Abstract
Anaplasma phagocytophilum, an obligate intracellular bacterium, is the causative agent of human granulocytic anaplasmosis (HGA), a tickborne infection usually manifesting as fever, malaise, cytopenia, spleen enlargement, and hepatitis. Herein, we report a case of a 14-year-old girl with HGA whose whole-body magnetic resonance imaging (MRI) disclosed an unusual picture characterized by small, widespread punctuate millimetric nodules, hypointense on T1-weighted and hyperintense on STIR sequences. This firstly reported finding may represent an alternative tool for identifying atypical infectious diseases.
Introduction
Anaplasma phagocytophilum is a Gram-negative, obligate intracellular bacterium which causes human granulocytic anaplasmosis (HGA). Epidemiological studies have identified endemic areas mostly in rural areas: in the United States the highest incidence rates of HGA are in the northeastern and upper mid-western states: other endemic areas are known in Asia and in Europe.
Clinically HGA is characterized by long-term fever and malaise, arthralgia, headache, myalgia, and spleen enlargement: thrombocytopenia, leukopenia, and increased transaminase activities are commonly observed. More severe symptoms such as meningoencephalitis or myocarditis may occur.
Most systemic infections are diagnosed on the basis of the clinical symptoms (fever, asthenia, malaise) associated with positive bacterial culture and serological investigations, and are subsequently treated with intravenous antibiotics therapy. In some misleading cases, the diagnosis may be supported by radiologic examination methods. The purpose of the current report was to describe the whole-body magnetic resonance imaging (WB-MRI) pattern of lesions in a case of a systemic HGA infection.
Case report
A 14-year-old girl with an uneventful clinical history suddenly developed a severe syndrome characterized by intermittent-remittent fever with spikes up to 41℃, headache, meningismus, drowsiness, myalgia, and arthralgia without arthritis. A mild liver and spleen enlargement was present.
Routine investigations showed increased acute phase reactants (erythrocyte sedimentation rate [ESR], levels of 90 mm/h; C-reactive protein [CRP], 14.7 mg/dl), anemia [hemoglobin 9.6 g/dl], total white cell count 6.4 × 103/µL, thrombocytopenia [128 × 103/µL], and liver involvement [aspartate transaminase 322 UI/L, alanine transaminase 485 UI/L, and γ-GT 295 UI/L]). Serology for some hepatotropic viruses (i.e. hepatitis viruses A-B-C, Epstein-Barr virus, cytomegalovirus) was negative. The autoantibody panel showed a slight autoantibody positivity of anti-smooth muscle antibody (ASMA) (1:40) and perinuclear anti-neutrophil cytoplasmic antibodies (p-ANCA) with an atypical pattern.
Due to the increased transaminase and γ-GT plasma levels a liver biopsy was performed with the results being consistent with a non-specific hepatitis pattern. Moreover, brain MRI, cerebrospinal fluid analysis and bacterial culture ruled out encephalo-meningitis; bone marrow biopsy and bone scintigraphy were also negative. Antibiotics treatment (amoxicillin-clavulanate 90 mg/kg/day three times a day) was started without significant clinical response. Since fever spikes and myalgia/arthralgia persisted, the girl was admitted to our department 30 days after disease onset.
At admission, the patient complained of recurrent fever spikes and severe malaise, although according to the parents’ opinion the general conditions of the girl had been ameliorated. Laboratory investigations showed positive acute phase reactants (ESR 75 mm/h and CRP 3.9 mg/dl), hypergammaglobulinemia (1.73 g/dl) with increased IgM (241 mg/dl) and IgA (490 mg/dl) plasma concentrations: notably, transaminase activities were normal with persistently increased γ-GT 114 UI/L. As a diagnosis had not yet been reached, we performed a WB-MRI on a 1.5 T MRI scanner (Achieva Intera; Philips Medical Systems, Best, The Netherlands) using body coil with a field of view covering from head to toes. WB-MRI images were acquired (and displayed) in coronal plane using a turbo spin echo T1-weighted (T1W) sequence (TR, 630 ms; TE, 17 ms; slice thickness, 5 mm; gap, 0.5 mm; NSA, 1) and a turbo spin echo STIR sequence (TR, 2800 ms; TE, 165 ms; TI, 64 ms; slice thickness, 5 mm; gap, 0.5 mm; NSA, 3). Total scan time was about 30 min. No contrast medium was administered during the examination.
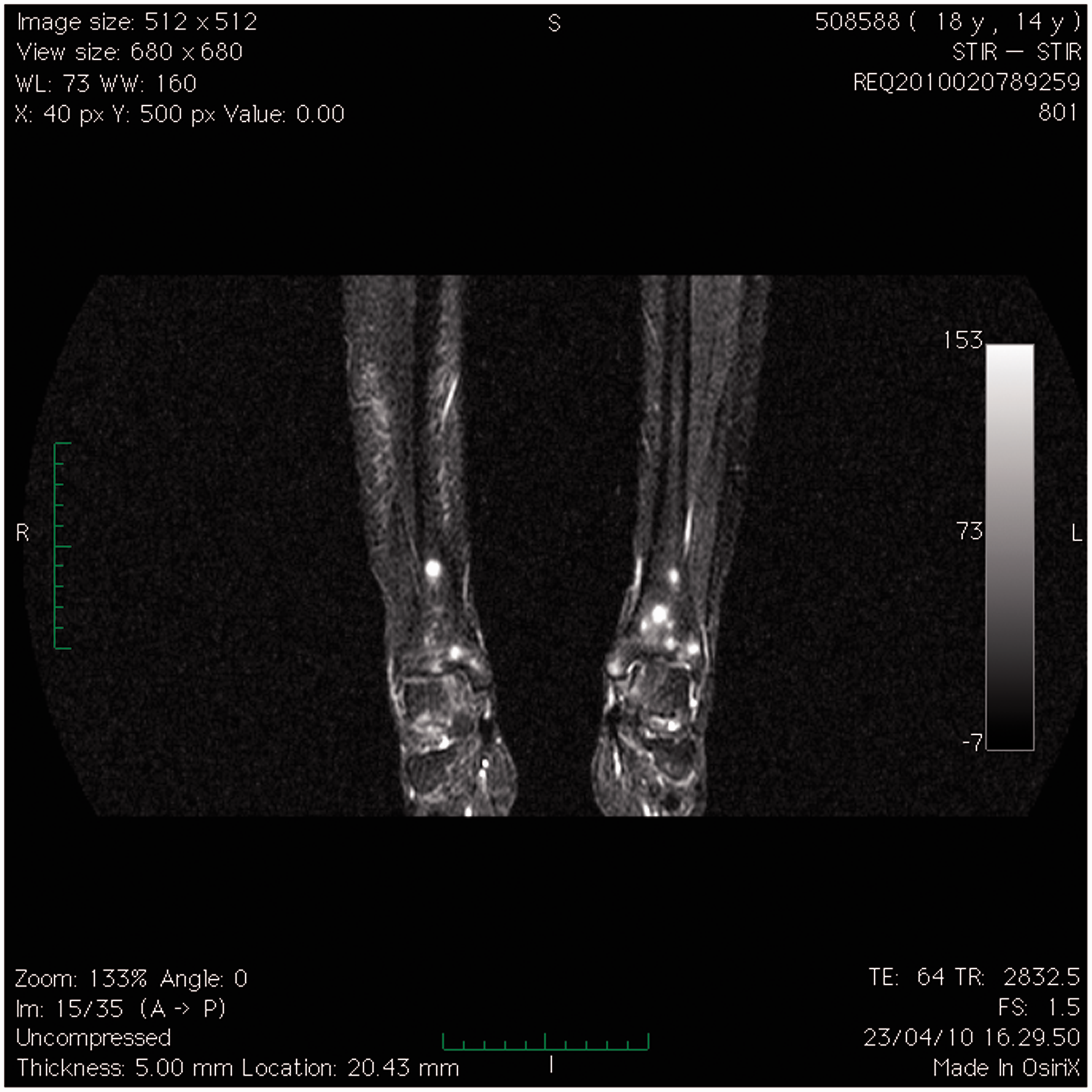
The MR investigation was carried out with the aim to rule out hemato-oncologic diseases and occult abscess. Surprisingly the WB-MRI showed an unusual pattern characterized by millimetric, widespread punctate nodules, respectively hypo-hyperintense in T1W and STIR sequences. These nodules were mainly localized in the meta-epiphyseal regions of the long bones of both legs (mostly at the level of the distal femur and proximal tibia) as further documented by an additional dedicated MRI of both knees (Figs. 1, 2a, 2b, and 3). According to the radiologist’s opinion, these images were possibly related to a hematologic intramedullary spread of an infectious process or to a post-infectious thrombotic microangiopathy.
Mobiview reconstruction of a coronal TSE STIR sequence acquired in a coronal plane. Notice the presence of multiple millimetric, widespread punctate nodules, hyperintense in STIR. These nodules are mainly localized in the meta-epiphyseal regions of long bones of both legs (mostly at the level of distal femur and proximal tibia). Coronal TSE STIR (a) and T1W (b) sequences over both knees, with a dedicated coil, better define the particular pattern of distribution in the meta-epiphyseal regions, mainly of distal femur, of these highly hyperintense (a) patchy nodules. Coronal TSE STIR sequence over both ankles reveals the presence of patchy meta-epiphyseal highly hyperintense nodules also in the distal tibia.

Upon this finding, and taking into account the apparent self-limiting disease course and the rural area where the patient lived, further investigations were conducted to rule out zoonosis. Serologic assay of anti-Anaplasma phagocytophilum showed an IgM titer of 1:40 (positive, >1:64) and an IgG titer of 1:160 (positive, >1:64). Hence intravenous ceftriaxone treatment (80 mg/kg daily) was started with a prompt clinical improvement (i.e. disappearance of fever, myalgia, malaise, and headache). The patient has not had a relapse after about 1 year of treatment.
Discussion
We report a pediatric patient affected with HGA who showed an unusual pattern of lesions at WB-MRI.
Anaplasmosis is caused by Anaplasma phagocytophilum, a Gram-negative, obligate intracellular bacterium with has a tropism for neutrophils.
This infectious disease (formerly known as human granulocytic ehrlichiosis) generally manifests itself as a non-specific febrile illness associated with non-specific signs and symptoms such as malaise, headache, myalgia, and arthralgia. Cytopenia and asymptomatic liver involvement with increased transaminase activity are commonly found. Rarely, life-threatening complications including encephalitis, myocarditis, acute respiratory distress syndrome, and shock-like syndrome have been reported with a mortality rate in the range of 2–5% (1).
The fever has a long-term course with phases of flare-up and remission (saddleback fever): this finding may be a misleading feature mimicking inflammatory diseases. It is of note that our patient came to our attention with the clinical suspicion of connectivitis.
Anaplasma phagocytophilum is transmitted to humans via the bite of a tick (Ixodes spp): infected reservoir hosts are small rodents and the white-tailed deer. Epidemiological studies have identified an endemic region for HGA in Italy, localized in the middle alpine region with higher seropositive rates for Anaplasma phagocytophilum (up to 8%) in the Isarco river basin and South Tyrol (2). Notably, our patient was coming from this area. Alternative modes of transmission have been reported: they include transplacental infection of an infant (3) and blood inhalation or percutaneous blood exposure following butchering of white-tailed deer (4).
WB-MRI is a fast and accurate modality for detection and monitoring disease throughout the entire body. For pediatric use the technique is of two-fold special interest: first, it is a radiological method without radiation exposure; and second, it allows for whole-body staging thus reducing the number of examinations to be performed in sedation. In the pediatric population this technique is used for staging of oncological and non-oncological diseases (i.e. fever of unknown origin, osteonecrosis) and for disease severity assessment of syndromes affecting the whole body. Although it is not recommended as a first-line investigation in every patient, it may add important information in difficult clinical cases (5,6). In the current case a WB-MRI was conducted in order to exclude systemic hemato-oncologic diseases or possible occult abscess. Interestingly WB-MRI depicted an unusual systemic involvement of the bone marrow with a characteristic punctate pattern and a typical peri-meta-epiphyseal distribution. Patchy foci of low signal intensity on T1W and high signal intensity on fat-saturated T2-weighted and STIR sequences are commonly described in the marrow of the feet and wrists of asymptomatic children and adolescents and might represent foci of residual red marrow, however in this case the depicted nodules showed a higher signal intensity and a different pattern of distribution which ruled out the possibility of residual normal red bone marrow (7). We also ruled out “edema-like marrow lesion” which is quite typical of a variety of conditions (e.g. trauma, infection, avascular necrosis, or other inflammatory conditions like chronic recurrent multifocal osteomyelitis) with diffuse, uniform and symmetrical marrow involvement (7,8). The WB-MRI pattern was also not indicative of bone infarction conditions which are characterized by typical metaphyseal “geographic” lesions (with an outer rim of sclerosis, an inner rim of granulation tissue, and a central core of fatty marrow) (7). The bone marrow signal alteration revealed by MRI in this case was characterized by multiple well defined millimetric punctate peri-meta-epiphyseal nodules which may correspond to the hematologic spread of an infectious disease (in this case the intramedullary spread of a Gram-negative, obligate intracellular bacterium with a tropism for neutrophils). Although a specific diagnosis might not be possible on the basis of the imaging findings, recognition of this pattern of marrow signal abnormality can refine the differential diagnosis and direct clinical investigations to this family of zoonosis.
In conclusion, WB-MRI with STIR sequence could be a possible diagnostic tool in patients with unusual systemic infections.
Footnotes
Conflict of interest
None declared.
