Abstract
Background
Endurance exercise training induces cardio-protective effects, but athletes are not exempted from a myocardial infarction. Evidence from animal studies suggests that exercise training attenuates pathological left ventricular remodelling following myocardial infarction. We tested the hypothesis that lifelong exercise training is related to attenuated pathological left ventricular remodelling after myocardial infarction as evidenced by better left ventricular systolic function in veteran athletes compared to sedentary peers.
Design
This was a cross-sectional study.
Methods
Sixty-five males (60 ± 6 years) were included and allocated to four groups based on lifelong exercise training volumes: (a) athletes (
Results
Cardiac enzyme markers (creatine-kinase, creatinine, aspartate transaminase and lactate dehydrogenase) following myocardial infarction and infarct location did not differ between athletes + myocardial infarction and sedentary controls + myocardial infarction. Left ventricular ejection fraction was significantly higher in athletes (61% ± 4), athletes + myocardial infarction (58% ± 4) and sedentary controls (57% ± 6) compared to sedentary controls + myocardial infarction (51% ± 7;
Conclusion
These findings suggest that lifelong exercise training may preserve left ventricular systolic function and possibly attenuates or minimises the deleterious effects of pathological post-myocardial infarction left ventricular remodelling in veteran athletes.
Introduction
Regular exercise training leads to a favourable cardiovascular risk factor profile, 1 improves cardiovascular function2,3 and lowers the risk for cardiovascular disease.4,5 Despite the cardio-protective effects of exercise training, athletes are not exempted from acute coronary syndromes or myocardial infarction (MI). 6
After a MI, pathological left ventricular (LV) remodelling starts within hours.7,8 This process is characterised by LV wall thinning, LV wall dilatation, reduced ejection fraction and scar formation,7,8 and eventually leads to impaired LV function. Preventing or reversing these maladaptations is of utmost importance to recover and maintain LV function. Animal studies have demonstrated that exercise training before a MI attenuates pathological LV remodelling. 9 Trained rats had less cardiac damage after ligation of the left anterior descending artery and fewer changes in cardiomyocyte function. 9 These results suggest that a physically active lifestyle before a MI may attenuate pathological LV remodelling. Confirmation of these findings in humans is lacking.
The primary aim of the study was to determine whether lifelong exercise training is related to attenuated pathological left ventricular remodelling after MI. For this purpose, we collected echocardiographic images in veteran athletes with and without a MI and sedentary controls with and without a MI. We hypothesised that veteran athletes will have better LV systolic function compared to their sedentary peers after a MI.
Methods
Participants
Sixty-five male participants were included and stratified into four groups based on their lifelong physical activity patterns and cardiac medical history: (a) veteran athletes (ATH group,
Study design
Individuals that expressed interest in the study were first screened via telephone and completed a questionnaire regarding their lifelong exercise history. Participants visited our laboratory at the Radboud university medical center on two days during this cross-sectional study. On day 1, participants were medically screened for eligibility, followed by an incremental maximal cycling test. On day 2, participants underwent a comprehensive assessment of LV function using transthoracic echocardiography.
Measurements
Medical screening
A physician screened the participants by taking a detailed medical history, physical examination, and 12-lead electrocardiogram. Blood samples were obtained, under fasting conditions, from an antecubital vein for the analysis of total cholesterol, high-density lipoprotein (HDL), low-density lipoprotein (LDL), triglycerides, glucose and glycated haemoglobin (HbA1c).
Lifelong physical activity patterns
Lifelong physical activity patterns were queried via an exercise history questionnaire, distinguishing five age-periods: (a) 20–29 years, (b) 30–39 years, (c) 40–49 years, (d) 50–59 years, and (e) >60 years. Each category consisted of three queries: (a) type of activity (e.g
Incremental maximal cycling test
Cardiorespiratory fitness (peak oxygen uptake (VO2peak), ml O2/min/kg) was assessed via an incremental maximal cycling test. Participants cycled with 60–80 rotations/min while the workload increased with 20 watts/min for athletes and 10 watts/min for post-MI controls. Heart rate (12 lead-electrocardiogram), oxygen uptake (VO2 (ml/min)), carbon dioxide output (VCO2 (ml/min)), and respiratory exchange ratio (RER) were continuously measured (Cardio Pulmonary Exercise Test system, Cosmed v9.1b, Italy). 12 The anaerobic threshold was defined as a RER above 1.0. 12 Participants were verbally encouraged to stimulate maximal exercise performance. Lactate concentration (mmol/l) was measured via a capillary blood sample taken one-and-a-half minute after exercise cessation (Arkray, type LT-1730, Japan).
Cardiac medical history
MI characteristics were extracted from medical health records from the hospitals at which the patients were admitted. Specifically, clinical diagnosis of the MI, cardiac enzyme levels (troponin-I, creatine kinase (CK), creatinine (CREAT), aspartate transaminase (ASAT), and lactate dehydrogenase (LDH)), treatment strategy, and secondary events were identified and used for data analyses.
Echocardiography
Participants abstained from exercise 24 h before the echocardiography assessment. Two-dimensional Doppler and four-dimensional images were obtained by a single experienced cardiologist using an ultrasound system (Vivid E9, General Electric Healthcare, Norway) equipped with a M5-S and V4 probe. All measurements were performed according to the American Society of Echocardiography (ASE) guidelines 13 with the participant in the left lateral recumbent position. Images were taken at end-expiratory breath hold, carefully avoiding Valsalva manoeuvre. A continuous three-lead electrocardiogram registration was used to detect end-diastole time points (onset of QRS). Data were transferred to a workstation for offline analysis (EchoPac PC version 113, General Electric Healthcare, Norway). Data analysis of the echocardiographic images was performed by an independent, blinded expert.
Left ventricular systolic function
LV ejection fraction (LVEF) was calculated from the LV end-diastolic volume (LVEDV) and LV end-systolic volume (LVESV) using Simpson’s biplane method. Based on ASE guidelines, a LVEF below 52% was defined as an impaired LVEF.
13
Stroke volume was calculated by multiplying the time velocity integral and cross-sectional area of the LV outflow tract. Cardiac output was calculated by multiplying stroke volume with heart rate. Cardiac index was calculated by dividing cardiac output by body surface area. Body surface area was calculated using DuBois’ formula (equation 1).
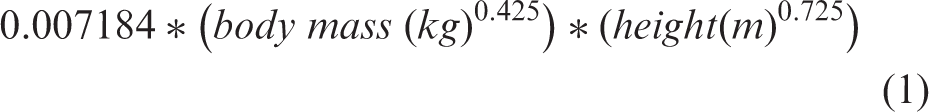
Pulsed-wave tissue Doppler imaging measurements of peak systolic annular tissue velocities were obtained at the septal and lateral mitral annulus from apical four-chamber images and an average of both sites is presented.
Left ventricular wall strain
Via the apical window, a 4D full volume R-wave six-beat gated dataset of the LV was acquired. Volume-rate was kept >30 Hz. The dataset was post-processed using the 4D automated LV quantification tool available in EchoPac to determine LV wall strain. From 4D images, endocardial border detection process was initialised by manual alignment of the apex and mitral valves in a long-axis view at both end-diastolic and end-systolic phase. The endocardial border was automatically generated throughout the cardiac cycle. The proposed contour was evaluated via short-axis cut-planes of the 3D volume at base, mid, and apex of the LV and cut-planes of the apical 4-, 3- and 2-chamber views. Only major deviations of the expected endocardial borders were operator corrected. Papillary muscles and major trabeculae were included in the LV cavity. The epicardial border was automatically generated by the software, which created a 3D region of interest of the LV wall. Speckle tracking was applied to determine global longitudinal, circumferential, area, and radial strain.
Left ventricular diastolic function
Diastolic function was assessed with LV-inflow pulsed-wave Doppler measurements at the mitral leaflet tips, including peak flow velocity of the early rapid filling wave (E-wave), peak flow velocity of the late filling wave due to atrial contraction (A-wave) and E/A ratio. Using pulsed-wave tissue Doppler measurements, the tissue velocity of the septal and lateral mitral annulus was registered. From these tracings, peak early mitral annular tissue velocity (e’-wave), and peak late mitral annular tissue velocity during atrial contraction (a’-wave) were measured. The ratio of E-wave and e’ (E/e’) was calculated.
Statistical analysis
Data is reported as mean ± standard deviation or median (interquartile range (IQR)). Categorical data were analysed using the Fisher’s exact test. Parameters were checked for normality using a Shapiro-Wilk test. Skewed variables were loge-transformed before analyses. Data that could not be transformed into Gaussian distribution were analysed using nonparametric tests. An independent Student’s
Results
Participant characteristics
Participants’ characteristics of the veteran athletes (ATH;
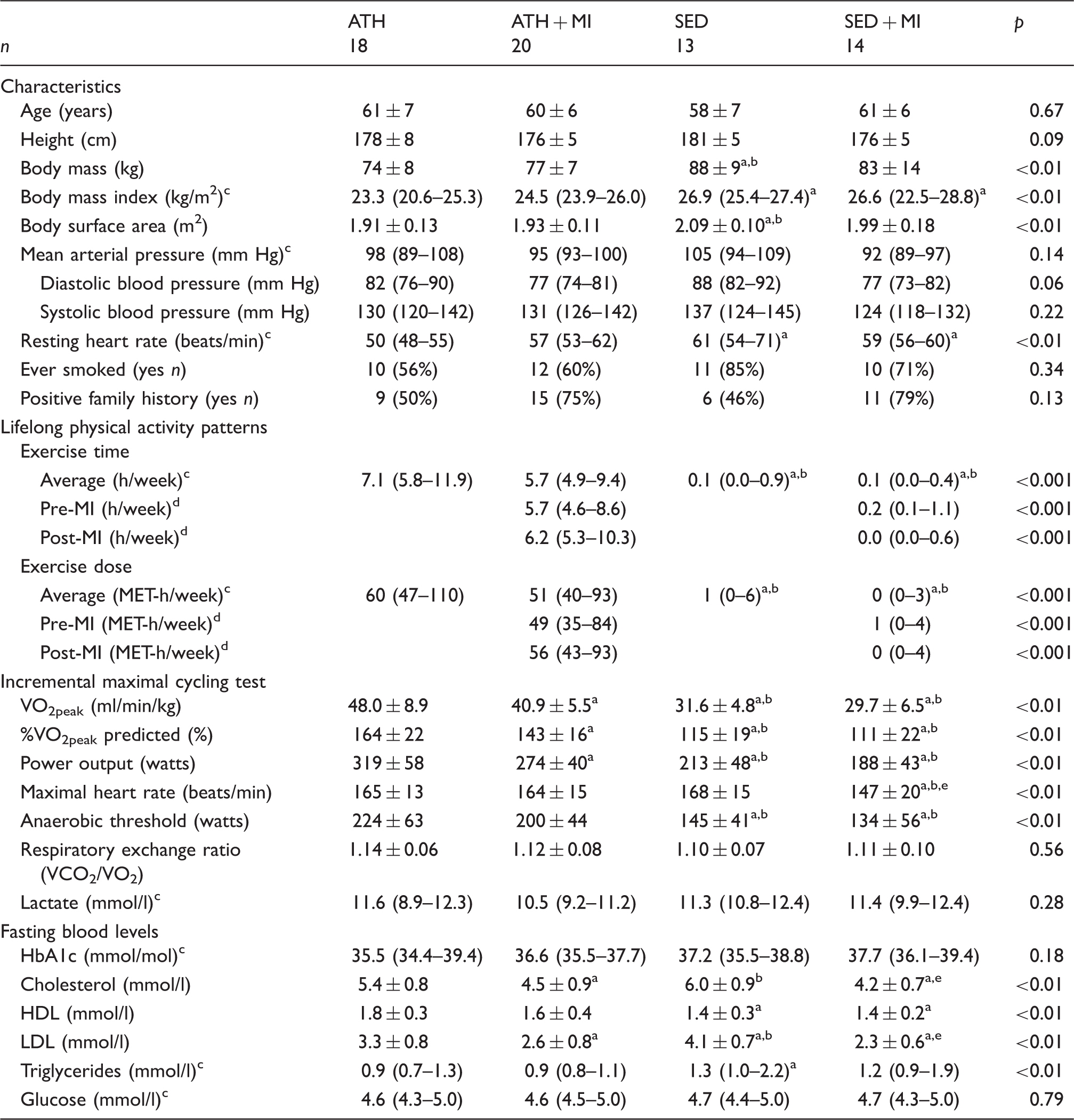
HbA1c: glycated haemoglobin; HDL: high-density lipoprotein; LDL: low-density lipoprotein: MET: metabolic equivalent of task.
Values of
Significantly different from ATH; bsignificantly different from ATH + MI; cKruskal-Wallis test; dMann-Whitney-U test; esignificantly different from SED.
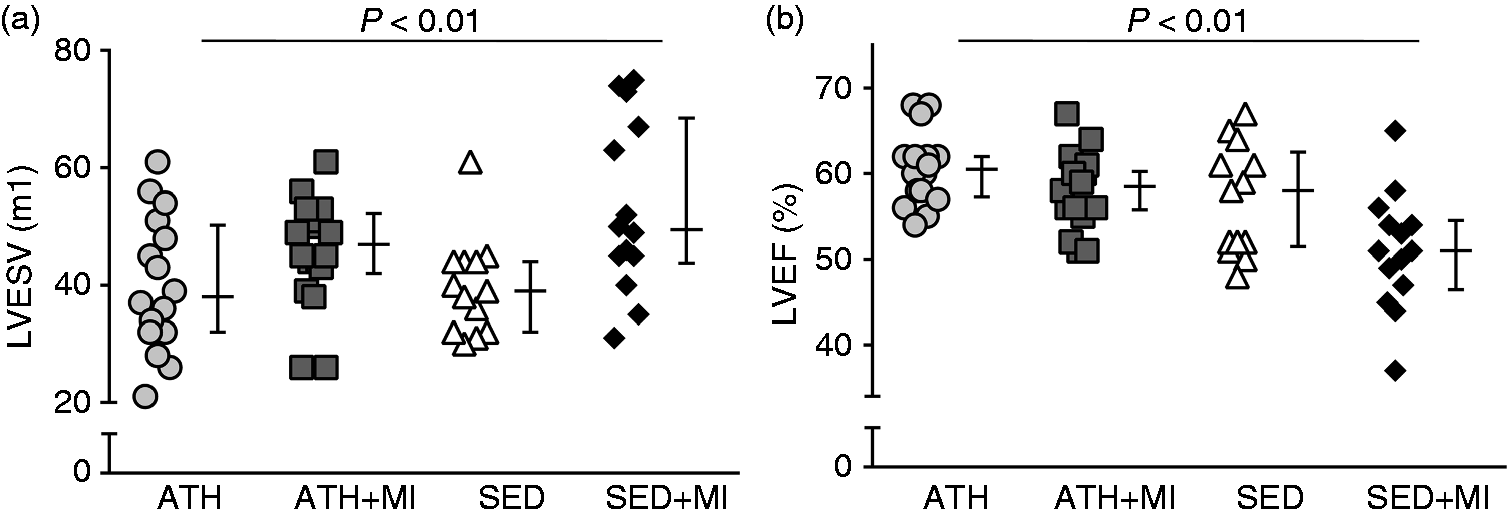
Individual and average values of (a) left ventricular (LV) end systolic volume (LVESV) and (b) left ventricular ejection fraction (LVEF) of the veteran athletes (ATH; circles), veteran post-MI athletes (ATH + MI; squares), sedentary controls (SED; triangles), and sedentary post-MI controls (SED + MI; diamonds). LVESV was significantly lower in ATH and SED groups compared to SED + MI group (
Medical history and medication usage
Cardiac medical history data of the veteran post-myocardial infarction (MI) athletes (ATH + MI,
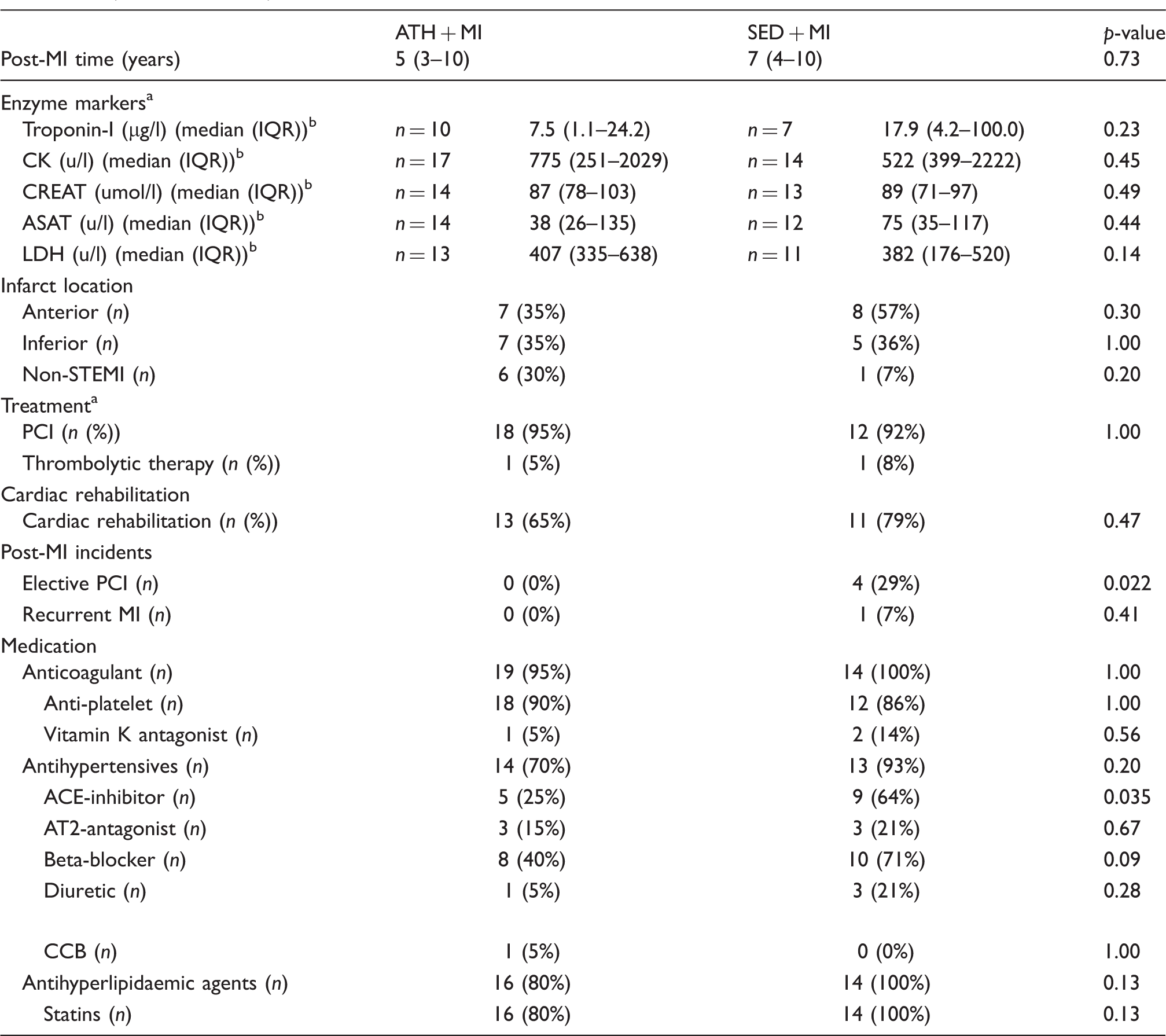
ACE: angiotensin-converting enzyme; ASAT: aspartate transaminase; AT: angiotensin; CCB: calcium channel blocker; CK: creatine kinase; CREAT: creatinine; IQR: interquartile range; LDH: lactate dehydrogenase; Non-STEMI: non-ST elevation myocardial infarction; PCI: percutaneous coronary intervention.
Based on a sub sample; hospital data were not available. Values of
Echocardiography
LV systolic function
Left ventricular systolic and diastolic function of the veteran athletes (ATH,
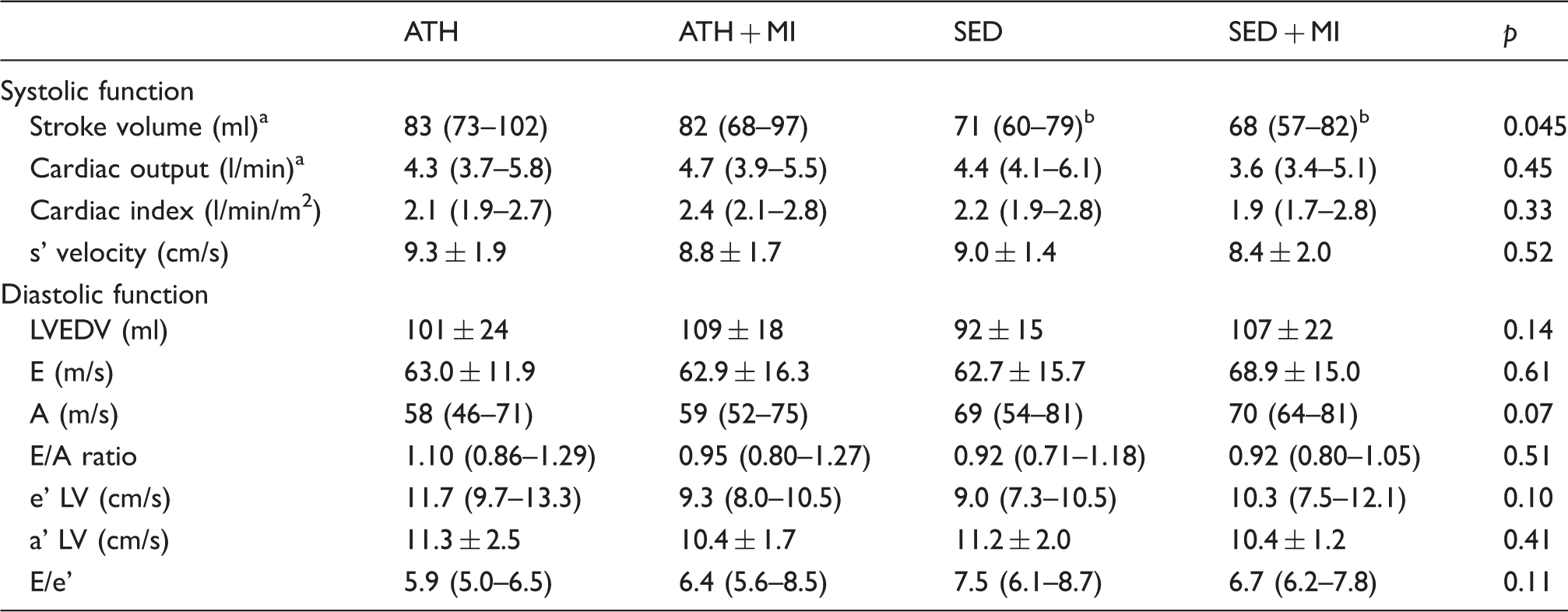
a’ LV: peak annular tissue velocity during late diastolic atrial contraction; A: peak flow velocity of the late filling wave at the mitral leaflet tips; e’ LV: peak annular tissue velocity during early filling; E: peak flow velocity of the early rapid filling wave at the mitral leaflet tips; E/e’: ratio of peak E velocity with e’; LVEDV: left ventricular end-diastolic volume; s’ velocity: peak systolic annular tissue velocity.
Values of
LV wall strain
LV longitudinal strain did not differ between ATH + MI (–13% (–18 to –10)), SED (–12% (–15 to –11)) and SED + MI group members (–11% (−15 to –6), Individual and average values of (a) left ventricular (LV) longitudinal strain, (b) circumferential strain, (c) area strain and (d) radial strain of the of the veteran athletes (ATH; circles), veteran post-MI athletes (ATH + MI; squares), sedentary controls (SED; triangles) and sedentary post-MI controls (SED + MI; diamonds). LV longitudinal strain did not differ between ATH + MI, SED and SED + MI groups, but LV longitudinal strain was superior (i.e. more negative strain) in ATH compared to SED + MI group. LV circumferential strain was superior in ATH, ATH + MI and SED groups compared to SED + MI group. LV area strain was superior in ATH and ATH + MI groups compared to SED + MI group, whereas LV area strain did not differ between SED and the other three groups. LV radial strain did not differ between ATH + MI, SED and SED + MI groups, but LV radial strain was superior in ATH compared to SED + MI group. Group averages are reported as median and interquartile range.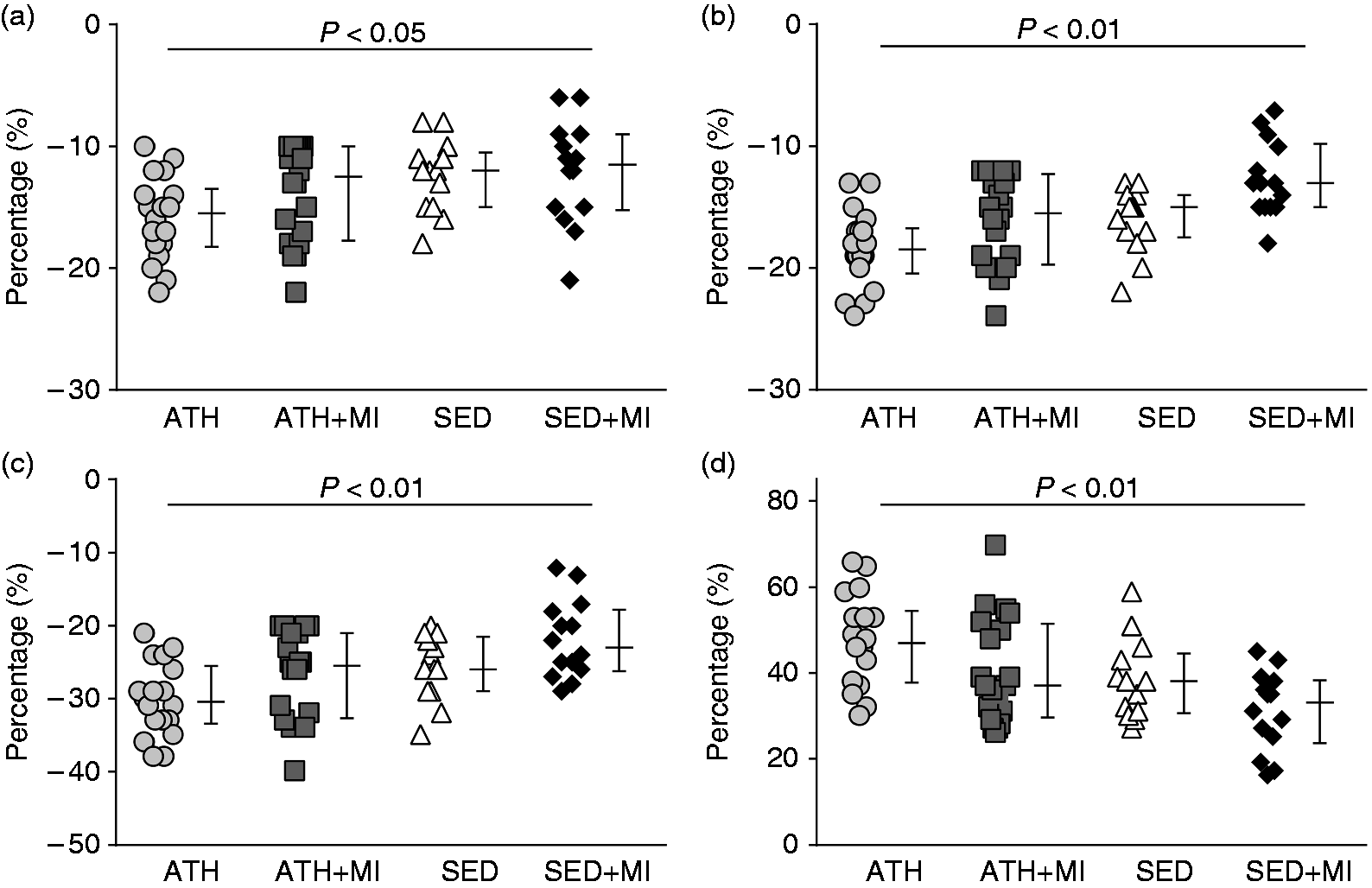
Left ventricular diastolic function
All diastolic function parameters (i.e. LVEDV, E-wave, A-wave, E/A ratio, e’ LV, a’ LV, and E/e’ ratio) did not differ across groups (Table 3).
Discussion
The major finding of this study is that ATH + MI group members had a better ejection fraction and a superior global LV wall strain compared to SED + MI group members. Ejection fraction and LV wall strain are important parameters for LV systolic function. 13 We found no differences in LV function between ATH and ATH + MI group members, whereas ejection fraction and circumferential strain differed between SED and SED + MI group members. These findings suggest that lifelong exercise training may protect against the deleterious effects of a MI and/or minimises the effects of pathological LV remodelling after a MI.
The magnitude of pathological LV remodelling is dependent on the severity of the MI, 14 clinical treatment (PCI), 15 medication use 16 and lifestyle changes following diagnosis. 16 We found no difference in cardiac enzyme levels, PCI treatment, infarct location, and medication (except ACE-inhibitors) between both post-MI groups, suggesting that MI size was comparable between ATH + MI and SED + MI groups. A potential explanation for the difference in ACE-inhibitors may relate to the physically active lifestyle of the ATH-MI group. Physical activity is related to a favourable blood pressure, 17 which may have enabled ATH-MI group members to reduce their medication. Interestingly, ATH + MI group members reported an increase in activity levels after the MI compared to before, whereas the SED + MI group members did not change their physical activity behaviour. These findings suggest that the ATH + MI group members and SED + MI group members did not differ in clinical characteristics, while their habitual exercise levels were significantly different.
Before the MI, ATH + MI group members were highly physically active (49 (35–84) MET-h/week), whereas SED + MI group members were inactive (1 (0–4) MET-h/week). Several studies support the hypothesis that exercise training induces preconditioning effects against ischaemia and reperfusion,18,19 which subsequently protects the myocardium against damage produced by ischaemia and reperfusion. 9 A reduction of the induced cardiac damage due to a MI will promote the healing process of the infarcted area. 9 Indeed, evidence from animal studies suggests that exercise training before a MI attenuates LV remodelling 9 and improves cardiac function 9 after MI. Findings from our study support this hypothesis as LV function (i.e. LVEF, global circumferential and area strain) was superior in ATH + MI group members compared to SED + MI group members. Our results are indicative that lifelong exercise training may improve infarct healing after MI.
An alternative explanation for the better LV systolic function in ATH + MI group members versus SED + MI group members may relate to their activity patterns after the MI. Most cardiovascular professional societies recommend post-MI patients to participate in a cardiac rehabilitation programme, and advise post-MI patients to remain physically active at a low-to-moderate endurance intensity level 16 to improve functional capacity and reduce (cardiovascular) mortality.20,21 An early start of cardiac rehabilitation and prolonged exercise training (>12 weeks) is associated with larger improvements in LV remodelling. 22 In the present study, ATH + MI group members continued and even increased their high-level physical activity patterns after MI, whereas SED + MI group members maintained their sedentary lifestyle. The VO2peak of our study population reinforces these observations; ATH + MI group members (40.9 ± 5.5 ml/min/kg) showed a substantially higher VO2peak uptake compared to SED + MI group members (29.8 ± 6.1 ml/min/kg). The physically active lifestyle after the MI may have contributed to the better LV systolic function in ATH + MI group members compared to SED + MI group members. In fact, these observations may represent optimal cardiac rehabilitation, as LV function of ATH + MI group members was not different from their non-MI peers.
In the current study, it is impossible to distinguish the independent effects of exercise training before and after the MI on LV function. To gain more information about post-infarction cardiac function and lifelong exercise training, we correlated the training of the different age periods with ejection fraction and the strain parameters. Overall, we observed that higher levels of physical activity were related to improved LV function, which is in line with the reported results of this study (Supplementary Material, Table 1). The combination of exercise training before and after MI may be superior to exercise training before or after MI only. One animal study suggests that the combination of exercise training before and after MI improves LV remodelling by reducing the inflammatory response and scar thinning process. 23 Another animal study demonstrated that the combination of exercise training before and after MI improved infarct healing and post-MI survival compared to no exercise training. 9 However, ameliorating effects on LV remodelling observed in mice that either exercised before or after MI were lost in mice that exercised before and after MI. 9 Absence of exercise benefits on LV remodelling in this combination group most likely relate to a very early start of post-MI exercise training accompanied with a high exercise intensity (∼7 km/day in the first week post-MI) in this particular study. 9 Indeed, there is evidence that vigorous post-MI exercise may cause further deterioration of the injured heart. 24 This negative effect seems to be dependent on the severity of the MI and timing of the exercise training. 25 Additional research is warranted to assess the relation between exercise before and after the MI in relation to LV remodelling in humans.
In contrast to LV systolic function, we did not observe statistical differences in diastolic function between ATH + MI and SED + MI group members. A potential explanation could relate to the fact that not all post-MI patients develop diastolic dysfunction after a MI.
26
Specific treatment to improve diastolic function following a MI is not available.
27
Potentially the long period between MI and study participation (Q50: six years (Q25: three to Q75: 10)), and adequate cardiac medication use may have contributed to the null findings of diastolic function between groups. Alternatively, aging has been associated with a progressive decline in diastolic function.28,29 Aging may lead to an impaired diastolic relaxation pattern28,29 and lifelong exercise training can only partially minimise the age-related decline.
28
Sub analysis of our results, revealed that indeed a higher age was associated with a significantly lower E/A ratio (
Clinical implications
In an event when exercise training ‘fails’ to prevent a MI, our data suggest that veteran athletes may restore and/or maintain their LV systolic function after a MI. Additional benefits are improved secondary prevention, since none of the ATH + MI group members had an elective PCI or recurrent MI. The information from the current study that exercise training improves LV remodelling after MI might be another reason to motivate sedentary post-MI patients or individuals at risk for cardiovascular disease to change their lifestyle and enjoy exercise training to improve cardiovascular health.
Limitations
Presence of recall bias regarding exercise history of the participants is a potential study limitation. To minimise this error, we did not elucidate our study hypothesis to the study participants. 30 Moreover, the time span of exercise history was similar between the three groups and it is likely that recall bias was similar across groups. This study was cross-sectional by design and is subject to the inherent limitations of that approach. It is likely that over the last 20 years, lifestyle habits have changed, and this might have influenced the risk for a MI (e.g. smoking or dietary habits). To avoid such concerns, a longitudinal study design is preferred, but such a study would take too much time for observations and tests. Ethical concerns would emerge during a longitudinal study design, because individuals clearly at risk for MI would receive preventative measures. These individuals may not endure a MI and will have no cardiac damage. Consequently, it would be impossible to study the protective effects of lifelong exercise training against pathological LV remodelling after the MI. Therefore, we used the cross-sectional approach, coupled with great effort to minimise bias. We could not retrieve information about other clinical markers (e.g. LVEF) than the reported cardiac enzyme markers, which may have limited the comparison of infarct size between post-MI groups. Although previous studies demonstrated that the cardiac enzyme markers reported in this study are related to infarct size,31–33 LVEF directly after the MI would have improved the comparison between post-MI groups. Unfortunately, these values could not be provided by the different hospitals of the patients that were included in the present study. Finally, it is important to keep in mind that of these results were generated from a relative small study population and future work needs to confirm our findings in a large sample size. Nonetheless, we believe that this study is a first step to confirm animal data that demonstrate that exercise may attenuate the deleterious effects of MI.
Conclusions
ATH + MI group members had better LV systolic function compared to SED + MI group members and similar LV systolic function compared to ATH group members. SED + MI group members had a lower LVEF and circumferential wall strain compared to SED group members. These findings suggest that lifelong exercise training may protect against the deleterious effects of a MI and/or minimises the effects of pathological LV remodelling after a MI in veteran athletes.
Footnotes
Author contribution
MM, TE, AD, MH contributed to the conception or design of the work. All authors contributed to the acquisition, analysis, and/or interpretation of data for the work. MM and TE drafted the manuscript. GS, AD, MH critically revised the manuscript. All gave final approval and agreed to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The authors MM, GS, AD and MH declare that they have no conflicts of interest that are directly relevant to the content of this article. TE was financially supported by a European Commission Horizon 2020 grant (Marie Sklodowska-Curie Fellowship 655502).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: this work was supported by a European Commission Horizon 2020 grant (Marie Sklodowska-Curie Fellowship 655502 to TE).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
