Abstract

Aortic aneurysms and the history of atherosclerosis
The word ‘aneurysm’ is related to the Greek word ‘aneurynein’, which means to dilate, supporting the definition of an abdominal aortic aneurysm (AAA) as a dilatation of the vessel with at least a 50% increase in diameter. 1 Rupture of the aneurysm is associated with bleeding in the perivascular tissue with a very high annual mortality of approximately 1–2% of deaths in the western world. 2
The existence of inflammation, which is a driving force in the process of aortic aneurysm formation, is mentioned in Egyptian papyrus notes 5000 years ago describing the characteristics of inflammation referring to heat and redness as concomitants of disease.
Aulus Cornelius Celsus further described the cardinal signs of inflammation including redness, swelling, heat and pain in the first century AD. Later on this was explained by a diapedesis of leukocytes from the blood into tissues. The great physiologist Rudolf Virchow (1821–1902) recognised the inflammatory nature of atherosclerosis: ‘In some, particularly violent cases the softening manifests itself even in the arteries not as the consequence of a really fatty process, but as a direct product of inflammation’. 3 He subsequently also recognised the first steps in calcification, bone formation in this process, thus suggesting a continuous process.
The mechanisms in inflammation were further elucidated by Paul Ehrlich and Ylia Mechnicov in their pioneering studies in immunity and host defences, but the application of these concepts to atherosclerosis lagged by almost a century. 4 However, the concept of dynamic interplay between mononuclear phagocytes and vascular cells during the process of atherosclerosis, once overlooked, has now become common knowledge.
The complexity of atherosclerosis as an immunological process was further understood after the discovery of adhesion molecules expressed by endothelial cells. 5 Poole and Florey in 1958 described that adhesion molecules like vascular cell adhesion molecules, bind to the monocytes and T lymphocytes in the early atherosclerotic plaque. 6 Later on the mononuclear cells receive chemoattractant signals such as monocyte chemoattractant protein-1 that beckon them to enter the intima, 7 where the monocytes mature into macrophages with scavenger receptors necessary for the uptake of modified lipoproteins and hence, foam cell formation. Pro-inflammatory cytokines promote the expression of matrix metalloproteinases (MMPs), interstitial collagenases by mononuclear phagocytes, revealing the transitional process from stable atherosclerosis to a dangerous state of plaque disruption, thrombosis and in aortic disease; rupture.
Aneurysmal disease
Atherosclerosis is affecting the whole cardiovascular system and might manifest itself also in a directly opposite manner of the obliterating disease – a dilating abdominal aortic process. The inflammatory process which underlies the aneurysm disease is more prone to affect the medial layer of the vessel with destruction of the elastic lamina accelerated by the overexpression of matrix degrading proteases including MMPs. Moreover, the dorsal surface of the infrarenal aorta is a predilection site for aneurysm formation due to the shortage of vasa vasorum to the media at this site of the aorta. In addition, experimental studies have shown that the tunica media of the thoracic aorta has a greater vasa vasorum density, with microvessels penetrating deeper towards the lumen than in the abdominal aorta confirming ischaemia as a contributing factor in the formation of AAAs.8,9 However, the mechanisms are complex involving processes initiated both in the media and the intima. Although several genetic disorders are associated with thoracic aortic aneurysms, this is less frequent in AAAs. However, 20% of patients with AAAs have a family history of AAAs. The discovery of ‘bone formation’ already by Virchow is further complicating the understanding of the atherosclerotic process both in the intima and media known as calcification.
Current issue
In the current issue of the European Journal of Preventive Cardiology, de Jong and coworkers are illuminating the abdominal aortic calcifications (AACs) as an independent contributor to and risk factor for cardiovascular disease. The authors refer to the HORUS study that evaluated ancient mummies from four different geographical regions. 10 Despite small differences related to gender and ethnicity, this disease was relatively frequent even in populations without modern urban risk factors. Moreover, this may indicate that arterial calcifications were already highly prevalent in ancient populations and that calcific aortic disease is not solely characteristic of any specific diet or life style. However, they also found that mummies with widespread calcification in peripheral vascular beds lived longer than those with less calcification; which might thus be a contradiction to the concept of AAC as an independent risk factor. 11
To assess the epidemiology of AACs and its relation to outcomes, the authors performed both a systematic and a manual search in recognised databases.
In the Pathobiological Determinants of Atherosclerosis in Youth (PDAY) study, the presence and extent of atherosclerotic lesions were studied on histology in 1532 deceased persons aged from 15 to 34 years. 12 In the youngest age group (15–19 years) 100% of the abdominal aortas had atherosclerotic lesions, whereas these lesions were found in only 50% of the coronary arteries. Calcification developed later, but was already present in 14% in the oldest age group (30–34 years). Furthermore, they found that AAC is highly prevalent in the general population and the prevalence and extent increases with age.
In the Framingham study AAC was already present in 15.5% of men and 7.8% in women in the age group under 45 years. In both groups, the prevalence increased to 100% in both men and women over 75 years. Agatston-like scores were higher in men in all age categories. 13
Prevalence and risk factors differ between men and women and different ethnicities. They include traditional cardiovascular risk factors and decreased bone mineral density, the later revealing the link to osteoporosis. The authors also found that AAC is shown to contribute to arterial stiffness and that it is a strong predictor of cardiovascular events and mortality per se. They conclude that AAC has existed in all eras and is increasingly acknowledged as an independent risk factor associated with cardiovascular disease. This is in accordance with the findings in the Multi-Ethnic Study of Atherosclerosis (MESA), which evaluated coronary and thoracic aortic calcification (TAC). The researcher found that traditional cardiovascular risk factors were related to both TAC incidence and progression. 14 Furthermore, conventional echocardiographic examination may offer an assessment of cardiac calcifications at different sites, such as the aortic valve and ascending aorta, that are associated with the clinical manifestation of atherosclerotic disease and are predictive of future cardiovascular events. 15
Patients with the chronic systemic inflammatory diseases of today, including systemic lupus erythematosus, rheumatoid arthritis (RA) and HIV infection, experience premature atherosclerosis and coronary events. This may indicate that the chronic inflammatory response of ancient times to infection have resulted in atherosclerosis. Smoke and dust inhalation from the use of open fires for daily cooking and illumination represents another potential cause that might have been more frequent in ancient times, but which is also increasingly acknowledged as a driver for cardiovascular disease in current polluted areas. The ongoing corona pandemic may give us some indications of the importance of these risk factors when most of the polluting transport mechanisms and polluting industry are globally almost totally locked down.
Despite lack of data, it is generally assumed that aortic calcifications are predominantly atherosclerotic in nature. However, whereas coronary atherosclerosis is primarily related to the intima, aortic atherosclerosis often includes processes in the medial layer. This might partially explain the difference in calcification between the two vascular beds.
Calcification of the arteries is increasingly seen as a protective factor, perceived as a response to injury to prevent plaque rupture in the coronaries or further aneurysm growth in the abdominal aorta. On the other hand it is also an end stage of a dangerous inflammatory process.
Mechanisms of calcification
The process of calcification shares many features with that of ordinary bone formation. Microparticles elaborated by macrophages may provide niduses for plaque calcification, yielding another link between inflammatory cells and cardiovascular calcification. 16 This process of calcification is very prominent in aortic aneurysmal disease.
The development of smooth muscle calcification shares inductive and inhibitory mechanisms underlying the regulation of bone remodelling indicating a temporal relationship between osteoporosis and mineral metabolism in the vessels. It is frequently found in autopsies, but the prognostic importance is less known. In the plaque, the secretion of cytokines (bone morphogenetic proteins) accelerates this calcification. Smooth muscle cell bone formation is mediated through the activation of receptor activator nuclear factor κB ligand (RANKL) via the bone morphogenetic protein-4-dependent pathway. 17 This activation can be decelerated by osteoprotegerin, 18 which is a soluble decoy receptor for RANKL and the tumour necrosic factor-related apoptosis-inducing ligand (TRAIL). Osteoprotegerin binds to RANKL and TRAIL, and inhibits the association with their receptors, which have been labelled as the receptor activator of nuclear factor κB (RANK).
The sustained release of osteoprotegerin from vascular endothelial cells (ECs) has been demonstrated in response to inflammatory proteins and cytokines, suggesting that the osteoprotegerin/RANKL/RANK system plays a modulatory role in vascular injury and inflammation. Therefore, for the development of potential therapeutic strategies targeting vascular calcification, critical consideration of the implications for bone metabolism must be taken into account to prevent potentially detrimental effects to bone metabolism.
Intima versus media calcification
Already in 1903 Mønckeberg described the differentiation between intima and media calcification, the later associated with chronic renal failure and imbalance in vitamin D metabolism.
Although all branches of the large vascular tree may calcify, only some of them develop atherosclerosis. The coronary arteries and the arteries of the abdomen, lower extremities and the aorta are well known vascular beds in which atherosclerosis is a major player. In contrast, others parts of the vascular tree are relatively resistant to the atheroma process, such as the arteries of the upper extremities. The susceptibility for developing atherosclerosis is strongly influenced by intrinsic differences in the vascular system. 19 One important factor is the diversity of smooth muscle cell lineage. 20 Media calcification can be induced experimentally by creating chronic renal failure and feeding pharmacological amounts of vitamin D, indicating that media calcification can occur in the absence of intima calcification and atherosclerosis. However, media calcium deposition is not a homogeneous condition; in diabetic arterial medial calcification hydroxyapatite is the predominant mineral, whereas it is whitlockite in vitamin D toxicity. 21 In the aorta both mechanisms are operative, indicating that the presence of AAC might have slightly different aetiology in ancient times and the present.
Thus, although ancient aortic calcification might mostly be due to genetic and perhaps renal failure-associated media calcification, the modern life style has added to the burden with atherosclerosis and intima calcification.
Therefore, therapy against the additional contributors to aortic disease and calcification is increasingly relevant.
Modern medical therapy
The authors of the current paper are finally asking if the process of arterial calcification, which for a time has been thought to be an irreversible endpoint of atherosclerotic disease, can be halted or even reversed. They elegantly line up potential medical therapies evaluated in AAC.
The focus of modern treatment of atherosclerosis and its manifestations is on hyperlipidaemia, thrombosis and inflammation, but not on the calcification process. This might be due to the perception of the problem of calcification as merely a reparative process. However, the disease is a progression from endothelial dysfunction to calcified arteriosclerosis with possibilities for acute complications in every step; from the early plaque rupture in what is perceived as normal coronary arteries to rupture of the diseased and calcified overextended AAA.
This clearly indicates the need for a change of view in accordance with the medical treatment of stable coronary artery disease. Prevention and medical intervention should be the focus and initiated at a much earlier step in the development, thereby targeting risk factors and not only the calcification per se. As in other diseases this might be a coincidental manifestation of the disease, related to mechanisms developed to protect the individual.
Prevention
A large number of patients with aortic diseases have risk factors and comorbidities such as smoking, concomitant coronary artery disease, chronic kidney disease, diabetes mellitus, dyslipidaemia and hypertension. Therefore treatment and prevention strategies must be similar to those indicated for the above diseases to avoid the development and progression of AAA to AAC.
Moderate physical activity prevents the progression of atherosclerosis. Recent research has now also shown that cardiac rehabilitation protects against the expansion of small AAAs and mitigates the risk associated with AAA repair, possibly because of the decreased elevation of blood pressure during exercise. 22 On the other hand, competitive sports should be avoided in patients with a diagnosed dilated aorta to prevent blood pressure spikes.
Smoking has been shown to be highly associated with the development of AAA, and this is further confirmed in the 7-year follow up in the Tromsø study. 23 The cessation of smoking is therefore very important, as studies have shown that self-reported current smoking induced a significantly faster AAA expansion (by approximately 0.4 mm/year). 24 Moreover the highest rate of increase of diameter in AAA is found in smokers and in patients with hypertension. 25
Hypertension is a risk factor for the development of atherosclerosis and subsequent calcification of the aorta. Treatment of hypertension per se is therefore important to slow down the process of aneurysm formation and prevent rupture and dissection. 26 In patients with Marfan syndrome, the prophylactic use of beta-blockers, angiotensin-converting enzyme inhibitors and angiotensin II receptor blockers seems to be able to reduce either the progression of the aortic dilation or the occurrence of complications. 27
In hyperlipidaemia small observational studies have suggested that statins may inhibit the expansion of aneurysms. 28 The use of statins has also been associated with significantly improved survival after AAA repair. 29 Moreover, statins have been shown to reduce extensive thoracic atheroma and lower lipid concentrations. 30 Future studies are evaluating the effect on aorta atheroma per se. 31
Originally developed to lower serum cholesterol, increasing evidence suggests that statins have potent anti-inflammatory effects that contribute to their beneficial effects in patients. 32 Recent research has indicated that elevated osteoprotegerin levels may reflect the vascular inflammation. 33 Preliminary results from the MICROS study indicate that osteoprotegerin might be altered after high-dose rosuvastatin therapy, indicating a possible statin- induced effect. On the other hand, 12-week aerobic interval training, although improving exercise capacity and endothelial function, did not alter osteoprotegerin in stable ischaemic heart failure patients on optimal medical therapy. 34
This brings us to the biological balance in life, for the current task named after the dangerous Bermuda triangle; the Bermuda triangle of atherosclerosis, osteoporosis and inflammation (see Figure 1). Non-inflammatory states, common conventional risk factors and low-grade inflammation may link atherogenesis and bone loss. In the case of an underlying inflammatory disease, such as RA, high-grade systemic inflammation may perpetuate the development of atherosclerosis and osteporois. 35 This adds systemic inflammatory disease such as RA to the risk factors for AAC, and osteoprotegerin as only one of the operative mediators.
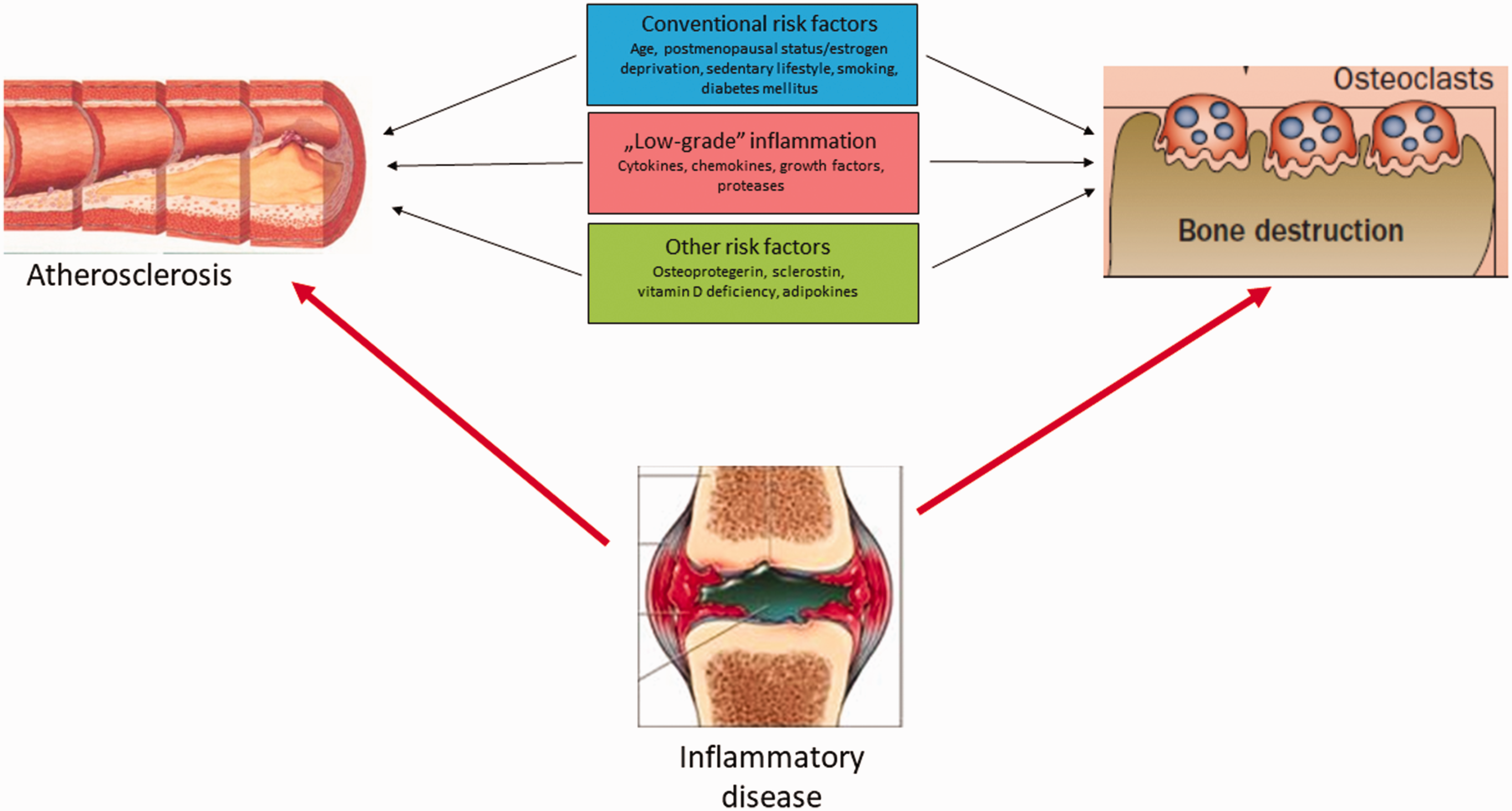
The ‘Bermuda triangle’ of atherosclerosis, osteoporosis, and inflammation.
Importantly, this highlights the value of a holistic approach, viewing the aorta as a ‘whole organ’ regardless of lesion localisation. Genetic disorders with tandem lesions of the aorta are important to reveal in order to give proper treatment, but the main bulk of patients is also in need of therapy against traditional risk factors. de Jong and co-workers have elegantly illuminated the forgotten issue of arotic calcification whether it is due to intima or media calcification. Whereas most of the interventional cardiology community is well aware of the complications associated with arterial and aortic calcification, the authors remind us cardiologists about the prognostic importance and the possibilities for medical therapy of the calcified aorta.
Footnotes
Declaration of conflicting interests
The author(s) declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article
