Abstract
Pulmonary artery sarcoma (PAS) is a rare and devastating disease. The diagnosis is often delayed, and optimal treatment remains unclear. The aim of this study is to report our experience in the surgical management of this disease. Between 2000 and 2018, 17 patients underwent operations for PAS at our center. The medical records were retrospectively reviewed to evaluate the clinical characteristics, operative findings, the postoperative outcomes, and the long-term results. The mean age at operation was 46.0 ± 12.4 years (range, 26–79 years), and eight (47.1%) patients were male. Six patients underwent tumor resection alone, whereas the other 11 patients received pulmonary endarterectomy (PEA). There were two perioperative deaths. Follow-up was completed for all patients with a mean duration of 23.5 ± 17.6 months (1–52 months). For all 17 patients, the median postoperative survival was 36 months, and estimated cumulative survival rates at 1, 2, 3, and 4 years were 60.0%, 51.4%, 42.9%, and 21.4%, respectively. The mean survival was 37.0 months after PEA and 14.6 months after tumor resection only (p = 0.046). Patients who had no pulmonary hypertension (PH) postoperatively were associated with improved median survival (48 vs. 5 months, p = 0.023). In conclusion, PAS is often mistaken for chronic pulmonary thromboembolism. The prognosis of this very infrequent disease remains poor. Early detection is essential for prompt and best surgical approach, superior to tumor resection alone, and PEA surgery with PH relieved can provide better chance of survival.
Introduction
Since the first description of pulmonary artery sarcoma (PAS) by Mandelstamm in 1923, 1 fewer than 450 cases have been reported with disastrous prognosis.2–4 Due to the rarity of PAS, only case reports and small case series have been published.
Because the clinical manifestations and conventional imaging features of this potentially lethal tumor are usually nonspecific and mimic those of chronic pulmonary thromboembolism, a definite diagnosis is often delayed until surgical exploration or necropsy. What is more, the management of this fatal disease remains controversial. Surgery remains the mainstay of treatment for PAS3–5; however, there is no widely accepted surgical approach for PAS until now. Prior studies have noted the importance of early radical resection for long-term survival,5–7 while pneumonectomy and pulmonary endarterectomy (PEA) represent the two mainstream choices, with the role of adjuvant therapy as yet not clearly defined.
There have been few reports regarding the long-term results of treatment for PAS. In this study, we retrospectively reviewed the early and late outcomes after treatment for 17 PAS patients at our center and reported our surgical experience with this orphan disease.
Methods
Study population
Between August 2000 and December 2018, 17 patients underwent operations and were histologically diagnosed with PAS at our center. Diagnosis was confirmed by histological examination and immunohistochemistry staining. The medical records were retrospectively reviewed to evaluate the clinical characteristics, operative findings, the postoperative outcomes, and the long-term results. This study followed the ethical standards of the World Health Organization’s Declaration of Helsinki. The Institutional Review Board of our center approved the protocol (16 January 2018; NO.: 2018–991).
Follow-up and statistical analysis
The patients were contacted by telephone interview or office visit. Overall survival was defined as the death before the end of follow-up (30 April 2020).
Continuous variables were described as the means ± standard deviation or medians with range, according to the normality of distribution assessed using the Kolmogorov–Smirnov test. Survival analysis was performed using the Kaplan–Meier method and the log-rank test. Surviving patients were censored at last contact. All analyses were performed using SPSS version 21.0. For all tests, p < 0.05 was considered statistically significant.
Results
Patient characteristics
The patient baseline characteristics are shown in Table 1. The mean age at operation was 46.0 ± 12.4 years (range, 26–79 years), and eight (47.1%) patients were male. The presenting symptoms included dyspnea (n = 16, 94.1%; New York Heart Association class III and IV in 52.9% of cases), chest pain (n = 8, 47.1%), and hemoptysis (n = 8, 47.1%), whereas syncope (n = 6, 35.3%) and prolonged fever (n = 3, 17.6%) were also observed. The median duration from the initial symptoms to surgery was six months. Median plasma D-dimer was 0.88 ug/ml (range, 0.12–2.76 ug/ml), with 10 (58.8%) patients exceeding the normal range (0.5 ug/ml). All the patients underwent at least one or two diagnostic tests among transthoracic echocardiography, computed tomography (CT) (Fig. 1c and d), positron emission tomography, pulmonary angiograms, and right heart catheterization. Eight (47.1%) patients exhibited pulmonary hypertension (PH), with the median systolic pulmonary artery pressure (sPAP) of 71.5 mmHg (range, 45–114 mmHg). On the basis of the findings of the imaging studies, 9 (52.9%) patients were suspected to have PAS, whereas 6 (35.3%) patients were misdiagnosed as having pulmonary embolism (PE) and underwent anticoagulation treatment before surgery; the remaining two patients were misdiagnosed as having pulmonary stenosis before operation.
Patient characteristics and therapeutic procedures.
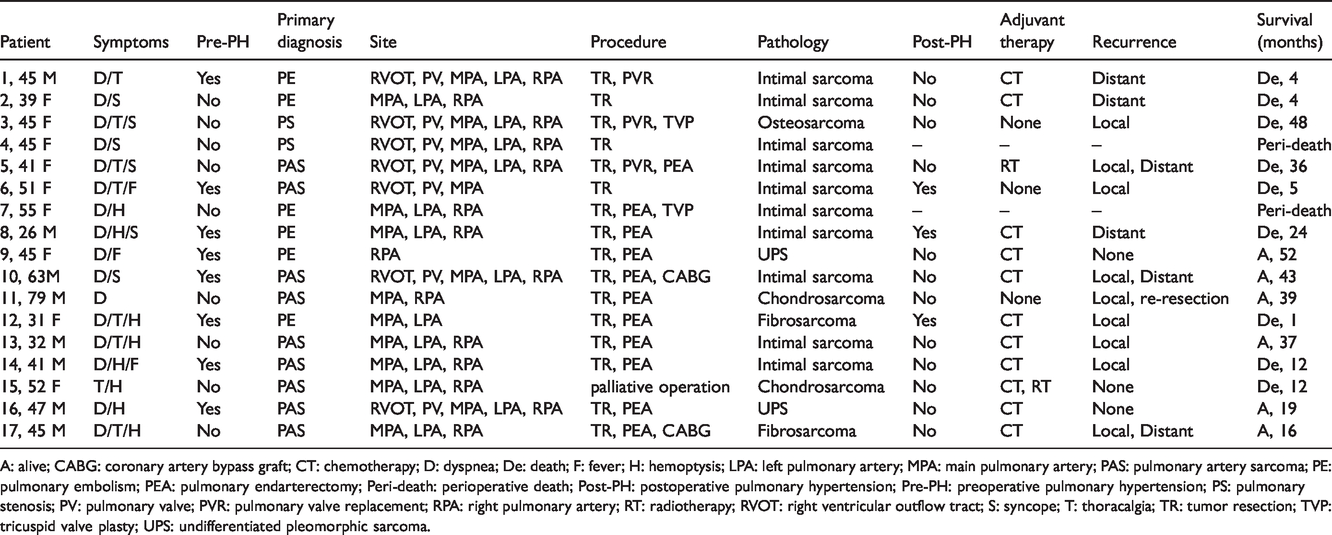
A: alive; CABG: coronary artery bypass graft; CT: chemotherapy; D: dyspnea; De: death; F: fever; H: hemoptysis; LPA: left pulmonary artery; MPA: main pulmonary artery; PAS: pulmonary artery sarcoma; PE: pulmonary embolism; PEA: pulmonary endarterectomy; Peri-death: perioperative death; Post-PH: postoperative pulmonary hypertension; Pre-PH: preoperative pulmonary hypertension; PS: pulmonary stenosis; PV: pulmonary valve; PVR: pulmonary valve replacement; RPA: right pulmonary artery; RT: radiotherapy; RVOT: right ventricular outflow tract; S: syncope; T: thoracalgia; TR: tumor resection; TVP: tricuspid valve plasty; UPS: undifferentiated pleomorphic sarcoma.
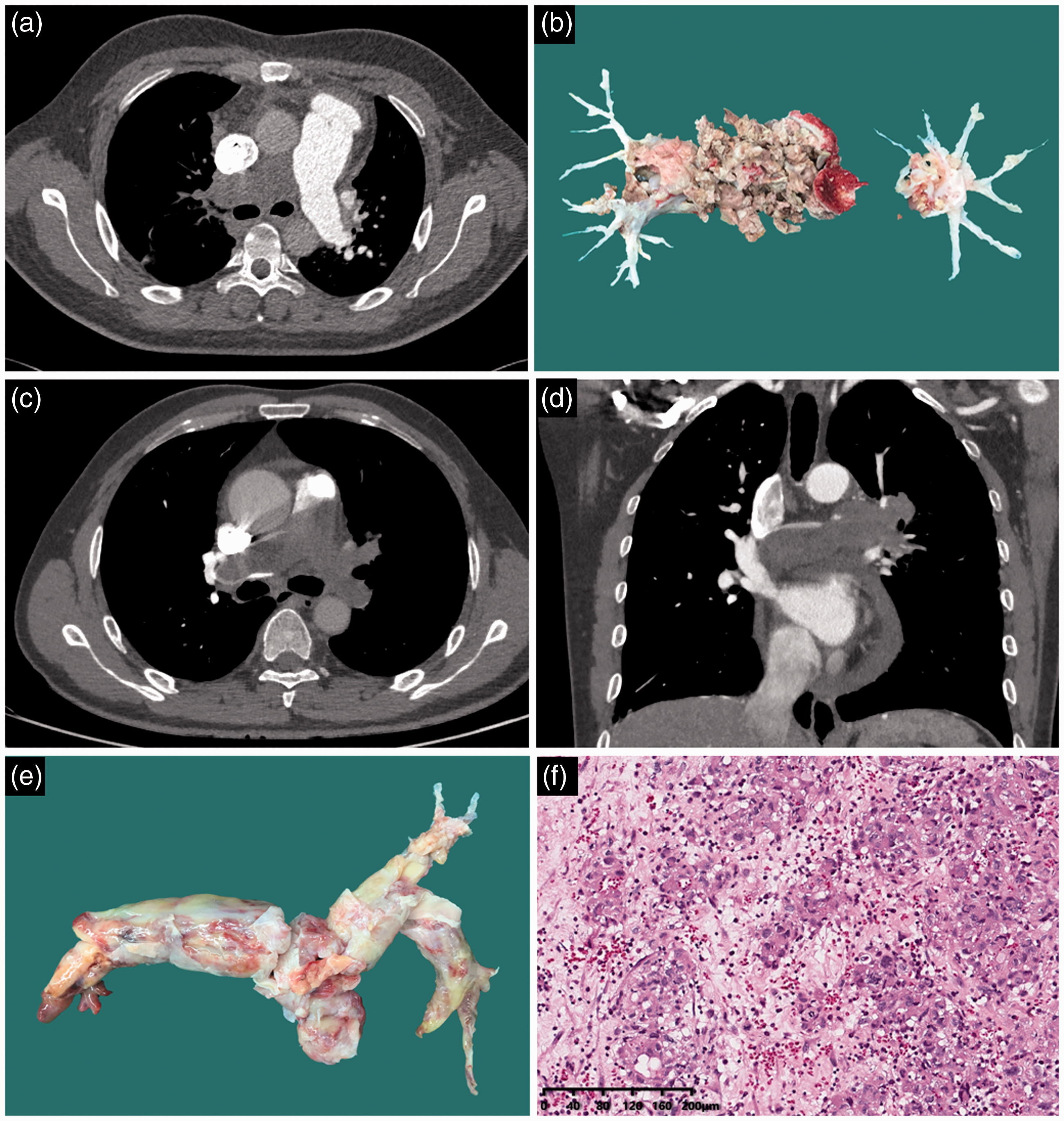
The CT images, surgical specimen, and histopathology features of CTEPH and PAS patients. (a) The CT image of a CTEPH patient shows a large cup-shaped mass in the right pulmonary artery; its branches are not shown. (b) Surgical specimen of a CTEPH patient. (c and d) The CT images of a PAS patient show a large and lobular filling defect in the main pulmonary trunk with continuous lesions straddling bilateral pulmonary arteries, extending medially into the truncus pulmonalis and distally into the lobar and segmental arteries of both lungs. (e) Surgical specimen of an undifferentiated pleomorphic PAS. (f) Hematoxylin and eosin staining of the tumor from the intima of the pulmonary artery, and high magnification demonstrates prominent pleomorphism and a storiform pattern.
Surgical procedures
As shown in Table 1, intraoperative examination confirmed tumors frequently involved pulmonary trunk (n = 15, 88.2%), pulmonary trunk into two branches (n = 12, 70.6%), and right ventricular outflow tract and pulmonary valve (n = 7, 41.2%). Interestingly, thrombosis was found simultaneously in three patients during the operation.
All operations were performed under general anesthesia and approached through a median sternotomy. All patients underwent tumor resection with the aid of cardiopulmonary bypass (CPB), and every effort was made to remove as much of the tumor as possible. In all patients with a sarcoma tumor that involved right ventricular outflow tract and pulmonary valve, the tumor was carefully peeled, with valve prosthesis replacement in three patients. As previously reported, 3 from 2004, PEA, in addition to PAS resection, was routinely undertaken in 10 patients. PEA was performed on both sides following the standard procedure with deep hypothermic circulatory arrest (DHCA) or deep hypothermic low flow established by the University of California San Diego (UCSD) group. 8 The process of CPB with DHCA for PEA had been described in detail previously. 9 Once CPB was initiated, active cooling was started with a heat-cool machine, a cooling head device, and cooling blanket. During the cooling period, some preliminary dissection was performed. When the nasopharyngeal temperature reached 18–20°C, a circulatory arrest was initiated. Endarterectomy was performed in a completely bloodless field on both sides during separate episodes of circulatory arrest (up to 20 min). Circulation was resumed for at least 10 min between arrest episodes.
In our series (Table 2), the mean CPB time and aortic clamping time were 226.5 ± 105.4 and 122.6 ± 58.0 min, respectively; DHCA was required in five patients (mean, 13.4 ± 2.7 min). Deep hypothermic low flow was applied in eight patients, whereas the other four patients received mild or moderate hypothermic CPB. Final histologic diagnosis and immunohistochemical results were available for all the 17 PAS patients (Table 1). The histopathological subclassification included intimal sarcoma in 10 (58.8%) patients, fibrosarcoma in 2 (11.8%) patient, undifferentiated pleomorphic sarcoma (Fig. 1e and f) in 2 (11.8%) patients, chondrosarcoma in 2 (11.8%) patients, and osteosarcoma in 1 (5.8%) patient.
Operative data and perioperative outcomes.
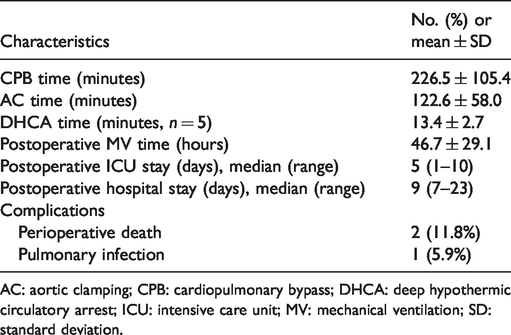
AC: aortic clamping; CPB: cardiopulmonary bypass; DHCA: deep hypothermic circulatory arrest; ICU: intensive care unit; MV: mechanical ventilation; SD: standard deviation.
Early results
The two (11.8%, patients 4 and 7) hospital deaths occurred before 2007, and both were related to unresolved severe PH after resection and failed to wean from bypass. The possible reasons included uncomplete removal of tumor and poor myocardial protection. No other patients suffered from significant complications related to the operations and were discharged uneventfully, except for one patient who developed pulmonary fungal infection but received timely and appropriate antibiotic therapy. As shown in Table 2, the mean length of mechanical ventilation time was 46.7 ± 29.1 h. The median lengths of intensive care unit stay and postoperative hospital stay were 5 days (range, 1–10 days) and 9 days (range, 7–23 days), respectively. Thrombosis was found simultaneously in three patients, all of whom underwent anticoagulation treatment (warfarin) after surgery. There were three patients (patients 6, 8, and 12) who had PH postoperatively, with the median sPAP of 49 mmHg (range, 46–53 mmHg). Patients 6 and 8 took sildenafil after discharge, but patient 12 did not receive pulmonary arterial hypertension–targeted pharmacotherapy due to his unwillingness. Eleven patients received chemotherapy or radiotherapy or both postoperatively, whereas the remaining four patients did not receive any adjuvant treatment (Table 1). The therapy regimens ranged from taxane plus carboplatin to ifosfamide plus epirubicin or pegylated liposomal doxorubicin regimens.
Late survival
Follow-up was completed for all the patients with a mean duration of 23.5 ± 17.6 months (range, 1–52 months). At the last follow-up, the sPAP of patients 6 and 8 were 44 and 50 mmHg, respectively. During follow-up, nine patients died after a mean survival time of 16.2 months (range, 1–48 months), and the cause of death was related to the recurrence or metastasis of PAS. For the six patients alive at follow-up, two (patients 11 and 13) already had local recurrence without metastasis, one (patient 10) had local recurrence with brain metastasis, and one (patient 17) had local recurrence with lung metastasis. In the two patients noted to have recurrent disease without metastasis, one (patient 11) underwent reoperation at the age of 81, two years after his first surgery.
For all 17 patients, the median postoperative survival was 36 months, and estimated cumulative survival rates at 1, 2, 3, and 4 years were 60.0%, 51.4%, 42.9%, and 21.4%, respectively (Fig. 2). The mean survival was 37.0 months after PEA and 14.6 months after tumor resection only (p = 0.046, Fig. 3a). Patients who had no PH postoperatively were associated with improved median survival (48 vs. 5 months, p = 0.023, Fig. 3b).
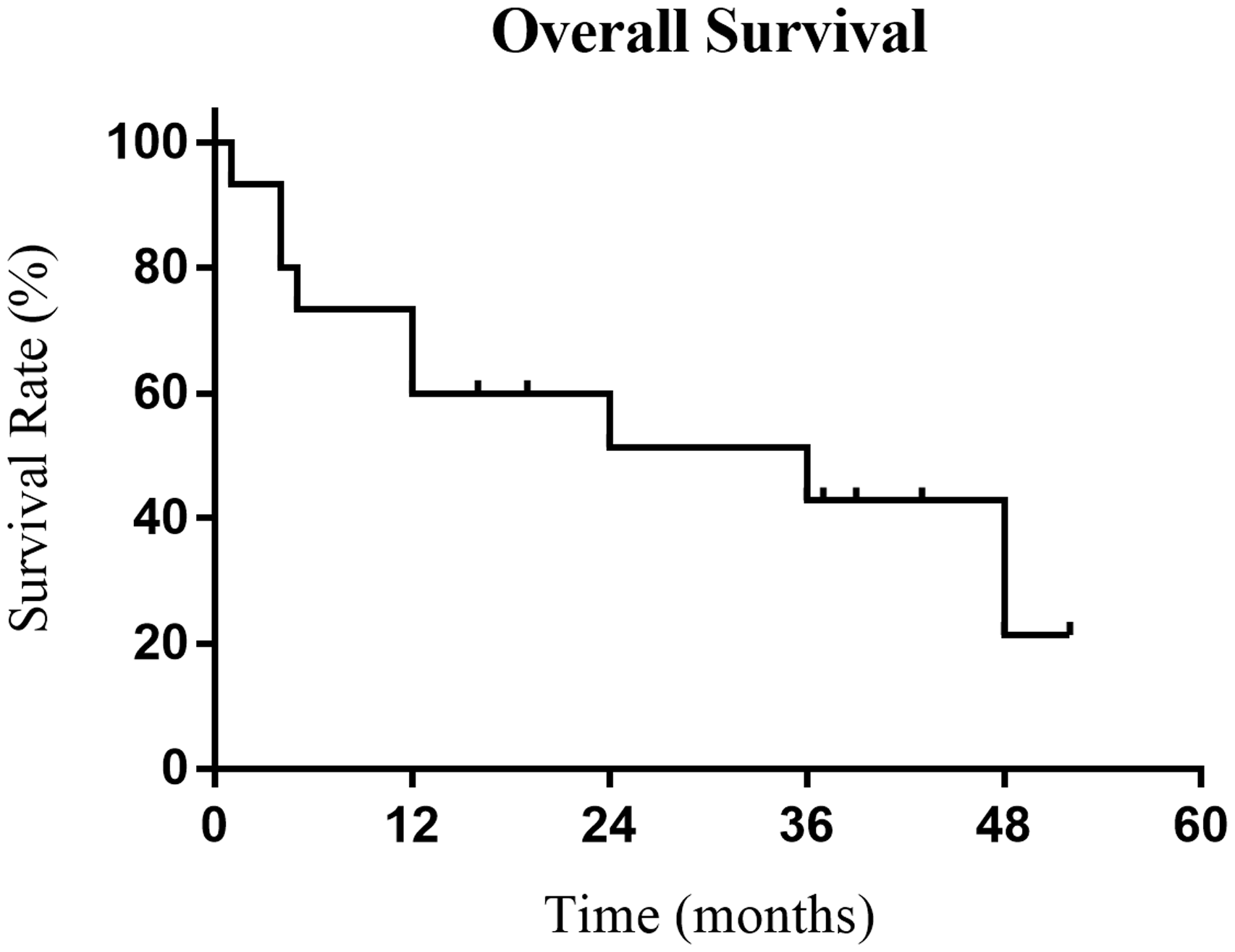
Statistical cumulative survival (Kaplan–Meier estimator) of all patients.
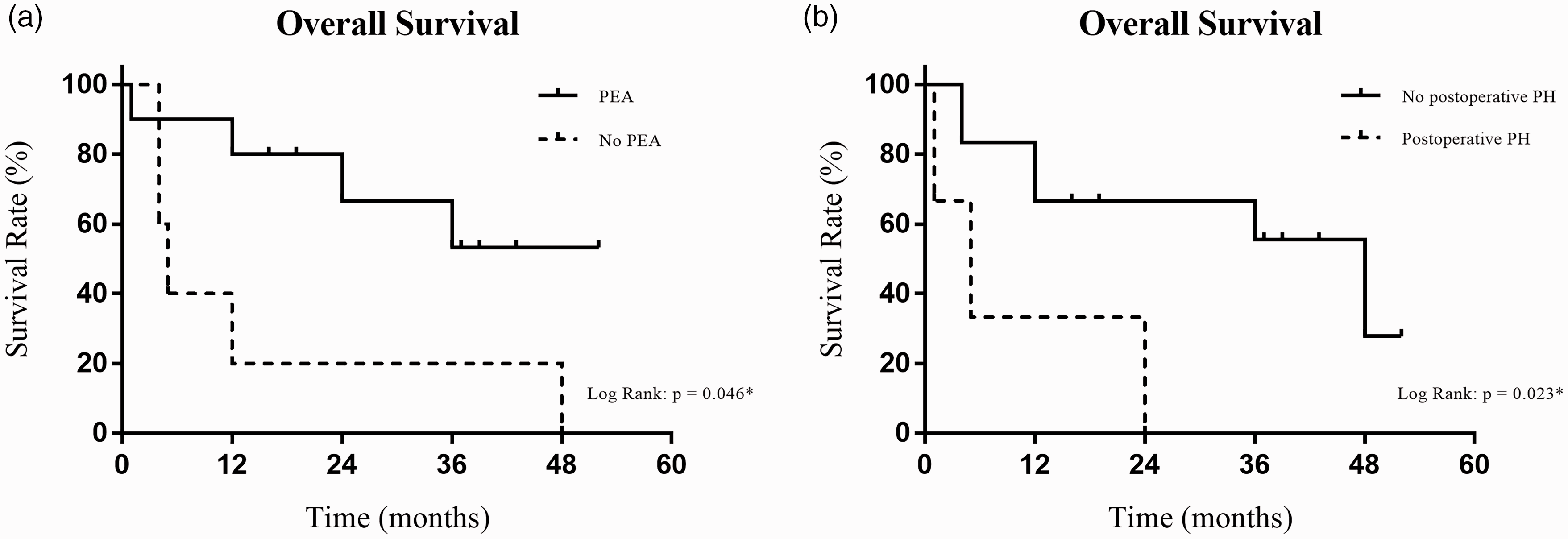
Statistical cumulative survival (Kaplan–Meier estimator) of patients. (a) Patients who underwent PEA compared to those who did not (p = 0.046*). (b) Patients who had PH postoperatively compared to those who did not (p = 0.023*).
Discussion
Our cohort of 17 patients is one of the biggest series, considering the infrequency of the disease, and limited studies reported more than 15 cases.5,7,10,11 Since PAS is likely to be mistaken for chronic thromboembolic pulmonary hypertension (CTEPH) due to the similar symptoms and imaging features,4,12,13 an early diagnosis is challenging, and patients are often initially treated with inappropriate therapy such as prolonged anticoagulation or thrombolysis. In the current study, the initial diagnosis was CTEPH in six patients and pulmonary stenosis in two patients. The median duration from the beginning of symptoms to surgery was six months, comparable to the results reported in previous studies.4,5,14
In recent years, heightened clinical awareness and advanced imaging methods have allowed the diagnosis to be made prior to surgery. Our experience3,15 indicates that PAS should be considered in the differential diagnosis of CTEPH patients in cases that (a) have no risk factors for PE such as deep vein thrombosis, (b) lack response to anticoagulation or thrombolysis therapy, and (c) the radiologic findings such as the involvement of right ventricular outflow tract and the pulmonary valve, pedunculated or lobulated masses, and extravascular invasion were observed. Other studies16,17 also support that the location (main or proximal pulmonary artery, extraluminal extension) and morphology (in full shape or expansive growth, and the proximal end of the tumor bulging or lobulated while aneurysm or grape-like distally with inhomogeneous enhancement) of lesions in CT or magnetic resonance imaging have great value for the suspected diagnosis of PAS. From 2014, positron emission tomography/computed tomography was performed in 10 patients, but only 4 of them were highly suspected of having PAS in recent series, as it is hard to differentiate between the inflammation of thrombus organization and malignant neoplasm. Other novel diagnostic methods currently being refined include the use of endovascular catheter biopsy 18 and endobronchial ultrasound-guided transbronchial needle aspiration. 19
Operation remains the mainstay of treatment for PAS.3–5 Prior studies that have noted the importance of early radical resection for long-term survival,5–7 while PEA and pneumonectomy represent the two mainstream choices. As Kruger et al. 20 pointed out, with an incidence of pulmonary metastases of 58%, pneumonectomy for PAS appeared to be the most reasonable oncological concept. According to the experience of UCSD,21,22 PAS was always bilateral if the patient presented with PH, and even in the absence of PH in an apparently unilateral case, bilateral seeding had probably occurred by the time the patient presented. So, they insist on PEA for palliative reasons and extended life expectancy and did not recommend pneumonectomy for this disease. With accumulating experience, Grazioli’s team 14 convinced that the presence of PH was usually associated with bilateral extension of PAS, even in the case of a unilateral CT pattern. Thus, pneumonectomy was indicated only in the case of unilateral presentation and the absence of PH in their center. As most patients had evidence of bilateral disease with eight patients with coexisting PH at baseline, we had chosen PEA and had not performed a pneumonectomy for PAS in our center. It indicates that the optimum surgical strategy for PAS patients should not only remove the tumor as completely as possible but also relieve the PH, and PEA should be given prior consideration. The residual PH post-PEA deserves attention. However, because of the small sample size, further studies are warranted.
Chemotherapy and/or radiotherapy is normally given postoperatively in most centers; however, the effect of postoperative adjuvant therapies remains controversial. Bandyopadhyay et al. 4 studied from their 10 PAS patients and another 381 cases reported in the literature and found that patients who received chemotherapy had a significantly improved survival. Yin et al. 2 recently observed the same effect of postoperative adjuvant therapy. However, Mussot et al., 5 who published the largest serial cohort case analysis of 31 PAS patients, showed that postoperative adjuvant therapy did not significantly impact survival outcome for PAS. Similar result was found in this series. Other than chemo- and radiotherapy, a number of reports have found that repeated surgical interventions could also be associated with prolonged survival in patients with recurrence or metastatic PAS.23,24 In our study, two patients noted to have recurrent disease without metastasis, especially one (patient 11), the oldest example for reoperation as we know, underwent secondary operation successfully at the age of 81.
Limitations
An inherent shortcoming with a study of such a rare disease is the limited patient volume. However, our cohort of 17 patients is one of the biggest series, considering the infrequency of the disease, and limited studies reported more than 15 cases. Another limitation of our study is the lack of complete data for some patients who were discharged back to their local hospitals. We do not have detailed information of postoperative chemo- and/or radiotherapy for two patients, which may have affected the survival and prognosis.
Conclusions
PAS is an uncommon malignancy, and the prognosis remains very poor even after surgical resection. Early detection is an essential prerequisite to allow for prompt and best surgical approach, and PEA surgery with PH relieved can provide the best chance of survival.
Footnotes
Acknowledgements
The authors thank Dr Xiao Chen and Dr Qian Zhao at the State Key Laboratory of Cardiovascular Disease, FuWai Hospital, National Center for Cardiovascular Diseases, Chinese Academy of Medical Sciences and Peking Union Medical College, Beijing, China for their kind assistance in specimen collection and pathological analysis and Dr Jiangping Song at FuWai Hospital, National Center for Cardiovascular Diseases, Chinese Academy of Medical Sciences and Peking Union Medical College, Beijing, China for his guidance on this study.
Conflict of interest
The author(s) declare that there is no conflict of interest.
Authors’ contributions
WS analyzed most of the data and prepared the manuscript; LD, JZ, and SZ collected the data and carried out additional analyses; HW concluded the image characters of patients; YS and SL designed and coordinated the study. All authors read and approved the final manuscript.
Ethical approval
The Institutional Review Board of Fuwai Hospital approved the protocol (16 January 2018; NO.: 2018-991).
Guarantor
Sheng Liu is the guarantor of this article and takes responsibility for the content of the manuscript, including the data and analysis.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Capital Clinical Feature Application Research and Achievements Promotion (Z171100001017215) and the Chinese Academy of Medical Sciences Innovation Fund for Medical Sciences (CIFMS) (2017-I2M-3–003).
