Abstract
Acute respiratory distress syndrome in patients with Coronavirus disease 19 is associated with an unusually high incidence of pulmonary embolism and microthrombotic disease, with evidence for reduced fibrinolysis. We describe seven patients requiring invasive ventilation for COVID-19-associated acute respiratory distress syndrome with pulmonary thromboembolic disease, pulmonary hypertension ± severe right ventricular dysfunction on echocardiography, who were treated with alteplase as fibrinolytic therapy. All patients were non-smokers, six (86%) were male and median age was 56.7 (50–64) years. They had failed approaches including therapeutic anticoagulation, prone ventilation (n = 4), inhaled nitric oxide (n = 5) and nebulised epoprostenol (n = 2). The median duration of mechanical ventilation prior to thrombolysis was seven (5–11) days. Systemic alteplase was administered to six patients (50 mg or 90 mg bolus over 120 min) at 16 (10–22) days after symptom onset. All received therapeutic heparin pre- and post-thrombolysis, without intracranial haemorrhage or other major bleeding. Alteplase improved PaO2/FiO2 ratio (from 97.0 (86.3–118.6) to 135.6 (100.7–171.4), p = 0.03) and ventilatory ratio (from 2.76 (2.09–3.49) to 2.36 (1.82–3.05), p = 0.011) at 24 h. Echocardiographic parameters at two (1–3) days (n = 6) showed right ventricular systolic pressure (RVSP) was 63 (50.3–75) then 57 (49–66) mmHg post-thrombolysis (p = 0.26), tricuspid annular planar systolic excursion (TAPSE) was unchanged (from 18.3 (11.9–24.5) to 20.5 (15.4–24.2) mm, p = 0.56) and right ventricular fractional area change (from 15.4 (11.1–35.6) to 31.2 (16.4–33.1)%, p = 0.09). At seven (1–13) days after thrombolysis, using dual energy computed tomography imaging (n = 3), average relative peripheral lung enhancement increased from 12.6 to 21.6% (p = 0.06). In conclusion, thrombolysis improved PaO2/FiO2 ratio and ventilatory ratio at 24 h as rescue therapy in patients with right ventricular dysfunction due to COVID-19-associated ARDS despite maximum therapy, as part of a multimodal approach, and warrants further study.
Keywords
Acute respiratory distress syndrome (ARDS) in patients with Coronavirus disease 19 (COVID-19) is associated with an unusually high incidence of pulmonary embolism (PE) and thrombosis, even in patients who have been fully anticoagulated. In addition to PE and deep vein thrombosis, autopsy specimens and imaging studies including dual energy computed tomography (DECT) imaging suggest an additional microthrombotic process.1,2 Microthrombosis has been previously described in ARDS and viral pneumonias, but appears to be more pronounced in COVID-19. 3 Patients may develop PE (‘macrothrombosis’), microthrombosis or a combination of these. 4 The onset of pulmonary hypertension (PH) and right ventricular dysfunction (RVD) is more common in patients with PE in COVID-19.5–7 RVD may be severe, 8 and associated with pulmonary microthrombosis early in the disease course. 9
Reduced fibrinolysis is frequently observed in severe COVID-19 cases, 1 and may result from high levels of plasminogen activator inhibitor (PAI)-1 release from infected, activated, necrotic endothelium and activated platelets. PAI-1 polymerises fibrin within the thrombus and can inhibit tissue plasminogen activator (tPA)-mediated fibrinolysis. This process is likely to contribute to the development of micro- and macrovascular thrombosis, 1 as well as alveolar fibrin deposition.
The rationale for tPA treatment for COVID-19-associated ARDS is therefore supported by the need for effective fibrinolysis. Cases so far describing the use of systemic tPA in patients with severe COVID-19 and ARDS have all had clinically suspected pulmonary thrombosis (with or without confirmed evidence of pulmonary thrombosis on CT imaging). These reported cases were refractory to interventions including prone ventilation, and had variable initial or durable improvement in their PaO2/FiO2 (P/F) ratio, without significant bleeding complications.10–12
The observed acute changes in ventilatory parameters may reflect an improvement in lung perfusion resulting in increased ventilatory efficiency, as well as a reduction in pulmonary vascular resistance and augmented cardiac output. We would like to extend these findings by reporting the experience from our centre, selecting patients with RVD and not just macrothrombotic PE on imaging, but also with microthrombosis. We describe short-term changes in repeated echocardiographic studies, and utilise follow-up DECT imaging to reflect potential changes to the pulmonary microcirculation.
We describe seven patients in a single UK intensive care unit requiring invasive mechanical ventilatory support for severe COVID-19 pneumonia. The study was approved by the Research Ethics Committee (reference number: 20/EE/0160). All patients lacked capacity, and the need for individual informed consent was waived for retrospective analysis of data collected prospectively for routine care, with no breach of privacy or anonymity. Patients selected were those with ARDS due to severe COVID-19 on maximum therapy with evidence of severe PH and/or RVD with pulmonary microvascular and/or macrovascular thrombosis on computed tomography pulmonary angiography and with DECT if possible. RVD or PH was assessed using transthoracic echocardiography and/or pulmonary artery catheterisation. Echocardiographic features were according to the latest international recommendations, with the presence of severe PH and RV dilatation, and reduced radial and long axis RV function (TAPSE < 17 mm, FAC < 35%). The presence of PH was defined on echo if TRv was available with a good Doppler trace, with systolic pulmonary artery pressure (SPAP) estimated using the following formula: SPAP = 4 × tricuspid regurgitation peak velocity 2 + right atrial pressure. PH was defined as SPAP > 35mmHg.13,14 Pulmonary arterial catheterisation used standard diagnostic criteria for PAH, using mPAP ≥ 25 mmHg rather than the 2018 adaptation of the mPAP cut off.
Clinical and laboratory parameters were analysed from electronic patient records. Physiological parameters were recorded pre- and 24 h post-thrombolysis. Measurements taken in patients receiving prone ventilation were taken in the same patient position at each time point. Where available, echocardiographic and CT measurements were assessed prior to and at the next available assessment following thrombolysis. Statistical analysis was performed using Graphpad Prism v8.3. Data are presented as mean (standard deviation) and median (interquartile range), as appropriate. Categorical variables were summarised as counts and percentages. Pre- and post-thrombolysis parameters were compared using paired analyses for non-parametric data (Wilcoxon paired tests), using p < 0.05 for statistical significance.
All patients were non-smokers, six (86%) were male and median age was 56.7 (interquartile range 50–64) years. None had prior vascular, autoimmune disease or malignancy. Pulmonary thromboembolic disease included one lobar occlusive PE, five segmental PE and three additional distal perfusion defects on DECT scan; in addition, one had distal defects only. All had either severe PH (confirmed on right heart catheterisation, n = 2) and/or RVD on echocardiography (severe in n = 5) despite vasopressor support (n = 5). One patient was commenced on veno-venous extracorporeal membrane oxygenation (VV-ECMO) two days prior to thrombolysis. They had failed conventional approaches including therapeutic anticoagulation with intravenous unfractionated heparin, prone ventilation (n = 4), inhaled nitric oxide (n = 5) and nebulised epoprostenol (n = 2). The median duration of mechanical ventilation prior to thrombolysis was seven (5–11) days.
Systemic alteplase was administered to six patients (50 or 90 mg bolus over 120 min) at 16 (10–22) days after symptom onset. All received therapeutic heparin pre- and post-thrombolysis, with no intracerebral haemorrhage or other major bleeding events. The patient on VV-ECMO had an occlusive proximal PE with severe RVD and underwent catheter-directed thrombolysis due to ECMO-related bleeding concerns.
Immediately after thrombolysis, D-dimer increased (from 3832 (1062–18829) to 16,011 (10,920–42,146) ng/ml, p = 0.10), and at 11 (10–14) h after thrombolysis, fibrinogen fell from 6.75 (5.95–8.83) g/L to 4.40 (3.88–6.60) g/L (p = 0.04). Other bloods showed no different in inflammatory markers over the same time period (C-reactive protein 280 mg/L (185–318) to 263 mg/L (158–373), p = 0.85; white blood cells 17.9 × 109 (12.9–22.2) to 16.3 × 109 (13.2–19.6), p = 0.73). At 24 h following thrombolysis, in the six non-ECMO patients, P/F ratio improved (from 97.0 (86.3–118.6) to 135.6 (100.7–171.4), p = 0.03) (Fig. 1a) and ventilatory ratio improved (from 2.76 (2.09–3.49) to 2.36 (1.82–3.05), p = 0.011) (Fig. 1b).
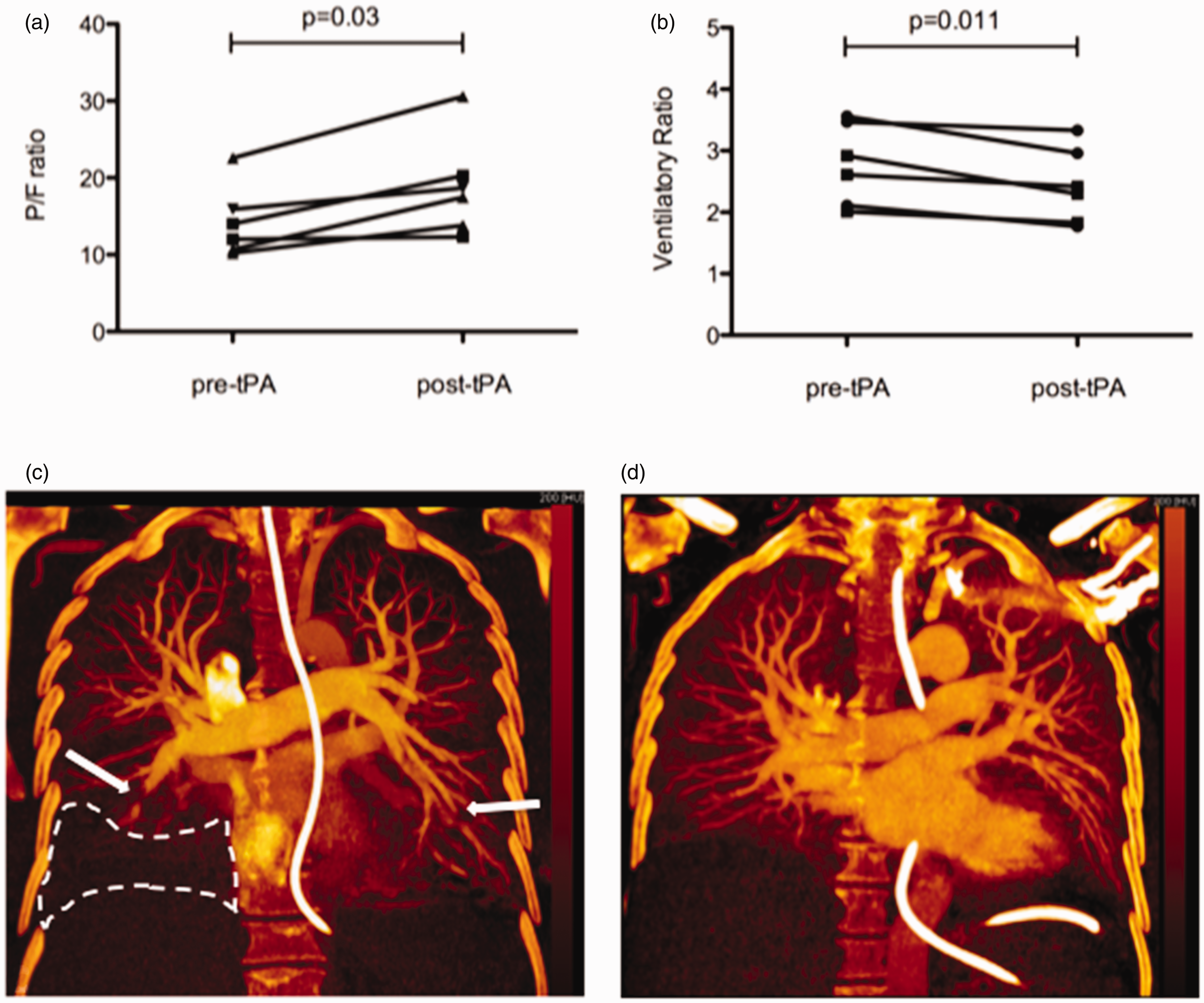
(a) P/F ratios of six patients on mechanical ventilation pre- and 24 h post-systemic thrombolysis with alteplase. (b) Ventilatory ratios of six patients on mechanical ventilation pre- and 24 h post-systemic thrombolysis with alteplase. (c) Coronal dual energy CT scan images of a 64-year-old male with severe COVID-19 pneumonia (c) pre- and (d) post-systemic thrombolysis with alteplase.
Six patients had echocardiographic follow-up data available at two (1–3) days after thrombolysis. In these patients, RVSP was 63 (50.3–75) then 57 (49–66) mmHg post-lysis (p = 0.26), TAPSE was unchanged (from 18.3 (11.9–24.5) to 20.5 (15.4–24.2) mm, p = 0.56) and RV fractional area change trended to improvement (from 15.4 (11.1–35.6) to 31.2 (16.4–33.1)%, p = 0.09). There were no differences in cardiac biomarkers at the 24-h time point (troponin 71 ng/L (13–137) to 68 ng/L (59–113), p = 0.84) (n = 7), brain natriuretic peptide 617 ng/L (176–646) to 224 ng/L (97–467), p = 0.28 (only available in four patients).
At follow-up CT (n = 5), nine (1–13) days following thrombolysis, despite 3/5 patients demonstrating worsening parenchymal lung disease, a decrease in proximal thrombus burden was seen in four patients. In patients with paired DECT analysis (n = 3) at a median of seven (1–13) days after thrombolysis, the average relative peripheral lung enhancement improved from 12.6 to 21.6% (p = 0.06), in keeping with a trend to improvement in distal perfusion (Fig. 1c and d). At the time of writing, three patients had died, and four patients survived to discharge from critical care.
We demonstrate that thrombolysis resulted in a significantly improved ventilatory and P/F ratio in a carefully selected group of patients with severe COVID-19 with RVD and evidence of thrombosis, including microthrombosis, who failed conventional ICU interventions; however, there was no significant change in RV function or cardiac biomarkers, suggesting the impact was on ventilation perfusion matching rather than improved cardiac output. This was a small, uncontrolled cohort and it is impossible to make conclusions to change practice, but our results suggest that thrombolysis used here as rescue therapy significantly improved ventilatory parameters as well as a trend to echocardiographic measures of RV function. Furthermore, in those who survived, thrombolysis, as part of a multi-modal approach, led to a sustained trend to improvement on the follow-up DECT scans available, in terms of micro- and macrovascular perfusion. This last observation was despite progression of parenchymal disease on CT.
Therapeutic strategies targeting the pulmonary circulation in COVID-19 are likely to require a multimodal approach. Thrombolysis has been previously shown to clear vascular obstructions in ARDS, 15 but this approach is not usually applied outside the setting of confirmed PE. However, with the profound immune activation and intravascular thrombosis seen in COVID-19, this approach is of interest, especially with the observation that microthrombosis is common. In a study with 60 ARDS patients without COVID-19 treated with nebulized heparin (10,000 IU/4 h), nebulized streptokinase (250,000 IU/4 h) and conservative management, P/F ratio was higher in the streptokinase group from day 1 to day 8. The streptokinase group also had improved lung compliance, pCO2 and lower ICU mortality. 16 Evidence does suggest that COVID-19 presents a sufficiently ‘pulmonary vascular’ ARDS phenotype with relevant haematological abnormalities that fibrinolysis is an avenue worthwhile exploring. The prevalent defects in microvascular perfusion suggested by DECT imaging in COVID-19 is an important recent observation, 1 and we have shown, albeit in small numbers, that this may a realistic method to re-examine the impact of pulmonary vascular interventions in the acute setting. Whether this is a realistic end point for studies in ARDS is of interest.
This description of a small retrospective cohort has obvious limitations, and conclusions are limited due to the small sample size, as well as the lack of a control group. In addition, DECT scanning was not available in all follow-up scans to determine the impact on microthrombotic disease. Major bleeding was not seen; however, the number of patients was too small to conclude safety.
PE is common in patients with COVID-19: with a resulting spectrum of thromboembolic disease (macrovascular, macro + microvascular or only microvascular disease). Given the prothrombotic, hypofibrinolytic state, we propose that thrombolysis might be a useful rescue therapy based on this small series of consecutive patients who were on maximum intensive care treatment with RVD, and were unlikely to survive. It is increasingly reported that PH on echo, RV dilatation and RVD are independent risk factors for mortality in patients with COVID-19,5,6 even in a non-ventilated cohort. 7
This is the first report to describe the impact of rescue thrombolysis on short-term follow-up echocardiography and imaging of the pulmonary microcirculation using DECT studies in patients with severe COVID-19 ARDS, thrombosis and RVD. Further to short-term improvement in oxygenation and ventilatory efficiency, we show a trend to improvement in perfusion score in a small group of follow-up DECT scans, in survivors, which needs further exploration.
Randomised controlled studies are needed to understand the impact of thrombolysis on micro- and macrothrombosis, ventilatory efficiency and RVD in these complex patients with COVID-19 ARDS, the potential survival benefit, safety and the possible role of thrombolysis in preventing longer-term pulmonary vascular disease.
Footnotes
Acknowledgements
Authors would like to thank Dr Thomas Semple, Dr Charlotte Briar, Dr Melissa Baldwin, Dr Aikaterini Vlachou, Dr Katherine Good, Dr Donna Hall, Dr Alex Rosenberg, Dr Nick Lees, Professor Simon Padley, Dr Brijesh V. Patel, Mr Christopher Remmington and all clinical staff in the adult intensive care unit, Royal Brompton & Harefield NHS Foundation Trust.
Author contributions
L.C.P. and D.J.A. were involved in design of the study, literature search, data collection, interpretation of the data and writing the manuscript. B.G. was involved in data collection, interpretation of the data and writing the manuscript. A.K. and C.M. were involved in data collection. C.B. was involved in data collection, interpretation and analysis. C.A.R. was involved in figure and data interpretation. S.P., C.C. and S.J.W. were involved in design of the study. All authors approved the final version of the manuscript.
Conflict of interest
D.J.A. has received funding for COVID-19 haematology studies. Otherwise none.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
