Abstract
Background
Trends in interventional radiology and urology training are orientated towards reducing costs and increasing efficiency. In order to comply with the trends, we propose training on inexpensive patient-specific kidney phantoms.
Purpose
To develop a new kidney phantom for puncture and drainage training in interventional urology and radiology, and to evaluate their anatomical correctness and suitability for training compared to the traditional way of training on home-made phantoms.
Material and Methods
A case study for validation of kidney phantoms was conducted with nine radiology students divided into two groups: one trained on standard home-made training phantom (n = 4) and the other on our kidney phantoms (n = 5). Another test phantom was used to evaluate the effectiveness of the training of the two groups. The tests were video recorded and analyzed. Duration of the procedure was used as the primary indicator of procedure’s quality. Comparison tests were also conducted with professional radiologists. Anatomical correctness of the kidney phantom was evaluated by comparing the post mortem kidney scans with reconstructed models from CT scans. Subjective feedback was also collected from the participants. Wider use of kidney phantoms was analyzed.
Results
The average volumetric difference between post mortem kidney scans and reconstructed CT kidney models was 4.70 ± 3.25%. All five students practicing on the kidney phantom improved their performance and the results were almost equal to the results of the professional radiologist while in the other group two students out of four trained on standard home-made training phantoms failed to improve their performance. However, the small number of test subjects prevents us from drawing general conclusions about the efficiency of the new practice. The kidney phantoms were found usable also for nephrostomy catheter placement training under fluoroscopy.
Conclusion
The feedback from radiologists showed that the anatomically correct features of the phantom is an added value for the training and has a potential to increase the quality of minimally invasive procedures in general.
Introduction
Interventional radiology is a rapidly developing area of minimally invasive procedures (1). The vast amount of skills that urology and radiology residents or other specialty residents dealing with minimally invasive procedures must learn during their training period, makes phantoms (artificial replica of human organs) a desired test platform so that the residents can get their first hands-on experience before everyday practice (2). Phantoms are not only useful for skill development in needle guidance and ultrasound (US) probe handling for residents but also for practicing radiologists to improve their skills with new techniques and ever changing equipment (3–5) as well as to increase their confidence before procedures on patients.
In our previous work we developed an anatomically shaped kidney phantom for interventional radiology trainees (6) suitable for practicing variable needle thickness biopsies using US guidance. That kidney phantom prototype did not have drainage functionality. In this work we widen the applicability of our patient specific phantoms for nephrostomy drainage by introducing a kidney phantom with calyxes and fluid flow.
The purpose of this study was to develop anatomically correct kidney phantoms with calyxes for training interventional urology and radiology. The goal was to evaluate the kidney phantoms’ suitability for training in the 1-week introductive course to interventional radiology compared to the traditional way of training on home-made phantoms and to evaluate the kidney phantoms’ anatomical correctness by comparing the postmortem scans of physical kidneys retrieved from pathologists with preoperative kidney models reconstructed from computed tomography (CT) scans.
Material and Methods
Phantom materials
In our previous work (6) we compared different phantom materials and chose to use gelatin gels as the tissue-mimicking material as they are self-supportive solids with a proper range of achievable elasticity, US characteristics, and a relatively simple manufacturing process. A thorough characterization of the material was performed (composition versus properties).
Phantoms’ description
In this work we used three different phantoms, two of them were made from gelatin mixture by using the recipe described in our previous work (6) (referred to as kidney phantom (A) and test phantom (B) further on) and one was created by the radiology students in the hospital (referred to as home-made phantom (C)).
The kidney phantoms (A) and home-made phantoms (C) were used in the training of two groups of residents. The test phantom (B), which was not known to the residents before the tests, was designed to evaluate the groups’ performances after the training.
Kidney phantoms (A)
A patient-specific kidney phantom was created based on a CT scan. The model of the kidney was created by segmenting the CT images and creating an STL model using 3D Slicer software. Using the kidney model, a two-sided mold was modeled using 3D CAD design software Solidworks. The mold was made of PLA plastic (Poly Lactic Acid) using rapid prototyping technique.
Prior to the casting of the kidney phantoms, three reservoirs representing the kidney calyxes were prepared from air balloons with a diameter of 20–23 mm and placed into the mold. The calyxes were connected with silicone pipes (inner diameter, 3 mm) to outer reservoirs (made of 12 mL syringes) after preparing the kidney phantom. The kidney was placed into a box (20 × 10 × 10 cm) and surrounded with an additional gelatin medium. Before casting the surrounding medium, air balloons were removed from the kidneys. The upper surface of the phantom was covered with a pigmented silicone layer (EcoFlex 0030). The three calyxes and corresponding reservoirs were filled with differently colored water. Different colors were used for that it would be possible to check that the correct calyx was punctured and drained. The design of the phantom can be seen in Fig. 1.
Kidney phantom model and prepared kidney training phantoms.
Test phantom (B)
For comparative experiments between the two groups of radiology trainees, another test phantom was prepared as well. This test phantom did not have an anatomically correct form but had the same basic functionality as the kidney phantom (permitting puncturing calyxes filled with different colored liquids).
Using the same mixtures described in (6), a rectangular box shape (9.5 × 9.5 × 5.0 cm) test phantom having four 20–23 mm calyxes connected to four reservoirs was prepared. The box-shaped phantom was placed in the middle of a larger box (18.5 × 16.0 × 10.0 cm) and the surrounding space was filled with a clear gelatin medium. The upper surface of the test phantom was covered with a pigmented silicone layer (EcoFlex 0030).
Home-made phantom (C)
The control group of the radiology residents used their standard training materials – simple home-made phantoms.
The home-made phantom that the residents prepared consisted of fruits and vegetables (grapes, pieces of cucumber and tomato, etc. representing tumors and lesions) casted into a bowl filled with a regular food gelatin mixed with water. The home-made phantom was placed into a refrigerator for hardening. A thin layer of opaque plastic sheet was placed on the surface of gelatin to prevent any direct guidance of the needle.
Evaluation of the kidney phantoms
Quality assessment of the reconstruction of the patient-specific kidney phantoms (A) and their usability tests are described in the sections below.
Anatomical reconstruction validation of the patient-specific kidney phantoms
In order to validate the reconstruction quality of the patient-specific kidney phantoms we recorded postmortem scans of patient kidneys (by using DAVID-Laserscanner software with a Logitech C920 webcam and an Optoma ML300 LED video-projector) and compared the results with reconstructed models from CT data.
Inclusion criteria were set for the patients. The time between the autopsy and recording of the CT scan had to be 14 days or less. Only kidneys without acute pathology, without solid tumors and cysts less than 3 cm were included.
The computer models of kidneys were built using the following procedure:
Patient kidneys were removed from the abdomen and excess fat around the kidney was removed; The ureter, renal vein, and the renal artery were cut off close to the renal pelvis; Each kidney was hung with a rotating hook to a portable frame and scanned 12 times from different angles to maximize the covered surface; The obtained scans were semi-automatically merged together into a single mesh. The mesh defined the surface of the kidney model.
The volumes of the physical kidneys and kidneys segmented from CT scans were compared. An approval from Tallinn Medical Research Ethics Committee was granted before the work with patients CT scans and postmortem organs (decision number 2527).
Training protocol
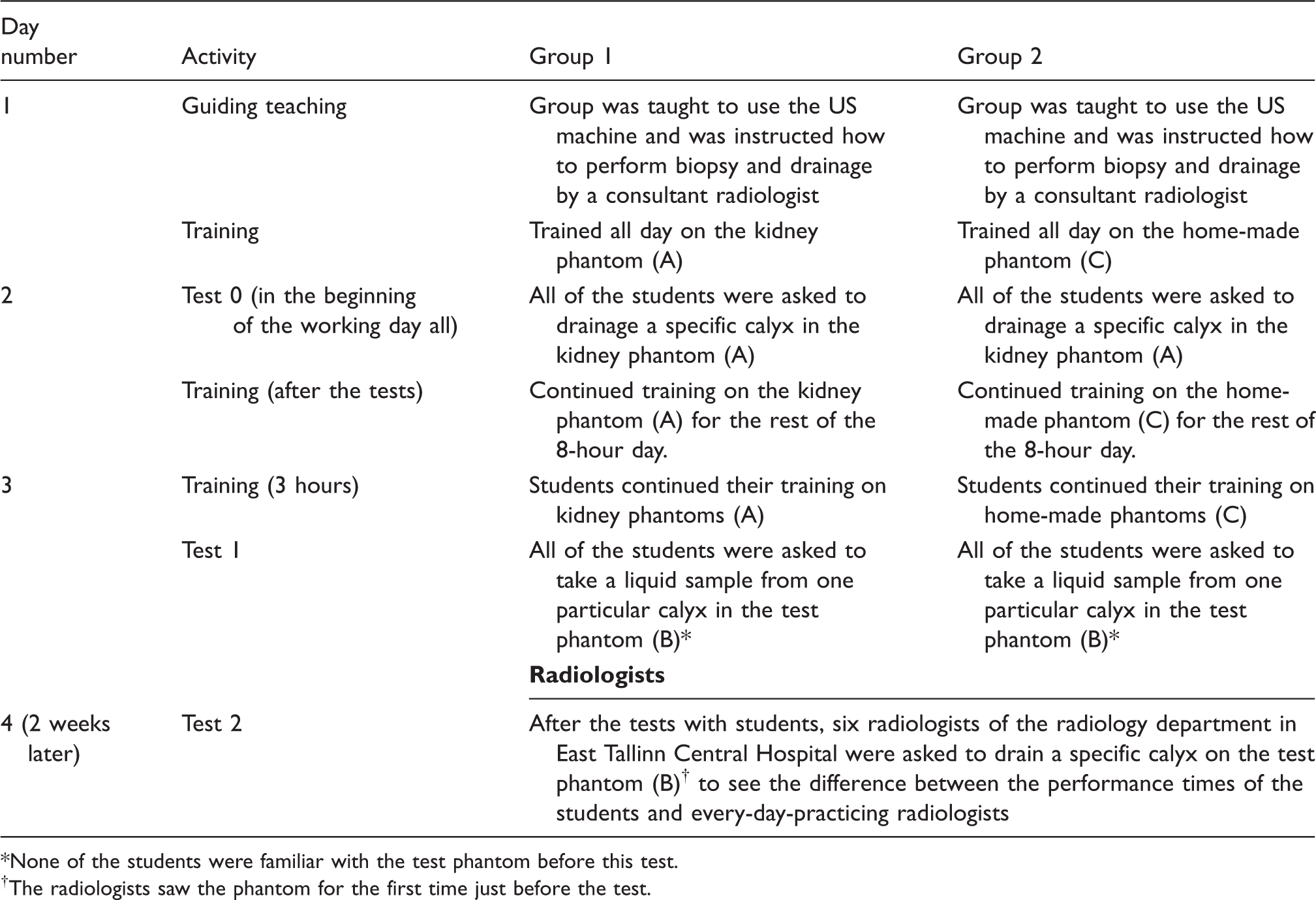
Radiology second year residents were validating our kidney phantoms (A) during a 5-day interventional radiology training as an introduction to interventional radiology of their 5-year radiology residency curriculum. In order to quantitatively validate the progress of the trainees and to study if there was any difference between training on anatomically shaped kidney phantoms (A) and home-made phantoms (C), we prepared a case study where:
We divided the residents into two groups: Group 1 consisting of five residents trained on the kidney phantom (A) and Group 2 consisting of four residents trained on the home-made phantom (C); We video-recorded the tests; The duration of the puncture procedure (time from identifying the target until extraction of the liquid) was measured from the recordings; Comparison of the puncture procedure’s duration before and after the 3-day hands-on practice were compared; All tests were performed using Philips US equipment (Philips Medical Systems, Best, The Netherlands) with the linear 5–8 MHz probe; A questionnaire was conducted to evaluate the suitability of the phantoms. Training protocol overview. None of the students were familiar with the test phantom before this test. The radiologists saw the phantom for the first time just before the test.
The outline of the tests is described in Table 1.
The puncture procedure was done by using freehand US guidance method with following steps of the procedure:
Adding US gel to the surface of the phantom and examining the phantom with linear probe; Choosing the target point, deciding on the depth of the procedure, and choosing the needle; Keeping the US probe in place, puncturing the skin with the needle from the correct side and in the same plane of the US probe; Inserting the needle into the target while keeping the target and the needle visible in the US image; Extracting liquid from the target.
Alternatively to the freehand technique, biopsy guiding systems embedded to the US probe could be used to perform this training. Guiding system fix the needle to the probe in a way that the needle follows US image plane and does not allow adjusting the course of the needle after the insertion through the skin. This method would allow the residents to perform the puncture procedure with better results but it would leave them without practical skills of hand–eye coordination that is necessary in freehand technique.
After the test we asked the participants to fill in a questionnaire. The results of the questionnaire and training performance analysis are shown in the Results section.
Improvement and wider use of the kidney phantoms
In order to test if the kidney phantoms had other applications besides the puncture and drainage functionality, we tested different imaging and treatment techniques used in radiology.
Also throughout the training of the residents and during the session with radiologist, feedback was received to improve the phantom’s realism.
Results
Evaluation of the phantoms
Comparative results of the home-made phantom and the kidney phantom.

Rated on scale 1 (not important) to 5 (very important).
Standard deviation in brackets.
Probability that KP phantoms score higher than HMP.
HMP, home-made phantom; KP, kidney drainage phantom.
The results show that the kidney phantom is more suitable for practicing drainage than the home-made phantom (C) (4.40 vs. 1.60), the consistency is better for training (4.90 vs. 3.80), and that the kidney phantom could be used in addition to the training cycle of the residents in everyday practice for longer periods of time (5.00 vs. 4.00). In other aspects no notable differences between the phantoms A and C were found.
Training results
In Group 1, two subjects failed to improve their time of drainage comparing Test 1 to Test 0. Two residents in Test 1 (one from both Groups 1 and 2) were identifying the drainable calyx longer than in Test 0.
Overall, 3-day training test results revealed that Group 1 improved their average drainage time as it is shown by comparing the Test 0 and Test 1 73.60 (77.96) s (mean value (standard deviation) s) (from 117.80 s to 44.20 s). The mean drainage time of the Group 2 improved during the 3-day training 3.00 (11.34) s (from 48.25 s to 45.25 s). Both groups’ average times of target identification in the phantom improved: Group 1 by 6.40 (4.59) s (from 20.40 s to 14.00 s) and Group 2 by 1.00 (1.87) s (from 12.00 s to 11.00 s).
Radiologists’ average time of completing the drainage took 47.33 (19.11) s (average identification time of the target 10.67 (3.73) s).
Anatomical correctness
The average volumetric difference between the postmortem kidney scans and the reconstruction from CT data of eight kidneys was 4.70 ± 3.25%. An image of the scanned kidney, its corresponding CT model and the postmortem scanned model are shown in Fig. 2.
Reconstruction of kidneys (A – postmortem kidney; B – postmortem kidney scan; C – model from CT scan).
Wider use of the kidney phantoms
The calyxes of the kidney-phantoms (A) can be filled with radio-opaque X-ray contrast medium through the connecting tubes to make the desired calyx visible in fluoroscopy. A similar method is used in an operating room in real life to visualize calyxes. Another option is to inject X-ray contrast medium under the US guidance into the fluid-filled phantom calyx after the antegrade puncture of the phantom calyx and verify the correct position of the needle tip in fluoroscopy. This can be followed by the insertion of a guide wire, dilator, and finally nephrostomy catheter simulating the real-life procedure.
The feedback from radiologists revealed that the size and proportions of the kidney phantom were appropriate for the training. The resistivity and stiffness of the surface, surrounding material, kidney, and collecting system of the phantom during the needle insertion were close to a real-life situation. Also US and X-ray properties were well accepted.
Improved version of the kidney phantom
According to the suggestions from the radiologists and residents, a kidney phantom with six calyxes was prepared (7). The walls of the calyxes were made out of long narrow air balloons (to enhance the tactile feedback) and were placed into the kidney mold prior to casting. The connecting tubes to the calyxes were orientated and bound together in a way that they represent the declining ureter. The reservoirs made out of syringes were placed outside of the phantom (Fig. 3).
Kidney phantom with six calyxes.
The US image of the improved version of the kidney phantom with visible calyxes can be seen in Fig. 4.
US image of the improved kidney phantom.
Discussion
We have produced an affordable disposable patient-specific puncture and drainage training kidney phantom with the possibility to practice nephrostomy insertion. The affordable price and disposable content of the kidney phantom are valuable properties that could motivate urology and radiology departments to use phantoms not only for the training of residents but also encourage experienced specialists to use phantoms before performing rarely used techniques or new equipment. This can enhance the quality of minimally invasive procedures. Our kidney phantom was well accepted by the radiology students and staff who preferred to use our kidney phantoms (A) in training and in everyday practice over home-made phantoms (C). The low score in thick needle usage of the new kidney phantom can be resolved by using the results of our previous work.
As of the training analysis, video recordings proved to be a very useful tool not just for time recording but also for post-training analysis of the correct methods and the posture of handling the syringe and the US probe by the students.
The analysis of the training showed that the group practicing on our kidney phantom (Group 1) that had much lower scores in the beginning of the training, improved significantly (73.60 s) compared to students practicing on the home-made phantom (Group 2) (3.00 s). In the end of the training Group 1 performance had caught up and somewhat exceeded Group 2 by 1.05 s. Professional radiologists performed 70.47 s faster than Group 1 and 0.91 s faster than Group 2 in the initial test (comparing Test 0 and the test with the radiologists), which shows that the radiologists are well prepared in hand–eye coordination when it comes to working with new kind of objects.
The results of the comparison of the volumetric data of a cadaveric kidney reconstruction with CT scan reconstructions show that the kidney phantom produced by using 3D printed molds constructed from the CT scan data simulates reliably the real-life situation.
There are limitations of our study. The small size of the group restricts us from making general conclusions about the results but it can be expected that students who train on anatomically correctly constructed phantoms gain experience faster. Some changes in the kidney phantom could also improve the training quality. For example, the US image quality of the phantom could be improved by increasing the attenuation of the surrounding medium of the kidney, the problem with needle marks left after needle extraction need attention, and the reality of the phantom could be increased by introducing pulsating blood vessels.
In conclusion, our phantom was well received by the residents and staff. The tests showed that our kidney phantom is suitable for objectively evaluating the work and training of the radiology residents. According to the feedback from the radiology students and staff we further developed the kidney phantom by improving the tactile feedback of needle insertion and anatomical complexity and correctness.
Footnotes
Acknowledgements
The authors thank East Tallinn Hospital for using their equipment, radiologists and residents for participating in the tests.
Funding
This work was funded by European Union FP7 projects SAFROS and ISUR.
