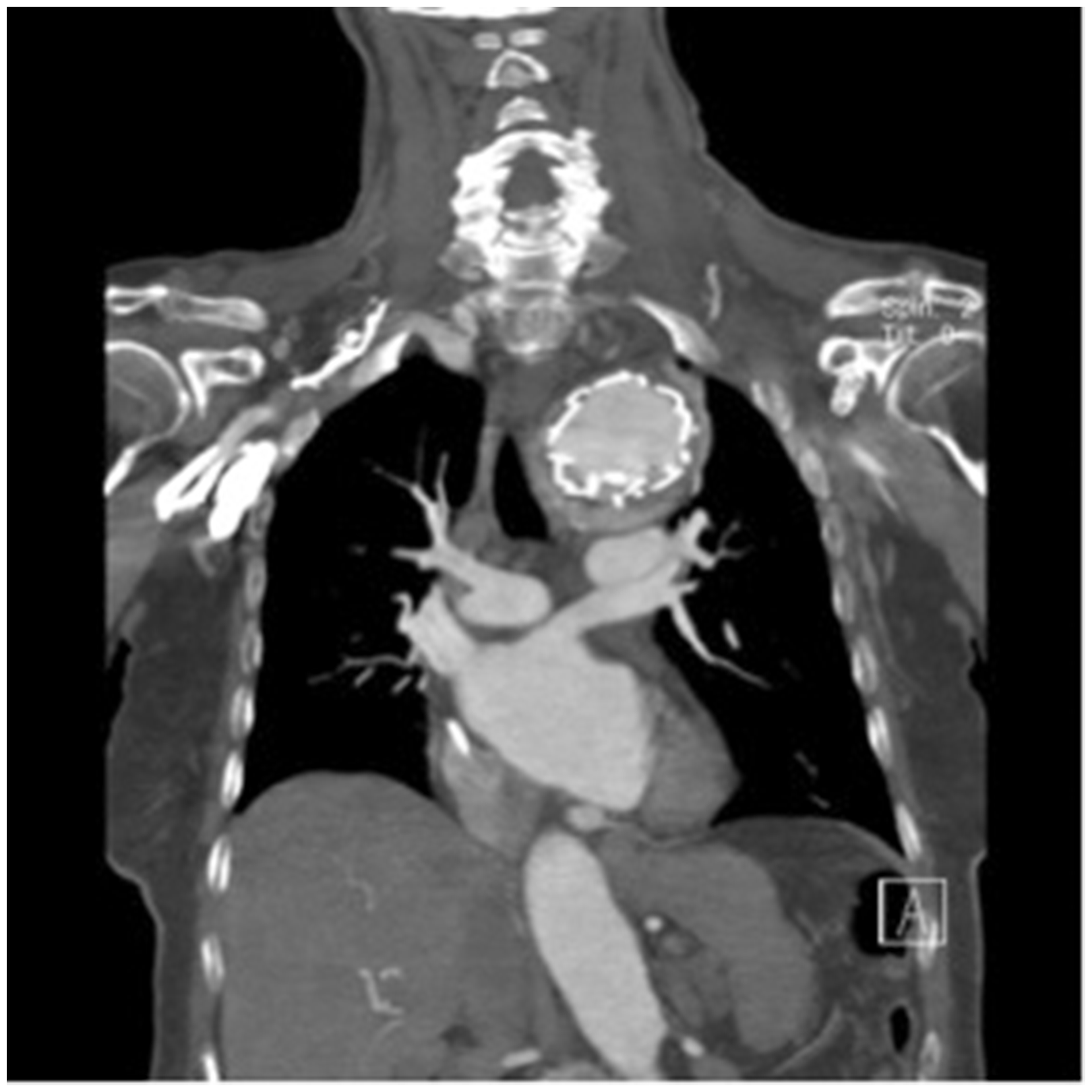
The RCCA was isolated through a right laterocervical incision, while LSA and LCCA through the supraclavicular one with scalenotomy and phrenic nerve conservation. Using a 8 mm Dacron prosthesis, we manufactured a carotid-carotid right–left and carotid-subclavian sequentially by-passes; during the entire procedure, we monitored EEG and PESS. We performed termino-lateral anastomosis between RCCA and prosthesis, lateral-terminal anastomosis between prosthesis and LCCA and terminal-lateral anastomosis between prosthesis and LSA. The proximal stump of LCCA was ligated and sutured. Under controlled hypotension produced by cardiac pacing, we deployed Backup Meir guidewire Captivia Valiant (44-44 200). During the release, there was a ‘sail effect’ causing the dislocation of the prosthesis about 4 cm far from the origin of innominate artery. For this reason, we proceeded to proximal relining with another Captivia Valiant (44-44-200). The procedure ended with the occlusion of LSA positioning Amplatzer Plug. The completion angiography was optimal (Figure 2). The final oversize in the proximal and distal landing were 10% and 15%, respectively. In the second post-operative day, the patient developed a right laterocervical tumefaction. The neck and thoracic CT scan showed an important hematoma enclosing and compressing the RCCA and the origin of bypass extended to the posterior cervical triangle. This study also showed a type III endoleak (EL) in the concavity of aortic arch rising from the overlapping zone of the two prostheses (Figures 3 and 4). We decided to reopen the previous right surgical incision in the neck and evacuate hematoma. No active blood losses were found. EL III was treated with balloon angioplasty. Completion angiography showed a good result. The patient was scheduled for an accurate follow-up at our institute with CT scan after six months, then annually and DopplerUS of bypass every six months. Six-month CT scan was optimal. Eighteen months’ control indeed presented a regular patency of bypass, the persistent absence of EL but the appearance of intraluminal ulcerated thrombus ascending on the external side of the endoprosthesis. A deep ulcer in the thrombus perfused its caudal part. At this level, aortic diameter was increased from 51 to 61 mm (Figure 5). This finding led to another endovascular procedure on the patient which was planned to extend the distal aortic coverage with a tapered Captivia Valiant (44-40-150). The oversize was 10%. The procedure was performed after two weeks from the previous CT scan. The last control was scheduled in September 2016 after 21 months from the first treatment. It showed the reduction of covered aortic diameter and no leak was revealed. However, it presented the enlargement of suprarenal tract untreated (which reached the size of 50 mm) and the presence of endoluminal thrombus with an irregular surface due to ulceration. Also, the infrarenal tract of aorta was increasing in diameter (40 mm) (Figure 6). The patient still remained asymptomatic, and she has no limitation to her life. We decided to observe the evolution of the present lesion with a close CT scan.
Completion angiography showing the aneurysmal exclusion and the patency of the by-passes. III P.O. day El type III on the concavity of aortic arch. III P.O. day El type III on the concavity of aortic arch. 18 Months after the procedure showing development of intraluminal ulcerated thrombus ascending on the external side of the endoprosthesis. 21 Months after the first treatment showing development of ulcerated thrombus and enlargement of infrarenal aorta.



Conclusion
The progression of aortic lesion after thoracic endovascular aortic repair (TEVAR) might be related to the changes in the aortic wall due to the implantation of the device.1–3 There are different physical proprieties of the stent graft when compared with the blood vessel. 4 Stent grafts are several orders of magnitude stiffer than the native aorta notably in the longitudinal axis, which implies the increase of wall stress in the segment adjacent to the stent graft caused by a compliance mismatch. 3 The impact of stent graft oversize reduced the wall strength. 5 In addition to the local impact, TEVAR might affect the cardiovascular system on a systematic level by damping the high pulsatile flow coming from left ventricle with an implication for cardiovascular disease development.6,7 Furthermore, histologic and immunohistochemical studies advocate the structural disarrangement of the aortic wall after the insertion of an endoprosthesis by reducing the number of muscle and elastic fibres. 2 In our case, the patient with a fragile aorta, the modifications induced from the implantation of TEVAR probably have brought to the enlargement of the distal uncovered part of aorta and the development of intramural hematoma. The displacement of prosthesis during the first procedure has caused an unexpected increase of oversize with the further reduction in wall strength. In conclusion, planning of procedure before TEVAR is crucial. In degenerative disease, it is important to use devices with adequate oversize and radial forces to minimize aortic physical and mechanical changes. Follow-up must be continuous and accurate in order to precociously find every further lesion.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The patients gave her consent to the publication.
Guarantor
EC.
Contributorship
EB EC wrote the paper. GM Literature search. SB FN Perform surgical procedure.
