Abstract
Treprostinil is a prostacyclin approved for the treatment of pulmonary arterial hypertension. Commercial data sets indicate that approximately 20–25% of patients are prescribed a higher dose than the maximum recommended dosage of nine breaths per treatment session (bps) (54 μg), four times a day (QID) and numerous studies have demonstrated the safety of doses >9 bps QID. This phase 4, retrospective analysis of specialty pharmacy records assessed the effects of inhaled treprostinil at doses >9 bps QID. Patients receiving inhaled treprostinil between September 2009 and June 2018 were included, and a random sampling of 5000 patients was selected for further analysis. Subjects were grouped based on the highest dose reached for ≥2 months within a rolling six-month window and were followed for up to three years. Of the total of 5000 patients analyzed, 28.5% received >9 bps QID. Survival rates were significantly higher in the >9 bps QID dosing group for years one, two, and three (
Introduction
Treprostinil for inhalation (Tyvaso) is a prostacyclin mimetic approved for the treatment of pulmonary arterial hypertension (PAH), a rare and fatal disease characterized by progressive elevation in pulmonary artery pressure and pulmonary vascular resistance, ultimately leading to right ventricle failure. 1 –5
Treatment with prostacyclins, such as inhaled treprostinil, can improve patients’ symptoms and exercise tolerance. 4 Treprostinil acts by triggering direct vasodilation of the pulmonary and systemic arterial vascular beds. It also inhibits platelet aggregation and has in-vitro antiproliferative properties. 6 , 7 Treprostinil is available via subcutaneous (SC), intravenous (IV), inhaled, and oral formulations. The parenteral versions of the drug are administered in increasing doses over time with no defined upper limit. In contrast, the recommended target dose for inhaled treprostinil is nine breaths per treatment session (bps) (54 μg), four times a day (QID). 1 In the TRIUMPH study, 52% of patients receiving treprostinil improved on a 6-min walking test by 20 m or greater, with 31% improving greater than 50 m. 4 In the long term, open-label extension for the TRIUMPH study which included 206 patients, 42% achieved a dose of 12 bps QID (72 μg), and the adverse events (AEs) were similar to those observed in a 12-week placebo-controlled trial. The duration and magnitude of reduction in pulmonary vascular resistance have been shown to be dose dependent. 8
However, the efficacy of inhaled treprostinil at doses exceeding 9 bps QID has not been well characterized in the literature. In this retrospective analysis, we assess the effects of inhaled treprostinil at doses >9 bps QID on survival, drug persistence, and time to transition to SC/IV therapy using records obtained from the specialty pharmacy distributors.
Methods
This study was a phase 4, retrospective, real-world analysis that utilized anonymized, specialty pharmacy data to assess the effects of inhaled treprostinil at doses >9 bps (54 µg) QID. Patients were assigned a unique ID number by the specialty pharmacy which was maintained even if they switched between different treprostinil products. As a result, it was possible to calculate the time from treprostinil discontinuation to the transition to SC/IV treprostinil therapy using specialty pharmacy shipment records.
Patients who received inhaled treprostinil between September 2009 and June 2018 and met the study’s inclusion criteria were included in the study. From the overall population, a random sample of 5000 patients was selected for further analysis. Subjects were grouped by dose (i.e. ≤9 vs. >9 bps QID) based on the highest dose reached for ≥2 months in a rolling six-month timeframe. Patients who did not have a single highest dose for ≥2 months in any six-month rolling window were assigned the dose corresponding to their median dose achieved through their entire course of therapy. As a result, some patients had non-whole numbers for doses, a byproduct of the statistical median.
Patients were excluded from the analysis if there was omitted data for dose or shipment date, if the patient transferred pharmacies, if the patient was prescribed treprostinil at a frequency other than QID, or if the patient enrolled in a clinical study.
The primary objective of the study was to assess the survival rate of patients on doses of >9 bps (54 μg) QID after one, two, and three years. For this study, survival was defined as time on therapy prior to discontinuation of inhaled treprostinil due to death.
The secondary objectives were (1) to assess the efficacy of inhaled treprostinil at doses >9 bps QID, as measured by time to transition to SC/IV treprostinil therapy as a surrogate for disease progression and (2) to assess the drug persistence of inhaled treprostinil at doses >9 bps QID, as a surrogate for tolerability. Time to transition to SC/IV therapy was defined as the absolute length of time between the first treprostinil shipment and the first SC/IV shipment among patients who initiated SC/IV treatment 60 days within discontinuing inhaled treprostinil. Time to transition is a commonly used indicator of disease worsening and is robustly captured within the specialty pharmacy dataset. 9 –12
The exploratory objectives were to assess the effects of doses ≥12 bps (72 μg) and ≥15 bps (90 μg) QID on survival, time to transition to SC/IV treprostinil therapy, and drug persistence.
For continuous variables, descriptive statistics included the number of observations, mean, standard deviation, median, minimum, and maximum, if appropriate. For categorical variables, descriptive statistics included the frequency and percentage, if appropriate. Survival rate and tolerability of drug persistence at years one, two, and three were calculated and summarized. Kaplan-Meier curves and log-rank tests, hazard ratios (HRs), and a chi-squared test were used to compare the >9 bps vs. ≤9 bps dose groups. The time to transition to SC/IV treprostinil therapy was summarized. Mann-Whitley U tests were used to compare the >9 bps vs. ≥9 bps dose groups. Survival rate, time to transition to SC/IV treprostinil therapy, and drug persistence rates at years one, two, and three were also summarized and compared, as described above for the ≥12 bps vs. <12 bps and ≥15 bps vs. 9 bps dose groups.
The authors did not explore alternative analytic approaches to bias study outcomes. Preliminary results have been presented previously. 13 This study was supported by United Therapeutics Corporation. As no patient or provider contact was made and patient information was de-identified, institutional review board approval was not required.
Results
Patient demographics
Overall, 6709 patients met all the study eligibility criteria. From this group of eligible patients, a random sample of 5000 patients was selected for further analysis using simple random sampling with equal probability. Table 1 displays the number of patients at each inhaled treprostinil dose after applying the pre-specified algorithm described in the methods.
Treprostinil dosing in the analysis population.

Of the 5000 patients included in the analysis, the majority (71.5%; 3577/5000) were taking treprostinil at ≤9 bps QID, 28.5% (1423/5000) were taking >9 bps QID, 25.5% (1276/5000) were taking ≥12 bps QID, and 6.1% (303/5000) were taking ≥15 bps QID (see Fig. 1). Demographic information such as age, sex, ethnicity, etc. are not included in the specialty pharmacy dataset utilized in this real-world analysis.
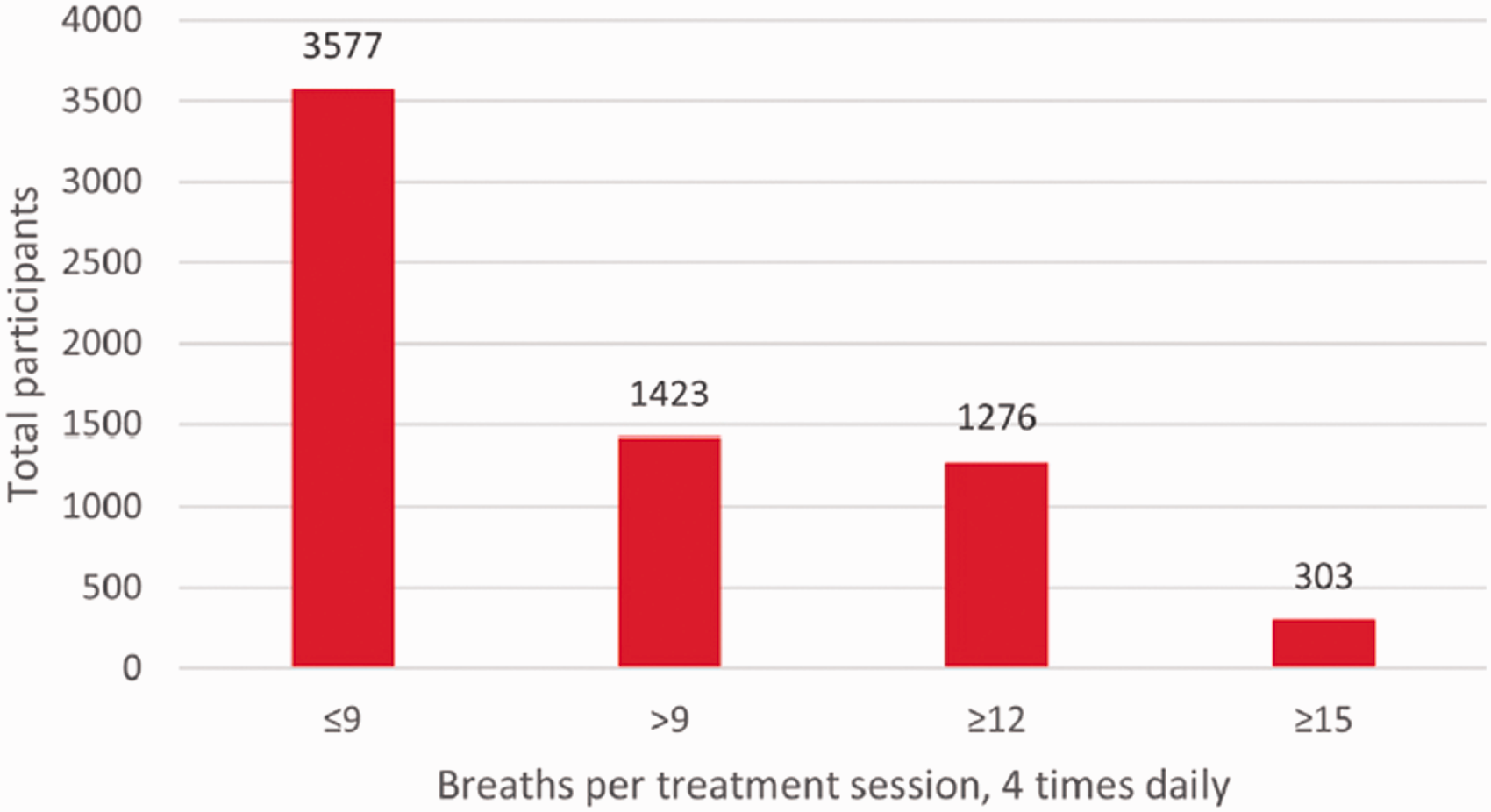
Number of patients at each inhaled treprostinil dose after applying the pre-specified algorithm described in the methods.
Survival
Survival rates at years one, two, and three for treprostinil doses are presented in Table 2 and Fig. 2. At all three timepoints, survival rates were significantly higher in the >9 bps QID dosing groups (
Survival rate.
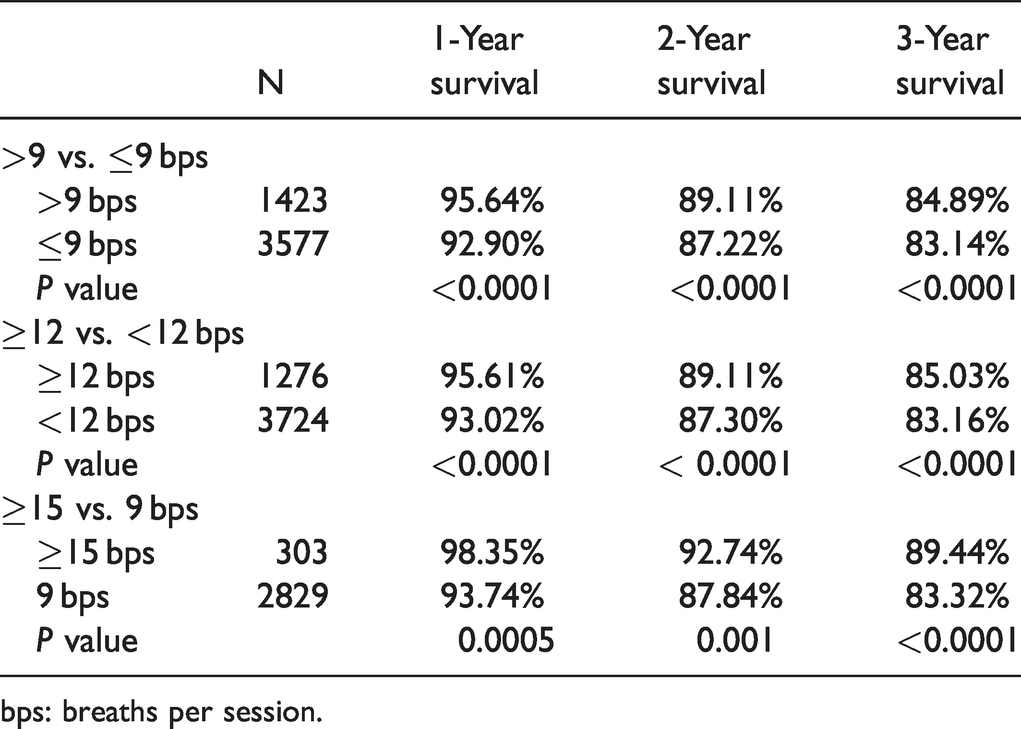
bps: breaths per session.
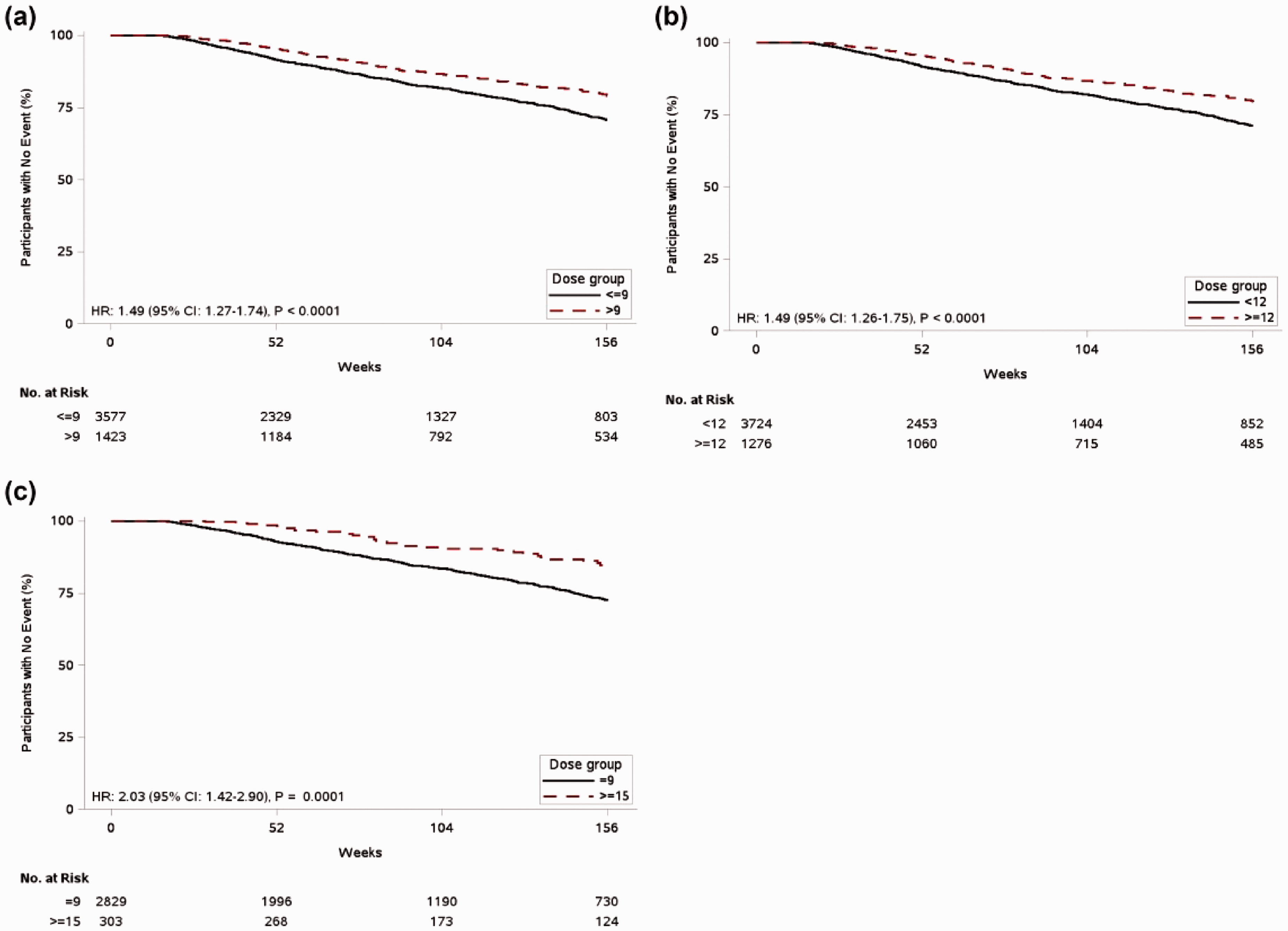
Survival rates at years one, two, and three for treprostinil doses A) >9 vs ≥9 bps, B) ≥12 vs. <12 bps, and C) ≥15 bps vs. 9 bps.
The HRs for survival were 1.49 for >9 bps vs. ≤9 bps (95% confidence interval (CI) 1.27 to 1.74;
Time to transition to SC/IV treprostinil
Transitioning from inhaled treprostinil to SC/IV therapy is a surrogate measure for disease progression. Of the 5000 patients included in this analysis, a total of 136 transitioned from inhaled treprostinil to SC/IV therapy. The time to transition to SC/IV treprostinil was significantly longer for those taking treprostinil at doses >9 bps compared to doses ≤9 bps (17.5 vs. 9.5 months,
Time to transition to SC/IV treprostinil.
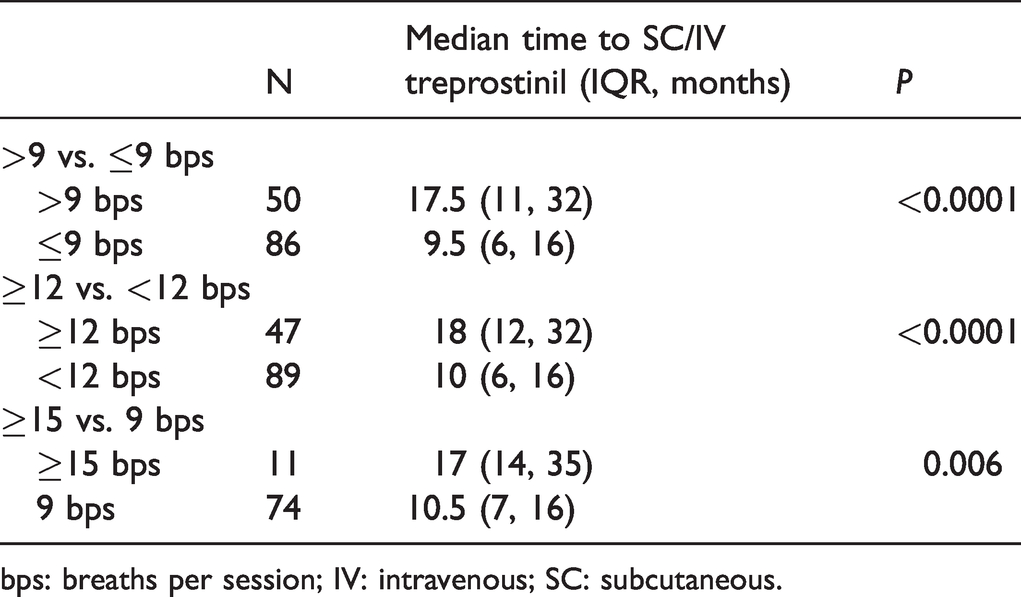
bps: breaths per session; IV: intravenous; SC: subcutaneous.
Safety evaluation
The secondary objective of this study was to assess the drug persistence of inhaled treprostinil at doses >9 bps QID, as a surrogate for safety and tolerability. The secondary safety/tolerability endpoint of the drug persistence rate at years one, two, and three was calculated and summarized for the various dose comparisons in Table 4.
Drug persistence rate.
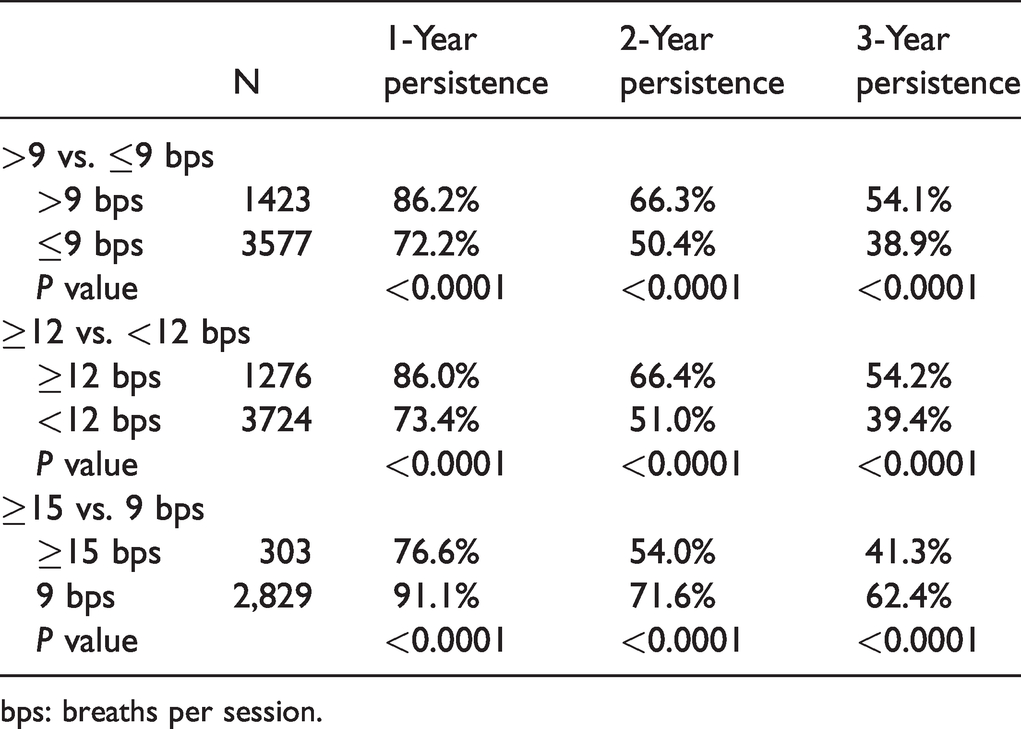
bps: breaths per session.
Drug persistence was significantly higher for those taking >9 bps at years 1, 2, and 3 (
The HRs for drug persistence were 1.76 for >9 vs. ≤9 bps (95% CI 1.61 to 1.92,
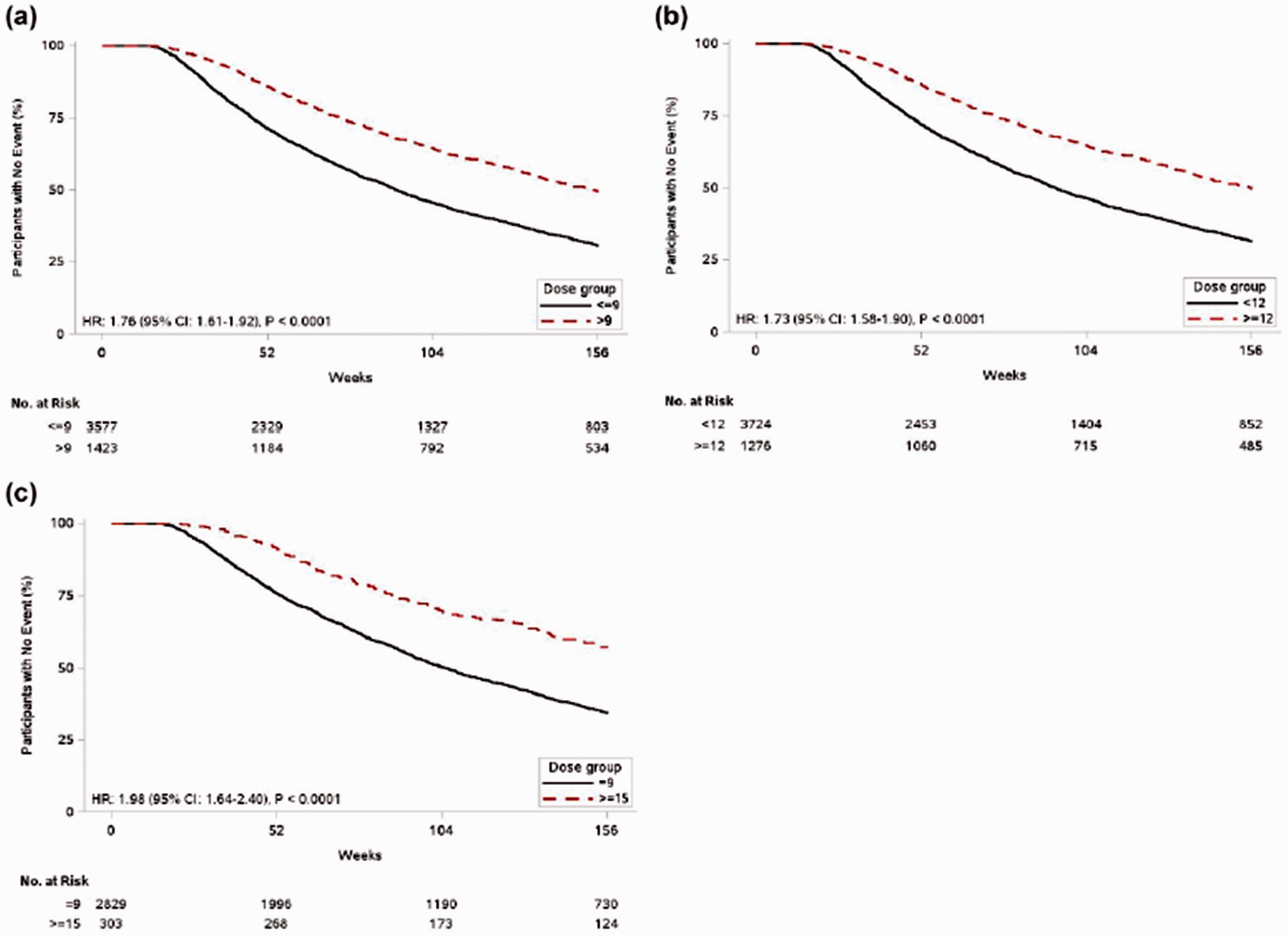
Drug persistence rates at years one, two, and three for treprostinil doses (a) >9 vs. ≥9 bps, (b) ≥12 vs. <12 bps, and (c) ≥15 bps vs. 9 bps.
Discussion
This study was conducted to assess the efficacy of treprostinil at doses >9 bps QID as measured by survival rate and time to transition to SC/IV treprostinil. We also assessed the tolerability of treprostinil at doses >9 bps as measured by drug persistence. Using real-world data, this analysis generated real-world evidence to support the hypothesis that higher doses of treprostinil are associated with improved survival, delayed time to transition to SC/IV therapy, which may be a sign of disease worsening, and increased drug persistence.
Commercial datasets indicate that approximately 20–25% of patients are prescribed a dose that is higher than the recommended 9 bps, and studies have demonstrated the safety of doses exceeding 9 bps QID. 4 ,14– 18 In the 5000 patients randomly selected for analysis, those receiving higher doses of inhaled treprostinil had significantly higher rates of drug persistence and survival over three years. The improved persistence rates associated with higher doses of inhaled treprostinil may act as a surrogate for reduced disease progression. This finding was supported by our analysis of time to parenteral treprostinil therapy, which was approximately twice as long in patients at higher doses. While previous findings support the safety of higher doses, the data presented here suggest that increased efficacy may also be evident. Further analysis of drug discontinuations and prospective controlled studies of higher doses are warranted to confirm these findings.
Death occurred less frequently among patients receiving higher doses of treprostinil. In all three comparison groups, patients receiving higher doses of treprostinil had a statistically significant greater one-, two-, and three-year survival and overall lower risk of death. Additionally, in all three dose comparison groups (>9 vs. ≤9 bps, ≥12 vs. <12 bps, ≥15 vs. 9 bps), patients receiving higher doses of treprostinil had a statistically significantly greater one-, two-, and three-year drug persistence, as well as a statistically lower overall risk of discontinuing therapy. Although clinical parameters such as 6-min walk distance (6MWD), functional class, and biomarker data were not available in this analysis, these findings suggest that higher doses of inhaled treprostinil are associated with maintaining clinical stability. The delayed time to SC/IV treprostinil transition further supports this concept.
Our findings are consistent with studies of other treprostinil formulations that demonstrated increased clinical benefit associated with increasing doses. In a pooled analysis of oral and SC/IV treprostinil, Ramani et al. found that higher doses were associated with significantly delayed time to all-cause and pulmonary hypertension-related hospitalization, as well as improvements in 6MWD. 19 Other studies 20 , 21 of treprostinil have demonstrated that higher doses of SC/IV treprostinil are associated with increased survival, but to our knowledge, this present analysis is the first published finding of a dose–response relationship for the inhaled formulation of treprostinil. 21 , 22 Our findings are consistent with a study by Tonelli et al. that demonstrated that inhaled treprostinil had beneficial effects on risk status, which was prognostic for survival. The median dose of inhaled treprostinil at their follow-up timepoints was 9 bps; thus, half of their cohort was receiving a dose of ≥9 bps. 23
These results complement the existing literature on the safety and tolerability of inhaled treprostinil at doses greater than eight breaths per session. In 2012, a retrospective review of patients at a large PAH center receiving doses exceeding 9 bps QID was completed, in which 20 patients were not meeting their treatment goals and were instructed to increase their dose to a maximum of 18 bps QID. 24 Patients were concomitantly taking PDE5I monotherapy (n = 3) or PDE5I with an ERA (n = 17). The median maximum dose reached was 12 bps QID. Despite the higher doses evaluated in this study, the AEs encountered were similar to those found in TRIUMPH 1. 4
The safety and tolerability of >9 bps QID were also assessed in a retrospective cohort study of pulmonary hypertension (PH) patients classified by the World Health Organization (WHO) as being in groups 1–5 (n = 80). 17 Patients were given increasing daily doses until they reached 12 bps QID. A total of 52% of patients had group 1 PAH and 32% had group 3 PH secondary to lung disease. The maximum dose of 12 bps QID was achieved by 97.5% of patients. Cough (38.8%) and headache (28.8%) were the most common AEs at baseline, and these decreased to 17.9% and 12.8%, respectively, at follow-up visit 2 (20.3 months). The authors concluded that inhaled treprostinil was well tolerated in their cohort of patients.
The most recent assessment was the INCREASE study, a phase 3, randomized, placebo-controlled, 16-week trial in patients with PH associated with interstitial lung disease. The study met its primary endpoint of change in the 6-minute walk test (6MWT), as well as secondary endpoints including changes in N-terminal pro b-type natriuretic peptide (NT-proBNP) and time to clinical worsening. 16 In a pre-specified subgroup analysis of dose, a total of 78 patients received >9 bps of treprostinil QID and 94 received placebo. Those taking 7–9 bps QID had a 6MWD improvement of 17.7 m compared to 33.7 m for those taking 10–12 bps QID, suggesting a dose–response relationship in patients with PH associated with interstitial lung disease. 18
This analysis is limited by a lack of blinding, placebo control, and the retrospective nature. While treprostinil is approved for the treatment of WHO group 1 PAH and more recently WHO group 3 PH, we cannot draw conclusions specific to PH subtypes due to uncertainties about the makeup of our study cohort. The specialty pharmacy dataset does not contain the clinical parameters necessary for confirming a patient’s diagnosis. Additionally, concomitant medications and background PAH therapies are also not present in the dataset. As a result, differences in clinical characteristics between the different dose groups being studied cannot be tested. Also, it is possible that clinical differences between the dose groups led to bias. Lastly, specialty pharmacy data does not include clinical variables such as 6MWD or functional class so controlling for bias was not possible (e.g. using propensity score matching), and we were unable to confirm improvement in these clinical parameters to confirm our positive findings related to therapy persistence, survival, and delayed time to parenteral therapy. These clinical parameters would have enhanced our analysis, but unfortunately, they are not a part of the specialty pharmacy data used in this analysis.
Lastly, the definition of survival utilized for this analysis has limitations. Because deaths are only recorded for patients who discontinue treprostinil due to death, patients who discontinue treprostinil for other reasons (e.g. tolerability, drug cost) and experience death later on are excluded from meeting this endpoint. However, there is no evidence in the literature to suggest that there is a bias toward patient mortality after discontinuing treprostinil at any specific dose. Despite these limitations, the results suggest that patients may benefit clinically from higher doses of inhaled treprostinil in the context of drug tolerability and adverse effect management.
Conclusions
In this analysis, patients receiving higher doses of inhaled treprostinil had significantly higher rates of drug persistence and survival over three years. Higher doses were also associated with a significantly longer time to parenteral therapy, which is an indicator of disease worsening. While previous findings support the tolerability and safety of higher doses of inhaled treprostinil, the present data suggest highly relevant clinical benefits may also be evident. These data suggest that the clinical implications of the development of a more convenient way to deliver higher inhaled doses (e.g. dry powder inhalers) would allow for easier titration and treatment of patients who do not respond to the current maximum dose recommendations, without having to switch them to SC or IV administration. The data suggest that higher doses are more efficacious and raise the question of whether it might be possible to get further benefit by increasing the amount of drug which can be administered in an inhaled preparation.
Footnotes
Conflict of interest
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: SS has received grants from Reata Pharmaceuticals, United Therapeutics, Lung RX, Gossamer, and Liquidia. SM is a paid consultant and speaker for United Therapeutics and Bayer. RRJ has received grants from and serves on advisory boards and speaker bureaus for United Therapeutics, Bayer, and Johnson & Johnson. ES, MB, YR, and ACN are employees of United Therapeutics.
Acknowledgements
The authors thank Dorothy Keine, PhD of 3Prime Medical Writing, LLC for providing medical writing support, compensated by United Therapeutics Corporation.
Authors’ contribution
SS, SM, RJ, ES, MB, and ACN made substantial contributions to the conception and design of the work. Data were analyzed by SS, SM, RJ, ES, MB, and ACN. DL was responsible for the statistical analysis. SS, SM, and RJ contributed to the interpretation of the data. SS, SM, RJ, and ES contributed to drafting and revising of the article. ES, MB, and DL have accessed and verified the data.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by United Therapeutics Corporation.
