Abstract

Chronic heart failure is diagnosed in 1–2% of the adult population, with a prevalence ≥10% among individuals > 80 years of age. 1 Heart failure is defined as cardiac dysfunction combined with symptoms such as exercise intolerance and attenuation of habitual physical activity. 2 Professional guidelines recommend supervised exercise training for heart failure patients to reduce the risk for hospital admittance and improve quality of life. 3 Nevertheless, prognosis of heart failure is highly variable and influenced by health/disease characteristics and exercise parameters. 4 Risk prediction among cardiovascular patients is an emerging topic of interest.5–7 To gain insight into the prognosis of heart failure, several clinical risk models were developed, such as the Meta-Analysis Global Group in Chronic heart failure (MAGGIC) and Metabolic Exercise data combined with Cardiac and Kidney Indexes (MECKI) score.8,9 Both MAGGIC and MECKI scores are internally and externally validated,8–11 but their clinical use is limited to specific settings. Furthermore, it is unclear whether these risk scores and their predictive ability change after completion of a cardiac rehabilitation programme. The aim of the present study is to examine the change in MAGGIC and MECKI score after cardiac rehabilitation supervised exercise training and to identify the predictive power of these scores.
Heart failure patients who attended an exercise-based cardiac rehabilitation programme at the Isala Clinic (Zwolle, Netherlands) between August 2009 and January 2017 were eligible to participate in this retrospective cohort study. Patients were excluded if they did not perform a post-rehabilitation exercise test and if they had missing values for MAGGIC or MECKI scores due to absence of baseline or post-rehabilitation characteristics. Electronic patient files were used to collect baseline and post-rehabilitation patient characteristics including age, sex, body mass index, current smoking status, diabetes mellitus and chronic obstructive pulmonary disease. Furthermore, heart failure characteristics such as disease aetiology, New York Heart Association (NYHA) classification, medication use and the presence of medical devices were aggregated. Serum sodium, potassium, creatinine and haemoglobin levels were collected from laboratory results at baseline and post-rehabilitation. Glomerular filtration rate was calculated using Modification of Diet in Renal Disease score. 12 Left ventricular ejection fraction (LVEF) was assessed using echocardiography. At baseline and upon completion of the cardiac rehabilitation programme, heart failure patients underwent cardiopulmonary exercise testing on a bicycle ergometer to obtain physical fitness level. Breath-by-breath analysis measured oxygen uptake, carbon dioxide production, and ventilation. After a 3-min warm-up (0 W), a personalized ramp protocol was performed to achieve a test duration of 8–12 min. Workload increased every minute and subjects were verbally encouraged to achieve maximum exertion. Maximal VO2 uptake (VO2-peak) and VE/VCO2 slope were determined. Primary end points were all-cause mortality, cardiovascular mortality and urgent heart transplantation.
Descriptive statistics for pre- and post-rehabilitation variables of included heart failure patients to calculate MAGGIC and MECKI score.
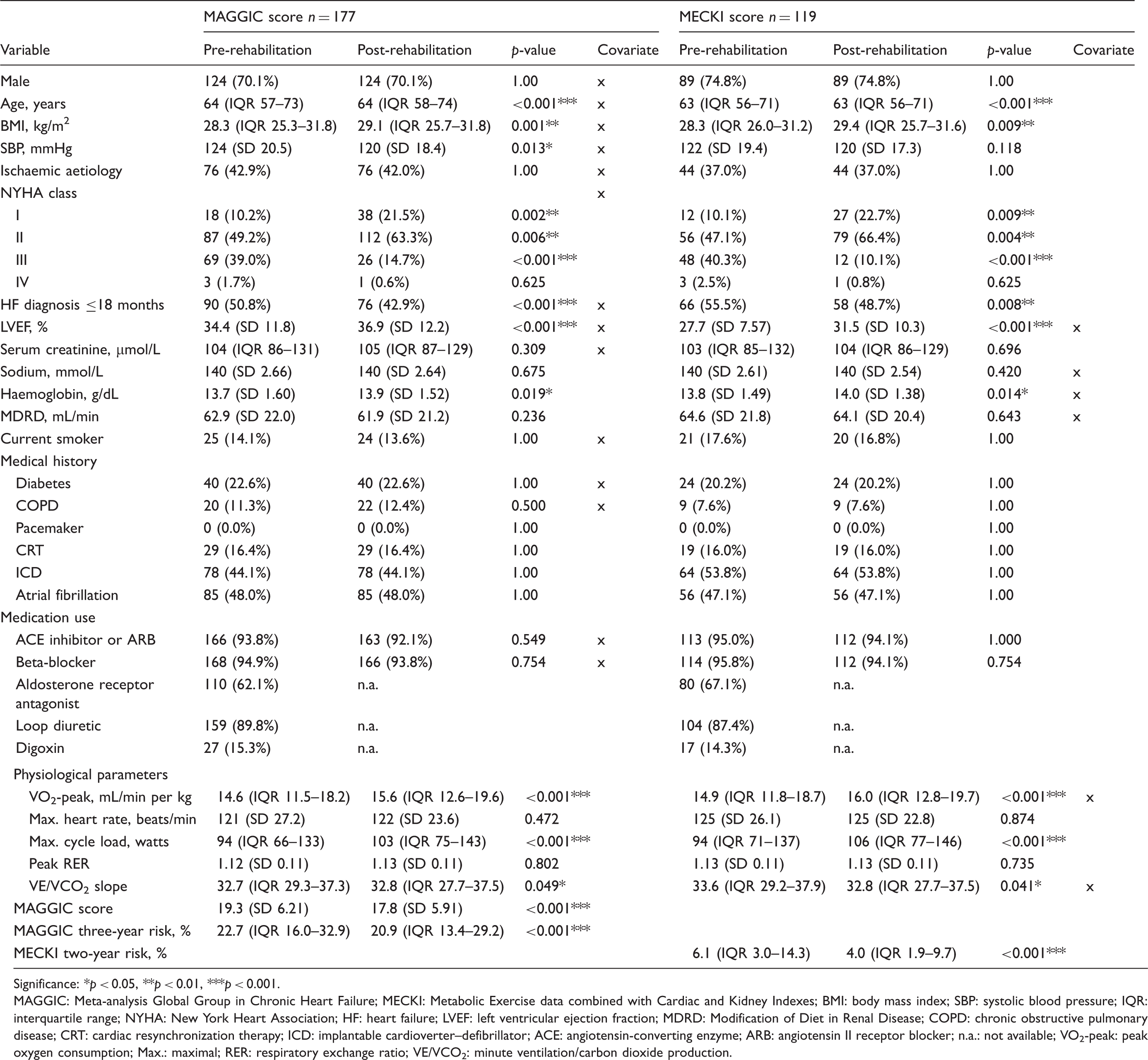
Significance: *p < 0.05, **p < 0.01, ***p < 0.001.
MAGGIC: Meta-analysis Global Group in Chronic Heart Failure; MECKI: Metabolic Exercise data combined with Cardiac and Kidney Indexes; BMI: body mass index; SBP: systolic blood pressure; IQR: interquartile range; NYHA: New York Heart Association; HF: heart failure; LVEF: left ventricular ejection fraction; MDRD: Modification of Diet in Renal Disease; COPD: chronic obstructive pulmonary disease; CRT: cardiac resynchronization therapy; ICD: implantable cardioverter–defibrillator; ACE: angiotensin-converting enzyme; ARB: angiotensin II receptor blocker; n.a.: not available; VO2-peak: peak oxygen consumption; Max.: maximal; RER: respiratory exchange ratio; VE/VCO2: minute ventilation/carbon dioxide production.
A total of 177 and 119 heart failure patients were included for analyses of the MAGGIC and MECKI score, respectively. Baseline and post-rehabilitation characteristics of heart failure patients are summarized in Table 1. During a median follow-up of 45 (interquartile range 28–73) months, 30 (16.9%) patients died. No patients received an urgent heart transplantation. AUC for MAGGIC three-year was 0.66 at baseline and increased to 0.70 at post-rehabilitation. MECKI two-year showed a discriminative performance of 0.60 at baseline and improved to 0.82 after cardiac rehabilitation (Figure 1). So, model performance of MECKI score improved from fair at baseline to excellent after CR, whereas model performance of the MAGGIC score remained fair to acceptable. Our findings suggest that the MECKI score can be a valuable tool to monitor changes in risk estimates for adverse outcomes in a population of heart failure patients.
Discriminative performance of the (a) MAGGIC three-year score for all-cause mortality in heart failure patients (n = 177) and (b) MECKI two-year score for cardiovascular mortality in patients with heart failure with reduced ejection fraction (n = 119). Red lines represent the pre-rehabilitation performance, whereas green lines represent post-rehabilitation performances of MAGGIC and MECKI. The AUC for mortality prediction increased after cardiac rehabilitation, with the largest increase for MECKI two-year from 0.60 (95% CI 0.34–0.86) at pre-rehabilitation to 0.82 (95% CI 0.72–0.91).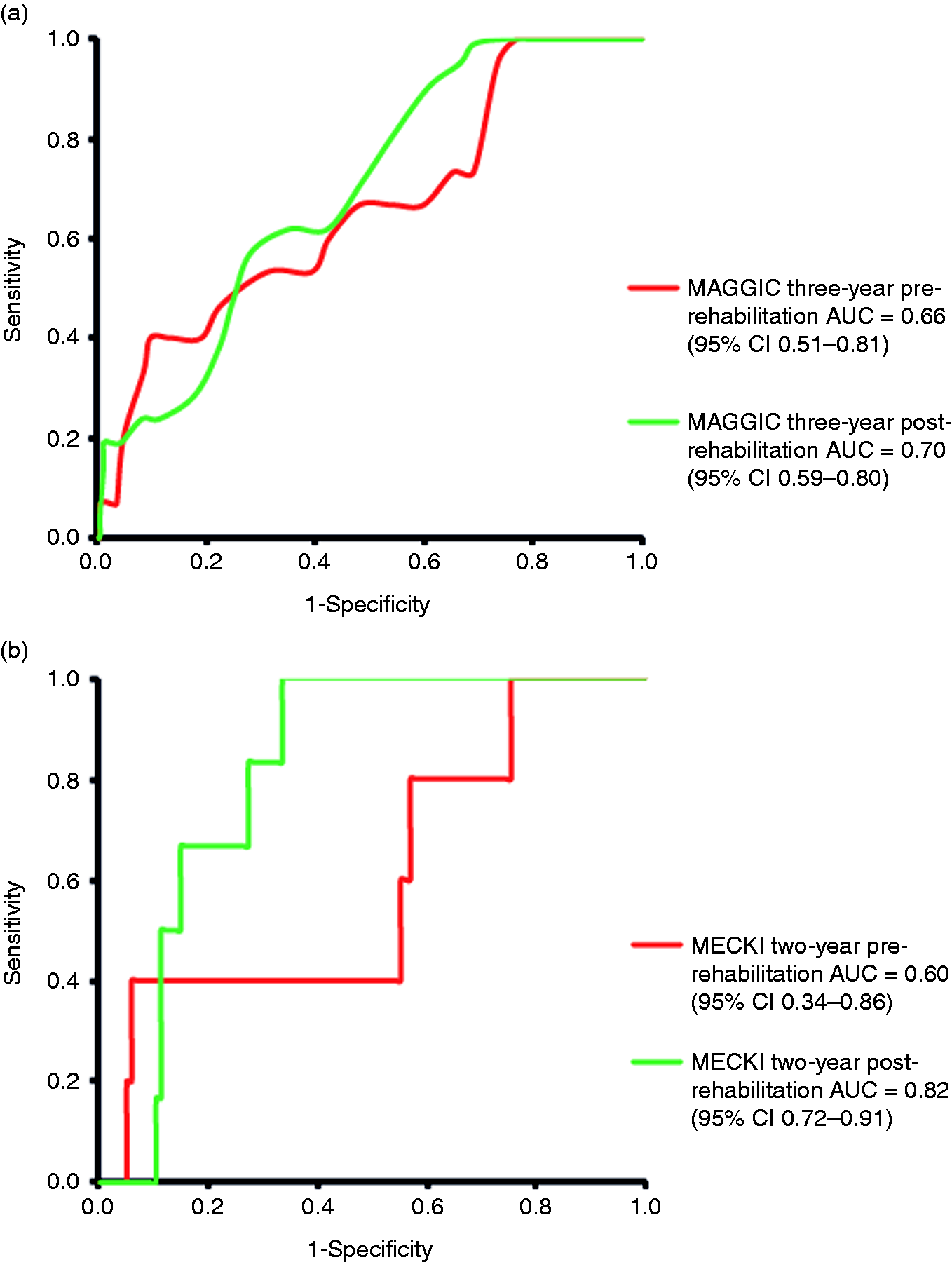
A previous study examining the MAGGIC score showed a higher AUC (0.781) for all-cause mortality after three years of follow-up compared with our results at baseline (AUC: 0.66) and post-rehabilitation (AUC: 0.70). 10 Another study validated the MECKI score in an Italian heart failure population and showed a higher AUC (0.76) compared with our results at baseline (AUC: 0.60), 11 but our AUC was slightly higher after cardiac rehabilitation (AUC: 0.82). Moreover, Freitas et al. showed recently that MECKI emerged as the best risk predictor with very good discriminative power for their original endpoints compared with Heart Failure Survival Score, Seattle Heart Failure Model and MAGGIC. 13 The difference in model performance between MECKI and MAGGIC might be due to different combinations of clinical variables, biomarkers and exercise capacity. The MECKI score includes exercise capacity parameters, which are important prognostic factors, 14 whereas the MAGGIC score is solely based on subject and disease characteristics. Another explanation for the difference in predictive ability might be the use of NYHA class in the MAGGIC score rather than VO2-peak as a measure of functional capacity. Finally, the difference in prediction time between MAGGIC and MECKI might also explain the observed divergence.
In conclusion, the MECKI score was better able to detect changes in cardiovascular risk after cardiac rehabilitation, with an excellent model performance in Dutch heart failure patients. In contrast, the predictive capacity of the MAGGIC score was only fair and did not improve after cardiac rehabilitation. These observations emphasize that the MECKI score can appropriately monitor time-dependent changes in risk estimates for adverse outcomes in heart failure patients.
Footnotes
Acknowledgement
The authors thank Annemiek Huis in ’t Veld for data collection and data entry.
Author contribution
TMHE, EAB, JAS and IJMK contributed to the conception and design of the work. TMHE, EAB and IJMK contributed to the acquisition, analysis and interpretation of data for the work. JAS contributed to the acquisition and interpretation of data for the work. MJMC contributed to the interpretation of data for the work. IJMK drafted the research letter. TMHE, EAB, JAS and MJMC critically revised the research letter. All gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: TMHE is funded by the Dutch Heart Foundation (Senior E-Dekker grant #2017T051).
