Abstract

The metabolic syndrome (MetS) represents a cluster of risk factors for cardiovascular disease (CVD) and type 2 diabetes, including central obesity, reduced high-density lipoprotein (HDL) cholesterol, elevated triglycerides, blood pressure and fasting plasma glucose. Numerous studies have demonstrated that MetS is associated with an increased risk of dementia and all-cause mortality.1,2 Insomnia is characterised by difficulties to initiate, maintain or consolidate sleep in addition to impaired daytime function. Emerging evidence has suggested that insomnia is associated with hypothalamic–pituitary–adrenal axis dysregulation, increased sympathetic activity, systemic inflammation and endothelial dysfunction, which in turn increase the risk of cardiovascular and metabolic disease. 3 We aimed to study the relationship between insomnia disorder and MetS in the Swedish CArdioPulmonary bioImage Study (SCAPIS) pilot cohort. 4
In this cross-sectional study, 830 subjects, aged 50–64 years, stratified by low and high socioeconomic residential area were selected from the population registry in Gothenburg, Sweden. Participants underwent extensive examinations including anthropometric, biochemistry and questionnaires on health and lifestyle. Moderate to vigorous intensity physical activity (MVPA) and time spent sedentary (SED) were assessed by triaxial accelerometers worn for 7 days. The MetS was classified using the National Cholesterol Education Program’s Adult Treatment Panel III latest report (2001) on MetS. The insomnia severity index (ISI) questionnaire was used for classification of insomnia (cut-off: ISI score ≥15). 5 Smoking status was defined as ‘non-smoker’ or ‘smoker’, which includes occasional/former/current active smoker.
MetS was found in 17.1% and insomnia in 12.4% of the population (Table 1). The prevalence of MetS was 24.3% and 16.1% in the insomnia and non-insomnia groups, respectively (P = 0.039). In a generalised binary logistic regression model controlling for sex, age, body mass index, socioeconomic status (SES), university education, employment status, smoking, risky alcohol consumption, subjective short sleep time, physician diagnosed sleep apnoea, chronic stress, depression symptoms, SED and MVPA, insomnia was associated with an approximately two-fold increased risk of MetS (odds ratio (OR) 1.97, 95% confidence interval (CI) 1.00–3.86, P = 0.049). Male sex (OR 2.62, 95% CI 1.61–4.26, P < 0.001) and body mass index (OR 1.31, 95% CI 1.24–1.39, P < 0.001) were associated with an increased risk of MetS whereas MVPA (OR 0.92, 95% CI 0.84–1.00, P = 0.041) was associated with a reduced risk of MetS. Smoking and depression symptoms tended to increase the risk of MetS (P = 0.060 and P = 0.065, respectively).
Characteristics of the study population.
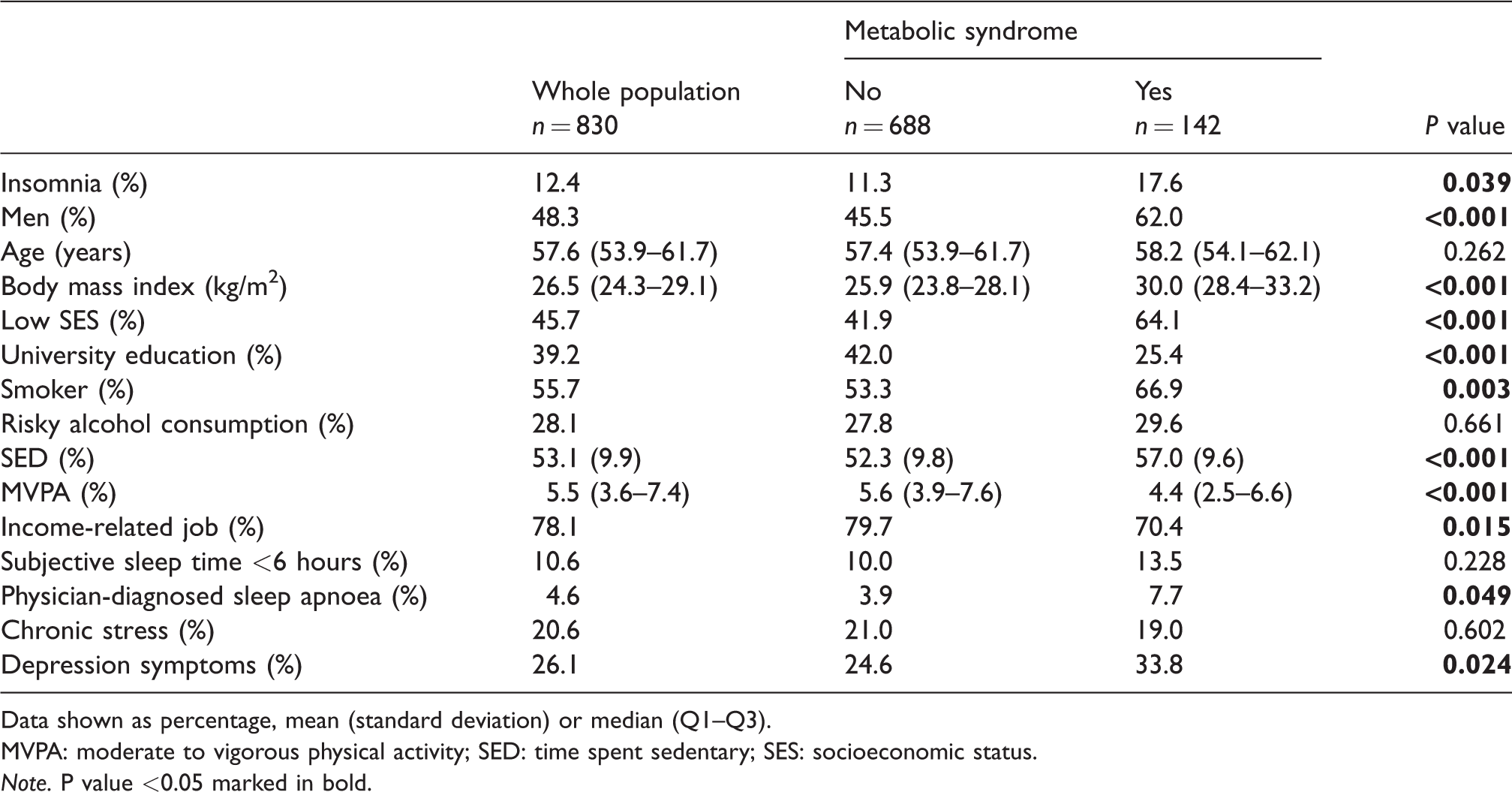
Data shown as percentage, mean (standard deviation) or median (Q1–Q3).
MVPA: moderate to vigorous physical activity; SED: time spent sedentary; SES: socioeconomic status.Note. P value <0.05 marked in bold.
In a univariate analysis, high serum triglyceride and low HDL-cholesterol were more prevalent in the insomnia compared with the non-insomnia group (high triglycerides 33.0% vs. 18.8%, low HDL-cholesterol 17.5% vs. 9.1%, P = 0.001 and P = 0.008, respectively). The proportions of high blood pressure, high fasting plasma glucose and high waist circumference were similar between the insomnia and non-insomnia groups (Figure 1). On average, non-insomnia subjects fulfilled 1.28 (1.19–1.36) MetS criteria, whereas the insomnia subjects fulfilled 1.57 (1.32–1.83) criteria (P = 0.032).
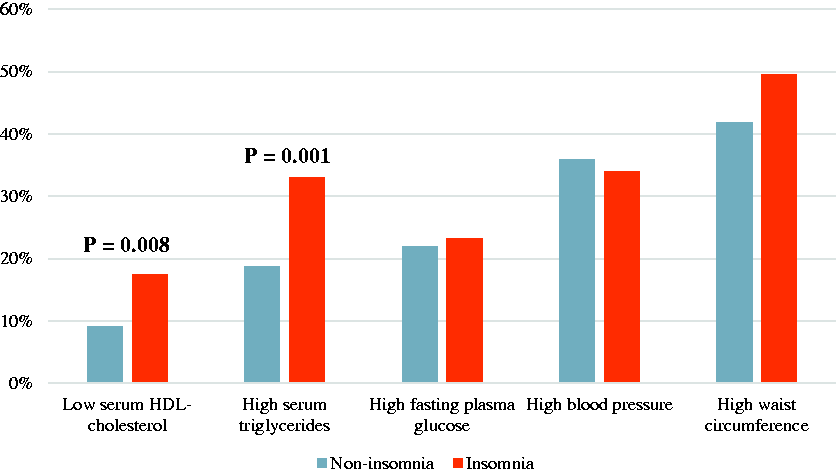
Percentage of subjects who fulfill each of the metabolic syndrome components in the insomnia and non-insomnia groups. (P value for the comparison between insomnia and non-insomnia groups.)
In this middle-aged population, we observed an independent association between insomnia disorder and MetS. To the best of our knowledge, this is the first study on insomnia and MetS to include ISI as a diagnostic instrument, while also using accelerometer-based information on MVPA and SED. A recent meta-analysis has shown that poor sleep quality is associated with elevated blood pressure, abnormal lipid profile, unhealthy food intake and impaired glycaemic control, which attribute to different components of the MetS. 6 The finding that insomnia was associated with high triglycerides and low HDL-cholesterol in the current study suggested that dyslipidaemia as a hallmark of underlying insulin resistance could be influenced by stress, cortisol, insomnia and other neuroendocrine perturbations. 7 However, the link between insomnia and MetS is still not fully established in longitudinal studies. Chen et al. 8 used the Athens insomnia scale to define insomnia and conducted a longitudinal 1-year follow-up study in an elderly population. They demonstrated that insomniacs had more than twice the risk of developing MetS compared with those without insomnia. 8 Troxel et al. 9 performed a 3-year follow-up in 812 middle-aged individuals and found difficulties initiating sleep, non-restorative sleep and loud snoring were associated with an increased risk of developing MetS. However, there was no significant risk increase in those with clinically diagnosed insomnia. 9 The discrepancy in the results may be due to the population selection, or to the insomnia/MetS definitions applied in the studies.
Unhealthy lifestyle is closely associated with MetS and it is well known that physical activity is an important component of a healthy lifestyle to reduce the risk of CVD and type 2 diabetes. Accelerometers allow for more detailed and continuous assessment of individuals’ SED and physical activity than do self-reported methods. Previous studies on insomnia and MetS have used self-reported information on physical activity and sedentary time. 6 As expected, we found that accelerometer-based MVPA was independently associated with a reduced risk of MetS. Although SED was greater in MetS group compared with the non-MetS group, sedentary time was not associated with MetS in the multivariate model.
Economic and social vulnerability is associated with MetS. The current cohort was stratified for SES by socioeconomic residential area and we observed an accumulation of subjects with low SES status in the MetS group. This is in line with a previous report of a more adverse CVD risk factor profile in participants with low SES in the SCAPIS pilot cohort. 10 In the multivariate analysis, living in a low SES area was no longer significantly associated with an increased MetS risk probably due to the small sample size. Another potential explanation may be that the effect of SES on MetS risk is mediated by other established risk factors (e.g. low physical activity).
The limitations of this study include the relatively narrow age range, which may limit the generalisability of the results. Information regarding ethnicity, concomitant medications, specific insomnia symptoms (e.g. difficulty initiating/maintaining sleep, early morning awakening) and shift work were not addressed. Finally, poor health and comorbid conditions may even have synergetic effects on insomnia. Hence, no conclusion on causality between the two conditions could be drawn from the current cross-sectional analysis.
In conclusion, insomnia, one of the most common sleep disorders, was associated with MetS in a middle-aged population. Longitudinal studies are needed to establish further the link between insomnia and the MetS. With increased recognition of the role of sleep in public health, sleep complaints (e.g. insomnia) should be considered as a modifiable risk factor for CVD and metabolic disease and, thus, may be implemented in risk assessment of MetS. 11
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The main funding body of the SCAPIS is the Swedish Heart and Lung Foundation. The study is also funded by the Knut and Alice Wallenberg Foundation, the Swedish Research Council and VINNOVA (Sweden’s innovation agency). In addition, the SCAPIS pilot study received support from the Sahlgrenska Academy at the University of Gothenburg and Region Västra Götaland. Work by GB was supported by the Swedish Heart and Lung Foundation (20180324) and the Swedish Research Council (2019-01140). Work by JH (20180585) and MB (20180379) was supported by the Swedish Heart and Lung Foundation.
