Abstract
Background
The import of the cavum variation and its prevalence rate in healthy individuals is still not clear, likewise in neurologically diseased patients.
Purpose
To evaluate the frequency and pattern of caval variations in neurologically diseased patients.
Material and Methods
The presence or absence of the cavum septum pellucidum (CSP), cavum vergae (CV), or cavum velum interpositum (CVI) was reviewed from successive cranial computerized tomography (CT) images of patients who were aged 6 months and above. Two hundred and seventeen cranial CT images were reviewed.
Results
At least a cavum variation was noted in 130 (59.9%) of the CT scan images reviewed. The CV, CVI, and CSP were noted in 86 (39.6%), 53 (24.4%), and 50 images (23%), respectively. Caval multiplicity was noted in 102 patients (47%). There was no significant difference in the rate of occurrence of cavum variations in patients with congenital brain diseases and acquired brain conditions (P = 0.484), neither was there a significant difference in the frequency of cavum variation in children aged older than 6 months compared to adults (P = 0.101).
Conclusion
Cava variations are relatively common in neurological brain diseases. Patients with congenital brain diseases did not have a higher frequency of cava variation when compared with those that had acquired lesions. The most common type of cavum variation noted in this study was the vergae variety, while the CSP is the rarest.
Introduction
The cavum septum pellucidum (CSP) is part of normal development. It is a fluid-filled cavity located between the membranes of the septi pellucidi and usually isolated from the ventricular system (1). In normal fetuses, the CSP can be visualized between 18 and 37 weeks gestation. It is usually found in all premature and persists shortly after birth in approximately 50% of term neonates (2). A CSP is present in 10–40% of adults (3,4).
Anteriorly the CSP is demarcated by the genu of the corpus callosum, posteriorly by the columns and body of the fornix, superiorly by the body of the corpus callosum, and inferiorly by the rostrum of the corpus callosum (5,6). The cavum vergae (CV), on the other hand, is bordered anteriorly by the posterior border of the CSP, inferiorly by the body of the fornix, and superiorly and posteriorly by the corpus callosum. The CSP may also communicate with the cavum vergae through the aqueductus caudae septi or aqueductus ventriculi vergae (7). In time past the CSP and CV were considered as the fifth and sixth cerebral ventricles. Another cavity, known as the cavum velum interpositum (CVI), is part of the leptomeningeal space and intervenes between the roof of the third ventricle and the body of the fornices. When the corpus callosum is absent, the cavum septi becomes unroofed and, therefore, unidentifiable.
The significance of the finding of a cava variation and its prevalence rate in healthy individuals are still not clear, likewise in neurologically diseased patients. Some studies have suggested that the persistence of the CSP and CV beyond the neonatal period may be a marker of cerebral dysgenesis. Spillane first incidentally noted the association of a CSP and boxers (8). Studies by DeGreef et al. originally described the increased prevalence of the CSP in schizophrenia (9,10). Many researchers have since made similar conclusions (5,11–13). In schizophrenia the presence of a CSP is one marker that has been consistently found in imaging studies and neuropathological reports (14–17).
There is scanty or no information on the frequency and persistence of CSP, CV, and CVI in neurologically diseased patients in the literature generally. Our aim was to study the frequency and pattern of caval variations in neurologically diseased patients.
Material and Methods
A cohort, prospective study of 217 patients was done. The study was conducted at Lagos State University Teaching Hospital, Ikeja, Lagos, South Western Nigeria; a tertiary care referral center in the state. The target population for this study were a cohort of 217 consecutive Nigerian patients (adults and children aged older than 6 months), who were sent for cranial computerized tomography (CT) scan on account of various neurological and neurosurgical pathologies (congenital and acquired.). A GE Hi-Speed Dual CT scanner® (GE Healthcare, Milwaukee, WI, USA) was used. All study subjects were positioned supine in the scanner gantry and scanned from skull base to the vertex with contiguous axial slices parallel to the inferior orbitomeatal line, at 5 mm slice thickness at intervals of 3 mm.
The CT scan images were reviewed by all the authors. The presence or absence of the CSP, CV, or CVI were noted and documented in a proforma form. Information regarding demography and indication for cranial CT was obtained from their referral forms for documentation. Patients aged less than 18 years were classified as children and the patients CT diagnosis was documented and further classified into congenital and acquired causes.
The data collected were checked for errors prior to analysis. Data analysis was performed using the SPSS version 16 (SPSS Inc., Chicago, IL, USA). The association between categorical variables was investigated by means of chi-square (or Fisher’s exact test) and means compared by independent T-test. A P value less than 0.05 was considered statistically significant. The study followed principles in the Declaration of Helsinki.
Results
Two hundred and seventeen CT scan images were reviewed. There were 121 male patients and 96 female patients. The patients’ ages ranged from 6 months to 87 years, with a mean and median of 39.3 (male patients, 40.1 years; female patients, 38.3 years) and 41 years (male patients, 42.0 years; female patients 40.0 years), respectively. Children accounted for 55 of all patients with a mean age of 6.1 years (median, 6.0 years), while adult mean age was 50.1 years (median, 52.0 years). Patients with cava variations were older than those without any form of caval variation (Fig. 1); however, there was no statistical difference (P = 0.713). There were no statistical significant age difference in patients with or without CSP (P = 0.623), CVI (P = 0.600), or CV (P = 0.702).
Box-plot of age of patients and the presence or absence of cavum variations.
Indication for cranial CT compared with presence or absence of cavum anomaly.

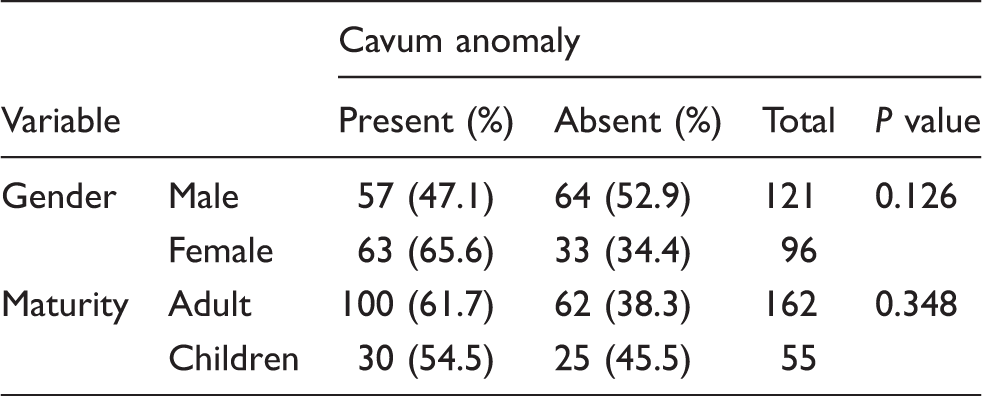
At least one cava variation was noted in 130 (59.9%) of the patient CT scan images reviewed (Fig. 2). One hundred and two of the images had multiple cava variations (47% of all patients). The CV, CVI, and CSP were noted in 86 images (39.6%), 53 images (24.4%), and 50 images (23%), respectively. Patients with congenital brain diseases did not have significant caval variation rates compared to those with acquired brain diseases (P = 0.481). The presence or absence of a caval variation was not dependent on gender (P = 0.126), or whether the patient was a child or adult (Table 2); neither was it significantly related to the etiology of the brain pathology (P = 0.783).
Cranial CT scan images of CSP, CVI, and CV. Cavum anomaly presence in patients.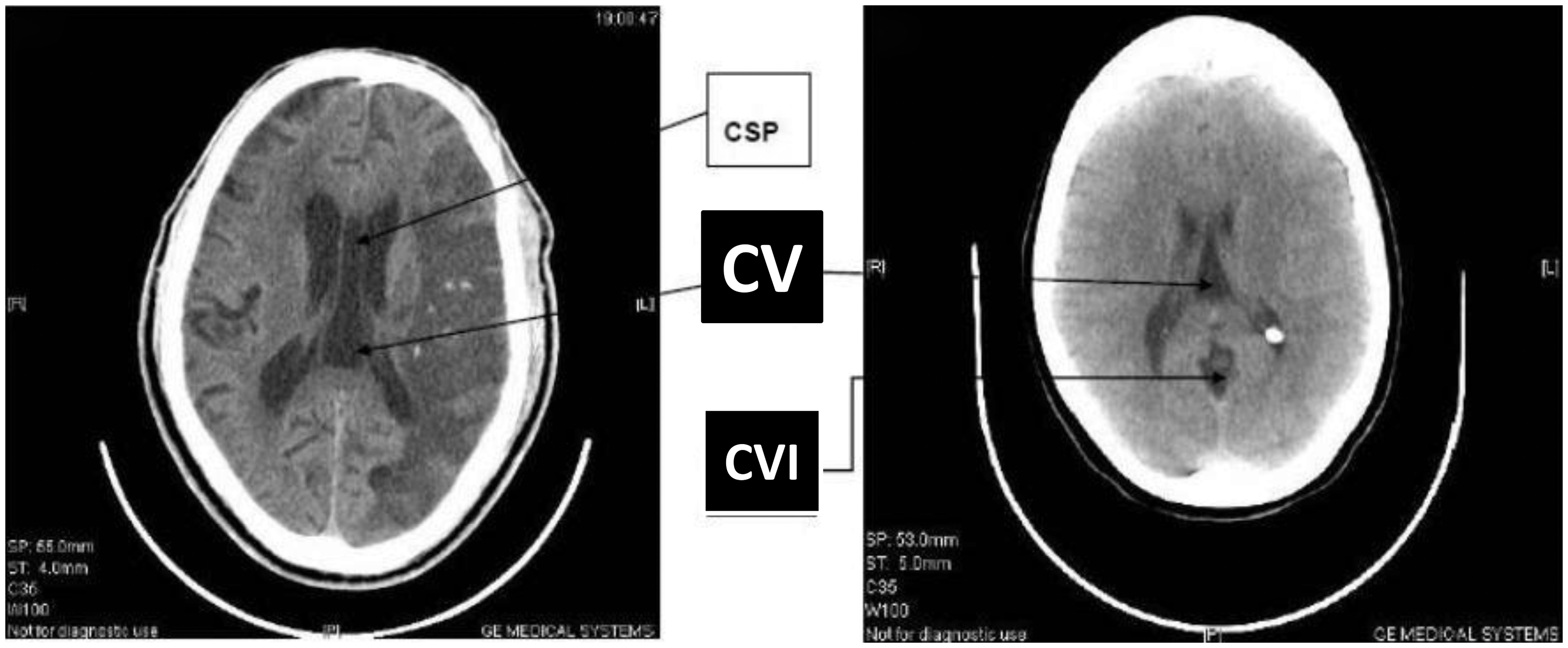
There was no significant relationship between the presence of CVI with respect to the patients’ gender (P = 0.860). The presence of CVI was significantly higher in adults (adults, 27.8%; children, 14.5%; P = 0.048) but not in those with congenital brain malformations (congenital brain lesions, 28.6%; acquired brain lesions, 24.1%; P = 0.460) (Fig. 3).
Cavum septum interpositi presence and absence in the CT scan images.
The CV was more common in female patients (47.9%) compared to male patients 33% (P = 0.030) while CSP was more common in male patients (male patients, 24.8%; female patients, 20.8%; P = 0.491). The presence of CV and CSP was not significantly related to whether the patient was a child or adult (CV, P = 0.118; CSP, P = 0.535) or whether the pathology was congenital or acquired (CV, P = 0.746; CSP, P = 0.591). The presence of cava variation multiplicity was also not significantly related to gender (P = 0.120), or etiology of disease (P = 0.830). However, there was a significantly higher rate of caval multiplicity in adult patients (P = 0.041).
Discussion
One of the first evolving processes in brain is the development of the ventricular system and the associated septum separating the two chambers (15). Normally, from the second to the fifth months of embryonic and fetal development, the lateral ventricles elongate and expand laterally away from the midline along with the expansion of the cerebral cortex. The frontal, occipital, and temporal horns as well as the bodies of the lateral ventricles become delineated and increase in size through the seventh month of development. The septum pellucidum forms a medial wall between the body and the anterior horn of the lateral ventricles. Incomplete fusion of the laminae can manifest as one or two separate cavities: a CSP and/or a CV.
The presence of CSP has been recognized at least since the 15th century; and the CV since the 19th century (5,18,19). It is sometimes called the fifth ventricle. The term fifth ventricle is, however, a misnomer, because unlike the ventricular cavities the caval variations do not usually contain cerebrospinal fluid. Communication with the ventricular system occurs only when the caval leaves are ruptured or fenestrated. Hence its content may originate from either the ependymal secretion of the leaflets or from a communication with the third ventricle (20). Hsu et al. noted that the most common normal caval variation in neonates was the presence of cavum septum pellucidum (1979 cases, 62.1%) (21).
Controversy exists over the significance of the isolated finding of caval variations and its prevalence rate in healthy individuals is not well elucidated. Spontaneous enlargement of the cavum septi pellucidi may impede CSF circulation and cause hydrocephalus (22).
Computerized tomographic (CT) imaging, magnetic resonance imaging (MRI), and postmortem brain studies have reported a wide variance in the range of 0.1–85% in the prevalence of the CSP in normal adults (12,23–28). In Hagino et al.’s study of 79 volunteered normal subjects (44 men and 35 women; mean age, 24.0 years, SD, 6.1), they noted that the prevalence of CSP was 74.7% (29). All these 79 subjects were physically healthy, and none had a history of head trauma, serious medical or surgical illness, or substance abuse.
In this present study, the probability of having any variety of caval anomalies on neuroimaging was greater than 50%, irrespective of the brain pathology (Table 1). Though vascular pathologies showed higher frequencies of caval variation, this was not statistically significant. We also noted a higher percentage of cava variations in adults, however, not statistically significant. Our own study revealed that 24.4% of participants had CSP. This is low compared to the series of 164 patients by Aviv et al. but higher compared to Galarza et al. studies (3,4). Aviv et al. noted a higher incidence of CSP in boxers (a CSP was present in 40% of controls and 49% of boxers), and an association between progressive scan images and increased CSP size over time in boxers, independent of age (P = 0.05) (3); this study also noted no difference in CSP size between the two groups (P = 0.43), and eight boxers demonstrated a CSP on a subsequent scan not seen on an earlier scan. In our study, none of the patients was a boxer.
Cavum vergae was the most common form of cava variation in our study. It is a cavity posterior to the septum pellucidum whose clinical significance remains unclear. Donauer et al. described four patients with presumed CV cysts that caused hydrocephalus via compression of the cerebral aqueduct (30). Wester et al. in 1995 reported three children with hydrocephalus and CSP and CV in whom hydrocephalus predated the cava (31). They suggested that caval expansion might be the result, rather than the cause, of the hydrocephalus. In our series, none of our patients had a predated hydrocephalus or ongoing hydrocephalus.
Recurrence of a previously closed cavum septi has been reported following the placement of an Ommaya reservoir for administration of chemotherapeutics with the tip against the septum (32). This suggests that previously closed cavum can re-open and thus may explain a relatively higher frequency of cava in our adult population.
The CSP, which is anterior, is separated from the more posterior CV by the anterior columns of the fornix. During development, these spaces obliterate postero-anteriorly: the CV followed by the CSP. Because of this ordered obliteration, it implies that a CV will almost always accompany a CSP. However, exceptions have been noted, suggesting that the obliteration of the spaces may not always occur in the same sequence. We noted a higher incidence of CV compared to CSP. This may lend credence to the fact that the manner of obliteration of the cavum is not necessarily postero-anteriorly or that a re-opening of previously closed cava occurs in adults.
Hemorrhage into the cavum has been reported in preterm infants with coexisting intraventricular hemorrhage; report in adults was secondary to trauma and hypertension (33,34). Tumors arising from the cavum have also been reported (35). Hemorrhage into the cavum was not noted in any of the patients we studied.
Our patients with congenital brain diseases did not have a significant different frequency of CSP and CV variation rates when compared with those that have acquired brain lesions. This suggests that the caval variations does not significantly predispose to various acquired variations. This study, however, showed that of CVI was significantly higher in adults when compared with children (P = 0.048); proposing a possible association with acquired lesions and that it is unlikely to be a normal anatomical variant. This may need further study to delineate what acquired lesions.
The limitation of this study is the low number of images, which resulted from the exclusion criteria in the material and methods, coupled with the low rate of neuroimaging in our environment due to cost of neuroimaging in low socioeconomic environment where medical insurance is virtually non-operational in most cases.
In conclusion, cava variations are relatively common in neurological brain diseases. Patients with congenital brain diseases did not have a higher frequency of cava variation when compared with those that had acquired lesions. There was also no statistically significant difference in the frequency of cava variation in children aged more than 6 months compared to adults. The most common type of cava variation noted in our study was the vergae variety while the CSP is the rarest form.
Footnotes
Conflict of interest
None declared.
