Abstract
A 15-year-old girl was referred to a dentist complaining of parageusia, bad taste in the mouth, which started 9 months ago. Panoramic X-ray and non-enhanced computed tomography scan revealed multiple bilateral unilocular cysts in the mandible and maxilla, along with calcification of anterior part of the falx cerebri. She was eventually diagnosed with Gorlin-Goltz syndrome based on imaging and histopathologic finding of keratocystic odontogenic tumor.
Introduction
Gorlin-Goltz syndrome (GGS) is a rare genetic disorder which could be inherited or occur as a result of a new mutation. GGS can have different manifestations, varying from dermatologic or radiologic findings to intracranial and other various types of neoplasms (1,2).
Here, we report a case of a 15-year-old girl with GGS.
Case report
A 15-year-old girl presented to a dentist with the chief compliant of a gradually progressive parageusia without halitosis, which started 9 months ago. Initial examination of the mouth did not reveal any mucosal lesions or dental cavities. On physical examination of the head and neck, she had a coarse face, mild acne, and sporadic macular lesions on the posterior neck. Similar lesions were found in the abdominal area.
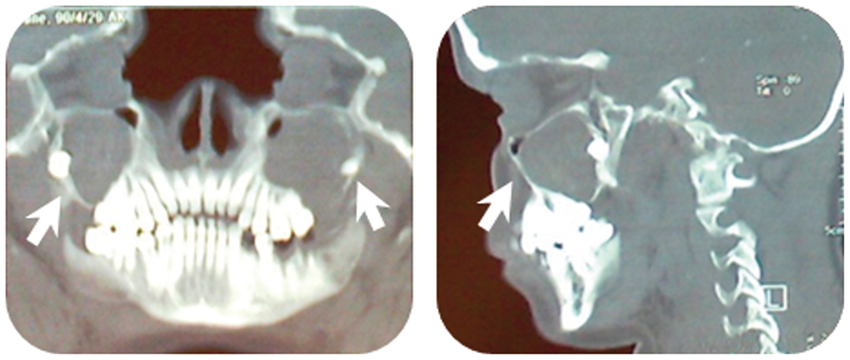
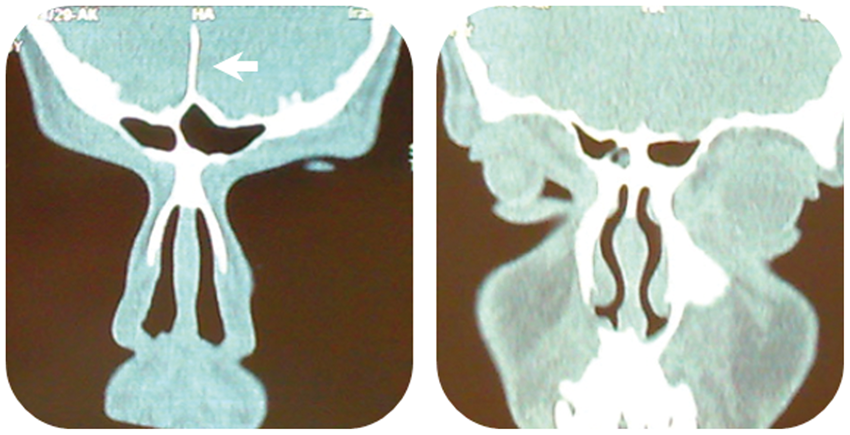
During the initial evaluation, panoramic x-ray revealed multiple bilateral unilocular cysts in the mandible (Fig. 1), and her chest X-ray showed bilateral bifid ribs (Fig. 2). Non-enhanced computed tomography (CT) scan demonstrated bilateral odontogenic cysts of the mandible and maxilla (Figs. 3 and 4), along with calcification of anterior part of the falx cerebri (Fig. 5). The patient underwent a biopsy of the left maxillary cyst under local analgesia and histopathologic evaluation revealed odontogenic keratocyst (OKC), nowadays better known as keratocystic odontogenic tumor (KCOT). She was eventually diagnosed with Gorlin-Goltz syndrome based on histopathologic and genetic analysis findings.
Panoramic radiography revealed multiple bilateral unilocular cysts (white arrow) in the mandibular bone. Bilateral bifid ribs (white arrow) are demonstrated by chest X-ray. Bilateral odontogentic cysts of the mandibule (white arrow). Coronal views show these cysts in the body of the mandibular bone. Expansions of cysts are seen into the gonial angles and the ramus of mandible. Bilateral odontogentic cysts of the maxilla (white arrow). Coronal view of anterior part of the maxilla (left) and sagittal view (right), which also shows sporadic tentorial calcifications. Calcification of the falx cerebri in anterior part (white arrow) is shown in coronal views of CT image. Coronal views of the anterior part (left), and posterior part of the falx cerebri (right), respectively.


Discussion
GGS is inherited in an autosomal dominant fashion. The diagnosis is made based on definitive criteria (1,3). A mutation in a tumor suppressor gene, which is located in the long arm of the chromosome 9, has been confirmed as the etiology of GSS. About 35–50% of cases result from new mutations (2). Other associated clinical presentations include basal cell carcinoma (BCC), KCOT, skeletal anomalies (hemivertebrae, scoliosis, syndactyly, polydactyly, or shortened fourth metacarpal bone), prognathism, and calcification of the falx and tentorium. Patients are predisposed to a higher risk for various neoplasms, such as medulloblastoma, meningioma, fibrosarcoma, and cardiac fibroma (1,4). Therefore, early diagnosis, appropriate workup and genetic counseling are necessary to help preventing possible fatal complications.
Multiple mandibular or maxillary cystic lesions should raise suspicion for KCOT. Dentists are frequently the first medical professional encountered such patients, who could be easily diagnosed by an oral panoramic radiograph. The cystic nature of KCOT has been debated in the literature, and some investigators classify KCOT as a benign tumor. Although in 1992, the World Health Organization (WHO) histologic typing of odontogenic tumors listed OKC as the preferred terminology for such cysts with a keratinized lining (5). In recent years, the term KCOT has replaced the term OKC to better reflect the neoplastic nature of the lesion. Several characteristics of cystic lesions including aggressive behavior, high mitotic activity, and associated genetic and chromosomal abnormalities, served as the basis for this new classification (6).
Multiple imaging modalities can be used to detect the components of GGS. Head and neck radiography can reveal calcifications of the falx cerebri, tentorium cerebelli, and diaphragma sellae, and routine chest X-ray may simply show bifid, hypoplastic, fused, partially missing, or splayed ribs in up to 60% of the patients; the third, fourth and fifth ribs are most commonly affected. Other possible imaging findings include malformations of the occipitovertebral junction, fusion of the cervical or upper thoracic vertebrae and small, pseudocystic or lytic bone lesions presenting as “flame-shaped lucencies” in the hands and feet (7).
The treatment of the KCOT is still controversial and various treatment options have been proposed including decompression and marsupialization, enucleation with/without adjuncts, enucleation and application of Carnoy solution to reconstruct the bony defect, enucleation and liquid nitrogen cryotherapy, and block resection with/without preservation of the jaw continuity (8). Factors affecting treatment outcome are the location and size of the cysts, presence of teeth, mucosal involvement, the development of infection, and association with the GGS (9). Altogether and considering these influencing factors, the treatment and prognosis of KCOT must be managed individually.
In conclusion, awareness of the pathognomonic radiologic findings (odontogenic cysts in the jaw, calcifications of the falx cerebri) will allow early detection and definite diagnosis of GGS. These imaging findings are accurately recognizable in a CT scan of the head and neck.
Footnotes
Ethics
The written informed consent was obtained from the patient and her family for submission of this manuscript for publication. This report followed the ethical standards supervised by the ethical committee of Tabriz University of Medical Sciences, Tabriz, Iran (consistent with the revised Helsinki Declaration in 2000).
Acknowledgments
Authors would like to thank Dr. Nafi Aygun (Johns Hopkins Hospital) and Dr. Behzad Salari (Harvard University) for their help.
Conflict of interest
None declared.
