Abstract
A late preterm infant had pulmonary hypertension caused by a variety of mechanisms leading to complex management. This child had complete atrioventricular septal defect associated with mild left ventricular hypoplasia and Down syndrome diagnosed prenatally. The mother had been treated by antiretroviral HIV treatment during pregnancy. Aortic coarctation was diagnosed and rapidly repaired. After surgery, he required noninvasive ventilation for persisting elevated PCO2. Pulmonary CT scan showed normal bronchial tree, lung parenchymal abnormalities with mosaic aspect and hyperlucent zones, and indirect signs of lung hypoplasia with peripheral microbubbles. During follow-up, severe pulmonary hypertension was diagnosed on echocardiography without recoarctation, significant intracardiac shunting or diastolic dysfunction. The patient died after four months unable to be weaned from noninvasive ventilation. Post mortem lung biopsy showed abnormally muscularized arterioles with intimal fibrosis and pulmonary immaturity. Gentetic screening identified a BMPR-2 mutation. This patient illustrates the multifactorial origin of pulmonary hypertension in the neonatal period. The respective contribution of left-to-right shunt, post-capillary obstruction, and abnormally elevated pulmonary vascular resistances led to perform right heart catheterization to exclude excessive shunting and restrictive physiology of the left heart. Subjects with Down syndrome are also highly susceptible to decreased lung vascular and alveolar growth, which may increase the risk for pulmonary hypertension and lung hypoplasia. This case highlights two issues. The first one is that right heart catheterization should be discussed in neonates with unexplained pulmonary hypertension and the second is to extend indications of genetic testing for pulmonary hypertension genes in neonates who have unusual course of neonatal pulmonary hypertension, particularly in the setting of associated congenital heart disease (CHD).
Keywords
Introduction
It is not exceptional that multiple factors contribute to the development of pulmonary hypertension (PH) in children. We report a case of a late preterm infant with PH caused or aggravated by a variety of mechanisms leading to inextricable decision making.
Case report
A late preterm male neonate (35 weeks, birth weight 2.1 (fifth percentile)) with prenatal diagnosis of complete atrioventricular septal defect (AVSD) associated with mild left ventricular hypoplasia and Down syndrome (DS) was admitted at birth. The mother had been treated by antiretroviral HIV treatment (ritonavir, darunavir, and emtricitabine/tenofovir) during pregnancy. The child was started on zidovudine for one month. His HIV-PCR was negative. Complete AVSD with mild left heart hypoplasia and small ventricular septal defect (VSD) was confirmed. Aortic coarctation was rapidly diagnosed and was repaired through left thoracotomy. After surgery, the child required noninvasive ventilation for persisting elevated PCO2. Pulmonary CT scan showed normal bronchial tree, lung parenchymal abnormalities located in the left upper lobe and right inferior lobe with mosaic aspect of the lung and hyperlucent zones, and indirect signs of lung hypoplasia with peripheral microbubbles. During follow-up, severe PH was diagnosed on transthoracic echocardiography with an estimated systolic pulmonary arterial pressure of 100 mmHg. Right heart catheterization confirmed severe PH (with pulmonary artery pressure 72/25/44 mmHg and aortic pressure 70/39/50 mmHg) without recoarctation, significant intracardiac shunting nor diastolic dysfunction. Right atrial pressure was 10 mmHg, as well as left atrial pressure (large ostium primum). Based on the Fick Principle, indexed pulmonary vascular resistance (PVRi) was 7.6 WU × m2, QP/QS: 0.7, and cardiac index: 6 l/min/m2. He was treated by sildenafil two months after diagnosis without significant improvement. The patient died after four months unable to be weaned from noninvasive ventilation. Post mortem lung biopsy showed abnormally muscularized arterioles with intimal fibrosis (Fig. 1). The intimal lesions were highly cellularized indicating an active process. Venous vessels and lymphatic were normal as well as vessel organization. Alveoli wall were thickened as observed in pulmonary immaturity. As current practice in atypical cases with lung development anomalies, a PH genetic panel was performed in our dedicated PH lab and identified a heterozygote BMPR-2 mutation (c.1969C>T).
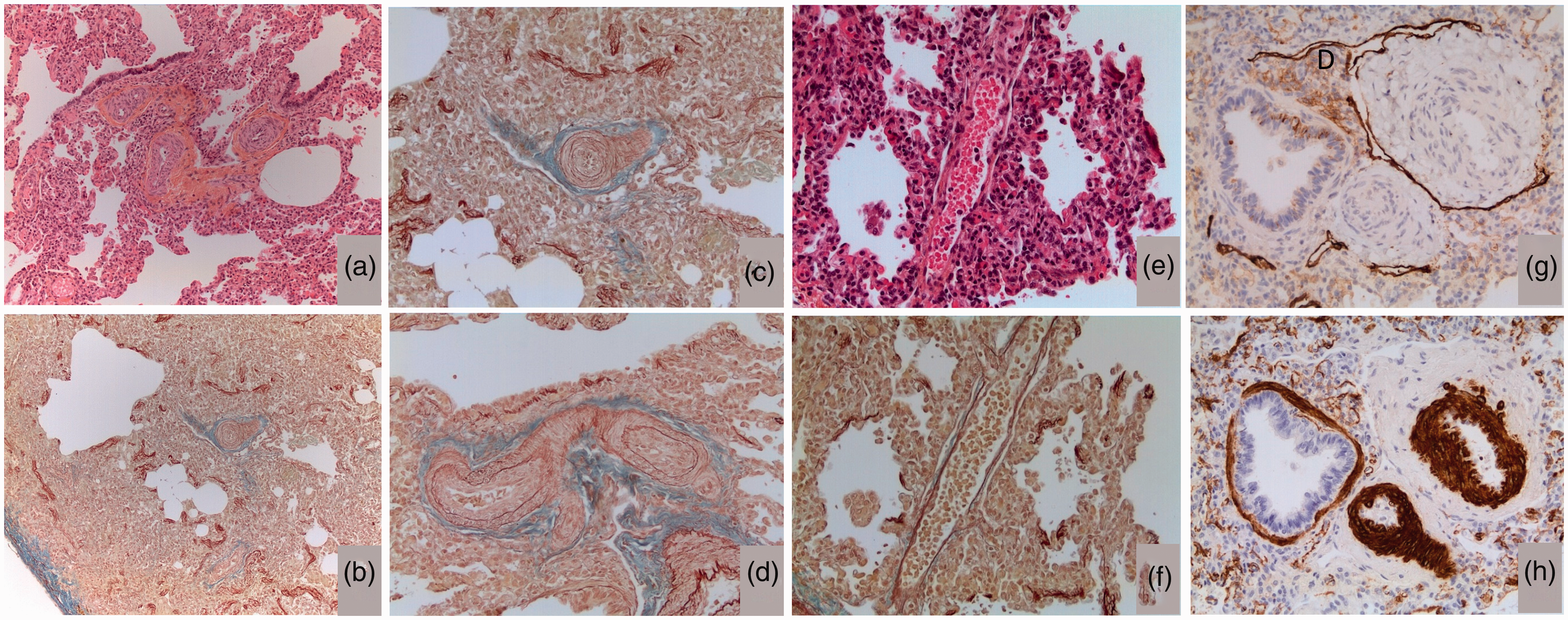
Post mortem lung biopsy showing severe pulmonary arterial lesions and thickening of alveolar walls but without anomalies of pulmonary veins nor bronchial circulation. (a) and (b): HES and orcein staining showing ×10 showing few pulmonary alveoli and thick alveolar wall indicating immature lung; (c) and (d): Orcein staining ×10 and ×25 showing thick-walled intra-acinar pulmonary arteries with severe intimal proliferation and occluded lumen. Note that the surrounding parenchyma exhibits thick-walled and poor ventilated alveoli; (e) and (f): HES and orcein staining ×40 showing normal pulmonary veins without intimal fibrosis nor muscularized wall; (g) and (h): D2-40 immunostaining and actin ×25 showing that vessels surrounding occluded pulmonary arteries are lymphatics and not veins, nor bronchial arteries.
Discussion
This patient illustrates the multifactorial origin of PH in the neonatal period. Indeed, the AVSD is an obvious cause of PH but the VSD was small and should have been restrictive if pulmonary resistances had normally dropped after birth. The role of the atrial shunting aggravated by the small size of the left ventricle was a possible mechanism for severe neonatal PH. 1 The association of elevated PVR with left heart obstructive disease is also known in neonates. 1 The respective contribution of left-to-right shunt, abnormally elevated PVR, and post-capillary obstruction led to perform right heart catheterization in this infant to exclude excessive shunting and restrictive physiology of the left heart.
Newborn infants with DS are at high risk of developing severe persistent PH of the newborn. 2 They also have more aggressive pulmonary vascular disease secondary to congenital heart defects compared to children without DS. 2 While the mechanisms that increase PH susceptibility of infants with DS are incompletely understood, lung hypoplasia with decreased alveolarization, peripheral lung cysts, and persistence of the double-capillary network has been described. 3 In this patient, we showed alveolar simplification (Fig. 1a and b) but we did not see double capillary network, prominent bronchial artery circulation nor intrapulmonary connections in form of bronchopulmonary anastomoses. Recent work has shown that three antiangiogenic genes are present on chromosome 21, and are each overexpressed in human fetal and infant lung tissue. 4 Experimentally, early disruption of angiogenic signaling decreases vascular growth and increases the risk for PH, and also impairs distal airspace growth. 5 Laboratory and clinical findings suggest that subjects with DS are highly susceptible to decreased lung vascular and alveolar growth, which may increase the risk for PH and lung hypoplasia as in our patient. The current WSPH Paediatric Task Force agreed that the phenotype of DS-related PH is variable and does not universally fit into a single classification group, but that children with DS will be classified as group 3 (“developmental lung disease”) in the absence of CHD. In our patient, chronic ventilation was necessary but PH did not improve while normal PCO2 and normal oxygenation were normalized.
Genetic factors underlying persistent PH of newborns in a cohort of Chinese neonates had been recently described. Nine patients were identified as harboring genetic variants, including three with pathogenic/likely pathogenic variants in TBX4 and BMPR-2 and six with variants of unknown significance in BMPR-2, SMAD9, TGFB1, KCNA5 and TRPC6. 6 Three single nucleotide polymorphisms (SNPs) in CPS1 and one SNP in NOTCH3 were significantly associated with persistent PH of the newborn. CPS1 and SMAD9 were identified as risk genes for persistent PH of the newborn. 6 Currently, no BMPR-2 mutation associated to DS had been reported in this setting.
In addition, it is known that perinatally HIV-exposed but uninfected children had cardiac effect after birth. 7 Zidovudine was also suggested to affect the structural development of the human fetal lung in vitro. Disentangling these different mechanisms and identifying the most important contributor is challenging, indeed impossible. Hitherto, however, this case highlights two issues to our opinion. The first one is that right heart catheterization should be discussed in all neonatal cases when non-invasive informations cannot confirm the role of the congenital heart defect in the persistence of high PVR during the neonatal period or when these informations are ambiguous. The second issue is to extend indications of genetic testing for mutations/variants of the known PH genes in neonates who have unusual course of neonatal PH, particularly in the setting of associated congenital heart disease. Here, we were surprised to find a mutation in the BMPR-2 gene as they have rarely been observed in congenital heart diseases with PH, 8 while TBX4 and SOX17 mutations are increasingly reported. 9 Rare variants in SOX17 are associated with pulmonary arterial hypertension with congenital heart disease. 10 This unusual case is an example of multifactorial origin of neonatal PH or an illustration of the multiple hits that could lead to irreversible pulmonary vascular disease or neonatal maladaptation.
Footnotes
Conflict of interest
The author(s) declare that there is no conflict of interest.
Author contributions
F.B. collected the data, drafted the manuscript, and revised the manuscript. S.M.-M., M.L., and D.B. critically reviewed and revised the manuscript. All authors approved the final manuscript as submitted and agree to be accountable for all aspects of the work.
Acknowledgements
We would like to thank the patient’s family for their consent to publish this report.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
