Abstract
Aims
European Society of Cardiology/European Atherosclerosis Society 2019 guidelines recommend more aggressive lipid targets in high- and very high-risk patients and the addition of adjuvant treatments to statins in uncontrolled patients. We aimed to assess (a) achievement of prior and new European Society of Cardiology/European Atherosclerosis Society lipid targets and (b) lipid-lowering therapy prescribing in a nationwide cohort of very high-risk patients.
Methods
We conducted a retrospective observational population study using linked health data in patients undergoing percutaneous coronary intervention (2012–2017). Follow-up was for one-year post-discharge.
Results
Altogether, 10,071 patients had a documented LDL-C level, of whom 48% had low-density lipoprotein cholesterol (LDL-C)<1.8 mmol/l (2016 target) and (23%) <1.4 mmol/l (2019 target). Five thousand three hundred and forty patients had non-high-density lipoprotein cholesterol (non-HDL-C) documented with 57% <2.6 mmol/l (2016) and 37% <2.2 mmol/l (2019). In patients with recurrent vascular events, fewer than 6% of the patients achieved the 2019 LDL-C target of <1.0 mmol/l. A total of 10,592 patients had triglyceride (TG) levels documented, of whom 14% were ≥2.3 mmol/l and 41% ≥1.5 mmol/l (2019). High-intensity statins were prescribed in 56.4% of the cohort, only 3% were prescribed ezetimibe, fibrates or prescription-grade N-3 fatty acids. Prescribing of these agents was lower amongst patients above target LDL-C, non-HDL-C and triglyceride levels. Females were more likely to have LDL-C, non-HDL-C and triglyceride levels above target.
Conclusion
There was a low rate of achievement of the new European Society of Cardiology/European Atherosclerosis Society lipid targets in this large post-percutaneous coronary intervention population and relatively low rates of intensive lipid-lowering therapy prescribing in those with uncontrolled lipids. There is considerable potential to optimise lipid-lowering therapy further through statin intensification and appropriate use of novel lipid-lowering therapy, especially in women.
Keywords
Introduction
The European Society of Cardiology (ESC)/European Atherosclerosis Society (EAS) 2019 guidelines for management of dyslipidaemia recommend a low-density lipoprotein cholesterol (LDL-C) reduction of ≥50% from baseline and a LDL-C goal of <1.4 mmol/l (<55 mg/dl) in very high cardiovascular disease risk patients (reduced from <1.8 mmol/l (70 g/dl) in the 2016 guidelines) and <1.0 mmol/l (<40 mg/dl) in patients with recurrent atherosclerotic cardiovascular events within the previous 2 years.1,2 These guidelines recommend the addition of ezetimibe to statin therapy and proprotein convertase subtilisin/kexin type 9 inhibitors in patients who are not at goal despite taking maximally tolerated statin dose ± ezetimibe. There is also an emphasis on the importance of considering non-high-density lipoprotein cholesterol (non-HDL-C) levels as secondary treatment targets, especially in those individuals with elevated triglycerides (TGs). Furthermore, in patients at high risk (or greater) with TGs between 1.5–5.6 mmol/l despite statin therapy, N-3 fatty acids (N-3) should be considered.
It is unknown what proportions of patients at very high CVD risk achieve, or do not achieve, these recommended levels of LDL-C (non-HDL-C) and/or TGs; and how these respective patient groups are treated. A better understanding of these relationships will characterise not only the therapeutic gap, but also the potential opportunity to optimise CVD risk management at an individual and population level. The objectives of this study were to document (a) lipid-lowering treatment and (b) achievement of prior and current ESC/EAS lipid targets in a contemporary national cohort of patients post-percutaneous coronary intervention (PCI) population.
Method
We undertook a retrospective observational cohort study using linked anonymised electronic health record (EHR) data for patients undergoing PCI in Wales, UK between January 2012–December 2017. Access to data and linkage was performed using the Secure Anonymised Information Linkage (SAIL) databank.3,4 Study subjects included those ≥18 years of age, with ≥90 days of follow-up. An index date was assigned to the date of the first PCI during the study period for each patient with follow-up for 1 year.
Lipid levels and prescriptions for lipid-modifying therapy
The time to the first lipid profile and the lowest LDL-C, non-HDL-C and TG level between 28–365 days post-discharge was documented. Prescriptions were identified during follow-up for lipid-lowering therapy (LLT) including statins, ezetimibe, fibrates and prescription-grade N-3.
LLT prescribed in the 90 days immediately post-discharge and within 90 days prior to the lowest LDL-C and non-HDL-C were classified as high-intensity statin (HI-statin; atorvastatin ≥40 mg/d and rosuvastatin ≥20 mg/d), non-high-intensity (NI-statin; any other statin prescription), combination of ezetimibe and/or fibrate with either HI- or NI-statin (combination statin) and other treatments including ezetimibe and/or fibrate (other treatment) without a co-prescription of statin, or no treatment.
LLT prescribed within the 90 days prior to the lowest TG level were classified as statin (either HI- or NI-statin); combination fibrate and/or N-3 with statin (combination statin), fibrate and/or N-3 (other), or no treatment.
We identified the number (and proportion) of patients achieving (a) LDL-C< or ≥: 1.4 and 1.8 mmol/l; (b) non-HDL-C< or ≥: 2.2 and 2.6 mmol/l (< or ≥: 65 and 100 mg/dl); (c) TG< or ≥: 1.5 and 2.3 mmol/l (135 and 200 mg/dl) and their respective LLT regimen.
We also evaluated the number (and proportion) of patients achieving LDL-C< or ≥1.0 mmol/l and non-HDL-C< or ≥1.8 mmol/l and their respective LLT regimens where the index PCI was a recurrent event within 2 years of a prior event.
Statistical analysis
Baseline variables are presented as mean (standard deviation (SD)) for continuous and frequency (percentage) for categorical variables. Comparisons between groups with and without lipid profiles documented during follow-up were performed using two-sample t-test and χ2 as appropriate. Multivariable binary logistic regression modelling was conducted to identify variables independently associated with the absence of a recorded lipid profile and achievement of lipid targets.
Model selection for all analyses was conducted using forward stepwise approach in SPSS (v22.0).
Results
Altogether, 15,203 patients met the study inclusion criteria: 14,577 patient years of follow-up with mean follow-up 349 ± 47 days (Table 1 and Supplementary Material Figure 1). Of the patients, 11,048 (72.7%) had a lipid profile documented within the study period, of whom 1154 (10.4%) had their first lipid profile within 28 days of discharge (Supplementary Material Figure 2). Overall, 8573 (56.4%) of patients were prescribed HI-statins with the prescribing rate of these agents considerably higher amongst those presenting with acute coronary syndrome (ACS) as index event (7269 (68.5%) vs 1304 (28.4%), p < 0.001). Patients with diabetes mellitus who were prescribed LLT were also more likely to have a lipid profile checked in the year post-discharge (Supplementary Material Table 1). Female sex, chronic kidney disease (CKD) stage 4+, dementia, ischaemic stroke, atrial fibrillation (AF) and peripheral vascular disease (PVD) were independently less likely to have lipid levels documented.
Cohort characteristic and comparison between those with and without a documented lipid profile in the first-year post-discharge post-percutaneous coronary intervention (PCI).
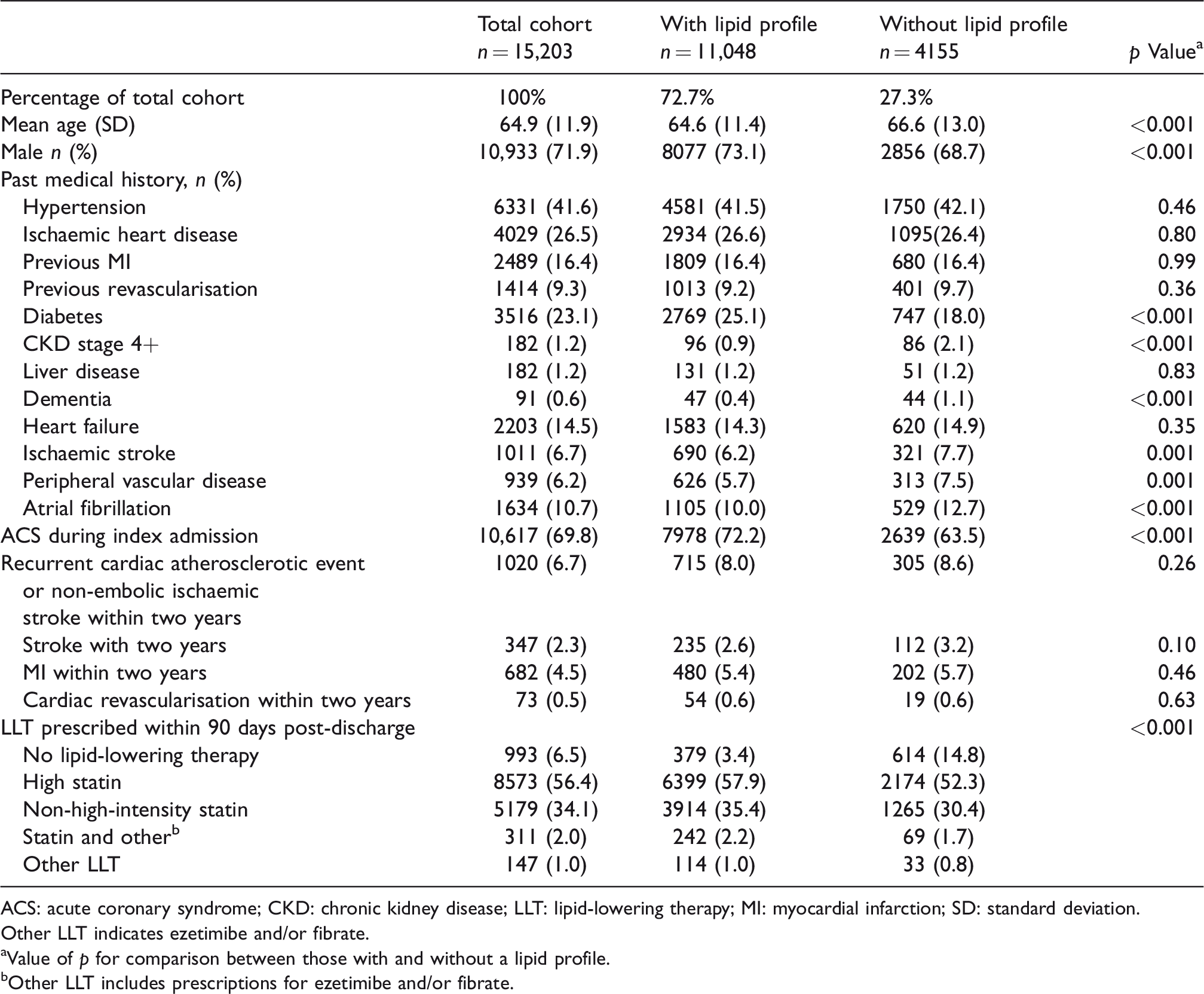
ACS: acute coronary syndrome; CKD: chronic kidney disease; LLT: lipid-lowering therapy; MI: myocardial infarction; SD: standard deviation.
Other LLT indicates ezetimibe and/or fibrate.
aValue of p for comparison between those with and without a lipid profile.
bOther LLT includes prescriptions for ezetimibe and/or fibrate.
LDL-C levels
Of the 11,048 with any lipid profile, 10,071 patients had an LDL-C documented between 28 days and 1 year (mean lowest LDL-C = 1.90 mmol/l, SD = 0.79) (Figure 1(a)). Of these, 4812 (47.8%) had achieved LDL-C levels below the 2016 EAS/ESC target of 1.8 mmol/l, but only 2353 (23.4%) were below the 2019 target of 1.4 mmol/l.
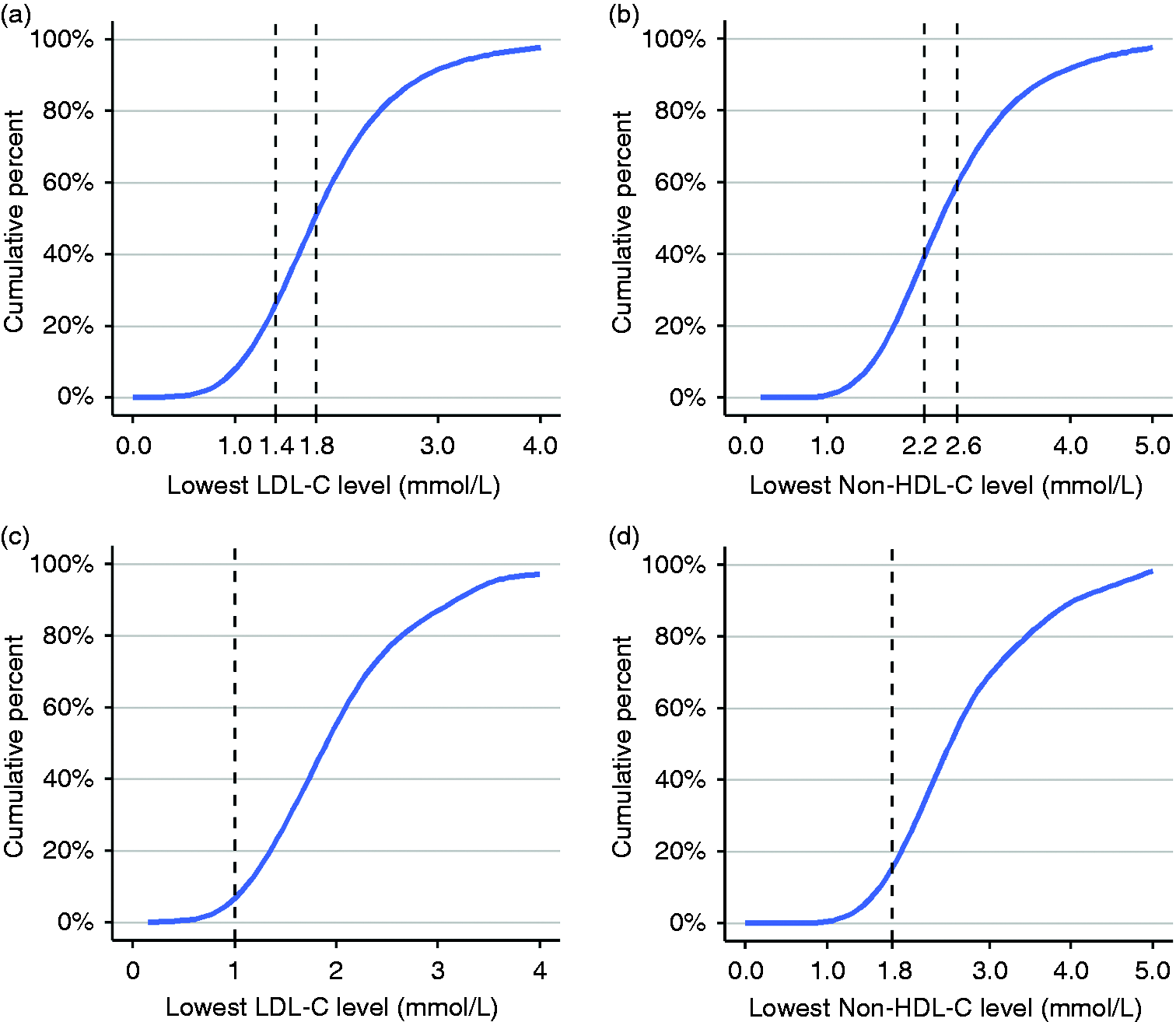
(a) Lowest low-density lipoprotein cholesterol (LDL-C) recorded between 28–365 days in 10,071 patients; (b) lowest non--high-density lipoprotein cholesterol (non-HDL-C) recorded between 28–365 days in 5340 patients; (c) lowest LDL-C recorded between 28–365 days in 627 patients with recurrent events; (d) lowest non-HDL-C recorded in 314 patients with recurrent events.
A total of 1020 (6.7%) of patients had experienced an atherosclerotic event within 2 years prior to the index PCI of whom 627 had documented LDL-C during the study period. Of these, only 33 (5.3%) had LDL-C levels below the 2019 target of 1.0 mmol/l (Figure 1(c)).
Considering patients with documented LDL-C levels greater than 1.8 and 1.4 mmol/, 2437 (46%) and 4082 (53%) respectively were prescribed HI-statins; only 117 (2.2%) and 146 (1.9%) respectively were prescribed a combination of ezetimibe and/or fibrate plus a statin, with 103 (2.0%) and 109 (1.4%) prescribed ezetimibe and/or fibrate without a statin (Figure 2).
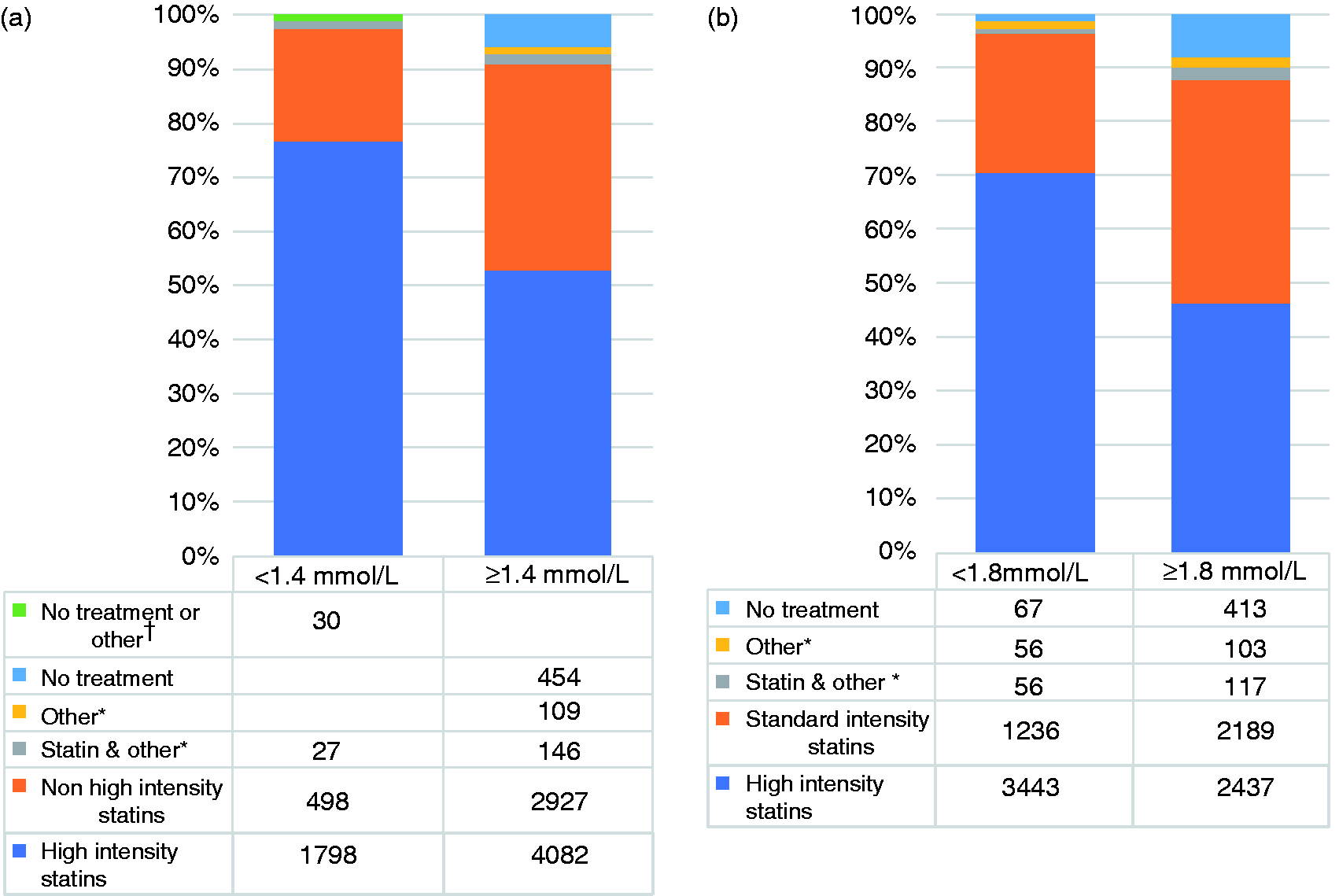
Prescribed lipid-lowering therapy in patients at or above (a) 2019 and (b) 2016 European Society of Cardiology/European Atherosclerosis Society (ESC/EAS) guideline low-density lipoprotein cholesterol (LDL-C) targets.
Of the 594 of patients with recurrent events within 2 years and LDL-C ≥1.0 mmol, 312 (52.5%) were prescribed HI-statins; 214 (36.0%) were prescribed standard-intensity statins, 19 (3.1%) were prescribed ezetimibe and/or a fibrate in addition to a statin, 12 (2.0%) were prescribed either ezetimibe and/or fibrate without a statin and 37 (6.2%) patients had no LLT prescription.
Characteristics independently associated with LDL-C ≥1.4 mmol/l included female sex and PVD. Diabetes mellitus, increasing age and the presence of an ACS during the index admission were independently associated with achieving the 2019 target LDL-C <1.4 mmol/l (Supplementary Material Table 2).
Non-HDL-C levels
Non-HDL-C levels were documented in 5340 patients (mean lowest non-HDL-C = 2.6 mmol/l, SD = 0.97) of whom 2286 (42.8%) had a non-HDL-C ≥2.6 mmol/l and 3366 (63.0%) ≥2.2 mmol/l (Figure 1(b)). Amongst those with recurrent atherosclerotic events, 314 patient had non-HDL-C results documented, of whom only 40 (12.7%) were below the threshold of 1.8 mmol/l (Figure 1(d)).
The LLT regimens in those achieving and not achieving target non-HDL-C levels were similarly distributed to those observed in patients at and not at LDL-C targets (Supplementary Material Figure 3).
Characteristics independently associated with non-HDL-C >2.2 mmol/l included female sex, hypertension and PVD (Supplementary Material Table 3). ACS during the index admission, increasing age and LLT, but not diabetes independently predicted non-HDL-C <2.2 mmol/l.
TG levels
A total of 10,592 patients had TG levels recorded between 28 days and 1 year (mean lowest TG = 1.54 mmol/l, SD = 1.01), of whom 1524 (14.4%) had TG ≥2.3 mmol/l and 4314 (40.7%) had TG ≥1.5 mmol/l (Figure 3).
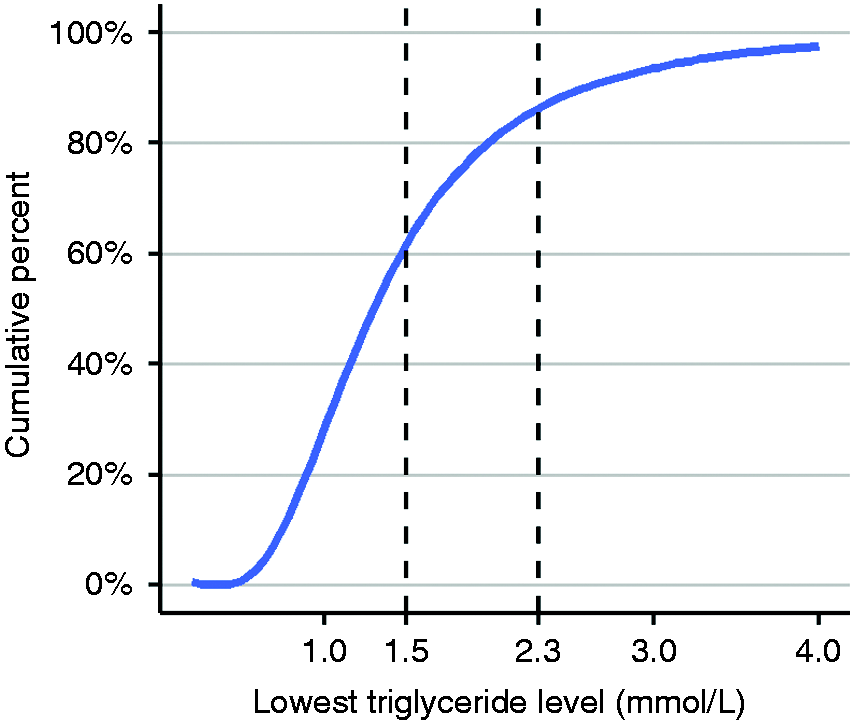
Lowest triglyceride recorded between 28–365 days in 10,592 patients.
Of those patients with TG ≥2.3 mmol/l and ≥1.5, 1313 (86.2%) and 3847 (89.2%) were prescribed statins respectively, 62 (4.1%) and 122 (2.0%) prescribed fibrates and/or N-3 and 133 (8.7%) and 312 (7.2%) were not prescribed any LLT (Figure 4).
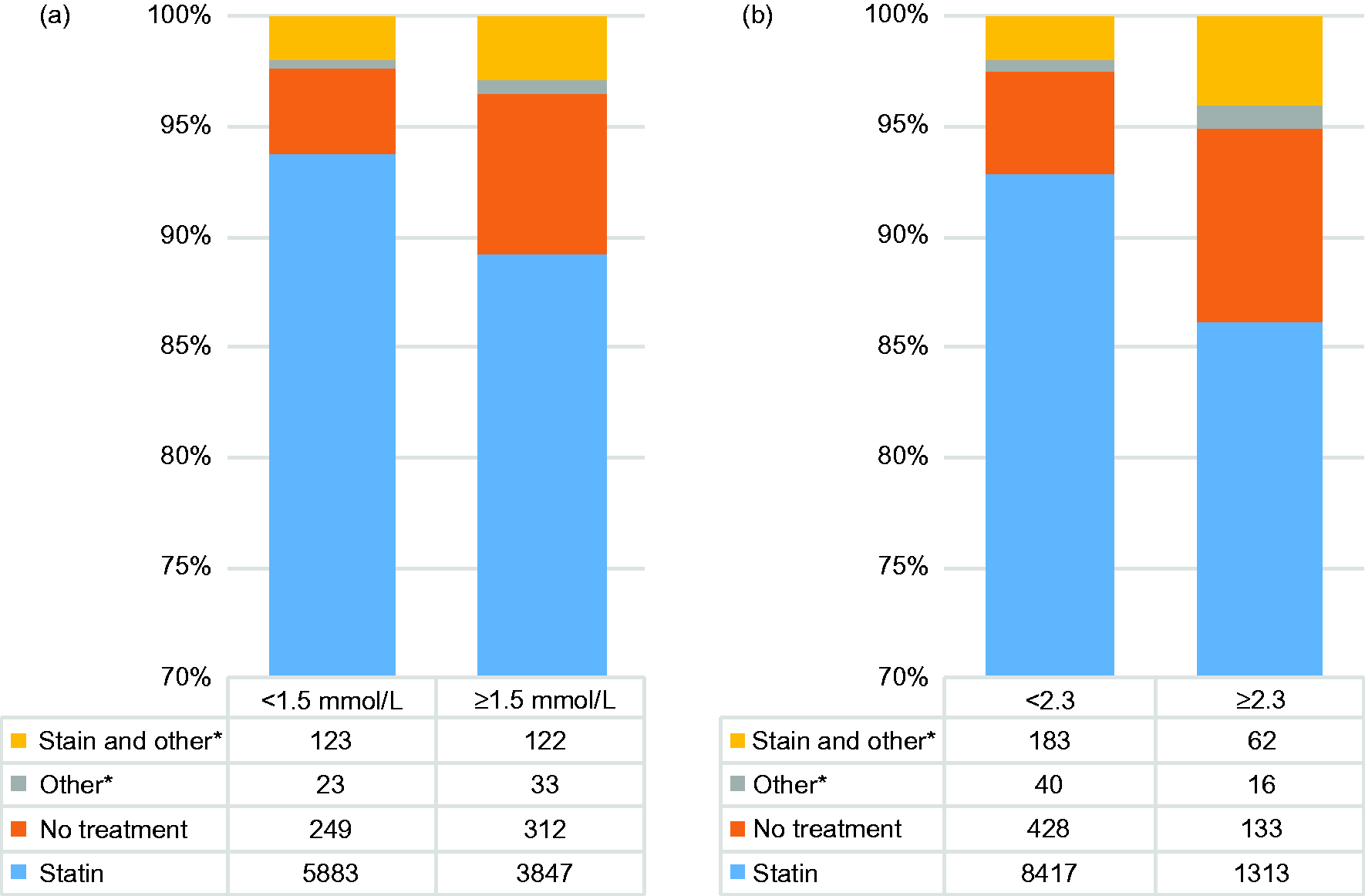
Prescribed lipid-lowering therapy according to triglyceride levels.
In a multivariable analysis, variables independently associated with TG>1.5 mmol/l included female sex, hypertension, CKD stage 4+, ischaemic stroke, PVD and diabetes mellitus.
Discussion
This is the first real-world study examining the prescribing of LLT and achieved lipid levels against the recently published ESC/EAS guideline targets in a national cohort of post-PCI patients. This analysis was conducted in order to examine the potential implications for changes in management strategy at a system level in this very-high-risk population.
Notably, just under half (47.8%) of these very-high-risk patients achieved LDL-C below the 2016 ESC/EAS target of 1.8 mmol/l in the year post-PCI, but fewer than a quarter (23.4%) were below the new 2019 target of 1.4 mmol/l. Furthermore, fewer than 6% of patients undergoing PCI within 2 years of a prior admission for an ACS, non-embolic ischaemic stroke or transient ischaemic attack (TIA) achieved the new target of below 1.0 mmol/l.
We also found that over 40% of patients had TG ≥1.5 mmol/l, the newly introduced recommendation for consideration of a high-dose N-3 prescription. In keeping with our findings, a recent Canadian real-world study has found that approximately 25% of patients with atherosclerotic cardiovascular disease had moderately elevated TG with controlled LDL-C, identifying an increased event rate in these patients compared to those with TG <1.0 mmol/l. 5
Prescribing of HI-statins was considerably lower amongst patients with LDL-C levels above target. Whilst it is possible that a proportion of these patients were intolerant of HI-statins, it is more likely that a greater proportion of patients prescribed NI-statins with uncontrolled LDL-C, would be tolerant of HI-statins, acknowledging prior reported levels of statin intolerance in just 10–20% of statin-treated patients.6–8 Furthermore, (co)prescribing of other LLTs, such as ezetimibe or fibrate, was also low.
Similarly, amongst those patients with recorded TG levels ≥2.3 mmol/l and ≥1.5 mmol/l, the prescribing of fibrate or N-3s was also low, with fewer than 3% of patients prescribed these treatments with or without statins. However, these therapies are not currently endorsed by the UK National Institute for Health and Care Excellence (NICE). 9
Although HI-statin prescription is recommended by NICE, only 56.4% of the patients in this study were receiving this treatment. Nevertheless, our observed HI-statin prescribing rate was considerably higher than documented in previous post-myocardial infarction studies, which have reported HI-statin therapy in only 4–31% of patients.6,10,11 However, these observations were in different healthcare systems,6,11 and in historical populations, 6 rather than a contemporary UK, post-PCI population.
To allow assessment of response to treatment and consideration that LDL-C levels tend to fall transiently after an ACS, we only analysed lipid profiles recorded beyond 28 days. For patients with ≥1 result we identified the lowest lipid levels recorded between 28–365 days and documented the LLT prescription in the preceding six-month period. Thus, our results do not represent longer-term lipid management and control beyond 1 year. However, it is well recognised that LLT prescribing, treatment concordance and lipid control decrease progressively over time.12,13
Just under three-quarters (73.1%) of our cohort had a lipid profile recorded during the first-year post-discharge. Females and those with a history of CKD stage 4+, ischaemic stroke, AF and/or PVD (clinical characteristics associated with worse outcomes in coronary patients) were not only less likely to have lipid levels documented, but also to have uncontrolled lipids. Although our findings provide further evidence of gender disparity in the management of CVD risk, they do highlight a specific, evidence-based opportunity to close this gap. 14
Notably patients with an ACS during the index admission were almost 2.5 times more likely to be prescribed HI-statins than those undergoing PCI for stable disease and more likely to have lipids checked within the first year. These observations are possibly due to the differences in clinical pathways between ACS patients and those undergoing PCI for stable coronary artery disease who are less likely to have changes made to longer-term CVD prevention regimens during an elective admission.
The 2016 non-HDL-C and LDL-C targets were achieved in 57% and 48% of patients, whereas the 2019 respective targets where achieved in only 37% and 23% of patients, with a similar clinical picture to that seen for LDL-C treatment and control. Importantly, whilst diabetic patients were more likely to achieve LDL-C targets this was not the case for non-HDL-C targets, emphasising the importance of addressing this measure in these very-high-risk individuals.
Two recent registry studies looked at the effectiveness of LDL-C control in populations with coronary artery disease (CAD).15,16 In both of these registries approximately 42% of ACS patients had LDL-C levels below the 2016 ESC/EAS target of <1.8 mmol/l, but only 28% of those with stable CAD achieved this.
Both of these studies emphasise the difficulty faced in achieving adequate LDL-C control across high-risk populations, even considering the less stringent 2016 targets. These registry findings are consistent with ours which also reflects the challenges in achieving the even more stringent 2019 targets. It is likely that increased use of novel therapies will be required, where appropriate, in order to close this therapeutic gap.
Strengths and limitations
By identifying important gaps in the achievement of ESC/EAS guideline target lipid levels, this study demonstrates considerable potential to improve secondary prevention of CVD through optimisation of evidence-based LLT. Although these data reflect practice prior to the publication of the 2019 guidelines, this is a recent, representative cohort of very-high-risk patients who would now qualify for more aggressive LDL-C, non-HDL-C and TG targets according to the new guidelines.
There are a number of potential limitations to this study. While we identified the prescribing of LLT, we were not able to identify the quantity of medication prescribed, whether it was dispensed or if the patient complied with therapy. This study makes no assumptions on medication compliance, which is often low in chronic conditions, particularly with statins.17,18 Therefore, the prescribing of LLT reported in this study likely describes a best-case management scenario.
We did not identify any patients who were prescribed PCSK9 inhibitors in our dataset. These agents were only approved for use within the UK National Health Service (NHS) in June 2016.19,20 Although the prescribing of these treatments is mainly provided though specialist hospital outpatient clinics, for which data was not available for this study, the uptake of PCSK9 inhibitors within Wales has been very low. Therefore, it is unlikely that the absence of the prescribing data for these treatments would have significantly influenced our results.
It was not possible to explore further why over a quarter of our patients did not have a lipid profile documented. It is unlikely that this was due to ‘loss to follow-up’ as we only included patients with ≥90 days follow-up (mean follow-up = 349 days), allowing sufficient time to record lipid profiles post-PCI. Patients without documented lipid levels typically had higher risk characteristics, but we cannot determine whether this difference is explained by confounding factors or a risk-treatment paradox.
It is possible that some patients had lipid profiles documented post-discharge within secondary care pathology datasets that were not available for this study. However, longer-term risk factor management is undertaken in primary care for the vast majority of patients in Wales in both the primary and secondary prevention settings.
Lipid profiles recorded during the index admission were also not available for this study. While we categorised patients at or above target levels, we were not able to calculate the percentage LDL-C reduction from baseline. It is therefore likely that an even greater number of patients would not have achieved a ≥50% reduction in LDL-C target levels.
In this study, 6.7% of patients were classified as having recurrent atherosclerotic events, including a previous hospital admission for an ACS, coronary revascularisation, non-embolic stroke or TIA within 2 years before the index PCI. Due to ambiguity of clinical coding of PVD within the secondary care hospital datasets, we were unable to accurately detect acute peripheral vascular events and therefore these were not included. We noted that a far greater number of our patient cohort had experienced prior clinical atherosclerotic events, but over 2 years before the index PCI.
It was beyond the scope of this study to evaluate the relationships between early post-PCI lipid levels, LLT strategy and clinical outcomes in the first year post-PCI. The purpose of this short report was to characterise the nature of LLT and achievement of lipid targets according to contemporary ESC/EAS2 guidance at the time of PCI. This identifies the potential for further optimisation of lipid management in this very-high-risk cohort with reference to the new guideline targets and recommended treatments. Therefore, future prospective evaluation will be required to evaluate the how changes in lipid management impact on lipid levels and clinical outcomes at a population level.
A last, but important, consideration is that the data for this study were obtained from patients treated in the Welsh NHS, where the cost of healthcare, including prescription drugs, is entirely free at the point of delivery. This mitigates against potential barriers of affordability of follow-up consultation, lipid monitoring and medication purchasing. These would be important considerations when comparing the findings of our study to other healthcare systems, where greater discrepancies in quality of care might be expected in more economically disadvantaged individuals and populations. 21
We have identified a relatively low rate of prescribing of HI-statins ± additional evidence-based LLT agents in a large post-PCI patient population, as well as a relatively low proportion of these patients achieving the new EAS/ESC lipid targets. Our data suggest that there is considerable potential to optimise LLT through statin intensification and appropriate use of novel LLT, which would be expected to improve clinical outcomes at both an individual and population level. However, the budgetary impact of novel management strategies and thus potential value to healthcare providers and funders will need to be carefully planned and evaluated.
Supplemental Material
CPR914115 Supplemental material - Supplemental material for Achievement of European guideline-recommended lipid levels post-percutaneous coronary intervention: A population-level observational cohort study
Supplemental material, CPR914115 Supplemental material for Achievement of European guideline-recommended lipid levels post-percutaneous coronary intervention: A population-level observational cohort study by Daniel E Harris, Arron Lacey, Ashley Akbari, Fatemeh Torabi, Dave Smith, Geraint Jenkins, Daniel Obaid, Alex Chase, Mike Gravenor and Julian Halcox in European Journal of Preventive Cardiology
Footnotes
Author contribution
DH, MG and JH designed the study. DH, AA, FT and AL contributed to the data acquisition. DH, AL, AA, FT, DS, GJ, DO, AC, MG & JH all contributed to the interpretation of data, revision of manuscript and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: All authors have completed the ICMJE uniform disclosure at ![]() and declare: DH, JH, DS and AC have received speaking and/or consulting fees from Amgen. JH has also received speaking/consulting fees from Sanofi Aventis, Abbott Products and Bayer. The authors have no other relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the article apart from those disclosed. No writing assistance was utilised in the production of this article.
and declare: DH, JH, DS and AC have received speaking and/or consulting fees from Amgen. JH has also received speaking/consulting fees from Sanofi Aventis, Abbott Products and Bayer. The authors have no other relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the article apart from those disclosed. No writing assistance was utilised in the production of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Swansea Bay University Health Board research and development department.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
