Abstract

Circulating cell-free DNA (cfDNA) is genomic DNA released as result of cell death mechanisms (e.g. apoptosis, necrosis and autophagy).1 Mostly investigated in oncology, cfDNA fragments have proven utility for early detection, treatment monitoring and risk stratification of cancer and this is known as the ‘liquid biopsy’ method.1–3 More recently, investigations in the cardiovascular field4–6 have shown associations of cfDNA with cardiovascular risk factors, 7 cardiovascular disease status (acute myocardial infarction and atrial fibrillation), 6 and for the early diagnosis of heart transplant rejection.8–10 However, despite knowing that cell death is one of the features of heart failure (HF) pathobiology, cfDNA has never been investigated in chronic HF. The aim of the present study was to assess, for the first time, the role of the liquid biopsy in HF, investigating cfDNA levels in HF patients and their associations with clinical status, morbidity and mortality.
Seventy-one consecutive chronic stable HF patients with ejection fraction (EF) < 50% were enrolled from our HF unit and compared with 64 healthy volunteers enrolled as controls from a preventive medicine programme. Inclusion criteria comprised no history of recent acute HF decompensation in the last 6 months, severe liver (cirrhosis Child-Pugh class) or kidney disease (serum creatinine >2.5 mg/dl); further, patients had to be on stable HF medications for at least 3 months. cfDNA was extracted from plasma using the QIAamp Blood DNA Mini Kit (Qiagen). Each sample was quantified in triplicate relative to a standard curve, generated from a serially diluted human genomic DNA template (Roche) of known concentration and an in-house locus-specific TaqMan assay targeting a 96 bp intronic sequence in the housekeeping gene, glyceraldehyde 3-phosphate dehydrogenase (GAPDH). 2 Primers were designed to target GAPDH as described previously. 3 B-type natriuretic peptide (BNP) levels were measured with the RapidPIA BNP kit (Sekisui Medical Co). The primary endpoint was defined as a composite of all-cause mortality and/or HF hospitalisation (death/HF) at 30 months. The associations between analysed variables and outcome were investigated using Cox proportional hazard regression analyses. Kaplan-Meier survival curves were generated and the Mantel-Cox log-rank test was used to compare event-free survival after stratification of cfDNA levels by the median. Outcome prediction accuracies were assessed by calculating the area under the curve (AUC) for receiver-operating characteristic (ROC) curve analysis and by using the Net Reclassification Index (NRI) for cfDNA and BNP levels, alone and/or combined, as adjusted for the multivariable linear model.
Patient demographics, blood chemistry measurements and clinical characteristics are described in Table 1. When cfDNA levels were compared, approximately six-fold higher cfDNA levels were observed in HF patients (median (interquartile range); HF: 19.4 (11.1–41.7), healthy: 3.4 (2.04–6.06), p < 0.001), with significant correlations between cfDNA levels and New York Heart Association (NYHA) class (rs = 0.294, p = 0.017) and biochemistry markers (albumin, calcium, creatine phosphokinase and beta-2 globulin) (rs = −0.267–0.298, p ≤ 0.048). No correlations were found with age, sex or echocardiographic parameters. For outcome analyses, cfDNA levels were not significantly associated with outcome as a continuous variable. However, when cfDNA levels were dichotomised according to the median value of the HF population (19.4 ng/ml) and also using Youden’s Index for optimal cut-off point from the ROC curve (20.8 ng/ml), with both methods of applying a cut-off value yielding identical categories (n = 34 patients with elevated cfDNA levels), univariate analysis showed that cfDNA levels were associated with death/HF hospitalisation at 30 months (hazard ratio (95% confidence interval) p value; 2.57 (1.36–4.87) p = 0.004). When adjusted for main clinical parameters (age, NYHA class, sex, aetiology, BNP and estimated glomerular filtration rate), cfDNA retained prediction abilities (2.12 (1.04–4.30) p = 0.038) (Table 2). ROC curve analysis using the same multivariable model, with and without BNP and cfDNA levels, showed that the combination of BNP and cfDNA gave a consistent gain in C-statistic for associations with outcome when compared to the same model with either BNP or cfDNA alone ((0.750 (0.619–0.881), 0.752 (0.632–0.872) and 0.796 (0.687–0.905) for BNP alone, cfDNA alone and combination of both biomarkers respectively, p < 0.001) (Table 3). For net reclassification analysis, when added to the clinical variables for the composite endpoint of death/HF, the combination of cfDNA and BNP (55.8 (10.3–101.3) p = 0.016) showed an improvement over BNP alone (52.9 (8.63–97.3) p = 0.019); however cfDNA alone was superior (63.8 (17.7–109.8) p = 0.006) suggesting that the cfDNA level is the weighing factor (Table 3). Kaplan-Meier survival analyses showed that HF patients with cfDNA levels above the median value have reduced survival (chi-square: 9.32; log rank test p = 0.002) (Figure 1(a)). In addition, when patients were categorised according to median levels of cfDNA and BNP (Figure 1(b)), patients with elevations in both biomarkers showed the lowest survival, significantly different to those with normal levels of both biomarkers (chi-square 17.83, log rank test p < 0.001) or only BNP elevated (chi square 4.52, p < 0.030). No differences were found when compared to patients with only cfDNA elevated which suggests that elevations in cfDNA levels is the weighing factor.
Patient demographics.
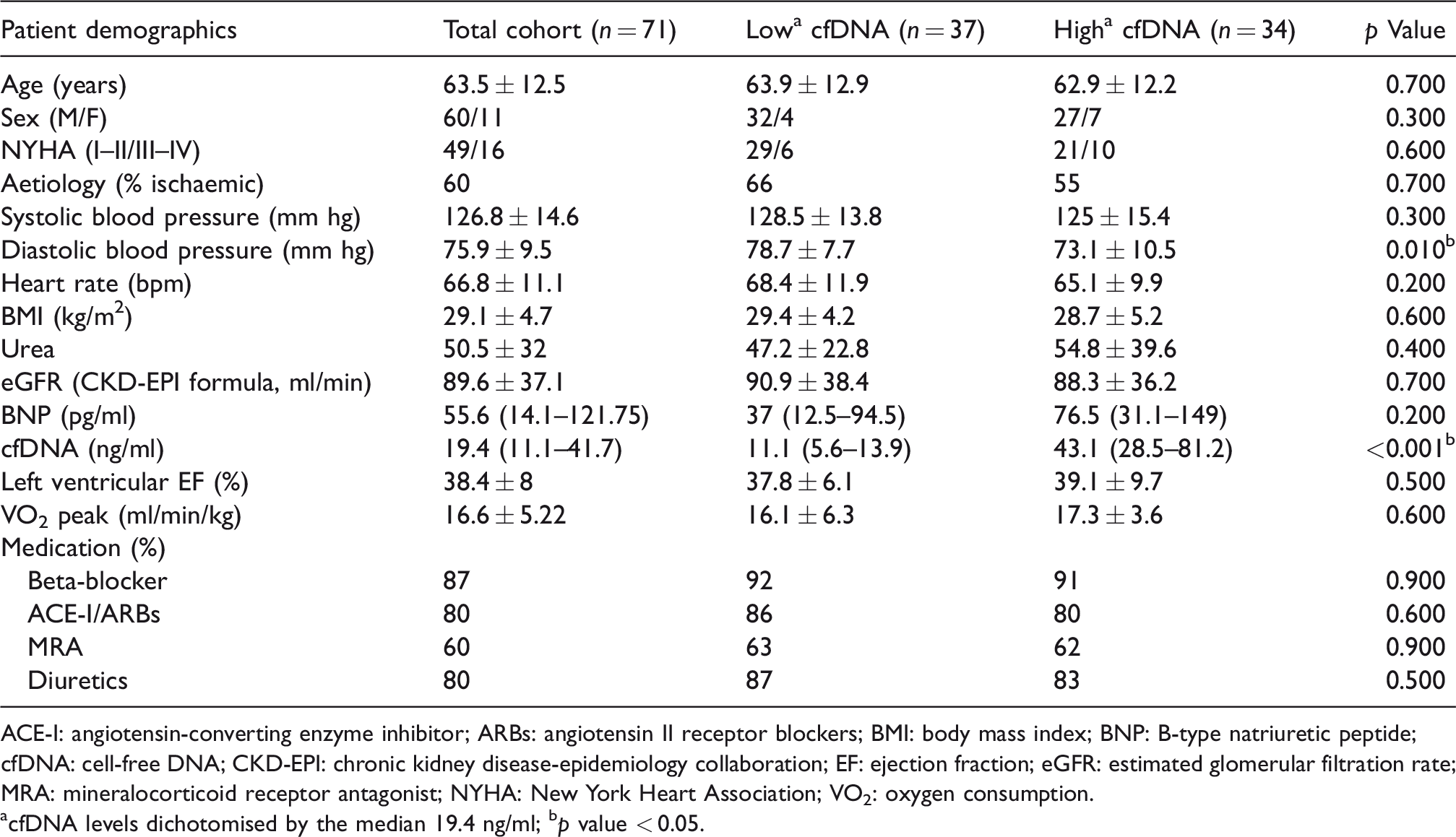
ACE-I: angiotensin-converting enzyme inhibitor; ARBs: angiotensin II receptor blockers; BMI: body mass index; BNP: B-type natriuretic peptide; cfDNA: cell-free DNA; CKD-EPI: chronic kidney disease-epidemiology collaboration; EF: ejection fraction; eGFR: estimated glomerular filtration rate; MRA: mineralocorticoid receptor antagonist; NYHA: New York Heart Association; VO2: oxygen consumption.
acfDNA levels dichotomised by the median 19.4 ng/ml; bp value < 0.05.
Cox proportional hazard regression analyses to establish association between analysed variables and outcome.
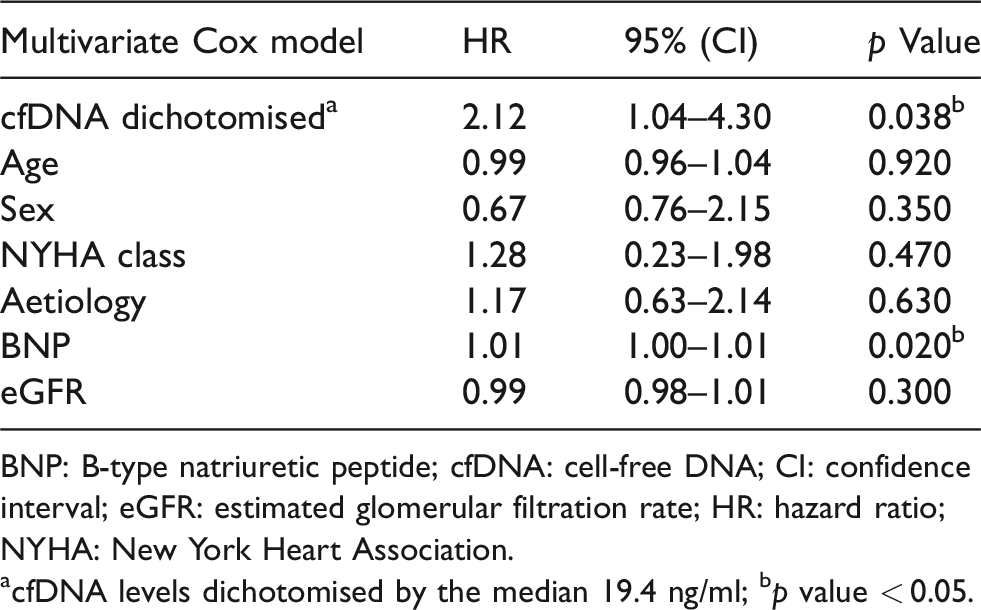
BNP: B-type natriuretic peptide; cfDNA: cell-free DNA; CI: confidence interval; eGFR: estimated glomerular filtration rate; HR: hazard ratio; NYHA: New York Heart Association.
acfDNA levels dichotomised by the median 19.4 ng/ml; bp value < 0.05.
Reclassification analysis using continuous reclassification of adding B-type natriuretic peptide (BNP), cell-free DNA (cfDNA) or both to the multivariable predictive model.
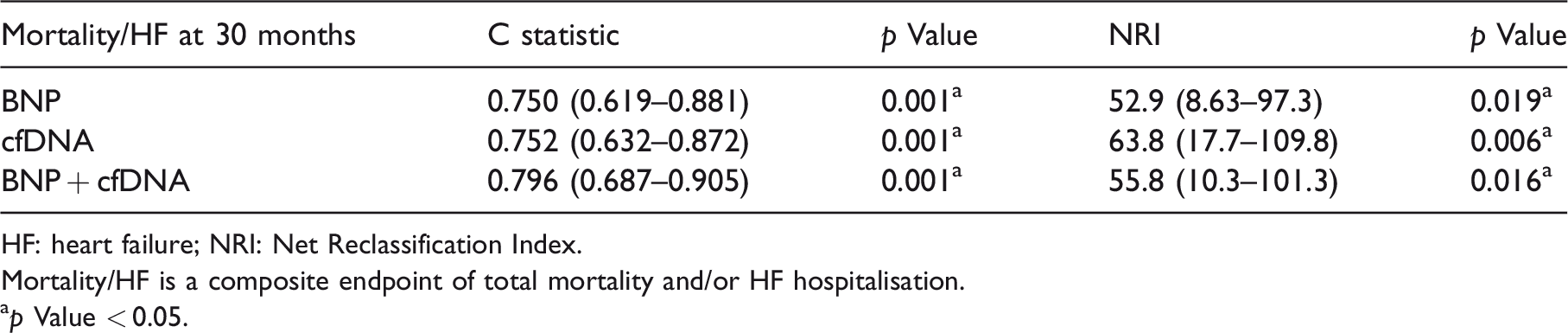
HF: heart failure; NRI: Net Reclassification Index.
Mortality/HF is a composite endpoint of total mortality and/or HF hospitalisation.
ap Value < 0.05.
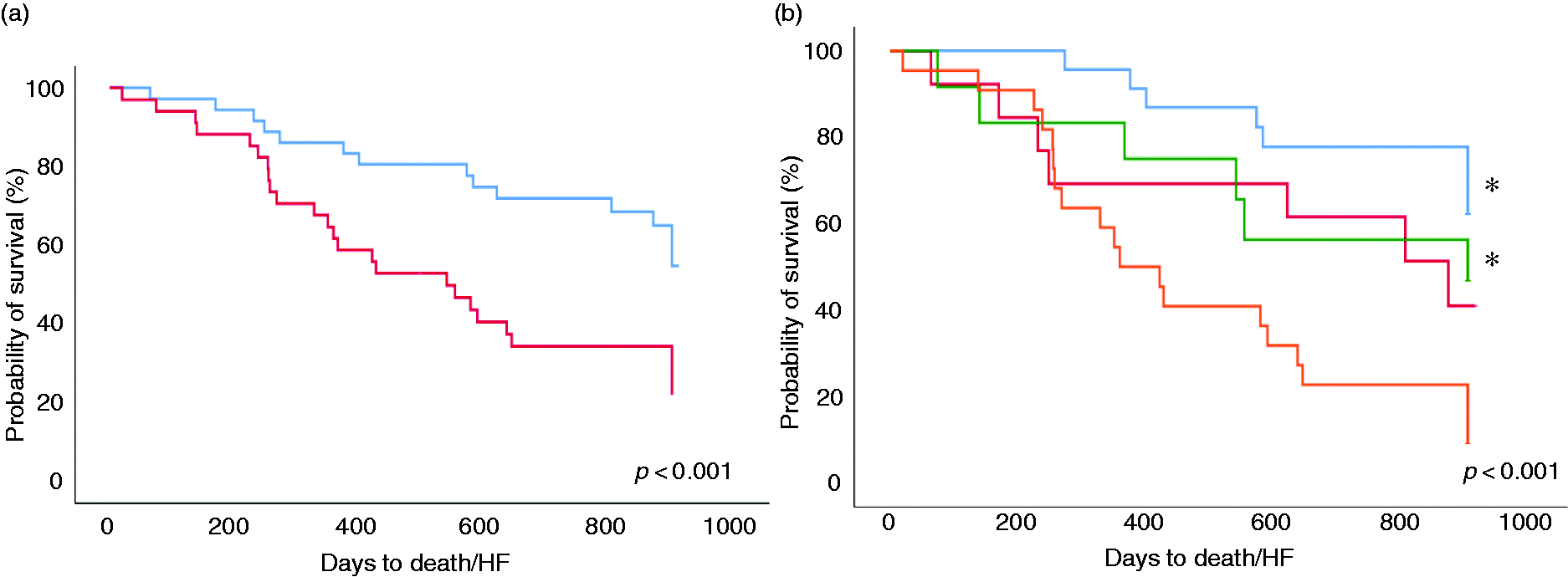
(a) Kaplan-Meier survival analysis for death and/or rehospitalisation due to heart failure (HF) stratified by cell-free DNA (cfDNA) median. Light blue: above the median; Red: under the median. (b) Kaplan-Meier survival analysis for death and/or rehospitalisation due to heart failure (death/HF) stratified by cfDNA and B-type natriuretic peptide (BNP) median. Blue: low cfDNA and low BNP; green: low cFDNA and high BNP; red: high cfDNA and low BNP; orange: high cfDNA and high BNP. *p < 0.05 when compared to high cfDNA and high BNP.
The present investigation showed, for the first time, that circulating cfDNA levels are elevated in HF patients compared to healthy subjects, similar to other cardiovascular diseases.6,11,12 In addition, cfDNA is associated with clinical status. When combined with BNP, cfDNA levels added to patient risk stratification, with cfDNA as the weighing factor, as shown by the Kaplan-Meier and net reclassification analyses. Despite the precise mechanisms of cfDNA in HF pathophysiology being unknown, it is plausible to envisage a combinatorial mechanism involving myocardial injury, chronic inflammation and thrombosis/platelet aggregation. Indeed, cfDNA is released from the heart upon reperfusion after prolonged ischaemia, and plays a critical role in ischaemia-reperfusion injury through a receptor for advanced glycation end products (RAGE)-toll like receptor 9. 13 Additionally, in cardiovascular diseases, cfDNA has been implicated in hyperactivation of polymorphonuclear cells which promote platelet aggregation and chronic thrombosis.4,6 Higher cfDNA levels have also been reported to be associated with systemic low-grade inflammation markers (e.g. interleukin (IL)-6, IL-8). 7 Furthermore, an association of cfDNA has been reported with increased neutrophil extracellular trap formation, which impacts the efficiency of fibrinolysis with enhanced peak thrombin generation, impaired fibrinolysis and decreased clot permeability. 14 As study limitations, patients were recruited from a single centre, with a relatively small sample size that does not allow extensive prediction modelling to be performed. However, the present report is hypothesis-generating, and further large-scale studies are warranted.
In conclusion, cfDNA levels are associated with morbidity and mortality in HF, suggesting that liquid biopsy may offer additional methods to monitor and stratify the risk of outcome in HF patients.
Footnotes
Acknowledgements
RapidPIA BNP kits were provided by Sekisui Medical Co.
Author contribution
AS and MZI contributed to conception, design, data acquisition, analysis and interpretation of the study, drafted and critically revised the manuscript. DFG contributed to design, data acquisition, analysis and interpretation of the study, drafted and critically revised the manuscript. LM contributed to design, data acquisition and analysis, drafted and critically revised the manuscript. RD contributed to data acquisition, drafted and critically revised the manuscript. AMM and MA contributed to data interpretation, drafted and critically revised the manuscript. YY, DB and SC contributed to analysis and interpretation, drafted and critically revised the manuscript. KP contributed to design, interpretation of the study, drafted and critically revised the manuscript. GR and EB contributed to data interpretation and critically revised the manuscript. AC contributed to conception, design, data acquisition and interpretation of the study, and critically revised the manuscript. JAS contributed to conception, design, data acquisition, analysis and critically revised the manuscript. TS contributed to the conception, design, data analysis and interpretation of the study and critically revised the manuscript. All authors gave final approval and agree to be accountable for all aspects of the work ensuring integrity and accuracy.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: A Salzano receives research grant support from CardioPath, Department of Advanced Biomedical Sciences, Federico II University, Naples, Italy, UniNA and Compagnia di San Paolo in the frame of the STAR (Sostengo Territoriale alla Attività di Ricerca) programme.
