Abstract
A 14-year-old girl, with no medical history except for a preterm birth at 7 months, presented with clinical and laboratory signs of pancytopenia. Radiological investigation, including abdominal ultrasound and magnetic resonance imaging revealed a focal irregular main portal vein wall and splenomegaly. Endoscopy showed major esophageal and gastric varices. Definitive diagnosis was made by direct, transhepatic portography revealing a focal stenosis associated with a dissection of the main portal vein. Percutaneous treatment including balloon angioplasty and stent placement resulted in complete re-expansion of the portal vein main branch. Clinical and radiological follow-up over 5 years showed complete disappearance of all clinical signs and normalization of laboratory data and splenic volume.
Focal, extrahepatic portal vein stenosis may result in severe symptoms of prehepatic portal hypertension, such as variceal bleeding, refractory ascites, and signs of hypersplenism. The underlying pathological mechanism of the stenosis can be inflammatory, such as in acute pancreatitis (1), radiation-induced (2) or related to tumoral invasion (3). In children, however, extrahepatic portal vein stenosis is most often seen after liver transplantation at the anastomosis of the recipient–donor portal vein (4). In this report, we describe the diagnosis and percutaneous treatment of a focal, portal venous stenosis identified in an adolescent and resulting in severe symptoms of prehepatic portal hypertension.
Case report
Laboratory data before and after portal vein stenting
Under general anesthesia, access to the right portal vein was made using ultrasound guidance. Venography clearly revealed a moderate focal stenosis of the main portal vein, potentially associated with a focal intimal dissection. Additionally very large gastric and esophageal variceal collaterals were also clearly visible (Fig. 1a). Pressure measurements over the stenosis were: 16 mmHg proximal and 6 mmHg distal to the stenosis resulting in a pressure gradient of 10 mmHg. Balloon angioplasty with a 10-mm diameter balloon did not result in sufficient re-expansion of the stenosed segment. It was decided to insert a self-expanding nitinol stent with a length of 4 cm and a nominal diameter of 12 mm (Zilverstent, Cook Medical, Bloomington, IN, USA) which was postdilated up to 10 mm diameter. Pressure measurements after stenting were: 12 mmHg proximal and 10 mmHg distal to the stented segment resulting in a residual pressure gradient of 2 mmHg. Completion venography confirmed the correct appositioning and expansion of the stent and absence of any collateral flow (Fig. 1b). Patient recovered well after the procedure and could leave the hospital 2 days later. Follow-up was performed by clinical, ultrasound, and laboratory evaluation at regular intervals: every 3 months in the first year and yearly thereafter. Clinically, all signs of chronic anemia disappeared and gastroscopy was normal without visible esophageal varices or hypertensive gastropathy. Ultrasound evaluation could demonstrate a normal volume of the spleen with maximal diameters after 1, 2, 3, 4, and 5 years of 13.2 cm, 13.2 cm, 13.6 cm, 13.8 cm, and 14 cm, respectively, which is at the upper limit of normal values (12–14 cm). Duplex-ultrasound flow measurements were on all evaluations between 40 and 60 cm/s over a 5-year follow-up period. Finally, values of red blood cell, white blood cell, and platelet count normalized during follow-up (Table).
(a) Transhepatic direct portography confirms the moderate focal stenosis (arrow), potentially associated with focal, intimal dissection. Note also the large fundal varices (arrowheads) originating proximally to the stenosis. (b) After stent placement (arrows) there is restored flow in the portal venous system without opacification of any variceal collaterals. The stent is bridging the thin left portal vein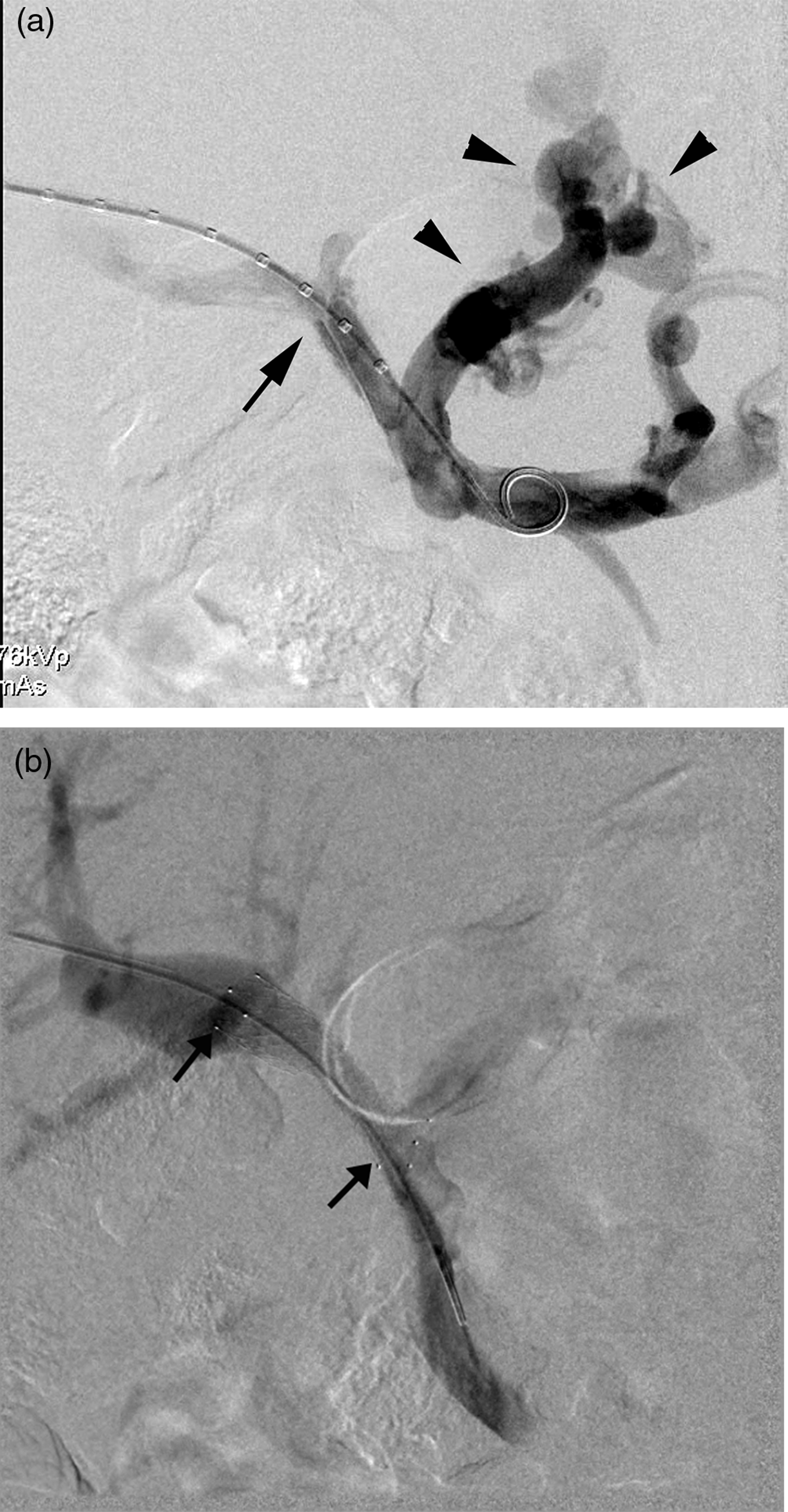
Discussion
We suggest that the underlying focal, extrahepatic portal vein stenosis is related to the anomalous placement of a venous umbilical catheter into the main portal vein, as umbilical catheter insertion was the only relevant event in the patient's medical history. Additionally, it is well-known that umbilical catheter may be associated with hepatic and portal vein complications. Schlesinger et al. (5) described the presence of a hepatic hematoma in a neonate as a result of intrahepatic vessel wall perforation due to umbilical catheter misplacement and these authors also noted the presence of a venous umbilical catheter, misdirected into the left, right, or main portal vein or even in veins that merge to form the portal vein: the superior mesenteric and splenic vein.
In this case it can be hypothesized that incorrect placement of the umbilical catheter with its tip into the main portal vein (Fig. 2) resulted in a focal dissection or even in a portal vein thrombosis which recanalized later on, but with a residual membranous stenosis in situ. These complications can occur in the neonatal period without clear symptoms.
Schematic view of correct placement (full line) of an umbilical catheter with its tip into the right atrium and an incorrect placement (dotted line) of an umbilical catheter with its tip in the portal vein main branch. dv, ductus venosus; ivc, inferior vena cava; lpv, left portal vein; mpv, main portal vein; ra, right atrium; rhv, right hepatic vein; rpv = right portal vein; ur, umbilical recess; uv, umbilical vein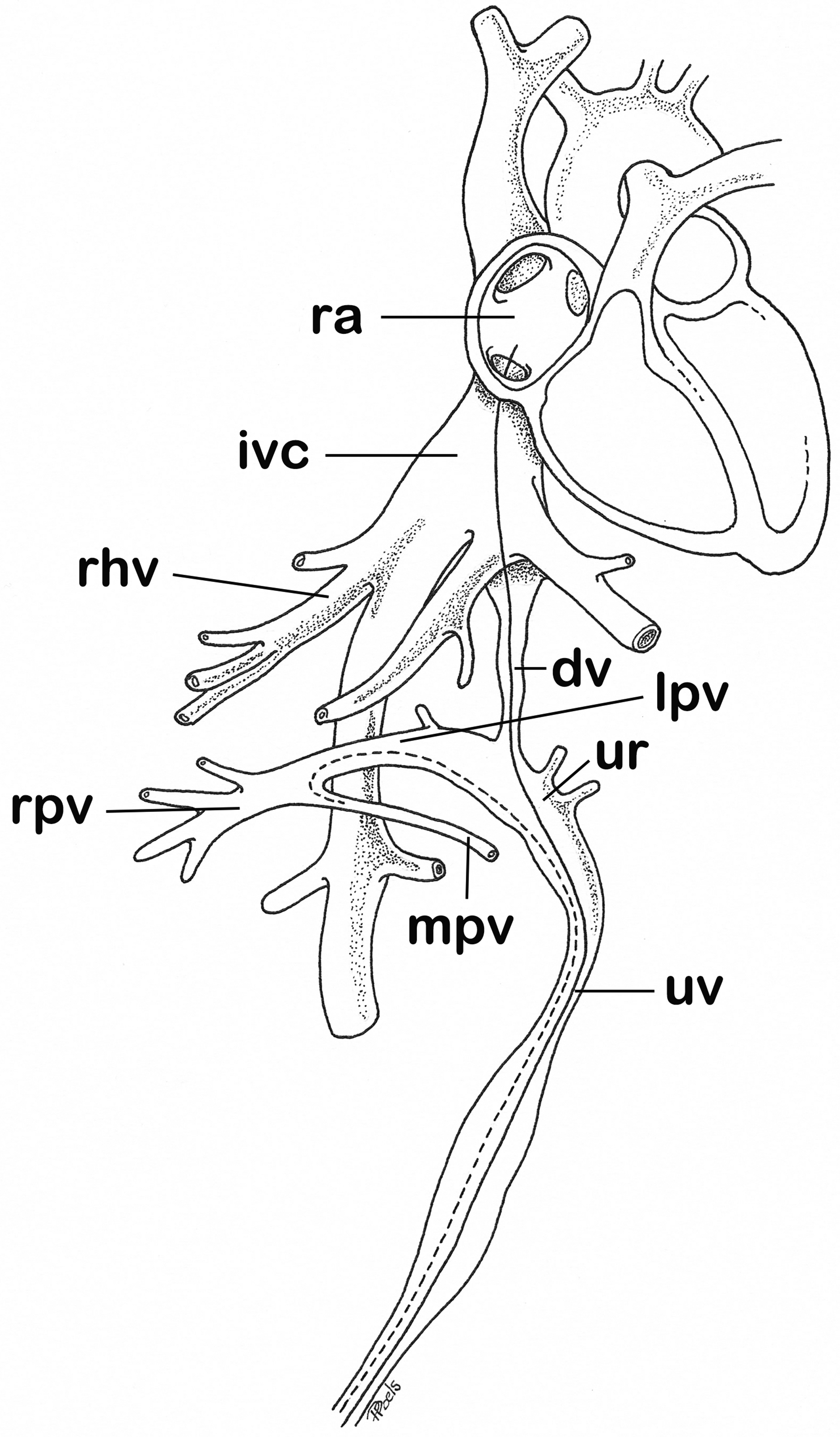
Finally, as umbilical catheters are inserted and later on retrieved by the pediatrician without image guidance, discrete vessel wall damage at the catheter tip may occur silently, but complications due to complicated insertion might become symptomatic later on. However, it is still unclear why portal vein damage in the neonatal period may become symptomatic at a very late stage (e.g. 14 years).
Stent placement in the main portal vein may lead to instent restenosis or even stent thrombosis. However, no anticoagulation treatment was prescribed afterwards, which is in agreement with Novellas et al. (6). These authors prescribed anticoagulation only in case of decreased portal flow and not in cases of normalized flow in the portal vein after stenting.
Finally, insertion of a stent into the portal vein may compromise a later liver transplantation. In this case, however, we decided to stent, as the patient's liver function was completely normal without any sign of cirrhosis and judged that late stent dysfunction would be unlikely, based on results of stent insertion after liver transplantation in adolescents (4).
In conclusion, we describe a rare case of focal, extrahepatic portal vein stenosis in a female adolescent, most probably related to a malpositioned umbilical catheter immediately after preterm birth and becoming symptomatic at a much later stage (age of 14 years). Definitive diagnosis and treatment were performed by minimally invasive catheter and stent techniques which are identical to the percutaneous treatment of inflammatory or malignant focal portal vein stenosis (1–3, 6). Late follow-up (up to 5 years) remains good with complete and durable disappearance of all signs and symptoms of portal hypertension. In children or adolescents with gradual onset of signs of portal hypertension and without any parenchymal liver disease, a focal portal vein stenosis must be excluded, especially if these children have had umbilical catheters immediately after birth.
